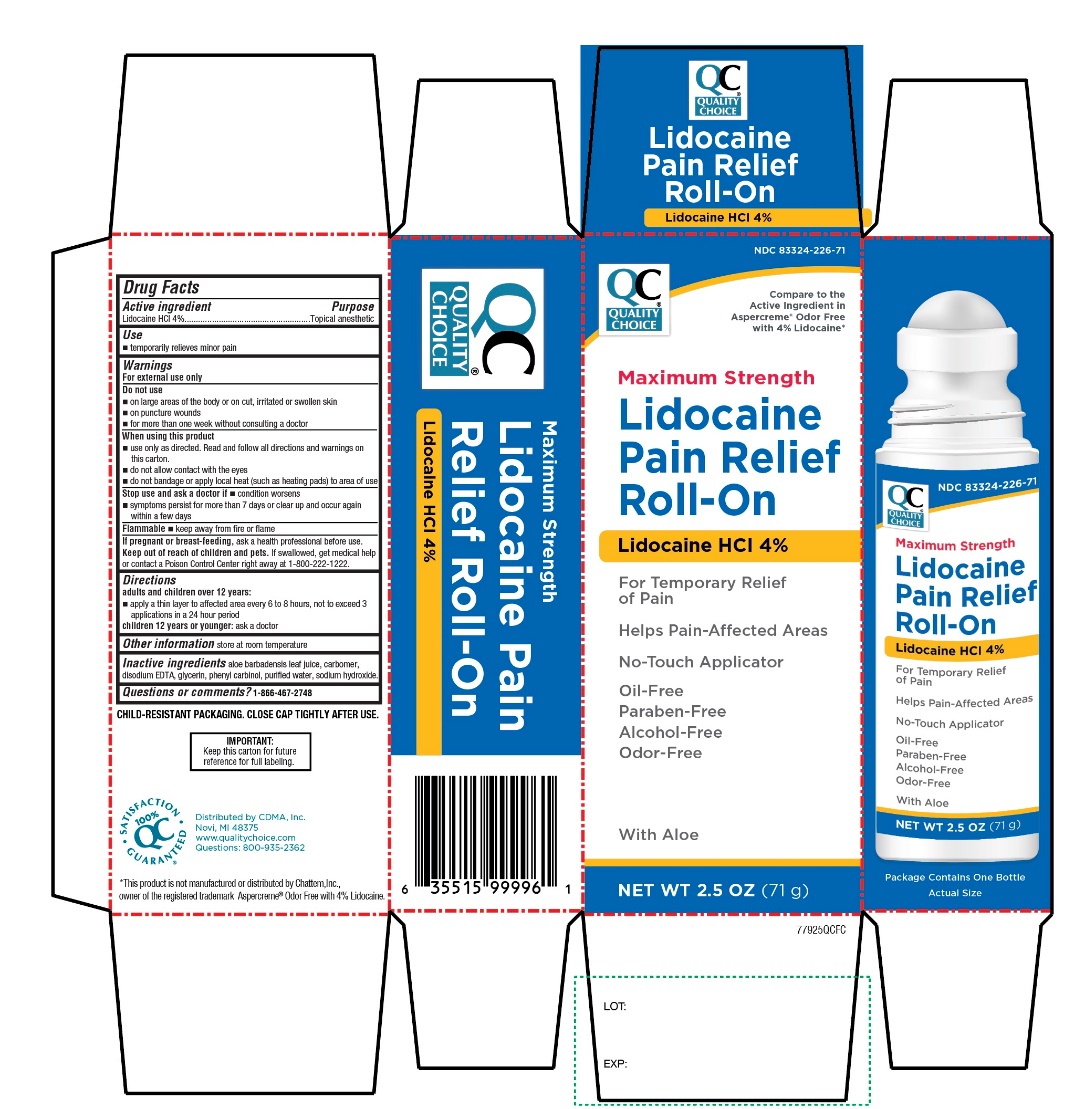
Quality Choice Maximum Strength Lidocaine Pain Relief Roll-on Drug Facts
7b3ff4c1-0b60-4f7a-8aa4-c84b7c7137c4
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine HCI 4%
Purpose
Topical anesthetic
Medication Information
Purpose
Topical anesthetic
Description
Lidocaine HCI 4%
Uses
- temporarily relieves minor pain
Warnings
For external use only
Flammable
- Keep away from fire or flame
Directions
adults and children over 12 years:
- apply a thin layer to affected area every 6 to 8 hours, not to exceed 3 applications in a 24 hour period
children 12 years and younger: ask a doctor
Do Not Use
- on large areas of the body or on cut, irritated or swollen skin
- on puncture wounds
- for more than one week without consulting a doctor
Active Ingredient
Lidocaine HCI 4%
Other Information
Store at room temperature
Inactive Ingredients
aloe barbadensis leaf juice. Carbomer, disodium EDTA, glycerin, phenyl carbinol, purified water, sodium hydroxide.
Questions Or Comments?
1-866-467-2748
100% SATISFACTION GUARANTEED
Principal Display Panel
NDC# 83324-226-71
QUALITY CHOICE®
Compare to the Active Ingredient in Aspercreme® Odor Free with 4 % lidocaine*
Maximum Strength
Lidocaine
Pain Relief
Roll-On
Lidocaine HCl 4%
For Temporary Relief of Pain
Helps pain-affected areas
No-touch applicator
Oil-Free
Paraben-Free
Alcohol-Free
Odor-Free
With Aloe
NET WT 2.5 OZ (71 g)
Distributed by: CDMA, Inc.
Novi, MI 48375
*This product is not manufactured or distributed by Chattem, Inc., owner of the registered trademark Aspercreme® Odor Free with 4% Lidocaine.
When Using This Product
- use only as directed. Read and follow all directions and warnings on this carton.
- do not allow contact with the eyes
- do not bandage or apply local heat (such as heating pads) to the area of use
Stop Use and Ask A Doctor If
- condition worsens
- symptoms persist for more than 7 days or clear up and occur again within a few days
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children and Pets.
If swallowed, get medical help or contact a Poison Control Center right away at 1-800-222-1222.
Structured Label Content
Uses
- temporarily relieves minor pain
Purpose
Topical anesthetic
Warnings
For external use only
Flammable
- Keep away from fire or flame
Directions
adults and children over 12 years:
- apply a thin layer to affected area every 6 to 8 hours, not to exceed 3 applications in a 24 hour period
children 12 years and younger: ask a doctor
Do Not Use (Do not use)
- on large areas of the body or on cut, irritated or swollen skin
- on puncture wounds
- for more than one week without consulting a doctor
Active Ingredient (Active ingredient)
Lidocaine HCI 4%
Other Information
Store at room temperature
Inactive Ingredients (Inactive ingredients)
aloe barbadensis leaf juice. Carbomer, disodium EDTA, glycerin, phenyl carbinol, purified water, sodium hydroxide.
Questions Or Comments? (Questions or comments?)
1-866-467-2748
100% SATISFACTION GUARANTEED
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC# 83324-226-71
QUALITY CHOICE®
Compare to the Active Ingredient in Aspercreme® Odor Free with 4 % lidocaine*
Maximum Strength
Lidocaine
Pain Relief
Roll-On
Lidocaine HCl 4%
For Temporary Relief of Pain
Helps pain-affected areas
No-touch applicator
Oil-Free
Paraben-Free
Alcohol-Free
Odor-Free
With Aloe
NET WT 2.5 OZ (71 g)
Distributed by: CDMA, Inc.
Novi, MI 48375
*This product is not manufactured or distributed by Chattem, Inc., owner of the registered trademark Aspercreme® Odor Free with 4% Lidocaine.
When Using This Product (When using this product)
- use only as directed. Read and follow all directions and warnings on this carton.
- do not allow contact with the eyes
- do not bandage or apply local heat (such as heating pads) to the area of use
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens
- symptoms persist for more than 7 days or clear up and occur again within a few days
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children and Pets. (Keep out of reach of children and pets.)
If swallowed, get medical help or contact a Poison Control Center right away at 1-800-222-1222.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:30.604515 · Updated: 2026-03-14T23:09:19.585027