These Highlights Do Not Include All The Information Needed To Use harliku Safely And Effectively. See Full Prescribing Information For Harliku.
7cf67e24-d46b-4376-bbd0-0b973c406d0d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
HARLIKU TM is indicated for the reduction of urine homogentisic acid (HGA) in adult patients with alkaptonuria (AKU).
Indications and Usage
HARLIKU TM is indicated for the reduction of urine homogentisic acid (HGA) in adult patients with alkaptonuria (AKU).
Dosage and Administration
The recommended dosage of HARLIKU is 2 mg administered orally, once daily. ( 2.1 )
Warnings and Precautions
Ocular Symptoms and Hyperkeratotic Plaques Due To Elevated Plasma Tyrosine Levels : Inadequate restriction of tyrosine and phenylalanine intake can lead to elevations in plasma tyrosine and levels above 500 micromol/L may lead to ocular signs and symptoms or painful hyperkeratotic plaques on the soles and palms. ( 5.1 ) Assess plasma tyrosine levels in patients presenting with ocular signs and symptoms. ( 5.1 ) Obtain slit-lamp examination prior to treatment and regularly thereafter. ( 5.1 ) Implement diet restriction and/or treatment interruption as appropriate. ( 5.1 ) Leukopenia and Severe Thrombocytopenia: Monitor platelet and white blood cell counts. ( 5.2 )
Contraindications
None.
Adverse Reactions
Most common adverse reactions (>1%) are elevated tyrosine levels, keratitis and thrombocytopenia. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Cycle Pharmaceuticals Ltd at 1-855-831-5413 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Sensitive CYP2C9 Substrates: Reduce dosage of co-administered drug metabolized by CYP2C9 by half. ( 7.1 ) OAT1/OAT3 Substrates : Avoid concomitant use of HARLIKU with OAT1/OAT3 substrates. Concomitant use with OAT1/OAT3 substrates may increase the risk of adverse reactions related to the co-administered drug. ( 7.1 )
Storage and Handling
How Supplied HARLIKU (nitisinone) tablet is white to beige, round, flat which may display light yellow to brown speckles, debossed with the “strength” in mg on one side and “L” on the other side. Each tablet contains 2 mg nitisinone. HARLIKU is supplied in a high-density polyethylene (HDPE) square bottle with a child-resistant tamper-evident polypropylene (PP) screw cap. Each bottle contains 60 tablets. 2 mg tablets: NDC 70709-112-60 Storage and Handling Store HARLIKU tablets at room temperature between 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. Pharmacist: Dispense in tight and light resistant container as defined in USP.
How Supplied
How Supplied HARLIKU (nitisinone) tablet is white to beige, round, flat which may display light yellow to brown speckles, debossed with the “strength” in mg on one side and “L” on the other side. Each tablet contains 2 mg nitisinone. HARLIKU is supplied in a high-density polyethylene (HDPE) square bottle with a child-resistant tamper-evident polypropylene (PP) screw cap. Each bottle contains 60 tablets. 2 mg tablets: NDC 70709-112-60 Storage and Handling Store HARLIKU tablets at room temperature between 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. Pharmacist: Dispense in tight and light resistant container as defined in USP.
Medication Information
Warnings and Precautions
Ocular Symptoms and Hyperkeratotic Plaques Due To Elevated Plasma Tyrosine Levels : Inadequate restriction of tyrosine and phenylalanine intake can lead to elevations in plasma tyrosine and levels above 500 micromol/L may lead to ocular signs and symptoms or painful hyperkeratotic plaques on the soles and palms. ( 5.1 ) Assess plasma tyrosine levels in patients presenting with ocular signs and symptoms. ( 5.1 ) Obtain slit-lamp examination prior to treatment and regularly thereafter. ( 5.1 ) Implement diet restriction and/or treatment interruption as appropriate. ( 5.1 ) Leukopenia and Severe Thrombocytopenia: Monitor platelet and white blood cell counts. ( 5.2 )
Indications and Usage
HARLIKU TM is indicated for the reduction of urine homogentisic acid (HGA) in adult patients with alkaptonuria (AKU).
Dosage and Administration
The recommended dosage of HARLIKU is 2 mg administered orally, once daily. ( 2.1 )
Contraindications
None.
Adverse Reactions
Most common adverse reactions (>1%) are elevated tyrosine levels, keratitis and thrombocytopenia. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Cycle Pharmaceuticals Ltd at 1-855-831-5413 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Sensitive CYP2C9 Substrates: Reduce dosage of co-administered drug metabolized by CYP2C9 by half. ( 7.1 ) OAT1/OAT3 Substrates : Avoid concomitant use of HARLIKU with OAT1/OAT3 substrates. Concomitant use with OAT1/OAT3 substrates may increase the risk of adverse reactions related to the co-administered drug. ( 7.1 )
Storage and Handling
How Supplied HARLIKU (nitisinone) tablet is white to beige, round, flat which may display light yellow to brown speckles, debossed with the “strength” in mg on one side and “L” on the other side. Each tablet contains 2 mg nitisinone. HARLIKU is supplied in a high-density polyethylene (HDPE) square bottle with a child-resistant tamper-evident polypropylene (PP) screw cap. Each bottle contains 60 tablets. 2 mg tablets: NDC 70709-112-60 Storage and Handling Store HARLIKU tablets at room temperature between 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. Pharmacist: Dispense in tight and light resistant container as defined in USP.
How Supplied
How Supplied HARLIKU (nitisinone) tablet is white to beige, round, flat which may display light yellow to brown speckles, debossed with the “strength” in mg on one side and “L” on the other side. Each tablet contains 2 mg nitisinone. HARLIKU is supplied in a high-density polyethylene (HDPE) square bottle with a child-resistant tamper-evident polypropylene (PP) screw cap. Each bottle contains 60 tablets. 2 mg tablets: NDC 70709-112-60 Storage and Handling Store HARLIKU tablets at room temperature between 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. Pharmacist: Dispense in tight and light resistant container as defined in USP.
Description
HARLIKU TM is indicated for the reduction of urine homogentisic acid (HGA) in adult patients with alkaptonuria (AKU).
Section 42229-5
Section 51945-4
Principal Display Panel - 2 mg Carton Label
NDC 70709-112-60
HARLIKUTM
(nitisinone) tablets
2 mg
Rx only 60 tablets
11 Description
HARLIKU (nitisinone) is a hydroxyphenyl-pyruvate dioxygenase inhibitor.
Nitisinone occurs as a white to yellowish-white, crystalline powder. It is practically insoluble in water, soluble in 2M sodium hydroxide and in methanol, and sparingly soluble in alcohol.
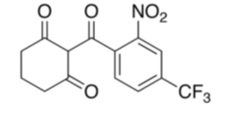
The chemical name of nitisinone is 2-(2-nitro-4-trifluoromethylbenzoyl) cyclohexane-1,3-dione. The molecular formula is C14H10F3NO5 and the molecular weight is 329.23. The structural formula is:
Each HARLIKU (nitisinone) tablet contains 2 mg of nitisinone to be administered orally. The inactive ingredients are glyceryl dibehenate and lactose monohydrate.
8.4 Pediatric Use
The safety and effectiveness of HARLIKU have not been established in pediatric patients with AKU.
8.5 Geriatric Use
There was 1 patient 65 years of age and older in the clinical studies for AKU [see Clinical Studies (14)]. Of the total number of HARLIKU-treated patients in these studies, 1 (2.5 %) were 65 years of age and older, while 0 (0%) were 75 years of age and older.
Clinical studies of HARLIKU did not include sufficient numbers of patients 65 years of age and older to determine whether they respond differently from younger adult patients.
14 Clinical Studies
4 Contraindications
None.
6 Adverse Reactions
Most common adverse reactions (>1%) are elevated tyrosine levels, keratitis and thrombocytopenia. ( 6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Cycle Pharmaceuticals Ltd at 1-855-831-5413 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
Sensitive CYP2C9 Substrates: Reduce dosage of co-administered drug metabolized by CYP2C9 by half. (7.1)
OAT1/OAT3 Substrates: Avoid concomitant use of HARLIKU with OAT1/OAT3 substrates. Concomitant use with OAT1/OAT3 substrates may increase the risk of adverse reactions related to the co-administered drug. (7.1)
12.2 Pharmacodynamics
In patients with AKU, HGA accumulates in various tissues and urine. In an open-label, single center, randomized, no-treatment controlled trial nitisinone treatment resulted in reduction of urinary HGA concentrations in patients with AKU [see Clinical Studies (14)].
Nitisinone exposure-response relationship and time course of pharmacodynamic response for the effectiveness have not been fully characterized.
Nitisinone inhibits catabolism of the amino acid tyrosine and can result in elevated plasma levels of tyrosine in patients with AKU. Treatment with nitisinone does not require routine dietary restriction in patients with AKU; however, patients who develop keratopathies should be monitored and dietary restriction of tyrosine and phenylalanine should be implemented [see Warnings and Precautions (5.1)].
12.3 Pharmacokinetics
The single-dose pharmacokinetics of nitisinone tablets have been studied in healthy adult subjects. Nitisinone pharmacokinetic parameters are presented as geometric mean [range] unless otherwise specified. Nitisinone maximum concentration (Cmax) and area under the curve from time 0 to 120 hours (AUC0-120h) were 1278 [780 to 1649] ng/mL and 77874 [42335 to 104211] ng•h/mL following oral administration of 10 mg (5 times the recommended dosage) nitisinone under fasting conditions.
2.1 Recommended Dosage
The recommended dosage of HARLIKU is 2 mg administered orally, once daily.
Administer HARLIKU with or without food [see Clinical Pharmacology (12.3)].
Missed Dose
If a dose of HARLIKU is missed, do not administer two doses at once to make up for a missed dose. Take the next dose at the scheduled time.
1 Indications and Usage
HARLIKUTM is indicated for the reduction of urine homogentisic acid (HGA) in adult patients with alkaptonuria (AKU).
12.1 Mechanism of Action
Nitisinone is a competitive inhibitor of hydroxyphenyl-pyruvate dioxygenase, an enzyme upstream of homogentisate 1,2-dioxygenase (HGD) in the tyrosine catabolic pathway.
5 Warnings and Precautions
Ocular Symptoms and Hyperkeratotic Plaques Due To Elevated Plasma Tyrosine Levels: Inadequate restriction of tyrosine and phenylalanine intake can lead to elevations in plasma tyrosine and levels above 500 micromol/L may lead to ocular signs and symptoms or painful hyperkeratotic plaques on the soles and palms. (5.1)
- Assess plasma tyrosine levels in patients presenting with ocular signs and symptoms. (5.1)
- Obtain slit-lamp examination prior to treatment and regularly thereafter. (5.1)
- Implement diet restriction and/or treatment interruption as appropriate. (5.1)
Leukopenia and Severe Thrombocytopenia: Monitor platelet and white blood cell counts. (5.2)
2 Dosage and Administration
The recommended dosage of HARLIKU is 2 mg administered orally, once daily. (2.1)
3 Dosage Forms and Strengths
Tablets: 2 mg white to beige, round, flat tablets, which may display light yellow to brown speckles, debossed with “2” mg on one side and “L” on the other side.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of HARLIKU was evaluated in Trial 1, a three-year, open-label, randomized, no-treatment controlled trial in 40 patients with AKU. Patients were between 38 and 68 years of age (27 male, 13 female) [see Clinical Studies (14)]. Patients received either HARLIKU 2 mg orally once daily or no treatment [see Dosage and Administration (2)].
The serious adverse reactions reported with HARLIKU were ocular/visual complaints associated with elevated tyrosine levels (keratitis) [see Warnings and Precautions (5.1)]. Keratitis led to permanent treatment discontinuation in 1 (5%) treated patient.
The most common adverse reactions (>1%) reported in Trial 1 are summarized in TABLE 1.
TABLE 1. Most Common Adverse Reactions* in Patients with AKU Treated with Nitisinone**
|
Adverse Reactions |
Nitisinone (N=20) n (%) |
No Treatment (N=20) n (%) |
|
Elevated tyrosine levels |
19 (95) |
0 (0) |
|
Keratitis*** |
3 (15) |
0 (0) |
|
Thrombocytopenia |
2 (10) |
0 (0) |
* reported in at least 1% of patients; ** another oral formulation of nitisinone;
*** keratitis also includes eye irritation, eye pain and photophobia.
17 Patient Counseling Information
Ocular Symptoms and Hyperkeratotic Plaques Due to Elevated Plasma Tyrosine Levels
Advise the patient or caregiver to report any unexplained ocular or other symptoms promptly to their healthcare provider [see Warnings and Precautions (5.1)].
How Supplied/Storage and Handling
Advise the patient or caregiver to store HARLIKU in the container that it is dispensed in and keep the container tightly closed [see How Supplied/Storage and Handling (16)].
16 How Supplied/storage and Handling
How Supplied
HARLIKU (nitisinone) tablet is white to beige, round, flat which may display light yellow to brown speckles, debossed with the “strength” in mg on one side and “L” on the other side. Each tablet contains 2 mg nitisinone.
HARLIKU is supplied in a high-density polyethylene (HDPE) square bottle with a child-resistant tamper-evident polypropylene (PP) screw cap. Each bottle contains 60 tablets.
2 mg tablets: NDC 70709-112-60
Storage and Handling
Store HARLIKU tablets at room temperature between 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
Pharmacist: Dispense in tight and light resistant container as defined in USP.
7.1 Effects of Harliku On Other Drugs
Sensitive CYP2C9 Substrates
Reduce the dosage of the co-administered drug metabolized by CYP2C9 by half. Additional dosage adjustments may be needed to maintain therapeutic drug concentrations where minimal concentration changes may lead to serious adverse reactions. See prescribing information for those drugs.
Nitisinone is a moderate CYP2C9 inhibitor. Nitisinone may increase exposure of co-administered drugs metabolized by CYP2C9 [see Clinical Pharmacology (12.3)].
OAT1/OAT3 Substrates
The concomitant use of HARLIKU with OAT1/OAT3 substrates may increase the risk of adverse reactions related to the co-administered drug. See prescribing information for those drugs.
Nitisinone is an OAT1/OAT3 inhibitor which can lead to increased exposure of the co-administered drug. [ see Clinical Pharmacology (12.3)] .
5.2 Leukopenia and Severe Thrombocytopenia
In clinical trials, patients with hereditary tyrosinemia type 1 (HT-1) treated with another oral formulation of nitisinone and dietary restriction developed reversible leukopenia (3%), thrombocytopenia (3%), or both (1.5%). Ten percent of patients in Trial 1 developed thrombocytopenia [see Adverse Reactions (6.1)]. No patients developed infections or bleeding as a result of the episodes of leukopenia and thrombocytopenia. Monitor platelet and white blood cell counts during HARLIKU therapy.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic potential of nitisinone was assessed in a 26-week oral (gavage) carcinogenicity study in Tg.rasH2 mice. There were no drug-related neoplastic findings in male or female Tg.rasH2 mice at doses up to 100 mg/kg/day nitisinone (approximately 243 times the maximum recommended human daily dose of 2 mg/day).
Nitisinone was not genotoxic in the Ames test and the in vivo mouse liver unscheduled DNA synthesis (UDS) test. Nitisinone was mutagenic in the mouse lymphoma cell (L5178Y/TK+/-) forward mutation test and in an in vivo mouse bone marrow micronucleus test.
5.1 Ocular Symptoms and Hyperkeratotic Plaques Due to Elevated Plasma Tyrosine Levels
Treatment with HARLIKU may cause elevated plasma tyrosine levels in patients with AKU. Tyrosine levels greater than 500 micromol/L may lead to the following:
- Ocular signs and symptoms including keratitis, corneal opacities, corneal irritation, corneal ulcers, conjunctivitis, eye pain, and photophobia. These ocular adverse reactions have been reported in patients treated with nitisinone [see Adverse Reactions (6.1)]. In a clinical trial in the AKU population, without dietary restriction and reported tyrosine levels > 500 micromol/L, both symptomatic and asymptomatic keratopathies have been observed. Perform a baseline ophthalmologic examination including slit-lamp examination prior to initiating HARLIKU treatment and regularly thereafter. Patients who develop photophobia, eye pain, or signs of inflammation such as redness, swelling, or burning of the eyes or tyrosine levels are > 500 micromol/L during treatment with HARLIKU should undergo slit-lamp re-examination and immediate measurement of the plasma tyrosine concentration.
- Painful hyperkeratotic plaques on the soles and palms.
There is no routine dietary restriction requirement for AKU patients taking HARLIKU. However, in patients who develop keratopathies, monitor plasma tyrosine levels, and implement a diet restricted in tyrosine and phenylalanine to keep the plasma tyrosine level below 500 micromol/L. Consider temporarily interrupting HARLIKU until resolution of symptoms.
Structured Label Content
Section 42229-5 (42229-5)
Section 51945-4 (51945-4)
Principal Display Panel - 2 mg Carton Label
NDC 70709-112-60
HARLIKUTM
(nitisinone) tablets
2 mg
Rx only 60 tablets
11 Description (11 DESCRIPTION)
HARLIKU (nitisinone) is a hydroxyphenyl-pyruvate dioxygenase inhibitor.
Nitisinone occurs as a white to yellowish-white, crystalline powder. It is practically insoluble in water, soluble in 2M sodium hydroxide and in methanol, and sparingly soluble in alcohol.
The chemical name of nitisinone is 2-(2-nitro-4-trifluoromethylbenzoyl) cyclohexane-1,3-dione. The molecular formula is C14H10F3NO5 and the molecular weight is 329.23. The structural formula is:
Each HARLIKU (nitisinone) tablet contains 2 mg of nitisinone to be administered orally. The inactive ingredients are glyceryl dibehenate and lactose monohydrate.
8.4 Pediatric Use
The safety and effectiveness of HARLIKU have not been established in pediatric patients with AKU.
8.5 Geriatric Use
There was 1 patient 65 years of age and older in the clinical studies for AKU [see Clinical Studies (14)]. Of the total number of HARLIKU-treated patients in these studies, 1 (2.5 %) were 65 years of age and older, while 0 (0%) were 75 years of age and older.
Clinical studies of HARLIKU did not include sufficient numbers of patients 65 years of age and older to determine whether they respond differently from younger adult patients.
14 Clinical Studies (14 CLINICAL STUDIES)
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (>1%) are elevated tyrosine levels, keratitis and thrombocytopenia. ( 6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Cycle Pharmaceuticals Ltd at 1-855-831-5413 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
Sensitive CYP2C9 Substrates: Reduce dosage of co-administered drug metabolized by CYP2C9 by half. (7.1)
OAT1/OAT3 Substrates: Avoid concomitant use of HARLIKU with OAT1/OAT3 substrates. Concomitant use with OAT1/OAT3 substrates may increase the risk of adverse reactions related to the co-administered drug. (7.1)
12.2 Pharmacodynamics
In patients with AKU, HGA accumulates in various tissues and urine. In an open-label, single center, randomized, no-treatment controlled trial nitisinone treatment resulted in reduction of urinary HGA concentrations in patients with AKU [see Clinical Studies (14)].
Nitisinone exposure-response relationship and time course of pharmacodynamic response for the effectiveness have not been fully characterized.
Nitisinone inhibits catabolism of the amino acid tyrosine and can result in elevated plasma levels of tyrosine in patients with AKU. Treatment with nitisinone does not require routine dietary restriction in patients with AKU; however, patients who develop keratopathies should be monitored and dietary restriction of tyrosine and phenylalanine should be implemented [see Warnings and Precautions (5.1)].
12.3 Pharmacokinetics
The single-dose pharmacokinetics of nitisinone tablets have been studied in healthy adult subjects. Nitisinone pharmacokinetic parameters are presented as geometric mean [range] unless otherwise specified. Nitisinone maximum concentration (Cmax) and area under the curve from time 0 to 120 hours (AUC0-120h) were 1278 [780 to 1649] ng/mL and 77874 [42335 to 104211] ng•h/mL following oral administration of 10 mg (5 times the recommended dosage) nitisinone under fasting conditions.
2.1 Recommended Dosage
The recommended dosage of HARLIKU is 2 mg administered orally, once daily.
Administer HARLIKU with or without food [see Clinical Pharmacology (12.3)].
Missed Dose
If a dose of HARLIKU is missed, do not administer two doses at once to make up for a missed dose. Take the next dose at the scheduled time.
1 Indications and Usage (1 INDICATIONS AND USAGE)
HARLIKUTM is indicated for the reduction of urine homogentisic acid (HGA) in adult patients with alkaptonuria (AKU).
12.1 Mechanism of Action
Nitisinone is a competitive inhibitor of hydroxyphenyl-pyruvate dioxygenase, an enzyme upstream of homogentisate 1,2-dioxygenase (HGD) in the tyrosine catabolic pathway.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Ocular Symptoms and Hyperkeratotic Plaques Due To Elevated Plasma Tyrosine Levels: Inadequate restriction of tyrosine and phenylalanine intake can lead to elevations in plasma tyrosine and levels above 500 micromol/L may lead to ocular signs and symptoms or painful hyperkeratotic plaques on the soles and palms. (5.1)
- Assess plasma tyrosine levels in patients presenting with ocular signs and symptoms. (5.1)
- Obtain slit-lamp examination prior to treatment and regularly thereafter. (5.1)
- Implement diet restriction and/or treatment interruption as appropriate. (5.1)
Leukopenia and Severe Thrombocytopenia: Monitor platelet and white blood cell counts. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dosage of HARLIKU is 2 mg administered orally, once daily. (2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 2 mg white to beige, round, flat tablets, which may display light yellow to brown speckles, debossed with “2” mg on one side and “L” on the other side.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of HARLIKU was evaluated in Trial 1, a three-year, open-label, randomized, no-treatment controlled trial in 40 patients with AKU. Patients were between 38 and 68 years of age (27 male, 13 female) [see Clinical Studies (14)]. Patients received either HARLIKU 2 mg orally once daily or no treatment [see Dosage and Administration (2)].
The serious adverse reactions reported with HARLIKU were ocular/visual complaints associated with elevated tyrosine levels (keratitis) [see Warnings and Precautions (5.1)]. Keratitis led to permanent treatment discontinuation in 1 (5%) treated patient.
The most common adverse reactions (>1%) reported in Trial 1 are summarized in TABLE 1.
TABLE 1. Most Common Adverse Reactions* in Patients with AKU Treated with Nitisinone**
|
Adverse Reactions |
Nitisinone (N=20) n (%) |
No Treatment (N=20) n (%) |
|
Elevated tyrosine levels |
19 (95) |
0 (0) |
|
Keratitis*** |
3 (15) |
0 (0) |
|
Thrombocytopenia |
2 (10) |
0 (0) |
* reported in at least 1% of patients; ** another oral formulation of nitisinone;
*** keratitis also includes eye irritation, eye pain and photophobia.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Ocular Symptoms and Hyperkeratotic Plaques Due to Elevated Plasma Tyrosine Levels
Advise the patient or caregiver to report any unexplained ocular or other symptoms promptly to their healthcare provider [see Warnings and Precautions (5.1)].
How Supplied/Storage and Handling
Advise the patient or caregiver to store HARLIKU in the container that it is dispensed in and keep the container tightly closed [see How Supplied/Storage and Handling (16)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
How Supplied
HARLIKU (nitisinone) tablet is white to beige, round, flat which may display light yellow to brown speckles, debossed with the “strength” in mg on one side and “L” on the other side. Each tablet contains 2 mg nitisinone.
HARLIKU is supplied in a high-density polyethylene (HDPE) square bottle with a child-resistant tamper-evident polypropylene (PP) screw cap. Each bottle contains 60 tablets.
2 mg tablets: NDC 70709-112-60
Storage and Handling
Store HARLIKU tablets at room temperature between 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
Pharmacist: Dispense in tight and light resistant container as defined in USP.
7.1 Effects of Harliku On Other Drugs (7.1 Effects of HARLIKU on Other Drugs)
Sensitive CYP2C9 Substrates
Reduce the dosage of the co-administered drug metabolized by CYP2C9 by half. Additional dosage adjustments may be needed to maintain therapeutic drug concentrations where minimal concentration changes may lead to serious adverse reactions. See prescribing information for those drugs.
Nitisinone is a moderate CYP2C9 inhibitor. Nitisinone may increase exposure of co-administered drugs metabolized by CYP2C9 [see Clinical Pharmacology (12.3)].
OAT1/OAT3 Substrates
The concomitant use of HARLIKU with OAT1/OAT3 substrates may increase the risk of adverse reactions related to the co-administered drug. See prescribing information for those drugs.
Nitisinone is an OAT1/OAT3 inhibitor which can lead to increased exposure of the co-administered drug. [ see Clinical Pharmacology (12.3)] .
5.2 Leukopenia and Severe Thrombocytopenia
In clinical trials, patients with hereditary tyrosinemia type 1 (HT-1) treated with another oral formulation of nitisinone and dietary restriction developed reversible leukopenia (3%), thrombocytopenia (3%), or both (1.5%). Ten percent of patients in Trial 1 developed thrombocytopenia [see Adverse Reactions (6.1)]. No patients developed infections or bleeding as a result of the episodes of leukopenia and thrombocytopenia. Monitor platelet and white blood cell counts during HARLIKU therapy.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic potential of nitisinone was assessed in a 26-week oral (gavage) carcinogenicity study in Tg.rasH2 mice. There were no drug-related neoplastic findings in male or female Tg.rasH2 mice at doses up to 100 mg/kg/day nitisinone (approximately 243 times the maximum recommended human daily dose of 2 mg/day).
Nitisinone was not genotoxic in the Ames test and the in vivo mouse liver unscheduled DNA synthesis (UDS) test. Nitisinone was mutagenic in the mouse lymphoma cell (L5178Y/TK+/-) forward mutation test and in an in vivo mouse bone marrow micronucleus test.
5.1 Ocular Symptoms and Hyperkeratotic Plaques Due to Elevated Plasma Tyrosine Levels
Treatment with HARLIKU may cause elevated plasma tyrosine levels in patients with AKU. Tyrosine levels greater than 500 micromol/L may lead to the following:
- Ocular signs and symptoms including keratitis, corneal opacities, corneal irritation, corneal ulcers, conjunctivitis, eye pain, and photophobia. These ocular adverse reactions have been reported in patients treated with nitisinone [see Adverse Reactions (6.1)]. In a clinical trial in the AKU population, without dietary restriction and reported tyrosine levels > 500 micromol/L, both symptomatic and asymptomatic keratopathies have been observed. Perform a baseline ophthalmologic examination including slit-lamp examination prior to initiating HARLIKU treatment and regularly thereafter. Patients who develop photophobia, eye pain, or signs of inflammation such as redness, swelling, or burning of the eyes or tyrosine levels are > 500 micromol/L during treatment with HARLIKU should undergo slit-lamp re-examination and immediate measurement of the plasma tyrosine concentration.
- Painful hyperkeratotic plaques on the soles and palms.
There is no routine dietary restriction requirement for AKU patients taking HARLIKU. However, in patients who develop keratopathies, monitor plasma tyrosine levels, and implement a diet restricted in tyrosine and phenylalanine to keep the plasma tyrosine level below 500 micromol/L. Consider temporarily interrupting HARLIKU until resolution of symptoms.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:21.090680 · Updated: 2026-03-14T22:29:33.822109