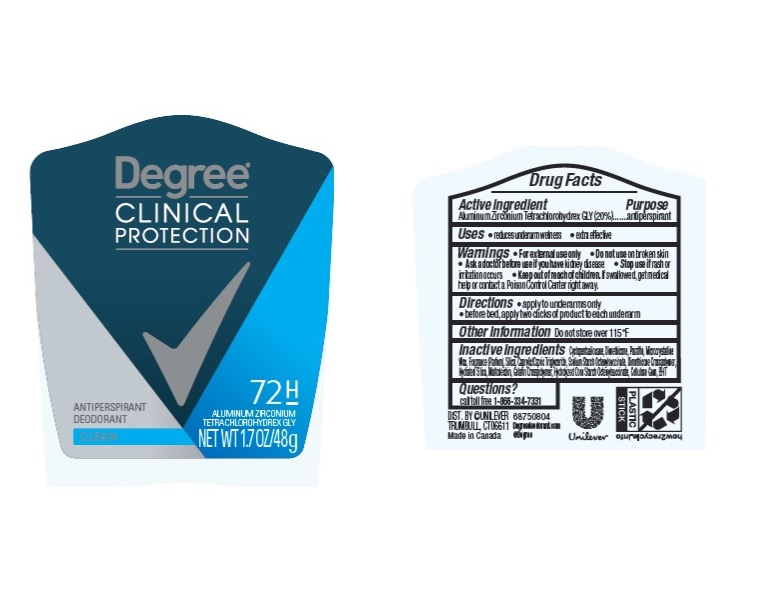
degree clinical protection clean 72h antiperspirant deodorant
7a307e8a-51ce-41a4-b12b-ecb44f0a2010
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredient Aluminum Zirconium Tetrachlorohydrex GLY (20%)
Medication Information
Warnings and Precautions
Warnings
For external use only
Active Ingredient
Active Ingredient
Aluminum Zirconium Tetrachlorohydrex GLY (20%)
Indications and Usage
Uses
▪ reduces underarm wetness
▪ 24 hour protection
▪ extra effective
Dosage and Administration
Directions
▪ apply to underarms only
▪ before bed, apply two clicks of product to each underarm
Description
Active Ingredient Aluminum Zirconium Tetrachlorohydrex GLY (20%)
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use if rash or irritation occurs
Section 50569-3
Ask a doctor before use if you have kidney disease
Section 50570-1
Do not use on broken skin
Section 51727-6
Inactive ingredients
Cyclopentasiloxane
Dimethicone
Paraffin
Microcrystalline Wax
Fragrance (Parfum)
Silica
Caprylic/Capric Triglyceride
Sodium Starch Octenylsuccinate
Dimethicone Crosspolymer
Hydrated silica
Maltodextrin
Gelatin crosspolymer
Hydrolyzed Corn Starch
Cellulose Gum
BHT
Section 53413-1
Questions? Call toll-free 1-866-334-7331
Section 55105-1
Purpose
anti-perspirant
Packaging
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
For external use only
Active Ingredient (55106-9)
Active Ingredient
Aluminum Zirconium Tetrachlorohydrex GLY (20%)
Indications and Usage (34067-9)
Uses
▪ reduces underarm wetness
▪ 24 hour protection
▪ extra effective
Dosage and Administration (34068-7)
Directions
▪ apply to underarms only
▪ before bed, apply two clicks of product to each underarm
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use if rash or irritation occurs
Section 50569-3 (50569-3)
Ask a doctor before use if you have kidney disease
Section 50570-1 (50570-1)
Do not use on broken skin
Section 51727-6 (51727-6)
Inactive ingredients
Cyclopentasiloxane
Dimethicone
Paraffin
Microcrystalline Wax
Fragrance (Parfum)
Silica
Caprylic/Capric Triglyceride
Sodium Starch Octenylsuccinate
Dimethicone Crosspolymer
Hydrated silica
Maltodextrin
Gelatin crosspolymer
Hydrolyzed Corn Starch
Cellulose Gum
BHT
Section 53413-1 (53413-1)
Questions? Call toll-free 1-866-334-7331
Section 55105-1 (55105-1)
Purpose
anti-perspirant
Packaging
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:11.580672 · Updated: 2026-03-14T23:12:21.703136