These Highlights Do Not Include All The Information Needed To Use Carvykti Safely And Effectively. See Full Prescribing Information For Carvykti.
7d040b91-3fb8-41db-ba7f-60a36f06e2c2
60684-8
CELLULAR THERAPY
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Cytokine Release Syndrome (CRS), including fatal or life-threatening reactions, occurred in patients following treatment with CARVYKTI. Do not administer CARVYKTI to patients with active infection or inflammatory disorders. Treat severe or life-threatening CRS with tocilizumab or tocilizumab and corticosteroids [see Dosage and Administration (2.2 , 2.3) , Warnings and Precautions (5.2) ] . Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS), which may be fatal or life-threatening, occurred following treatment with CARVYKTI, including before CRS onset, concurrently with CRS, after CRS resolution, or in the absence of CRS. Monitor for neurologic events after treatment with CARVYKTI. Provide supportive care and/or corticosteroids as needed [see Dosage and Administration (2.2 , 2.3) , Warnings and Precautions (5.3) ] . Parkinsonism and Guillain-Barré syndrome (GBS) and their associated complications resulting in fatal or life-threatening reactions have occurred following treatment with CARVYKTI [see Warnings and Precautions (5.3) ] . Hemophagocytic Lymphohistiocytosis/Macrophage Activation Syndrome (HLH/MAS), including fatal and life-threatening reactions, occurred in patients following treatment with CARVYKTI. HLH/MAS can occur with CRS or neurologic toxicities [see Warnings and Precautions (5.4) ]. Prolonged and/or recurrent cytopenias with bleeding and infection and requirement for stem cell transplantation for hematopoietic recovery occurred following treatment with CARVYKTI [see Warnings and Precautions (5.5) ] . Immune Effector Cell-associated Enterocolitis (IEC-EC), including fatal or life-threatening reactions, occurred following treatment with CARVYKTI [see Warnings and Precautions (5.9) ]. Secondary hematological malignancies, including myelodysplastic syndrome and acute myeloid leukemia, have occurred in patients following treatment with CARVYKTI. T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies, including CARVYKTI [see Warnings and Precautions (5.10) ] .
Indications and Usage
CARVYKTI is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma, who have received at least 1 prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent, and are refractory to lenalidomide.
Dosage and Administration
For autologous use only. For intravenous use only. Administer a lymphodepleting regimen of cyclophosphamide and fludarabine before infusion of CARVYKTI. ( 2.2 ) Do NOT use a leukodepleting filter. ( 2.2 ) Verify the patient's identity prior to infusion. ( 2.2 ) Premedicate with acetaminophen and an H1-antihistamine. ( 2.2 ) Avoid prophylactic use of systemic corticosteroids. ( 2.2 ) Confirm availability of tocilizumab prior to infusion. ( 2.2 , 5.2 ) Dosing of CARVYKTI is based on the number of chimeric antigen receptor (CAR)-positive viable T cells. ( 2.1 ) Recommended dose range is 0.5–1.0×10 6 CAR-positive viable T cells per kg of body weight, with a maximum dose of 1×10 8 CAR-positive viable T cells per single-dose infusion. ( 2.1 )
Warnings and Precautions
Prolonged and Recurrent Cytopenias : Patients may exhibit ≥Grade 3 cytopenias following CARVYKTI infusion. One or more recurrences of Grade 3 or higher cytopenias may occur after partial or complete recovery of cytopenias. Monitor blood counts prior to and after CARVYKTI infusion. Prolonged neutropenia has been associated with increased risk of infection. ( 5.5 ) Infections : Monitor patients for signs and symptoms of infection; treat appropriately. ( 5.6 ) Hypogammaglobulinemia : Monitor and consider immunoglobulin replacement therapy. ( 5.7 ) Hypersensitivity Reactions : Hypersensitivity reactions have occurred. Monitor for hypersensitivity reactions during infusion. ( 5.8 ) Immune Effector Cell-associated Enterocolitis: Patients may exhibit prolonged and severe diarrhea in the months following CARVYKTI infusion. Monitor for signs and symptoms of IEC-Enterocolitis after CARVYKTI infusion and manage per institutional guidelines. ( 5.9 ) Secondary Malignancies : Secondary hematological malignancies, including myelodysplastic syndrome and acute myeloid leukemia, have occurred. T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies, including CARVYKTI. In the event that a secondary malignancy occurs after treatment with CARVYKTI, contact Janssen Biotech, Inc. at 1-800-526-7736. ( 5.10 )
Contraindications
None.
Adverse Reactions
The most common nonlaboratory adverse reactions (incidence greater than 20%) are pyrexia, cytokine release syndrome, hypogammaglobulinemia, hypotension, musculoskeletal pain, fatigue, infections-pathogen unspecified, cough, chills, diarrhea, nausea, encephalopathy, decreased appetite, upper respiratory tract infection, headache, tachycardia, dizziness, dyspnea, edema, viral infections, coagulopathy, constipation, and vomiting. The most common Grade 3 or 4 laboratory adverse reactions (incidence greater than or equal to 50%) include lymphopenia, neutropenia, white blood cell decreased, thrombocytopenia, and anemia. ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact Janssen Biotech, Inc. at 1-800-526-7736 (1-800-JANSSEN) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
HIV and the lentivirus used to make CARVYKTI have limited, short spans of identical genetic material (RNA). Therefore, some commercial HIV nucleic acid tests (NATs) may yield false-positive results in patients who have received CARVYKTI.
Storage and Handling
CARVYKTI ® is supplied in one infusion bag containing a frozen suspension of genetically modified autologous T cells in 5% DMSO, either as a: 70 mL suspension in an infusion bag and metal cassette (NDC 57894-111-01) or 30 mL suspension in an infusion bag and metal cassette (NDC 57894-111-02) Each CARVYKTI infusion bag is individually packed in an aluminum cryo-cassette. Match the identity of the patient with the patient identifiers on the cassette and infusion bag upon receipt.
How Supplied
CARVYKTI ® is supplied in one infusion bag containing a frozen suspension of genetically modified autologous T cells in 5% DMSO, either as a: 70 mL suspension in an infusion bag and metal cassette (NDC 57894-111-01) or 30 mL suspension in an infusion bag and metal cassette (NDC 57894-111-02) Each CARVYKTI infusion bag is individually packed in an aluminum cryo-cassette. Match the identity of the patient with the patient identifiers on the cassette and infusion bag upon receipt.
Medication Information
Warnings and Precautions
Prolonged and Recurrent Cytopenias : Patients may exhibit ≥Grade 3 cytopenias following CARVYKTI infusion. One or more recurrences of Grade 3 or higher cytopenias may occur after partial or complete recovery of cytopenias. Monitor blood counts prior to and after CARVYKTI infusion. Prolonged neutropenia has been associated with increased risk of infection. ( 5.5 ) Infections : Monitor patients for signs and symptoms of infection; treat appropriately. ( 5.6 ) Hypogammaglobulinemia : Monitor and consider immunoglobulin replacement therapy. ( 5.7 ) Hypersensitivity Reactions : Hypersensitivity reactions have occurred. Monitor for hypersensitivity reactions during infusion. ( 5.8 ) Immune Effector Cell-associated Enterocolitis: Patients may exhibit prolonged and severe diarrhea in the months following CARVYKTI infusion. Monitor for signs and symptoms of IEC-Enterocolitis after CARVYKTI infusion and manage per institutional guidelines. ( 5.9 ) Secondary Malignancies : Secondary hematological malignancies, including myelodysplastic syndrome and acute myeloid leukemia, have occurred. T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies, including CARVYKTI. In the event that a secondary malignancy occurs after treatment with CARVYKTI, contact Janssen Biotech, Inc. at 1-800-526-7736. ( 5.10 )
Indications and Usage
CARVYKTI is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma, who have received at least 1 prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent, and are refractory to lenalidomide.
Dosage and Administration
For autologous use only. For intravenous use only. Administer a lymphodepleting regimen of cyclophosphamide and fludarabine before infusion of CARVYKTI. ( 2.2 ) Do NOT use a leukodepleting filter. ( 2.2 ) Verify the patient's identity prior to infusion. ( 2.2 ) Premedicate with acetaminophen and an H1-antihistamine. ( 2.2 ) Avoid prophylactic use of systemic corticosteroids. ( 2.2 ) Confirm availability of tocilizumab prior to infusion. ( 2.2 , 5.2 ) Dosing of CARVYKTI is based on the number of chimeric antigen receptor (CAR)-positive viable T cells. ( 2.1 ) Recommended dose range is 0.5–1.0×10 6 CAR-positive viable T cells per kg of body weight, with a maximum dose of 1×10 8 CAR-positive viable T cells per single-dose infusion. ( 2.1 )
Contraindications
None.
Adverse Reactions
The most common nonlaboratory adverse reactions (incidence greater than 20%) are pyrexia, cytokine release syndrome, hypogammaglobulinemia, hypotension, musculoskeletal pain, fatigue, infections-pathogen unspecified, cough, chills, diarrhea, nausea, encephalopathy, decreased appetite, upper respiratory tract infection, headache, tachycardia, dizziness, dyspnea, edema, viral infections, coagulopathy, constipation, and vomiting. The most common Grade 3 or 4 laboratory adverse reactions (incidence greater than or equal to 50%) include lymphopenia, neutropenia, white blood cell decreased, thrombocytopenia, and anemia. ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact Janssen Biotech, Inc. at 1-800-526-7736 (1-800-JANSSEN) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
HIV and the lentivirus used to make CARVYKTI have limited, short spans of identical genetic material (RNA). Therefore, some commercial HIV nucleic acid tests (NATs) may yield false-positive results in patients who have received CARVYKTI.
Storage and Handling
CARVYKTI ® is supplied in one infusion bag containing a frozen suspension of genetically modified autologous T cells in 5% DMSO, either as a: 70 mL suspension in an infusion bag and metal cassette (NDC 57894-111-01) or 30 mL suspension in an infusion bag and metal cassette (NDC 57894-111-02) Each CARVYKTI infusion bag is individually packed in an aluminum cryo-cassette. Match the identity of the patient with the patient identifiers on the cassette and infusion bag upon receipt.
How Supplied
CARVYKTI ® is supplied in one infusion bag containing a frozen suspension of genetically modified autologous T cells in 5% DMSO, either as a: 70 mL suspension in an infusion bag and metal cassette (NDC 57894-111-01) or 30 mL suspension in an infusion bag and metal cassette (NDC 57894-111-02) Each CARVYKTI infusion bag is individually packed in an aluminum cryo-cassette. Match the identity of the patient with the patient identifiers on the cassette and infusion bag upon receipt.
Description
Cytokine Release Syndrome (CRS), including fatal or life-threatening reactions, occurred in patients following treatment with CARVYKTI. Do not administer CARVYKTI to patients with active infection or inflammatory disorders. Treat severe or life-threatening CRS with tocilizumab or tocilizumab and corticosteroids [see Dosage and Administration (2.2 , 2.3) , Warnings and Precautions (5.2) ] . Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS), which may be fatal or life-threatening, occurred following treatment with CARVYKTI, including before CRS onset, concurrently with CRS, after CRS resolution, or in the absence of CRS. Monitor for neurologic events after treatment with CARVYKTI. Provide supportive care and/or corticosteroids as needed [see Dosage and Administration (2.2 , 2.3) , Warnings and Precautions (5.3) ] . Parkinsonism and Guillain-Barré syndrome (GBS) and their associated complications resulting in fatal or life-threatening reactions have occurred following treatment with CARVYKTI [see Warnings and Precautions (5.3) ] . Hemophagocytic Lymphohistiocytosis/Macrophage Activation Syndrome (HLH/MAS), including fatal and life-threatening reactions, occurred in patients following treatment with CARVYKTI. HLH/MAS can occur with CRS or neurologic toxicities [see Warnings and Precautions (5.4) ]. Prolonged and/or recurrent cytopenias with bleeding and infection and requirement for stem cell transplantation for hematopoietic recovery occurred following treatment with CARVYKTI [see Warnings and Precautions (5.5) ] . Immune Effector Cell-associated Enterocolitis (IEC-EC), including fatal or life-threatening reactions, occurred following treatment with CARVYKTI [see Warnings and Precautions (5.9) ]. Secondary hematological malignancies, including myelodysplastic syndrome and acute myeloid leukemia, have occurred in patients following treatment with CARVYKTI. T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies, including CARVYKTI [see Warnings and Precautions (5.10) ] .
Section 42229-5
Preparing the Patient for CARVYKTI Infusion
Confirm availability of CARVYKTI prior to starting the lymphodepleting chemotherapy regimen.
Section 42231-1
| MEDICATION GUIDE
CARVYKTI ®(car-vick-tee) (ciltacabtagene autoleucel) |
|---|
| Manufactured/Marketed by: Janssen Biotech, Inc., Horsham, PA 19044, USA. U.S. License Number 1864 |
| Marketed by: Legend Biotech, Somerset, NJ 08873, USA. For patent information: www.janssenpatents.com. |
| For more information, call 1-800-526-7736 or go to www.CARVYKTI.com. |
| © Johnson & Johnson and its affiliates 2022–2025 |
| This Medication guide has been approved by the U.S. Food and Drug Administration. |
| Read this Medication Guide before you start your CARVYKTI treatment. The more you know about your treatment, the more active you can be in your care. Talk with your healthcare provider if you have questions about your health condition or treatment. Reading this Medication Guide does not take the place of talking with your healthcare provider about your treatment. |
|
What is the most important information I should know about CARVYKTI?
CARVYKTI may cause side effects that are severe or life-threatening and can lead to death. Call your healthcare provider or get emergency help right away if you get any of the following:
|
What is CARVYKTI?
|
Before you receive CARVYKTI tell your healthcare provider about all your medical conditions, including if you have:
|
How will I receive CARVYKTI?
30 to 60 minutes before you are given CARVYKTI, you may be given other medicines. These may include:
After getting CARVYKTI,you will be monitored daily for at least 7 days after the infusion. You should plan to stay close to a healthcare facility for at least 2 weeks. Your healthcare provider will check to see that your treatment is working and help you with any side effects that may occur. You may be hospitalized if you develop serious side effects until your side effects are under control and it is safe for you to leave the hospital. Your healthcare provider will want to do blood tests to follow your progress. It is important that you have your blood tested. If you miss an appointment, call your healthcare provider as soon as possible to reschedule. |
What should I avoid after receiving CARVYKTI?
|
|
What are the possible or reasonably likely side effects of CARVYKTI?
The most common side effects of CARVYKTI include:
CARVYKTI can cause a very common side effect called cytokine release syndrome or CRS, which can be severe or fatal. Symptoms of CRS include fever, difficulty breathing, dizziness or lightheadedness, nausea, headache, fast heartbeat, low blood pressure, or fatigue. Tell your healthcare provider right away if you develop fever or any of these other symptoms after receiving CARVYKTI. CARVYKTI can increase the risk of life-threatening infections including COVID-19 that may lead to death. Tell your healthcare provider right away if you develop fever, chills, or any signs or symptoms of an infection. CARVYKTI can cause various neurologic side effects, some of which may be severe or fatal. Symptoms include but are not limited to confusion, disorientation, loss of consciousness, seizures, difficulty speaking, reading or writing, tremor, slower movements, changes in personality, depression, tingling and numbness of hands and feet, leg and arm weakness, and facial numbness. CARVYKTI can lower one or more types of your blood cells (red blood cells, white blood cells, or platelets [cells that help blood to clot]), which may make you feel weak or tired or increase your risk of severe infection or bleeding that may lead to death. After treatment, your healthcare provider will test your blood to check for this. Tell your healthcare provider right away if you get a fever, chills, or any signs or symptoms of an infection, are feeling tired, or have bruising or bleeding. CARVYKTI can cause serious gastrointestinal side effects, including severe or persistent diarrhea or ruptured bowel, which can be life-threatening and may lead to death. Tell your healthcare provider right away if you develop diarrhea, abdominal pain, weight loss, fever, chills, or any signs or symptoms of an infection. CARVYKTI may increase your risk of getting cancers including certain types of blood cancers. Your healthcare provider should monitor you for this. Having CARVYKTI in your blood may cause some commercial Human Immunodeficiency Virus (HIV) tests to incorrectly give you an HIV-positive result even though you may be HIV-negative. These are not all the possible side effects of CARVYKTI. Call your healthcare provider if you have any side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
General information about the safe and effective use of CARVYKTI
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. If you would like more information about CARVYKTI, talk with your healthcare provider. You can ask your healthcare provider for information about CARVYKTI that is written for health professionals. For more information go to www.CARVYKTI.com or call 1-800-526-7736. |
|
What are the ingredients in CARVYKTI?
Active ingredient: ciltacabtagene autoleucel Inactive ingredients:DMSO |
Revised: October 2025
Section 43683-2
| Boxed Warnings | 10/2025 |
| Dosage and Administration ( 2.2, 2.3) | 06/2025 |
| Warnings and Precautions ( 5.2, 5.3) | 06/2025 |
| Warnings and Precautions, CARVYKTI REMS (5.5) Removed | 06/2025 |
| Warnings and Precautions, Effects on Ability to Drive and Use Machines (5.11) Removed | 06/2025 |
| Warnings and Precautions ( 5.6, 5.9) | 10/2025 |
Section 44425-7
Store and transport below -120 °C, e.g., in a container for cryogenic storage in the vapor phase of liquid nitrogen.
Store CARVYKTI in the original packaging containing the cassette protecting the infusion bag.
Thaw CARVYKTI prior to infusion [see Dosage and Administration (2)] .
2.1 Dose
CARVYKTI is provided as a single dose for infusion containing a suspension of chimeric antigen receptor (CAR)-positive viable T cells in one infusion bag.
The recommended dose range is 0.5–1.0×10 6CAR-positive viable T cells per kg of body weight, with a maximum dose of 1×10 8CAR-positive viable T cells per single infusion.
15 References
- DW, Santomasso BD, Locke FL, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transplant 2019; 25: 625–638.
- National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) v 5.0; 2017.
11 Description
CARVYKTI ®(ciltacabtagene autoleucel) is a BCMA-directed genetically modified autologous T cell immunotherapy. CARVYKTI is prepared from the patient's peripheral blood mononuclear cells, which are obtained via a standard leukapheresis procedure. The mononuclear cells are enriched for T cells and genetically modified ex vivo by transduction with a replication-incompetent lentiviral vector to express a CAR comprising an anti-BCMA targeting domain, which consists of two single-domain antibodies linked to a 4-1BB costimulatory domain and a CD3-zeta signaling domain.
The transduced anti-BCMA CAR T cells are expanded in cell culture, washed, formulated into a suspension and cryopreserved. The product must pass a sterility test before release for shipping as a frozen suspension in a patient-specific infusion bag. The product is thawed and then infused back into the patient, where the anti-BCMA CAR T cells can recognize and eliminate BCMA-expressing target cells [see Dosage and Administration (2.2), How Supplied/Storage and Handling (16)].
In addition to T cells, CARVYKTI may contain Natural Killer (NK) cells. The formulation contains 5% dimethyl sulfoxide (DMSO).
5.6 Infections
CARVYKTI should not be administered to patients with active infection or inflammatory disorders. Severe, life-threatening, or fatal infections, occurred in patients after CARVYKTI infusion.
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, infections occurred in 57% (163/285), including ≥ Grade 3 in 24% (69/285) of patients. Grade 3 or 4 infections with an unspecified pathogen occurred in 12%, viral infections in 6%, bacterial infections in 5%, and fungal infections in 1% of patients. Overall, 5% (13/285) of patients had Grade 5 infections, 2.5% of which were due to COVID-19. Patients treated with CARVYKTI had an increased rate of fatal COVID-19 infections compared to the standard therapy arm [see Adverse Reactions (6.1)].
Monitor patients for signs and symptoms of infection before and after CARVYKTI infusion and treat patients appropriately. Administer prophylactic, pre-emptive and/or therapeutic antimicrobials according to the standard institutional guidelines. Febrile neutropenia was observed in 5% of patients after CARVYKTI infusion and may be concurrent with CRS. In the event of febrile neutropenia, evaluate for infection and manage with broad-spectrum antibiotics, fluids and other supportive care, as medically indicated.
Counsel patients on the importance of prevention measures. Follow institutional guidelines for the vaccination and management of immunocompromised patients with COVID-19.
8.4 Pediatric Use
Safety and effectiveness of CARVYKTI in pediatric patients have not been established.
8.5 Geriatric Use
Of the 97 patients in CARTITUDE-1 that received CARVYKTI, 28% were 65 to 75 years of age, and 8% were 75 years of age or older. CARTITUDE-1 did not include sufficient numbers of patients aged 65 and older to determine whether the effectiveness differs compared with that of younger patients. In 62 patients less than 65 years of age, all grade and Grade 3 and higher neurologic toxicities occurred in 19% (12/62) and 6% (4/62), respectively. Of the 35 patients ≥65 years of age, all grade and Grade 3 and higher neurologic toxicities occurred in 37% (13/35) and 20% (7/35), respectively.
Of the 188 patients in CARTITUDE-4 that received CARVYKTI, 38% were 65 to 75 years of age, and 2% were 75 years of age or older. In 112 patients less than 65 years of age, all grade and Grade 3 and higher neurologic toxicities occurred in 16% (18/112) and 3% (3/112) respectively. Of the 76 patients ≥65 years of age, all grade and Grade 3 and higher neurologic toxicities occurred in 34% (26/76) and 7% (5/76) respectively.
2.2 Administration
CARVYKTI is for autologous use only. For intravenous use only.The patient's identity must match the patient identifiers on the CARVYKTI cassette and infusion bag. Do not infuse CARVYKTI if the information on the patient-specific labels does not match the intended patient.
12.6 Immunogenicity
The immunogenicity of CARVYKTI has been evaluated using a validated assay for the detection of binding antibodies against the extracellular portion of the anti-BCMA CAR pre-dose, and at multiple timepoints post-infusion. In CARTITUDE-1, 19 of 97 (19.6%) patients were positive for anti-product antibodies. In CARTITUDE-4, 39 of 186 patients (21%) were positive for anti-CAR antibodies.
There was no clear evidence that the observed anti-product antibodies impact CARVYKTI kinetics of initial expansion and persistence, efficacy, or safety.
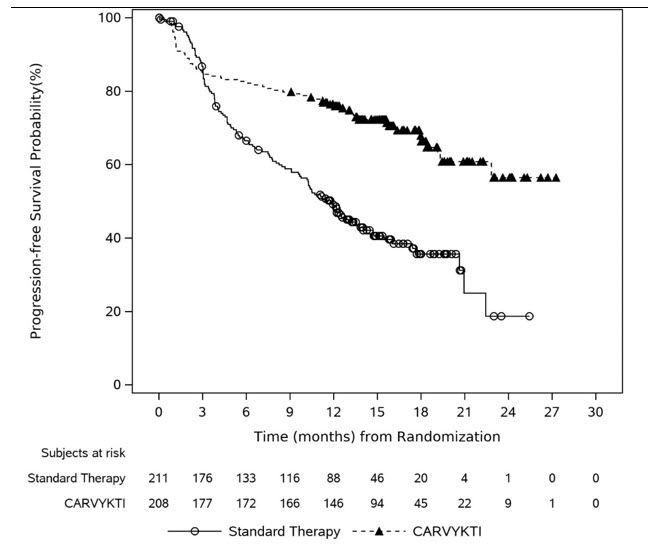
14 Clinical Studies
The efficacy of CARVYKTI was evaluated in two studies, CARTITUDE-4 (NCT04181827) and CARTITUDE-1 (NCT03548207), as described below.
4 Contraindications
None.
6 Adverse Reactions
The most common nonlaboratory adverse reactions (incidence greater than 20%) are pyrexia, cytokine release syndrome, hypogammaglobulinemia, hypotension, musculoskeletal pain, fatigue, infections-pathogen unspecified, cough, chills, diarrhea, nausea, encephalopathy, decreased appetite, upper respiratory tract infection, headache, tachycardia, dizziness, dyspnea, edema, viral infections, coagulopathy, constipation, and vomiting. The most common Grade 3 or 4 laboratory adverse reactions (incidence greater than or equal to 50%) include lymphopenia, neutropenia, white blood cell decreased, thrombocytopenia, and anemia. ( 6)
To report SUSPECTED ADVERSE REACTIONS, contact Janssen Biotech, Inc. at 1-800-526-7736 (1-800-JANSSEN) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions
HIV and the lentivirus used to make CARVYKTI have limited, short spans of identical genetic material (RNA). Therefore, some commercial HIV nucleic acid tests (NATs) may yield false-positive results in patients who have received CARVYKTI.
12.2 Pharmacodynamics
After a single infusion of CARVYKTI, expansion of CAR-positive T cells coincided with decreases of serum soluble BCMA, serum M-protein, and/or free light chains. Across all patients, levels of IL-6, IL-10, IFN-γ and IL-2 receptor alpha increased post-infusion and peaked at Days 7–14. The serum levels of all cytokines generally returned to baseline levels within 2–3 months post-infusion.
12.3 Pharmacokinetics
The pharmacokinetics (PK) of CARVYKTI was assessed in 285 adult patients with relapsed or refractory multiple myeloma in CARTITUDE-1 and CARTITUDE-4 receiving a single infusion at the median dose of 0.71×10 6CAR-positive viable T cells/kg (range: 0.41×10 6to 1.08×10 6cells/kg).
Following a single infusion, CARVYKTI exhibited an initial expansion phase followed by a rapid decline, and then a slower decline. However, high inter-individual variability was observed.
| Parameter | Summary Statistics | CARTITUDE-1
N=97 |
CARTITUDE-4
N=188 |
|---|---|---|---|
| C max(copies/µg genomic DNA) | Median (range), n | 47806 (7189 – 115234), 97 | 34891 (935 – 104861), 185 |
| t max(day) | Median (range), n | 12.7 (8.7 – 329.8), 97 | 12.8 (7.8 – 222.8), 185 |
| AUC 0–28d(copies*day/µg genomic DNA) | Median (range), n | 371569 (58691 – 2024126), 97 | 293490 (9215 – 1738455), 184 |
| t 1/2(day) | Median (range), n | 15.3 (3.0 – 95.4), 42 | 11.7 (4.1 – 179.6), 49 |
After the cell expansion, the persistence phase of CARVYKTI was observed for all patients. At the time of analysis in CARTITUDE-1 (n=65) and CARTITUDE-4 (n=87) studies, the median time for CAR transgene levels in peripheral blood to return to the pre-dose baseline level was approximately 100 days (range: 28 to 365 days) and 109 days (range: 29 to 366 days) post-infusion respectively.
Detectable CARVYKTI exposures in bone marrow indicate a distribution of CARVYKTI from systemic circulation to bone marrow. Similar to blood transgene levels, bone marrow transgene levels declined over time and exhibited high inter-individual variability.
Patients with higher CAR-T cell expansion tended to have higher rates of CRS. Some patients required tocilizumab, corticosteroids, and anakinra for the management of CRS. CARVYKTI continues to expand and persist following administration of tocilizumab, corticosteroids, and anakinra. In CARTITUDE-1, CARVYKTI median C maxand AUC 0–28din patients treated with tocilizumab (n=68) for CRS were 168% and 209% of those in patients (n=29) who did not receive tocilizumab for CRS, respectively. The median C maxand AUC 0–28dof CARVYKTI in patients who received corticosteroids (n=21) for CRS were 186% and 307% of those in patients who did not receive corticosteroids (n=76) for CRS, respectively. In addition, the median C maxand AUC 0–28dof CARVYKTI in patients who received anakinra (n=18) for CRS were 139% and 232% of those in patients who did not receive anakinra (n=79) for CRS, respectively. In CARTITUDE-4, the results related to tocilizumab and corticosteroid were consistent with CARTITUDE-1.
1 Indications and Usage
CARVYKTI is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma, who have received at least 1 prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent, and are refractory to lenalidomide.
12.1 Mechanism of Action
CARVYKTI is a BCMA-directed, genetically modified autologous T cell immunotherapy, which involves reprogramming a patient's own T cells with a transgene encoding a CAR that identifies and eliminates cells that express BCMA. The CARVYKTI CAR protein features two BCMA-targeting single-domain antibodies designed to confer high avidity against human BCMA, a 4-1BB co-stimulatory domain and a CD3-zeta (CD3ζ) signaling cytoplasmic domain. Upon binding to BCMA-expressing cells, the CAR promotes T cell activation, expansion, and elimination of target cells.
5.3 Neurologic Toxicities
Neurologic toxicities, which may be severe, life-threatening or fatal, occurred following treatment with CARVYKTI. Neurologic toxicities included ICANS, neurologic toxicity with signs and symptoms of parkinsonism, GBS, immune mediated myelitis, peripheral neuropathies and cranial nerve palsies. Counsel patients on the signs and symptoms of these neurologic toxicities, and on the delayed nature of onset of some of these toxicities. Instruct patients to seek immediate medical attention for further assessment and management if signs or symptoms of any of these neurologic toxicities occur at any time [see Patient Counseling Information (17)] .
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies for relapsed and refractory multiple myeloma, one or more neurologic toxicities occurred in 24% (69/285), including ≥ Grade 3 cases in 7% (19/285) of patients. The median time to onset was 10 days (range: 1 to 101) with 63/69 (91%) of cases developing by 30 days. Neurologic toxicities resolved in 72% (50/69) of patients with a median duration to resolution of 23 days (range: 1 to 544). Of patients developing neurotoxicity, 96% (66/69) also developed CRS. Subtypes of neurologic toxicities included ICANS in 13%, peripheral neuropathy in 7%, cranial nerve palsy in 7%, parkinsonism in 3%, and immune mediated myelitis in 0.4% of the patients [see Adverse Reactions (6.1)] .
5.7 Hypogammaglobulinemia
Hypogammaglobulinemia can occur in patients receiving treatment with CARVYKTI.
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, hypogammaglobulinemia adverse event was reported in 36% (102/285) of patients; laboratory IgG levels fell below 500 mg/dL after infusion in 93% (265/285) of patients. Hypogammaglobulinemia either as an adverse reaction or laboratory IgG level below 500 mg/dL, after infusion occurred in 94% (267/285) of patients treated. Fifty six percent (161/285) of patients received intravenous immunoglobulin (IVIG) post CARVYKTI for either an adverse reaction or prophylaxis [see Adverse Reactions (6.1)] .
Monitor immunoglobulin levels after treatment with CARVYKTI and administer IVIG for IgG <400 mg/dL. Manage per local institutional guidelines, including infection precautions and antibiotic or antiviral prophylaxis.
5 Warnings and Precautions
- Prolonged and Recurrent Cytopenias: Patients may exhibit ≥Grade 3 cytopenias following CARVYKTI infusion. One or more recurrences of Grade 3 or higher cytopenias may occur after partial or complete recovery of cytopenias. Monitor blood counts prior to and after CARVYKTI infusion. Prolonged neutropenia has been associated with increased risk of infection. ( 5.5)
- Infections: Monitor patients for signs and symptoms of infection; treat appropriately. ( 5.6)
- Hypogammaglobulinemia: Monitor and consider immunoglobulin replacement therapy. ( 5.7)
- Hypersensitivity Reactions: Hypersensitivity reactions have occurred. Monitor for hypersensitivity reactions during infusion. ( 5.8)
- Immune Effector Cell-associated Enterocolitis:Patients may exhibit prolonged and severe diarrhea in the months following CARVYKTI infusion. Monitor for signs and symptoms of IEC-Enterocolitis after CARVYKTI infusion and manage per institutional guidelines. ( 5.9)
- Secondary Malignancies: Secondary hematological malignancies, including myelodysplastic syndrome and acute myeloid leukemia, have occurred. T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies, including CARVYKTI. In the event that a secondary malignancy occurs after treatment with CARVYKTI, contact Janssen Biotech, Inc. at 1-800-526-7736. ( 5.10)
2 Dosage and Administration
For autologous use only. For intravenous use only.
- Administer a lymphodepleting regimen of cyclophosphamide and fludarabine before infusion of CARVYKTI. ( 2.2)
- Do NOT use a leukodepleting filter. ( 2.2)
- Verify the patient's identity prior to infusion. ( 2.2)
- Premedicate with acetaminophen and an H1-antihistamine. ( 2.2)
- Avoid prophylactic use of systemic corticosteroids. ( 2.2)
- Confirm availability of tocilizumab prior to infusion. ( 2.2, 5.2)
- Dosing of CARVYKTI is based on the number of chimeric antigen receptor (CAR)-positive viable T cells. ( 2.1)
- Recommended dose range is 0.5–1.0×10 6CAR-positive viable T cells per kg of body weight, with a maximum dose of 1×10 8CAR-positive viable T cells per single-dose infusion. ( 2.1)
5.10 Secondary Malignancies
Patients treated with CARVYKTI may develop secondary malignancies.
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, myeloid neoplasms occurred in 5% (13/285) of patients (9 cases of myelodysplastic syndrome, 3 cases of acute myeloid leukemia, and 1 case of myelodysplastic syndrome followed by acute myeloid leukemia). The median time to onset of myeloid neoplasms was 447 days (range: 56 to 870 days) after treatment with CARVYKTI. Ten of these 13 patients died following the development of myeloid neoplasms; 2 of the 13 cases of myeloid neoplasm occurred after initiation of subsequent antimyeloma therapy. Cases of myelodysplastic syndrome and acute myeloid leukemia have also been reported in the post marketing setting.
T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies, including CARVYKTI. Mature T-cell malignancies, including CAR-positive tumors, may present as soon as weeks following infusions, and may include fatal outcomes [see Boxed Warning, Adverse Reactions (6.1, 6.2)] .
Monitor life-long for secondary malignancies. In the event that a secondary malignancy occurs, contact Janssen Biotech, Inc. at 1-800-526-7736 for reporting and to obtain instructions on collection of patient samples.
3 Dosage Forms and Strengths
CARVYKTI is a cell suspension for intravenous infusion.
A single dose of CARVYKTI contains a cell suspension of 0.5–1.0×10 6CAR-positive viable T cells per kg body weight in one infusion bag up to a maximum of 1×10 8CAR-positive viable T cells [see How Supplied/Storage and Handling (16)] .
6.2 Postmarketing Experience
Because adverse events to marketed products are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to product exposure.
The following adverse events have been identified during postmarketing use of CARVYKTI.
Neoplasms:T cell malignancies including T-cell lymphoma of the gastrointestinal tract (including fatal cases).
Gastrointestinal disorders:immune effector cell-associated enterocolitis and gastrointestinal perforation (including fatal cases) [see Warnings and Precautions (5.9)] .
Immune System disorders:infusion related reactions.
Infections and infestations:John Cunningham (JC) virus progressive multifocal leukoencephalopathy (including fatal cases) [see Warnings and Precautions (5.6)].
5.1 Increased Early Mortality
In CARTITUDE-4, a randomized (1:1), controlled trial, there was a numerically higher percentage of early deaths in patients randomized to the CARVYKTI treatment arm compared to the control arm. Among patients with deaths occurring within the first 10 months from randomization, a greater proportion (29/208; 14%) occurred in the CARVYKTI arm compared to (25/211; 12%) in the control arm [see Clinical Studies (14)]. Of the 29 deaths that occurred in the CARVYKTI arm within the first 10 months of randomization, 10 deaths occurred prior to CARVYKTI infusion, and 19 deaths occurred after CARVYKTI infusion. Of the 10 deaths that occurred prior to CARVYKTI infusion, all occurred due to disease progression, and none occurred due to adverse events. Of the 19 deaths that occurred after CARVYKTI infusion, 3 occurred due to disease progression, and 16 occurred due to adverse events. The most common adverse events were due to infection (n=12).
5.2 Cytokine Release Syndrome
Cytokine release syndrome (CRS), including fatal or life-threatening reactions, occurred following treatment with CARVYKTI. Among patients receiving CARVYKTI for relapsed or refractory multiple myeloma in the CARTITUDE-1 and CARTITUDE-4 studies (N=285), CRS occurred in 84% (238/285), including ≥ Grade 3 CRS (ASTCT 2019) in 4% (11/285) of patients. The median time to onset of CRS, any grade, was 7 days (range: 1 to 23 days). Cytokine release syndrome resolved in 82% with a median duration of 4 days (range: 1 to 97 days). The most common manifestations of CRS in all patients combined (≥ 10%) included fever (84%), hypotension (29%) and aspartate aminotransferase increased (11%). Serious events that may be associated with CRS include pyrexia, hemophagocytic lymphohistiocytosis, respiratory failure, disseminated intravascular coagulation, capillary leak syndrome, and supraventricular and ventricular tachycardia [see Adverse Reactions (6.1)].
Cytokine release syndrome occurred in 78% of patients in CARTITUDE-4 (3% Grade 3 to 4) and in 95% of patients in CARTITUDE-1 (4% Grade 3 to 4).
Identify CRS based on clinical presentation. Evaluate for and treat other causes of fever, hypoxia, and hypotension. CRS has been reported to be associated with findings of HLH/MAS, and the physiology of the syndromes may overlap. HLH/MAS is a potentially life-threatening condition. In patients with progressive symptoms of CRS or refractory CRS despite treatment, evaluate for evidence of HLH/MAS [see Warnings and Precautions (5.4)].
Confirm that a minimum of two doses of tocilizumab are available prior to infusion of CARVYKTI.
Of the 285 patients who received CARVYKTI in clinical trials, 53% (150/285) patients received tocilizumab; 35% (100/285) received a single dose, while 18% (50/285) received more than 1 dose of tocilizumab. Overall, 14% (39/285) of patients received at least one dose of corticosteroids for treatment of CRS.
Monitor patients at least daily for 7 days following CARVYKTI infusion for signs and symptoms of CRS. Monitor patients for signs or symptoms of CRS for at least 2 weeks after infusion. At the first sign of CRS, immediately institute treatment with supportive care, tocilizumab, or tocilizumab and corticosteroids, as indicated in Table 1 [see Dosage and Administration (2.3)] .
Counsel patients to seek immediate medical attention should signs or symptoms of CRS occur at any time [see Patient Counseling Information (17)].
5.8 Hypersensitivity Reactions
Hypersensitivity reactions occurred following treatment with CARVYKTI.
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, hypersensitivity reactions occurred in 5% (13/285), all of which were ≤ Grade 2. Manifestations of hypersensitivity reactions included flushing, chest discomfort, tachycardia, wheezing, tremor, burning sensation, non-cardiac chest pain, and pyrexia.
Serious hypersensitivity reactions, including anaphylaxis, may be due to the dimethyl sulfoxide (DMSO) in CARVYKTI. Patients should be carefully monitored for 2 hours after infusion for signs and symptoms of severe reaction. Treat promptly and manage patients appropriately according to the severity of the hypersensitivity reaction.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety data described in the WARNINGS and PRECAUTIONS section reflect exposure to CARVYKTI in 285 patients with relapsed or refractory multiple myeloma: one randomized, open-label with 188 patients in CARTITUDE-4 and one single-arm, open-label study with 97 patients in CARTITUDE-1.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Inform patients of the risk of manufacturing failure [18%, (17/97 in the clinical study)]. In case of a manufacturing failure, a second manufacturing of CARVYKTI may be attempted. In addition, while the patient awaits the product, additional anticancer treatment (other than lymphodepletion) may be necessary and may increase the risk of adverse reactions during the pre-infusion period, which could delay or prevent the administration of CARVYKTI.
Advise patients that they will be monitored daily for the first 7 days following the infusion, and instruct patients to remain within proximity of a healthcare facility for at least 2 weeks following the infusion.
Prior to infusion, advise patients of the following risks and to seek immediate medical attention in the event of the following signs or symptoms:
16 How Supplied/storage and Handling
CARVYKTI ®is supplied in one infusion bag containing a frozen suspension of genetically modified autologous T cells in 5% DMSO, either as a:
- 70 mL suspension in an infusion bag and metal cassette (NDC 57894-111-01)
or
- 30 mL suspension in an infusion bag and metal cassette (NDC 57894-111-02)
Each CARVYKTI infusion bag is individually packed in an aluminum cryo-cassette.
Match the identity of the patient with the patient identifiers on the cassette and infusion bag upon receipt.
5.5 Prolonged and Recurrent Cytopenias
Patients may exhibit prolonged and recurrent cytopenias following lymphodepleting chemotherapy and CARVYKTI infusion.
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, Grade 3 or higher cytopenias not resolved by day 30 following CARVYKTI infusion occurred in 62% (176/285) of the patients and included thrombocytopenia 33% (94/285), neutropenia 27% (76/285), lymphopenia 24% (67/285) and anemia 2% (6/285). After Day 60 following CARVYKTI infusion 22%, 20%, 5%, and 6% of patients had a recurrence of Grade 3 or 4 lymphopenia, neutropenia, thrombocytopenia, and anemia respectively, after initial recovery of their Grade 3 or 4 cytopenia. Seventy-seven percent (219/285) of patients had one, two or three or more recurrences of Grade 3 or 4 cytopenias after initial recovery of Grade 3 or 4 cytopenia. Sixteen and 25 patients had Grade 3 or 4 neutropenia and thrombocytopenia, respectively, at the time of death [see Adverse Reactions (6.1)].
Monitor blood counts prior to and after CARVYKTI infusion. Manage cytopenias with growth factors and blood product transfusion support according to local institutional guidelines.
Principal Display Panel 30 Ml Bag Label
ciltacabtagene autoleucel
CARVYKTI™
Suspension for Intravenous Infusion
NDC 57894-111-02
Dose: One sterile bag for infusion.
Contents: A maximum of 1x10
8CAR-positive viable T cells in a 30 mL frozen suspension per
patient-specific infusion bag, containing 5% DMSO.
FOR AUTOLOGOUS USE ONLY.
FOR INTRAVENOUS USE ONLY.
Dosage: See Prescribing Information.
Storage: Store and transport in a vapor phase of
liquid nitrogen ≤ -120°C (-184°F). Thaw before using.
DO NOT re-freeze or refrigerate once thawed.
DO NOT irradiate.
DO NOT use a leukodepleting filter.
CULTURED, GENETICALLY MODIFIED.
NO U.S. STANDARD OF POTENCY.
NOT EVALUATED FOR INFECTIOUS SUBSTANCES.
NO PRESERVATIVE
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
One Sterile Bag for Infusion
Mfg./Mktd. by: Janssen Biotech, Inc., Horsham,
PA 19044, USA; U.S. License No. 1864
Mktd. by: Legend Biotech, Somerset, NJ 08873, USA
Upon receipt: Match the identity of
the patient with the patient
identifiers on the cassette and
infusion bag.
BAG ID: COI [followed by Bag Number]
LOT: XXXXXXXX
EXP: YYYY-MMM-DD
ORDER ID:
PATIENT
NAME:
DOB: YYYY-MMM-DD
MEDICAL RECORD NO.:
DIN:
© 2022 Janssen
10580600
LEGEND
BIOTECH
janssen
Principal Display Panel 70 Ml Bag Label
ciltacabtagene autoleucel
CARVYKTI™
Suspension for Intravenous Infusion
NDC 57894-111-01
Dose: One sterile bag for infusion.
Contents: A maximum of 1x10
8CAR-positive viable T cells in a 70 mL frozen suspension per
patient-specific infusion bag, containing 5% DMSO.
FOR AUTOLOGOUS USE ONLY.
FOR INTRAVENOUS USE ONLY.
Dosage: See Prescribing Information.
Storage: Store and transport in a vapor phase of
liquid nitrogen ≤ -120°C (-184°F). Thaw before using.
DO NOT re-freeze or refrigerate once thawed.
DO NOT irradiate.
DO NOT use a leukodepleting filter.
CULTURED, GENETICALLY MODIFIED.
NO U.S. STANDARD OF POTENCY.
NOT EVALUATED FOR INFECTIOUS SUBSTANCES.
NO PRESERVATIVE
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
One Sterile Bag for Infusion
Mfg./Mktd. by: Janssen Biotech, Inc., Horsham,
PA 19044, USA; U.S. License No. 1864
Mktd. by: Legend Biotech, Somerset, NJ 08873, USA
Upon receipt: Match the identity of
the patient with the patient
identifiers on the cassette and
infusion bag.
BAG ID: COI [followed by Bag Number]
LOT: XXXXXXXX
EXP: YYYY-MMM-DD
ORDER ID:
PATIENT
NAME:
DOB: YYYY-MMM-DD
MEDICAL RECORD NO.:
DIN:
© 2022 Janssen
10579200
LEGEND
BIOTECH
janssen
5.9 Immune Effector Cell Associated Enterocolitis
Immune effector cell-associated enterocolitis (IEC-EC) has occurred in patients treated with CARVYKTI [see Adverse Reactions (6.2)]. Manifestations include severe or prolonged diarrhea, abdominal pain and weight loss requiring parenteral nutrition. IEC-EC has been associated with fatal outcome from perforation or sepsis. Manage according to institutional guidelines including referral to gastroenterology and infectious disease specialists.
In cases of refractory IEC-EC, consider additional workup to exclude alternative etiologies, including T-cell lymphoma of the GI tract, which has been reported in the postmarketing setting [see Warnings and Precautions (5.10)and Adverse Reactions (6.2)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No genotoxicity or carcinogenicity studies have been performed with CARVYKTI as they were not indicated. In vitro studies with CARVYKTI manufactured from healthy donors and patients with multiple myeloma showed no evidence of cytokine independent growth and no preferential integration near genes associated with oncogenic transformation.
No studies have been conducted to evaluate the effects of CARVYKTI on fertility.
5.4 Hemophagocytic Lymphohistiocytosis (hlh)/macrophage Activation Syndrome (mas)
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, HLH/MAS occurred in 1% (3/285) of patients. All events of HLH/MAS had onset within 99 days of receiving CARVYKTI, with a median onset of 10 days (range: 8 to 99 days) and all occurred in the setting of ongoing or worsening CRS. The manifestations of HLH/MAS included hyperferritinemia, hypotension, hypoxia with diffuse alveolar damage, coagulopathy and hemorrhage, cytopenia and multi-organ dysfunction, including renal dysfunction and respiratory failure.
Patients who develop HLH/MAS have an increased risk of severe bleeding. Monitor hematologic parameters in patients with HLH/MAS and transfuse per institutional guidelines. Fatal cases of HLH/MAS occurred following treatment with CARVYKTI [see Adverse Reactions (6.1)].
HLH is a life-threatening condition with a high mortality rate if not recognized and treated early. Treatment of HLH/MAS should be administered per institutional standards.
Warning: Cytokine Release Syndrome, Neurologic Toxicities, Hlh/mas, Prolonged and Recurrent Cytopenia, and Secondary Hematological Malignancies
Cytokine Release Syndrome (CRS), including fatal or life-threatening reactions, occurred in patients following treatment with CARVYKTI. Do not administer CARVYKTI to patients with active infection or inflammatory disorders. Treat severe or life-threatening CRS with tocilizumab or tocilizumab and corticosteroids [see Dosage and Administration (2.2, 2.3), Warnings and Precautions (5.2)] .
Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS), which may be fatal or life-threatening, occurred following treatment with CARVYKTI, including before CRS onset, concurrently with CRS, after CRS resolution, or in the absence of CRS. Monitor for neurologic events after treatment with CARVYKTI. Provide supportive care and/or corticosteroids as needed [see Dosage and Administration (2.2, 2.3), Warnings and Precautions (5.3)] .
Parkinsonism and Guillain-Barré syndrome (GBS) and their associated complications resulting in fatal or life-threatening reactions have occurred following treatment with CARVYKTI [see Warnings and Precautions (5.3)] .
Hemophagocytic Lymphohistiocytosis/Macrophage Activation Syndrome (HLH/MAS), including fatal and life-threatening reactions, occurred in patients following treatment with CARVYKTI. HLH/MAS can occur with CRS or neurologic toxicities [see Warnings and Precautions (5.4)].
Prolonged and/or recurrent cytopenias with bleeding and infection and requirement for stem cell transplantation for hematopoietic recovery occurred following treatment with CARVYKTI [see Warnings and Precautions (5.5)] .
Immune Effector Cell-associated Enterocolitis (IEC-EC), including fatal or life-threatening reactions, occurred following treatment with CARVYKTI [see Warnings and Precautions (5.9)].
Secondary hematological malignancies, including myelodysplastic syndrome and acute myeloid leukemia, have occurred in patients following treatment with CARVYKTI. T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies, including CARVYKTI [see Warnings and Precautions (5.10)] .
Structured Label Content
Section 42229-5 (42229-5)
Preparing the Patient for CARVYKTI Infusion
Confirm availability of CARVYKTI prior to starting the lymphodepleting chemotherapy regimen.
Section 42231-1 (42231-1)
| MEDICATION GUIDE
CARVYKTI ®(car-vick-tee) (ciltacabtagene autoleucel) |
|---|
| Manufactured/Marketed by: Janssen Biotech, Inc., Horsham, PA 19044, USA. U.S. License Number 1864 |
| Marketed by: Legend Biotech, Somerset, NJ 08873, USA. For patent information: www.janssenpatents.com. |
| For more information, call 1-800-526-7736 or go to www.CARVYKTI.com. |
| © Johnson & Johnson and its affiliates 2022–2025 |
| This Medication guide has been approved by the U.S. Food and Drug Administration. |
| Read this Medication Guide before you start your CARVYKTI treatment. The more you know about your treatment, the more active you can be in your care. Talk with your healthcare provider if you have questions about your health condition or treatment. Reading this Medication Guide does not take the place of talking with your healthcare provider about your treatment. |
|
What is the most important information I should know about CARVYKTI?
CARVYKTI may cause side effects that are severe or life-threatening and can lead to death. Call your healthcare provider or get emergency help right away if you get any of the following:
|
What is CARVYKTI?
|
Before you receive CARVYKTI tell your healthcare provider about all your medical conditions, including if you have:
|
How will I receive CARVYKTI?
30 to 60 minutes before you are given CARVYKTI, you may be given other medicines. These may include:
After getting CARVYKTI,you will be monitored daily for at least 7 days after the infusion. You should plan to stay close to a healthcare facility for at least 2 weeks. Your healthcare provider will check to see that your treatment is working and help you with any side effects that may occur. You may be hospitalized if you develop serious side effects until your side effects are under control and it is safe for you to leave the hospital. Your healthcare provider will want to do blood tests to follow your progress. It is important that you have your blood tested. If you miss an appointment, call your healthcare provider as soon as possible to reschedule. |
What should I avoid after receiving CARVYKTI?
|
|
What are the possible or reasonably likely side effects of CARVYKTI?
The most common side effects of CARVYKTI include:
CARVYKTI can cause a very common side effect called cytokine release syndrome or CRS, which can be severe or fatal. Symptoms of CRS include fever, difficulty breathing, dizziness or lightheadedness, nausea, headache, fast heartbeat, low blood pressure, or fatigue. Tell your healthcare provider right away if you develop fever or any of these other symptoms after receiving CARVYKTI. CARVYKTI can increase the risk of life-threatening infections including COVID-19 that may lead to death. Tell your healthcare provider right away if you develop fever, chills, or any signs or symptoms of an infection. CARVYKTI can cause various neurologic side effects, some of which may be severe or fatal. Symptoms include but are not limited to confusion, disorientation, loss of consciousness, seizures, difficulty speaking, reading or writing, tremor, slower movements, changes in personality, depression, tingling and numbness of hands and feet, leg and arm weakness, and facial numbness. CARVYKTI can lower one or more types of your blood cells (red blood cells, white blood cells, or platelets [cells that help blood to clot]), which may make you feel weak or tired or increase your risk of severe infection or bleeding that may lead to death. After treatment, your healthcare provider will test your blood to check for this. Tell your healthcare provider right away if you get a fever, chills, or any signs or symptoms of an infection, are feeling tired, or have bruising or bleeding. CARVYKTI can cause serious gastrointestinal side effects, including severe or persistent diarrhea or ruptured bowel, which can be life-threatening and may lead to death. Tell your healthcare provider right away if you develop diarrhea, abdominal pain, weight loss, fever, chills, or any signs or symptoms of an infection. CARVYKTI may increase your risk of getting cancers including certain types of blood cancers. Your healthcare provider should monitor you for this. Having CARVYKTI in your blood may cause some commercial Human Immunodeficiency Virus (HIV) tests to incorrectly give you an HIV-positive result even though you may be HIV-negative. These are not all the possible side effects of CARVYKTI. Call your healthcare provider if you have any side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
General information about the safe and effective use of CARVYKTI
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. If you would like more information about CARVYKTI, talk with your healthcare provider. You can ask your healthcare provider for information about CARVYKTI that is written for health professionals. For more information go to www.CARVYKTI.com or call 1-800-526-7736. |
|
What are the ingredients in CARVYKTI?
Active ingredient: ciltacabtagene autoleucel Inactive ingredients:DMSO |
Revised: October 2025
Section 43683-2 (43683-2)
| Boxed Warnings | 10/2025 |
| Dosage and Administration ( 2.2, 2.3) | 06/2025 |
| Warnings and Precautions ( 5.2, 5.3) | 06/2025 |
| Warnings and Precautions, CARVYKTI REMS (5.5) Removed | 06/2025 |
| Warnings and Precautions, Effects on Ability to Drive and Use Machines (5.11) Removed | 06/2025 |
| Warnings and Precautions ( 5.6, 5.9) | 10/2025 |
Section 44425-7 (44425-7)
Store and transport below -120 °C, e.g., in a container for cryogenic storage in the vapor phase of liquid nitrogen.
Store CARVYKTI in the original packaging containing the cassette protecting the infusion bag.
Thaw CARVYKTI prior to infusion [see Dosage and Administration (2)] .
2.1 Dose
CARVYKTI is provided as a single dose for infusion containing a suspension of chimeric antigen receptor (CAR)-positive viable T cells in one infusion bag.
The recommended dose range is 0.5–1.0×10 6CAR-positive viable T cells per kg of body weight, with a maximum dose of 1×10 8CAR-positive viable T cells per single infusion.
15 References (15 REFERENCES)
- DW, Santomasso BD, Locke FL, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transplant 2019; 25: 625–638.
- National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) v 5.0; 2017.
11 Description (11 DESCRIPTION)
CARVYKTI ®(ciltacabtagene autoleucel) is a BCMA-directed genetically modified autologous T cell immunotherapy. CARVYKTI is prepared from the patient's peripheral blood mononuclear cells, which are obtained via a standard leukapheresis procedure. The mononuclear cells are enriched for T cells and genetically modified ex vivo by transduction with a replication-incompetent lentiviral vector to express a CAR comprising an anti-BCMA targeting domain, which consists of two single-domain antibodies linked to a 4-1BB costimulatory domain and a CD3-zeta signaling domain.
The transduced anti-BCMA CAR T cells are expanded in cell culture, washed, formulated into a suspension and cryopreserved. The product must pass a sterility test before release for shipping as a frozen suspension in a patient-specific infusion bag. The product is thawed and then infused back into the patient, where the anti-BCMA CAR T cells can recognize and eliminate BCMA-expressing target cells [see Dosage and Administration (2.2), How Supplied/Storage and Handling (16)].
In addition to T cells, CARVYKTI may contain Natural Killer (NK) cells. The formulation contains 5% dimethyl sulfoxide (DMSO).
5.6 Infections
CARVYKTI should not be administered to patients with active infection or inflammatory disorders. Severe, life-threatening, or fatal infections, occurred in patients after CARVYKTI infusion.
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, infections occurred in 57% (163/285), including ≥ Grade 3 in 24% (69/285) of patients. Grade 3 or 4 infections with an unspecified pathogen occurred in 12%, viral infections in 6%, bacterial infections in 5%, and fungal infections in 1% of patients. Overall, 5% (13/285) of patients had Grade 5 infections, 2.5% of which were due to COVID-19. Patients treated with CARVYKTI had an increased rate of fatal COVID-19 infections compared to the standard therapy arm [see Adverse Reactions (6.1)].
Monitor patients for signs and symptoms of infection before and after CARVYKTI infusion and treat patients appropriately. Administer prophylactic, pre-emptive and/or therapeutic antimicrobials according to the standard institutional guidelines. Febrile neutropenia was observed in 5% of patients after CARVYKTI infusion and may be concurrent with CRS. In the event of febrile neutropenia, evaluate for infection and manage with broad-spectrum antibiotics, fluids and other supportive care, as medically indicated.
Counsel patients on the importance of prevention measures. Follow institutional guidelines for the vaccination and management of immunocompromised patients with COVID-19.
8.4 Pediatric Use
Safety and effectiveness of CARVYKTI in pediatric patients have not been established.
8.5 Geriatric Use
Of the 97 patients in CARTITUDE-1 that received CARVYKTI, 28% were 65 to 75 years of age, and 8% were 75 years of age or older. CARTITUDE-1 did not include sufficient numbers of patients aged 65 and older to determine whether the effectiveness differs compared with that of younger patients. In 62 patients less than 65 years of age, all grade and Grade 3 and higher neurologic toxicities occurred in 19% (12/62) and 6% (4/62), respectively. Of the 35 patients ≥65 years of age, all grade and Grade 3 and higher neurologic toxicities occurred in 37% (13/35) and 20% (7/35), respectively.
Of the 188 patients in CARTITUDE-4 that received CARVYKTI, 38% were 65 to 75 years of age, and 2% were 75 years of age or older. In 112 patients less than 65 years of age, all grade and Grade 3 and higher neurologic toxicities occurred in 16% (18/112) and 3% (3/112) respectively. Of the 76 patients ≥65 years of age, all grade and Grade 3 and higher neurologic toxicities occurred in 34% (26/76) and 7% (5/76) respectively.
2.2 Administration
CARVYKTI is for autologous use only. For intravenous use only.The patient's identity must match the patient identifiers on the CARVYKTI cassette and infusion bag. Do not infuse CARVYKTI if the information on the patient-specific labels does not match the intended patient.
12.6 Immunogenicity
The immunogenicity of CARVYKTI has been evaluated using a validated assay for the detection of binding antibodies against the extracellular portion of the anti-BCMA CAR pre-dose, and at multiple timepoints post-infusion. In CARTITUDE-1, 19 of 97 (19.6%) patients were positive for anti-product antibodies. In CARTITUDE-4, 39 of 186 patients (21%) were positive for anti-CAR antibodies.
There was no clear evidence that the observed anti-product antibodies impact CARVYKTI kinetics of initial expansion and persistence, efficacy, or safety.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of CARVYKTI was evaluated in two studies, CARTITUDE-4 (NCT04181827) and CARTITUDE-1 (NCT03548207), as described below.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common nonlaboratory adverse reactions (incidence greater than 20%) are pyrexia, cytokine release syndrome, hypogammaglobulinemia, hypotension, musculoskeletal pain, fatigue, infections-pathogen unspecified, cough, chills, diarrhea, nausea, encephalopathy, decreased appetite, upper respiratory tract infection, headache, tachycardia, dizziness, dyspnea, edema, viral infections, coagulopathy, constipation, and vomiting. The most common Grade 3 or 4 laboratory adverse reactions (incidence greater than or equal to 50%) include lymphopenia, neutropenia, white blood cell decreased, thrombocytopenia, and anemia. ( 6)
To report SUSPECTED ADVERSE REACTIONS, contact Janssen Biotech, Inc. at 1-800-526-7736 (1-800-JANSSEN) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions (7 DRUG INTERACTIONS)
HIV and the lentivirus used to make CARVYKTI have limited, short spans of identical genetic material (RNA). Therefore, some commercial HIV nucleic acid tests (NATs) may yield false-positive results in patients who have received CARVYKTI.
12.2 Pharmacodynamics
After a single infusion of CARVYKTI, expansion of CAR-positive T cells coincided with decreases of serum soluble BCMA, serum M-protein, and/or free light chains. Across all patients, levels of IL-6, IL-10, IFN-γ and IL-2 receptor alpha increased post-infusion and peaked at Days 7–14. The serum levels of all cytokines generally returned to baseline levels within 2–3 months post-infusion.
12.3 Pharmacokinetics
The pharmacokinetics (PK) of CARVYKTI was assessed in 285 adult patients with relapsed or refractory multiple myeloma in CARTITUDE-1 and CARTITUDE-4 receiving a single infusion at the median dose of 0.71×10 6CAR-positive viable T cells/kg (range: 0.41×10 6to 1.08×10 6cells/kg).
Following a single infusion, CARVYKTI exhibited an initial expansion phase followed by a rapid decline, and then a slower decline. However, high inter-individual variability was observed.
| Parameter | Summary Statistics | CARTITUDE-1
N=97 |
CARTITUDE-4
N=188 |
|---|---|---|---|
| C max(copies/µg genomic DNA) | Median (range), n | 47806 (7189 – 115234), 97 | 34891 (935 – 104861), 185 |
| t max(day) | Median (range), n | 12.7 (8.7 – 329.8), 97 | 12.8 (7.8 – 222.8), 185 |
| AUC 0–28d(copies*day/µg genomic DNA) | Median (range), n | 371569 (58691 – 2024126), 97 | 293490 (9215 – 1738455), 184 |
| t 1/2(day) | Median (range), n | 15.3 (3.0 – 95.4), 42 | 11.7 (4.1 – 179.6), 49 |
After the cell expansion, the persistence phase of CARVYKTI was observed for all patients. At the time of analysis in CARTITUDE-1 (n=65) and CARTITUDE-4 (n=87) studies, the median time for CAR transgene levels in peripheral blood to return to the pre-dose baseline level was approximately 100 days (range: 28 to 365 days) and 109 days (range: 29 to 366 days) post-infusion respectively.
Detectable CARVYKTI exposures in bone marrow indicate a distribution of CARVYKTI from systemic circulation to bone marrow. Similar to blood transgene levels, bone marrow transgene levels declined over time and exhibited high inter-individual variability.
Patients with higher CAR-T cell expansion tended to have higher rates of CRS. Some patients required tocilizumab, corticosteroids, and anakinra for the management of CRS. CARVYKTI continues to expand and persist following administration of tocilizumab, corticosteroids, and anakinra. In CARTITUDE-1, CARVYKTI median C maxand AUC 0–28din patients treated with tocilizumab (n=68) for CRS were 168% and 209% of those in patients (n=29) who did not receive tocilizumab for CRS, respectively. The median C maxand AUC 0–28dof CARVYKTI in patients who received corticosteroids (n=21) for CRS were 186% and 307% of those in patients who did not receive corticosteroids (n=76) for CRS, respectively. In addition, the median C maxand AUC 0–28dof CARVYKTI in patients who received anakinra (n=18) for CRS were 139% and 232% of those in patients who did not receive anakinra (n=79) for CRS, respectively. In CARTITUDE-4, the results related to tocilizumab and corticosteroid were consistent with CARTITUDE-1.
1 Indications and Usage (1 INDICATIONS AND USAGE)
CARVYKTI is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma, who have received at least 1 prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent, and are refractory to lenalidomide.
12.1 Mechanism of Action
CARVYKTI is a BCMA-directed, genetically modified autologous T cell immunotherapy, which involves reprogramming a patient's own T cells with a transgene encoding a CAR that identifies and eliminates cells that express BCMA. The CARVYKTI CAR protein features two BCMA-targeting single-domain antibodies designed to confer high avidity against human BCMA, a 4-1BB co-stimulatory domain and a CD3-zeta (CD3ζ) signaling cytoplasmic domain. Upon binding to BCMA-expressing cells, the CAR promotes T cell activation, expansion, and elimination of target cells.
5.3 Neurologic Toxicities
Neurologic toxicities, which may be severe, life-threatening or fatal, occurred following treatment with CARVYKTI. Neurologic toxicities included ICANS, neurologic toxicity with signs and symptoms of parkinsonism, GBS, immune mediated myelitis, peripheral neuropathies and cranial nerve palsies. Counsel patients on the signs and symptoms of these neurologic toxicities, and on the delayed nature of onset of some of these toxicities. Instruct patients to seek immediate medical attention for further assessment and management if signs or symptoms of any of these neurologic toxicities occur at any time [see Patient Counseling Information (17)] .
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies for relapsed and refractory multiple myeloma, one or more neurologic toxicities occurred in 24% (69/285), including ≥ Grade 3 cases in 7% (19/285) of patients. The median time to onset was 10 days (range: 1 to 101) with 63/69 (91%) of cases developing by 30 days. Neurologic toxicities resolved in 72% (50/69) of patients with a median duration to resolution of 23 days (range: 1 to 544). Of patients developing neurotoxicity, 96% (66/69) also developed CRS. Subtypes of neurologic toxicities included ICANS in 13%, peripheral neuropathy in 7%, cranial nerve palsy in 7%, parkinsonism in 3%, and immune mediated myelitis in 0.4% of the patients [see Adverse Reactions (6.1)] .
5.7 Hypogammaglobulinemia
Hypogammaglobulinemia can occur in patients receiving treatment with CARVYKTI.
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, hypogammaglobulinemia adverse event was reported in 36% (102/285) of patients; laboratory IgG levels fell below 500 mg/dL after infusion in 93% (265/285) of patients. Hypogammaglobulinemia either as an adverse reaction or laboratory IgG level below 500 mg/dL, after infusion occurred in 94% (267/285) of patients treated. Fifty six percent (161/285) of patients received intravenous immunoglobulin (IVIG) post CARVYKTI for either an adverse reaction or prophylaxis [see Adverse Reactions (6.1)] .
Monitor immunoglobulin levels after treatment with CARVYKTI and administer IVIG for IgG <400 mg/dL. Manage per local institutional guidelines, including infection precautions and antibiotic or antiviral prophylaxis.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Prolonged and Recurrent Cytopenias: Patients may exhibit ≥Grade 3 cytopenias following CARVYKTI infusion. One or more recurrences of Grade 3 or higher cytopenias may occur after partial or complete recovery of cytopenias. Monitor blood counts prior to and after CARVYKTI infusion. Prolonged neutropenia has been associated with increased risk of infection. ( 5.5)
- Infections: Monitor patients for signs and symptoms of infection; treat appropriately. ( 5.6)
- Hypogammaglobulinemia: Monitor and consider immunoglobulin replacement therapy. ( 5.7)
- Hypersensitivity Reactions: Hypersensitivity reactions have occurred. Monitor for hypersensitivity reactions during infusion. ( 5.8)
- Immune Effector Cell-associated Enterocolitis:Patients may exhibit prolonged and severe diarrhea in the months following CARVYKTI infusion. Monitor for signs and symptoms of IEC-Enterocolitis after CARVYKTI infusion and manage per institutional guidelines. ( 5.9)
- Secondary Malignancies: Secondary hematological malignancies, including myelodysplastic syndrome and acute myeloid leukemia, have occurred. T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies, including CARVYKTI. In the event that a secondary malignancy occurs after treatment with CARVYKTI, contact Janssen Biotech, Inc. at 1-800-526-7736. ( 5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
For autologous use only. For intravenous use only.
- Administer a lymphodepleting regimen of cyclophosphamide and fludarabine before infusion of CARVYKTI. ( 2.2)
- Do NOT use a leukodepleting filter. ( 2.2)
- Verify the patient's identity prior to infusion. ( 2.2)
- Premedicate with acetaminophen and an H1-antihistamine. ( 2.2)
- Avoid prophylactic use of systemic corticosteroids. ( 2.2)
- Confirm availability of tocilizumab prior to infusion. ( 2.2, 5.2)
- Dosing of CARVYKTI is based on the number of chimeric antigen receptor (CAR)-positive viable T cells. ( 2.1)
- Recommended dose range is 0.5–1.0×10 6CAR-positive viable T cells per kg of body weight, with a maximum dose of 1×10 8CAR-positive viable T cells per single-dose infusion. ( 2.1)
5.10 Secondary Malignancies
Patients treated with CARVYKTI may develop secondary malignancies.
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, myeloid neoplasms occurred in 5% (13/285) of patients (9 cases of myelodysplastic syndrome, 3 cases of acute myeloid leukemia, and 1 case of myelodysplastic syndrome followed by acute myeloid leukemia). The median time to onset of myeloid neoplasms was 447 days (range: 56 to 870 days) after treatment with CARVYKTI. Ten of these 13 patients died following the development of myeloid neoplasms; 2 of the 13 cases of myeloid neoplasm occurred after initiation of subsequent antimyeloma therapy. Cases of myelodysplastic syndrome and acute myeloid leukemia have also been reported in the post marketing setting.
T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies, including CARVYKTI. Mature T-cell malignancies, including CAR-positive tumors, may present as soon as weeks following infusions, and may include fatal outcomes [see Boxed Warning, Adverse Reactions (6.1, 6.2)] .
Monitor life-long for secondary malignancies. In the event that a secondary malignancy occurs, contact Janssen Biotech, Inc. at 1-800-526-7736 for reporting and to obtain instructions on collection of patient samples.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
CARVYKTI is a cell suspension for intravenous infusion.
A single dose of CARVYKTI contains a cell suspension of 0.5–1.0×10 6CAR-positive viable T cells per kg body weight in one infusion bag up to a maximum of 1×10 8CAR-positive viable T cells [see How Supplied/Storage and Handling (16)] .
6.2 Postmarketing Experience
Because adverse events to marketed products are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to product exposure.
The following adverse events have been identified during postmarketing use of CARVYKTI.
Neoplasms:T cell malignancies including T-cell lymphoma of the gastrointestinal tract (including fatal cases).
Gastrointestinal disorders:immune effector cell-associated enterocolitis and gastrointestinal perforation (including fatal cases) [see Warnings and Precautions (5.9)] .
Immune System disorders:infusion related reactions.
Infections and infestations:John Cunningham (JC) virus progressive multifocal leukoencephalopathy (including fatal cases) [see Warnings and Precautions (5.6)].
5.1 Increased Early Mortality
In CARTITUDE-4, a randomized (1:1), controlled trial, there was a numerically higher percentage of early deaths in patients randomized to the CARVYKTI treatment arm compared to the control arm. Among patients with deaths occurring within the first 10 months from randomization, a greater proportion (29/208; 14%) occurred in the CARVYKTI arm compared to (25/211; 12%) in the control arm [see Clinical Studies (14)]. Of the 29 deaths that occurred in the CARVYKTI arm within the first 10 months of randomization, 10 deaths occurred prior to CARVYKTI infusion, and 19 deaths occurred after CARVYKTI infusion. Of the 10 deaths that occurred prior to CARVYKTI infusion, all occurred due to disease progression, and none occurred due to adverse events. Of the 19 deaths that occurred after CARVYKTI infusion, 3 occurred due to disease progression, and 16 occurred due to adverse events. The most common adverse events were due to infection (n=12).
5.2 Cytokine Release Syndrome
Cytokine release syndrome (CRS), including fatal or life-threatening reactions, occurred following treatment with CARVYKTI. Among patients receiving CARVYKTI for relapsed or refractory multiple myeloma in the CARTITUDE-1 and CARTITUDE-4 studies (N=285), CRS occurred in 84% (238/285), including ≥ Grade 3 CRS (ASTCT 2019) in 4% (11/285) of patients. The median time to onset of CRS, any grade, was 7 days (range: 1 to 23 days). Cytokine release syndrome resolved in 82% with a median duration of 4 days (range: 1 to 97 days). The most common manifestations of CRS in all patients combined (≥ 10%) included fever (84%), hypotension (29%) and aspartate aminotransferase increased (11%). Serious events that may be associated with CRS include pyrexia, hemophagocytic lymphohistiocytosis, respiratory failure, disseminated intravascular coagulation, capillary leak syndrome, and supraventricular and ventricular tachycardia [see Adverse Reactions (6.1)].
Cytokine release syndrome occurred in 78% of patients in CARTITUDE-4 (3% Grade 3 to 4) and in 95% of patients in CARTITUDE-1 (4% Grade 3 to 4).
Identify CRS based on clinical presentation. Evaluate for and treat other causes of fever, hypoxia, and hypotension. CRS has been reported to be associated with findings of HLH/MAS, and the physiology of the syndromes may overlap. HLH/MAS is a potentially life-threatening condition. In patients with progressive symptoms of CRS or refractory CRS despite treatment, evaluate for evidence of HLH/MAS [see Warnings and Precautions (5.4)].
Confirm that a minimum of two doses of tocilizumab are available prior to infusion of CARVYKTI.
Of the 285 patients who received CARVYKTI in clinical trials, 53% (150/285) patients received tocilizumab; 35% (100/285) received a single dose, while 18% (50/285) received more than 1 dose of tocilizumab. Overall, 14% (39/285) of patients received at least one dose of corticosteroids for treatment of CRS.
Monitor patients at least daily for 7 days following CARVYKTI infusion for signs and symptoms of CRS. Monitor patients for signs or symptoms of CRS for at least 2 weeks after infusion. At the first sign of CRS, immediately institute treatment with supportive care, tocilizumab, or tocilizumab and corticosteroids, as indicated in Table 1 [see Dosage and Administration (2.3)] .
Counsel patients to seek immediate medical attention should signs or symptoms of CRS occur at any time [see Patient Counseling Information (17)].
5.8 Hypersensitivity Reactions
Hypersensitivity reactions occurred following treatment with CARVYKTI.
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, hypersensitivity reactions occurred in 5% (13/285), all of which were ≤ Grade 2. Manifestations of hypersensitivity reactions included flushing, chest discomfort, tachycardia, wheezing, tremor, burning sensation, non-cardiac chest pain, and pyrexia.
Serious hypersensitivity reactions, including anaphylaxis, may be due to the dimethyl sulfoxide (DMSO) in CARVYKTI. Patients should be carefully monitored for 2 hours after infusion for signs and symptoms of severe reaction. Treat promptly and manage patients appropriately according to the severity of the hypersensitivity reaction.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety data described in the WARNINGS and PRECAUTIONS section reflect exposure to CARVYKTI in 285 patients with relapsed or refractory multiple myeloma: one randomized, open-label with 188 patients in CARTITUDE-4 and one single-arm, open-label study with 97 patients in CARTITUDE-1.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Inform patients of the risk of manufacturing failure [18%, (17/97 in the clinical study)]. In case of a manufacturing failure, a second manufacturing of CARVYKTI may be attempted. In addition, while the patient awaits the product, additional anticancer treatment (other than lymphodepletion) may be necessary and may increase the risk of adverse reactions during the pre-infusion period, which could delay or prevent the administration of CARVYKTI.
Advise patients that they will be monitored daily for the first 7 days following the infusion, and instruct patients to remain within proximity of a healthcare facility for at least 2 weeks following the infusion.
Prior to infusion, advise patients of the following risks and to seek immediate medical attention in the event of the following signs or symptoms:
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
CARVYKTI ®is supplied in one infusion bag containing a frozen suspension of genetically modified autologous T cells in 5% DMSO, either as a:
- 70 mL suspension in an infusion bag and metal cassette (NDC 57894-111-01)
or
- 30 mL suspension in an infusion bag and metal cassette (NDC 57894-111-02)
Each CARVYKTI infusion bag is individually packed in an aluminum cryo-cassette.
Match the identity of the patient with the patient identifiers on the cassette and infusion bag upon receipt.
5.5 Prolonged and Recurrent Cytopenias
Patients may exhibit prolonged and recurrent cytopenias following lymphodepleting chemotherapy and CARVYKTI infusion.
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, Grade 3 or higher cytopenias not resolved by day 30 following CARVYKTI infusion occurred in 62% (176/285) of the patients and included thrombocytopenia 33% (94/285), neutropenia 27% (76/285), lymphopenia 24% (67/285) and anemia 2% (6/285). After Day 60 following CARVYKTI infusion 22%, 20%, 5%, and 6% of patients had a recurrence of Grade 3 or 4 lymphopenia, neutropenia, thrombocytopenia, and anemia respectively, after initial recovery of their Grade 3 or 4 cytopenia. Seventy-seven percent (219/285) of patients had one, two or three or more recurrences of Grade 3 or 4 cytopenias after initial recovery of Grade 3 or 4 cytopenia. Sixteen and 25 patients had Grade 3 or 4 neutropenia and thrombocytopenia, respectively, at the time of death [see Adverse Reactions (6.1)].
Monitor blood counts prior to and after CARVYKTI infusion. Manage cytopenias with growth factors and blood product transfusion support according to local institutional guidelines.
Principal Display Panel 30 Ml Bag Label (PRINCIPAL DISPLAY PANEL - 30 mL Bag Label)
ciltacabtagene autoleucel
CARVYKTI™
Suspension for Intravenous Infusion
NDC 57894-111-02
Dose: One sterile bag for infusion.
Contents: A maximum of 1x10
8CAR-positive viable T cells in a 30 mL frozen suspension per
patient-specific infusion bag, containing 5% DMSO.
FOR AUTOLOGOUS USE ONLY.
FOR INTRAVENOUS USE ONLY.
Dosage: See Prescribing Information.
Storage: Store and transport in a vapor phase of
liquid nitrogen ≤ -120°C (-184°F). Thaw before using.
DO NOT re-freeze or refrigerate once thawed.
DO NOT irradiate.
DO NOT use a leukodepleting filter.
CULTURED, GENETICALLY MODIFIED.
NO U.S. STANDARD OF POTENCY.
NOT EVALUATED FOR INFECTIOUS SUBSTANCES.
NO PRESERVATIVE
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
One Sterile Bag for Infusion
Mfg./Mktd. by: Janssen Biotech, Inc., Horsham,
PA 19044, USA; U.S. License No. 1864
Mktd. by: Legend Biotech, Somerset, NJ 08873, USA
Upon receipt: Match the identity of
the patient with the patient
identifiers on the cassette and
infusion bag.
BAG ID: COI [followed by Bag Number]
LOT: XXXXXXXX
EXP: YYYY-MMM-DD
ORDER ID:
PATIENT
NAME:
DOB: YYYY-MMM-DD
MEDICAL RECORD NO.:
DIN:
© 2022 Janssen
10580600
LEGEND
BIOTECH
janssen
Principal Display Panel 70 Ml Bag Label (PRINCIPAL DISPLAY PANEL - 70 mL Bag Label)
ciltacabtagene autoleucel
CARVYKTI™
Suspension for Intravenous Infusion
NDC 57894-111-01
Dose: One sterile bag for infusion.
Contents: A maximum of 1x10
8CAR-positive viable T cells in a 70 mL frozen suspension per
patient-specific infusion bag, containing 5% DMSO.
FOR AUTOLOGOUS USE ONLY.
FOR INTRAVENOUS USE ONLY.
Dosage: See Prescribing Information.
Storage: Store and transport in a vapor phase of
liquid nitrogen ≤ -120°C (-184°F). Thaw before using.
DO NOT re-freeze or refrigerate once thawed.
DO NOT irradiate.
DO NOT use a leukodepleting filter.
CULTURED, GENETICALLY MODIFIED.
NO U.S. STANDARD OF POTENCY.
NOT EVALUATED FOR INFECTIOUS SUBSTANCES.
NO PRESERVATIVE
Attention: Dispense the enclosed Medication
Guide to each patient.
Rx only
One Sterile Bag for Infusion
Mfg./Mktd. by: Janssen Biotech, Inc., Horsham,
PA 19044, USA; U.S. License No. 1864
Mktd. by: Legend Biotech, Somerset, NJ 08873, USA
Upon receipt: Match the identity of
the patient with the patient
identifiers on the cassette and
infusion bag.
BAG ID: COI [followed by Bag Number]
LOT: XXXXXXXX
EXP: YYYY-MMM-DD
ORDER ID:
PATIENT
NAME:
DOB: YYYY-MMM-DD
MEDICAL RECORD NO.:
DIN:
© 2022 Janssen
10579200
LEGEND
BIOTECH
janssen
5.9 Immune Effector Cell Associated Enterocolitis (5.9 Immune Effector Cell-associated Enterocolitis)
Immune effector cell-associated enterocolitis (IEC-EC) has occurred in patients treated with CARVYKTI [see Adverse Reactions (6.2)]. Manifestations include severe or prolonged diarrhea, abdominal pain and weight loss requiring parenteral nutrition. IEC-EC has been associated with fatal outcome from perforation or sepsis. Manage according to institutional guidelines including referral to gastroenterology and infectious disease specialists.
In cases of refractory IEC-EC, consider additional workup to exclude alternative etiologies, including T-cell lymphoma of the GI tract, which has been reported in the postmarketing setting [see Warnings and Precautions (5.10)and Adverse Reactions (6.2)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No genotoxicity or carcinogenicity studies have been performed with CARVYKTI as they were not indicated. In vitro studies with CARVYKTI manufactured from healthy donors and patients with multiple myeloma showed no evidence of cytokine independent growth and no preferential integration near genes associated with oncogenic transformation.
No studies have been conducted to evaluate the effects of CARVYKTI on fertility.
5.4 Hemophagocytic Lymphohistiocytosis (hlh)/macrophage Activation Syndrome (mas) (5.4 Hemophagocytic Lymphohistiocytosis (HLH)/Macrophage Activation Syndrome (MAS))
Among patients receiving CARVYKTI in the CARTITUDE-1 and CARTITUDE-4 studies, HLH/MAS occurred in 1% (3/285) of patients. All events of HLH/MAS had onset within 99 days of receiving CARVYKTI, with a median onset of 10 days (range: 8 to 99 days) and all occurred in the setting of ongoing or worsening CRS. The manifestations of HLH/MAS included hyperferritinemia, hypotension, hypoxia with diffuse alveolar damage, coagulopathy and hemorrhage, cytopenia and multi-organ dysfunction, including renal dysfunction and respiratory failure.
Patients who develop HLH/MAS have an increased risk of severe bleeding. Monitor hematologic parameters in patients with HLH/MAS and transfuse per institutional guidelines. Fatal cases of HLH/MAS occurred following treatment with CARVYKTI [see Adverse Reactions (6.1)].
HLH is a life-threatening condition with a high mortality rate if not recognized and treated early. Treatment of HLH/MAS should be administered per institutional standards.
Warning: Cytokine Release Syndrome, Neurologic Toxicities, Hlh/mas, Prolonged and Recurrent Cytopenia, and Secondary Hematological Malignancies (WARNING: CYTOKINE RELEASE SYNDROME, NEUROLOGIC TOXICITIES, HLH/MAS, PROLONGED and RECURRENT CYTOPENIA, and SECONDARY HEMATOLOGICAL MALIGNANCIES)
Cytokine Release Syndrome (CRS), including fatal or life-threatening reactions, occurred in patients following treatment with CARVYKTI. Do not administer CARVYKTI to patients with active infection or inflammatory disorders. Treat severe or life-threatening CRS with tocilizumab or tocilizumab and corticosteroids [see Dosage and Administration (2.2, 2.3), Warnings and Precautions (5.2)] .
Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS), which may be fatal or life-threatening, occurred following treatment with CARVYKTI, including before CRS onset, concurrently with CRS, after CRS resolution, or in the absence of CRS. Monitor for neurologic events after treatment with CARVYKTI. Provide supportive care and/or corticosteroids as needed [see Dosage and Administration (2.2, 2.3), Warnings and Precautions (5.3)] .
Parkinsonism and Guillain-Barré syndrome (GBS) and their associated complications resulting in fatal or life-threatening reactions have occurred following treatment with CARVYKTI [see Warnings and Precautions (5.3)] .
Hemophagocytic Lymphohistiocytosis/Macrophage Activation Syndrome (HLH/MAS), including fatal and life-threatening reactions, occurred in patients following treatment with CARVYKTI. HLH/MAS can occur with CRS or neurologic toxicities [see Warnings and Precautions (5.4)].
Prolonged and/or recurrent cytopenias with bleeding and infection and requirement for stem cell transplantation for hematopoietic recovery occurred following treatment with CARVYKTI [see Warnings and Precautions (5.5)] .
Immune Effector Cell-associated Enterocolitis (IEC-EC), including fatal or life-threatening reactions, occurred following treatment with CARVYKTI [see Warnings and Precautions (5.9)].
Secondary hematological malignancies, including myelodysplastic syndrome and acute myeloid leukemia, have occurred in patients following treatment with CARVYKTI. T-cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T-cell immunotherapies, including CARVYKTI [see Warnings and Precautions (5.10)] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:13:06.033196 · Updated: 2026-03-14T23:19:41.168315