Llorens- Orasep Spray 504
7c50d986-6a7b-425f-90a0-a60e21283bdf
34390-5
HUMAN OTC DRUG LABEL
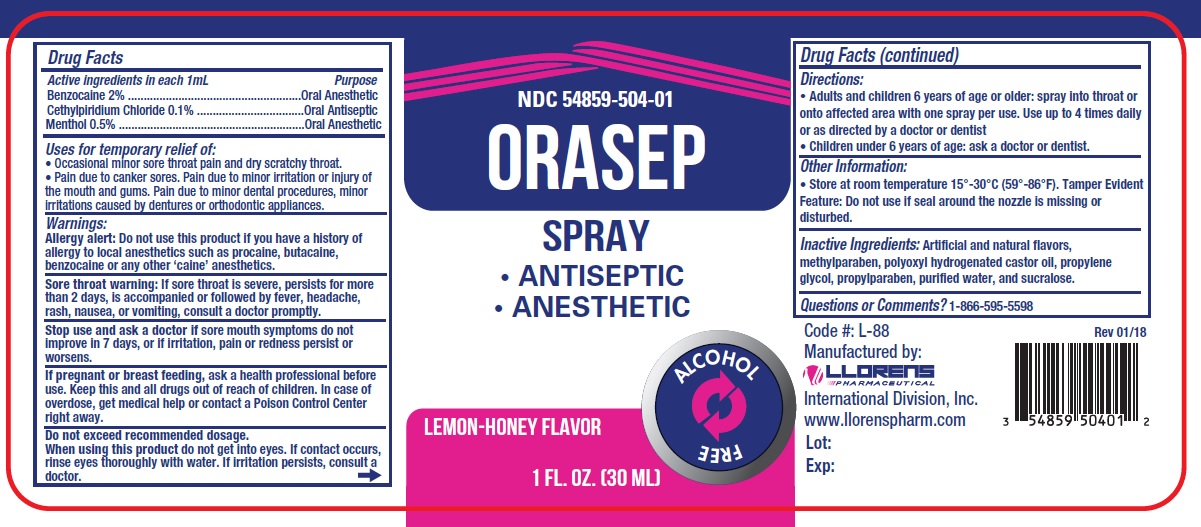
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients: Benzocaine...................... 2% Anesthetic Cetylpyridinium Chloride..........0.1% Antiseptic Menthol.................................0.5% Anesthetic
Medication Information
Warnings and Precautions
Warnings:
Allergy alert: Do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine or any other 'caine' anesthetics.
Sore throat warning: If sore throat is sever, persists for more than2 days, is accompanied or followed by fever, headache, rash, nausea, or vomitings, consult a doctor promptly.
Stop use and ask a doctor if sore mouth symptoms do not improve in 7 days, or if irritation, pain or redness persist or worsens.
Do not exceed recommended dosage.
When using this product do not get into eyes. If contact occurs, rinse eyes thoroughly with water. If irritation persists, consult a doctor.
Indications and Usage
Uses for temporary relief of:
- Occasional minor sore throat pain and dry scratch throat.
- Pain due to canker sores. Pain due to minor irritation or injury of the mouth and gums. Pain due to minor dental procedures, minor irritations cause by dentures or orthodontic appliances.
Dosage and Administration
Directions:
- Adults and children 6 years of age or older: spray into throat or onto affected area with one spray per use. Use up to 4 times daily or as directed by a doctor or dentist
- Children under 6 years of age: ask a doctor or dentist.
Description
Active Ingredients: Benzocaine...................... 2% Anesthetic Cetylpyridinium Chloride..........0.1% Antiseptic Menthol.................................0.5% Anesthetic
Section 50565-1
Keep this and all drugs out of the reach of children.In case of overdose, get medical help or contact a Poison Control Center right away.
Section 51727-6
Inactive Ingredients: Artificial and natural flavors, methylparaben, polyoxyl hydrogenated castor oil, propylene glycol, propylparaben, purified water, and sucralose.
Section 51945-4
Section 53413-1
Questions or comments? 1-866-595-5598
Section 53414-9
If pregnant or breast feeding,ask a health professional before use.
Section 55105-1
Purpose
Oral anesthetic
Oral antiseptic
Section 55106-9
Active Ingredients:
Benzocaine...................... 2% Anesthetic
Cetylpyridinium Chloride..........0.1% Antiseptic
Menthol.................................0.5% Anesthetic
Structured Label Content
Indications and Usage (34067-9)
Uses for temporary relief of:
- Occasional minor sore throat pain and dry scratch throat.
- Pain due to canker sores. Pain due to minor irritation or injury of the mouth and gums. Pain due to minor dental procedures, minor irritations cause by dentures or orthodontic appliances.
Dosage and Administration (34068-7)
Directions:
- Adults and children 6 years of age or older: spray into throat or onto affected area with one spray per use. Use up to 4 times daily or as directed by a doctor or dentist
- Children under 6 years of age: ask a doctor or dentist.
Warnings and Precautions (34071-1)
Warnings:
Allergy alert: Do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine or any other 'caine' anesthetics.
Sore throat warning: If sore throat is sever, persists for more than2 days, is accompanied or followed by fever, headache, rash, nausea, or vomitings, consult a doctor promptly.
Stop use and ask a doctor if sore mouth symptoms do not improve in 7 days, or if irritation, pain or redness persist or worsens.
Do not exceed recommended dosage.
When using this product do not get into eyes. If contact occurs, rinse eyes thoroughly with water. If irritation persists, consult a doctor.
Section 50565-1 (50565-1)
Keep this and all drugs out of the reach of children.In case of overdose, get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
Inactive Ingredients: Artificial and natural flavors, methylparaben, polyoxyl hydrogenated castor oil, propylene glycol, propylparaben, purified water, and sucralose.
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
Questions or comments? 1-866-595-5598
Section 53414-9 (53414-9)
If pregnant or breast feeding,ask a health professional before use.
Section 55105-1 (55105-1)
Purpose
Oral anesthetic
Oral antiseptic
Section 55106-9 (55106-9)
Active Ingredients:
Benzocaine...................... 2% Anesthetic
Cetylpyridinium Chloride..........0.1% Antiseptic
Menthol.................................0.5% Anesthetic
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:01.250657 · Updated: 2026-03-14T23:12:08.624404