Norelgestromin And Ethinyl Estradiol Transdermal System
7c2cc8ab-db6d-4a1c-ae74-e0ef27e2f3a9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
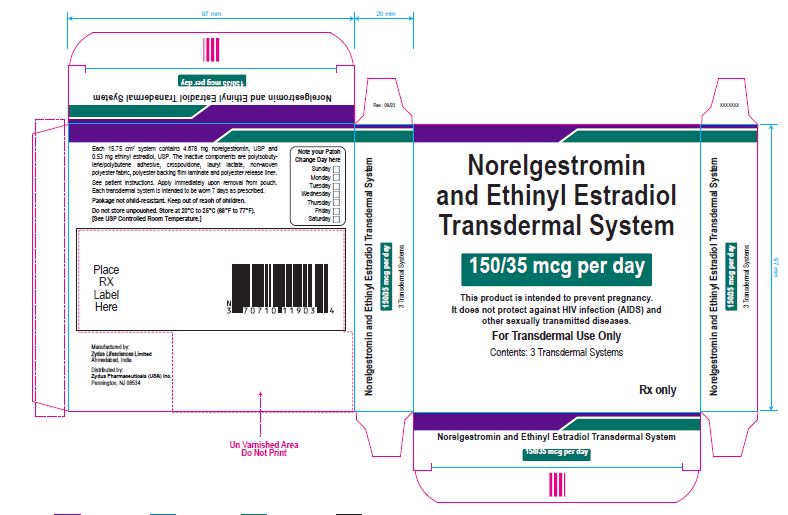
NDC 70771-1777-3 Norelgestromin and Ethinyl Estradiol Transdermal System 150/35 mcg per day This product is intended to prevent pregnancy. It does not protect against HIV infection (AIDS) and other sexually transmitted diseases. For Transdermal Use Only Contents: 3 Transdermal Systems Rx only Each 15.75 cm 2 system contains 4.678 mg norelgestromin, USP and 0.53 mg ethinyl estradiol, USP. The inactive components are polyisobutylene/polybutene adhesive, crospovidone, lauryl lactate, non-woven polyester fabric, polyester backing film laminate and polyester release liner. See patient instructions. Apply immediately upon removal from pouch. Each transdermal system is intended to be worn 7 days as prescribed. Package not child-resistant. Keep out of reach of children. Do not store unpouched. Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Manufactured by: Zydus Lifesciences Ltd. Ahmedabad, India Distributed by: Zydus Pharmaceuticals (USA) Inc. Pennington, NJ 08534 Rev.: 10/22
Medication Information
Description
NDC 70771-1777-3 Norelgestromin and Ethinyl Estradiol Transdermal System 150/35 mcg per day This product is intended to prevent pregnancy. It does not protect against HIV infection (AIDS) and other sexually transmitted diseases. For Transdermal Use Only Contents: 3 Transdermal Systems Rx only Each 15.75 cm 2 system contains 4.678 mg norelgestromin, USP and 0.53 mg ethinyl estradiol, USP. The inactive components are polyisobutylene/polybutene adhesive, crospovidone, lauryl lactate, non-woven polyester fabric, polyester backing film laminate and polyester release liner. See patient instructions. Apply immediately upon removal from pouch. Each transdermal system is intended to be worn 7 days as prescribed. Package not child-resistant. Keep out of reach of children. Do not store unpouched. Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Manufactured by: Zydus Lifesciences Ltd. Ahmedabad, India Distributed by: Zydus Pharmaceuticals (USA) Inc. Pennington, NJ 08534 Rev.: 10/22
Package Label.principal Display Panel
NDC 70771-1777-3
Norelgestromin and Ethinyl Estradiol Transdermal System 150/35 mcg per day
This product is intended to prevent pregnancy. It does not protect against HIV infection (AIDS) and other sexually transmitted diseases.
For Transdermal Use Only
Contents: 3 Transdermal Systems
Rx only
Each 15.75 cm2 system contains 4.678 mg norelgestromin, USP and 0.53 mg ethinyl estradiol, USP. The inactive components are polyisobutylene/polybutene adhesive, crospovidone, lauryl lactate, non-woven polyester fabric, polyester backing film laminate and polyester release liner.
See patient instructions. Apply immediately upon removal from pouch. Each transdermal system is intended to be worn 7 days as prescribed.
Package not child-resistant. Keep out of reach of children.
Do not store unpouched. Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Manufactured by:
Zydus Lifesciences Ltd.
Ahmedabad, India
Distributed by:
Zydus Pharmaceuticals (USA) Inc.
Pennington, NJ 08534
Rev.: 10/22
Structured Label Content
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
NDC 70771-1777-3
Norelgestromin and Ethinyl Estradiol Transdermal System 150/35 mcg per day
This product is intended to prevent pregnancy. It does not protect against HIV infection (AIDS) and other sexually transmitted diseases.
For Transdermal Use Only
Contents: 3 Transdermal Systems
Rx only
Each 15.75 cm2 system contains 4.678 mg norelgestromin, USP and 0.53 mg ethinyl estradiol, USP. The inactive components are polyisobutylene/polybutene adhesive, crospovidone, lauryl lactate, non-woven polyester fabric, polyester backing film laminate and polyester release liner.
See patient instructions. Apply immediately upon removal from pouch. Each transdermal system is intended to be worn 7 days as prescribed.
Package not child-resistant. Keep out of reach of children.
Do not store unpouched. Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Manufactured by:
Zydus Lifesciences Ltd.
Ahmedabad, India
Distributed by:
Zydus Pharmaceuticals (USA) Inc.
Pennington, NJ 08534
Rev.: 10/22
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:22.606761 · Updated: 2026-03-14T21:57:28.446364