Tom Whole Care®
7db8e49d-1073-4a81-a3a4-f3a19ebce921
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Anticavity
Medication Information
Purpose
Anticavity
Description
Drug Facts
Use
helps protect against cavities
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children under 6 years of age.
If more than used for brushing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Directions
| adults and children 2 years of age and older | brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or physician |
| children 2 to 6 years | use only a pea-sized amount and supervise child's brushing and rinsing (to minimize swallowing) |
| children under 2 years | ask a dentist or physician |
Questions?
1-800-FOR-TOMS (1-800-367-8667)
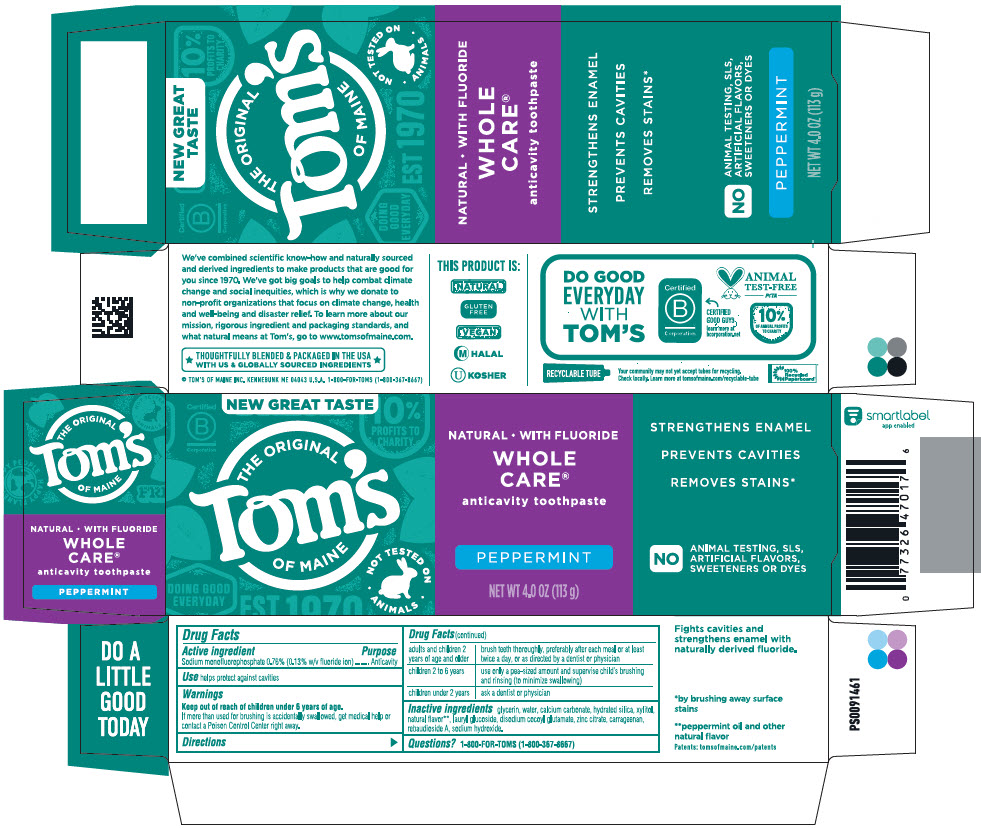
Active Ingredient
Sodium monofluorophosphate 0.76% (0.13% w/v fluoride ion)
Inactive Ingredients
glycerin, water, calcium carbonate, hydrated silica, xylitol, natural flavor
Principal Display Panel 113 G Tube Carton
NEW GREAT TASTE
THE ORIGINAL
Tom's
OF MAINE
NOT TESTED ON
ANIMALS
NATURAL ∙ WITH FLUORIDE
WHOLE
CARE®
anticavity toothpaste
PEPPERMINT
NET WT 4.0 OZ (113 g)
STRENGTHENS ENAMEL
PREVENTS CAVITIES
REMOVE STAINS*
NO
ANIMAL TESTING, SLS,
ARTIFICIAL FLAVORS,
SWEETENERS OR DYES
Structured Label Content
Use
helps protect against cavities
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children under 6 years of age.
If more than used for brushing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Anticavity
Directions
| adults and children 2 years of age and older | brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or physician |
| children 2 to 6 years | use only a pea-sized amount and supervise child's brushing and rinsing (to minimize swallowing) |
| children under 2 years | ask a dentist or physician |
Questions?
1-800-FOR-TOMS (1-800-367-8667)
Active Ingredient (Active ingredient)
Sodium monofluorophosphate 0.76% (0.13% w/v fluoride ion)
Inactive Ingredients (Inactive ingredients)
glycerin, water, calcium carbonate, hydrated silica, xylitol, natural flavor
Principal Display Panel 113 G Tube Carton (PRINCIPAL DISPLAY PANEL - 113 g Tube Carton)
NEW GREAT TASTE
THE ORIGINAL
Tom's
OF MAINE
NOT TESTED ON
ANIMALS
NATURAL ∙ WITH FLUORIDE
WHOLE
CARE®
anticavity toothpaste
PEPPERMINT
NET WT 4.0 OZ (113 g)
STRENGTHENS ENAMEL
PREVENTS CAVITIES
REMOVE STAINS*
NO
ANIMAL TESTING, SLS,
ARTIFICIAL FLAVORS,
SWEETENERS OR DYES
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:24.244486 · Updated: 2026-03-14T23:10:48.259693