Theo-24®
7a108fa0-c230-475f-be93-6f864b98a410
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Rx Only
Indications and Usage
Theophylline is indicated for the treatment of the symptoms and reversible airflow obstruction associated with chronic asthma and other chronic lung diseases, e.g., emphysema and chronic bronchitis.
Contraindications
Theo-24 is contraindicated in patients with a history of hypersensitivity to theophylline or other components in the product.
Adverse Reactions
Adverse reactions associated with theophylline are generally mild when peak serum theophylline concentrations are <20 mcg/mL and mainly consist of transient caffeine-like adverse effects such as nausea, vomiting, headache, and insomnia. When peak serum theophylline concentrations exceed 20 mcg/mL, however, theophylline produces a wide range of adverse reactions including persistent vomiting, cardiac arrhythmias, and intractable seizures which can be lethal (see OVERDOSAGE ). The transient caffeine-like adverse reactions occur in about 50% of patients when theophylline therapy is initiated at doses higher than recommended initial doses (e.g., >300 mg/day in adults and >12 mg/kg/day in children beyond 1 year of age). During the initiation of theophylline therapy, caffeine-like adverse effects may transiently alter patient behavior, especially in school age children, but this response rarely persists. Initiation of theophylline therapy at a low dose with subsequent slow titration to a predetermined age-related maximum dose will significantly reduce the frequency of these transient adverse effects (see DOSAGE AND ADMINISTRATION , Table V). In a small percentage of patients (<3% of children and <10% of adults) the caffeine-like adverse effects persist during maintenance therapy, even at peak serum theophylline concentrations within the therapeutic range (i.e., 10 - 20 mcg/mL). Dosage reduction may alleviate the caffeine-like adverse effects in these patients, however, persistent adverse effects should result in a reevaluation of the need for continued theophylline therapy and the potential therapeutic benefit of alternative treatment. Other adverse reactions that have been reported at serum theophylline concentrations <20 mcg/mL include diarrhea, irritability, restlessness, fine skeletal muscle tremors, and transient diuresis. In patients with hypoxia secondary to COPD, multifocal atrial tachycardia and flutter have been reported at serum theophylline concentrations ≥15 mcg/mL. There have been a few isolated reports of seizures at serum theophylline concentrations <20 mcg/mL in patients with an underlying neurological disease or in elderly patients. The occurrence of seizures in elderly patients with serum theophylline concentrations <20 mcg/mL may be secondary to decreased protein binding resulting in a larger proportion of the total serum theophylline concentration in the pharmacologically active unbound form. The clinical characteristics of the seizures reported in patients with serum theophylline concentrations <20 mcg/mL have generally been milder than seizures associated with excessive serum theophylline concentrations resulting from an overdose (i.e., they have generally been transient, often stopped without anticonvulsant therapy, and did not result in neurological residua). Table IV. Manifestations of theophylline toxicity.* Percentage of patients reported with sign or symptom Acute Overdose (Large Single Ingestion) Chronic Overdosage (Multiple Excessive Doses) Sign/Symptom Study 1 (n=157) Study 2 (n=14) Study 1 (n=92) Study 2 (n=102) * These data are derived from two studies in patients with serum theophylline concentrations >30 mcg/mL. In the first study (Study #1—Shanon, Ann Intern Med 1993;119:1161-67), data were prospectively collected from 249 consecutive cases of theophylline toxicity referred to a regional poison center for consultation. In the second study (Study #2—Sessler, Am J Med 1990;88:567-76), data were retrospectively collected from 116 cases with serum theophylline concentrations >30 mcg/mL among 6000 blood samples obtained for measurement of serum theophylline concentrations in three emergency departments. Differences in the incidence of manifestations of theophylline toxicity between the two studies may reflect sample selection as a result of study design (e.g., in Study #1, 48% of the patients had acute intoxications versus only 10 in Study #2) and different methods of reporting results. † NR =Not reported in a comparable manner. Asymptomatic NR† 0 NR† 6 Gastrointestinal Vomiting 73 93 30 61 Abdominal pain NR† 21 NR† 12 Diarrhea NR† 0 NR† 14 Hematemesis NR† 0 NR† 2 Metabolic/Other Hypokalemia 85 79 44 43 Hyperglycemia 98 NR† 18 NR† Acid/base disturbance 34 21 9 5 Rhabdomyolysis NR† 7 NR† 0 Cardiovascular Sinus tachycardia 100 86 100 62 Other supraventricular tachycardias 2 21 12 14 Ventricular premature beats 3 21 10 19 Atrial fibrillation or flutter 1 NR† 12 NR† Multifocal atrial tachycardia 0 NR† 2 NR† Ventricular arrhythmias with hemodynamic instability 7 14 40 0 Hypotension/shock NR† 21 NR† 8 Neurologic Nervousness NR† 64 NR† 21 Tremors 38 29 16 14 Disorientation NR† 7 NR† 11 Seizures 5 14 14 5 Death 3 21 10 4
How Supplied
Theo-24 (theophylline anhydrous) is supplied in extended-release capsules containing 100, 200, 300 or 400 mg of anhydrous theophylline. Theo-24 100 mg capsules are orange opaque and natural, with markings Theo-24, 100 mg, AP, and 2832, supplied as: NDC Number Size 52244-100-10 bottle of 100 Theo-24 200 mg capsules are orange opaque and clear, with markings Theo-24, 200 mg, AP, and 2842, supplied as: NDC Number Size 52244-200-10 bottle of 100 Theo-24 300 mg capsules are Swedish orange and natural, with markings Theo-24, 300 mg, AP, and 2852, supplied as: NDC Number Size 52244-300-10 bottle of 100 Theo-24 400 mg capsules are pink opaque and natural, with markings Theo-24, 400 mg, AP, and 2902, supplied as: NDC Number Size 52244-400-10 bottle of 100
Medication Information
Indications and Usage
Theophylline is indicated for the treatment of the symptoms and reversible airflow obstruction associated with chronic asthma and other chronic lung diseases, e.g., emphysema and chronic bronchitis.
Contraindications
Theo-24 is contraindicated in patients with a history of hypersensitivity to theophylline or other components in the product.
Adverse Reactions
Adverse reactions associated with theophylline are generally mild when peak serum theophylline concentrations are <20 mcg/mL and mainly consist of transient caffeine-like adverse effects such as nausea, vomiting, headache, and insomnia. When peak serum theophylline concentrations exceed 20 mcg/mL, however, theophylline produces a wide range of adverse reactions including persistent vomiting, cardiac arrhythmias, and intractable seizures which can be lethal (see OVERDOSAGE). The transient caffeine-like adverse reactions occur in about 50% of patients when theophylline therapy is initiated at doses higher than recommended initial doses (e.g., >300 mg/day in adults and >12 mg/kg/day in children beyond 1 year of age). During the initiation of theophylline therapy, caffeine-like adverse effects may transiently alter patient behavior, especially in school age children, but this response rarely persists. Initiation of theophylline therapy at a low dose with subsequent slow titration to a predetermined age-related maximum dose will significantly reduce the frequency of these transient adverse effects (see DOSAGE AND ADMINISTRATION, Table V). In a small percentage of patients (<3% of children and <10% of adults) the caffeine-like adverse effects persist during maintenance therapy, even at peak serum theophylline concentrations within the therapeutic range (i.e., 10 - 20 mcg/mL). Dosage reduction may alleviate the caffeine-like adverse effects in these patients, however, persistent adverse effects should result in a reevaluation of the need for continued theophylline therapy and the potential therapeutic benefit of alternative treatment.
Other adverse reactions that have been reported at serum theophylline concentrations <20 mcg/mL include diarrhea, irritability, restlessness, fine skeletal muscle tremors, and transient diuresis. In patients with hypoxia secondary to COPD, multifocal atrial tachycardia and flutter have been reported at serum theophylline concentrations ≥15 mcg/mL. There have been a few isolated reports of seizures at serum theophylline concentrations <20 mcg/mL in patients with an underlying neurological disease or in elderly patients. The occurrence of seizures in elderly patients with serum theophylline concentrations <20 mcg/mL may be secondary to decreased protein binding resulting in a larger proportion of the total serum theophylline concentration in the pharmacologically active unbound form. The clinical characteristics of the seizures reported in patients with serum theophylline concentrations <20 mcg/mL have generally been milder than seizures associated with excessive serum theophylline concentrations resulting from an overdose (i.e., they have generally been transient, often stopped without anticonvulsant therapy, and did not result in neurological residua).
| Percentage of patients reported
with sign or symptom |
||||
|---|---|---|---|---|
| Acute Overdose
(Large Single Ingestion) |
Chronic Overdosage
(Multiple Excessive Doses) |
|||
| Sign/Symptom | Study 1
(n=157) |
Study 2
(n=14) |
Study 1
(n=92) |
Study 2
(n=102) |
| * These data are derived from two studies in patients with serum theophylline concentrations >30 mcg/mL. In
the first study (Study #1—Shanon, Ann Intern Med 1993;119:1161-67), data were prospectively collected from 249 consecutive cases of theophylline toxicity referred to a regional poison center for consultation. In the second study (Study #2—Sessler, Am J Med 1990;88:567-76), data were retrospectively collected from 116 cases with serum theophylline concentrations >30 mcg/mL among 6000 blood samples obtained for measurement of serum theophylline concentrations in three emergency departments. Differences in the incidence of manifestations of theophylline toxicity between the two studies may reflect sample selection as a result of study design (e.g., in Study #1, 48% of the patients had acute intoxications versus only 10 in Study #2) and different methods of reporting results. |
||||
| † NR =Not reported in a comparable manner. | ||||
| Asymptomatic | NR† | 0 | NR† | 6 |
| Gastrointestinal | ||||
| Vomiting | 73 | 93 | 30 | 61 |
| Abdominal pain | NR† | 21 | NR† | 12 |
| Diarrhea | NR† | 0 | NR† | 14 |
| Hematemesis | NR† | 0 | NR† | 2 |
| Metabolic/Other | ||||
| Hypokalemia | 85 | 79 | 44 | 43 |
| Hyperglycemia | 98 | NR† | 18 | NR† |
| Acid/base disturbance | 34 | 21 | 9 | 5 |
| Rhabdomyolysis | NR† | 7 | NR† | 0 |
| Cardiovascular | ||||
| Sinus tachycardia | 100 | 86 | 100 | 62 |
| Other supraventricular | ||||
| tachycardias | 2 | 21 | 12 | 14 |
| Ventricular premature beats | 3 | 21 | 10 | 19 |
| Atrial fibrillation or flutter | 1 | NR† | 12 | NR† |
| Multifocal atrial tachycardia | 0 | NR† | 2 | NR† |
| Ventricular arrhythmias with | ||||
| hemodynamic instability | 7 | 14 | 40 | 0 |
| Hypotension/shock | NR† | 21 | NR† | 8 |
| Neurologic | ||||
| Nervousness | NR† | 64 | NR† | 21 |
| Tremors | 38 | 29 | 16 | 14 |
| Disorientation | NR† | 7 | NR† | 11 |
| Seizures | 5 | 14 | 14 | 5 |
| Death | 3 | 21 | 10 | 4 |
How Supplied
Theo-24 (theophylline anhydrous) is supplied in extended-release capsules containing 100, 200, 300 or 400 mg of anhydrous theophylline.
Theo-24 100 mg capsules are orange opaque and natural, with markings Theo-24, 100 mg, AP, and 2832, supplied as:
NDC Number
Size
52244-100-10 bottle of 100
Theo-24 200 mg capsules are orange opaque and clear, with markings Theo-24, 200 mg, AP, and 2842, supplied as:
NDC Number
Size
52244-200-10 bottle of 100
Theo-24 300 mg capsules are Swedish orange and natural, with markings Theo-24, 300 mg, AP, and 2852, supplied as:
NDC Number
Size
52244-300-10 bottle of 100
Theo-24 400 mg capsules are pink opaque and natural, with markings Theo-24, 400 mg, AP, and 2902, supplied as:
NDC Number
Size
52244-400-10 bottle of 100
Description
Rx Only
Section 34072-9
General:
Careful consideration of the various interacting drugs and physiologic conditions that can alter theophylline clearance and require dosage adjustment should occur prior to initiation of theophylline therapy, prior to increases in theophylline dose, and during follow up (see WARNINGS). The dose of theophylline selected for initiation of therapy should be low and,
if tolerated,
increased slowly over a period of a week or longer with the final dose guided by monitoring serum theophylline concentrations and the patient's clinical response (see DOSAGE AND ADMINISTRATION, Table V).
Section 34075-2
Effects on Laboratory Tests:
As a result of its pharmacological effects, theophylline at serum concentrations within the 10 - 20 mcg/mL range modestly increases plasma glucose (from a mean of 88 mg% to 98 mg%), uric acid (from a mean of 4 mg/dL to 6 mg/dL), free fatty acids (from a mean of 451 µEq/L to 800 µEq/L, total cholesterol (from a mean of 140 vs 160 mg/dL), HDL (from a mean of 36 to 50 mg/dL), HDL/LDL ratio (from a mean of 0.5 to 0.7), and urinary free cortisol excretion (from a mean of 44 to 63 mcg/24 hr). Theophylline at serum concentrations within the 10 - 20 mcg/mL range may also transiently decrease serum concentrations of tri-iodothyronine (144 before, 131 after one week and 142 ng/dL after 4 weeks of theophylline). The clinical importance of these changes should be weighed against the potential therapeutic benefit of theophylline in individual patients.
Section 34076-0
Information for Patients:
The patient (or parent/care giver) should be instructed to seek medical advice whenever nausea, vomiting, persistent headache, insomnia or rapid heart beat occurs during treatment with theophylline, even if another cause is suspected. The patient should be instructed to contact their healthcare professional if they develop a new illness, especially if accompanied by a persistent fever, if they experience worsening of a chronic illness, if they start or stop smoking cigarettes or marijuana, or if another healthcare professional adds a new medication or discontinues a previously prescribed medication. Patients should be informed that theophylline interacts with a wide variety of drugs (see Table II). The dietary supplement St. John's Wort (Hypericum perforatum) should not be taken at the same time as theophylline, since it may result in decreased theophylline levels. If patients are already taking St. John's Wort and theophylline together, they should consult their healthcare professional before stopping the St. John's Wort, since their theophylline concentrations may rise when this is done, resulting in toxicity. Patients should be instructed to inform all healthcare professionals involved in their care that they are taking theophylline, especially when a medication is being added or deleted from their treatment. Patients should be instructed to not alter the dose, timing of the dose, or frequency of administration without first consulting their healthcare professional. If a dose is missed, the patient should be instructed to take the next dose at the usually scheduled time and to not attempt to make up for the missed dose.
Patients should be instructed to take this medication each morning at approximately the same time and not to exceed the prescribed dose.
Patients who require a relatively high dose of theophylline should be informed of important considerations relating to time of drug administration and meal content (see PRECAUTIONS, Drug/Food Interactions; and DOSAGE AND ADMINISTRATION).
Section 34077-8
CATEGORY C:
In studies in which pregnant mice, rats and rabbits were dosed during the period of organogenesis, theophylline produced teratogenic effects.
In studies with mice, a single intraperitoneal dose at and above 100 mg/kg (approximately equal to the maximum recommended oral dose for adults on a mg/m2 basis) during organogenesis produced cleft palate and digital abnormalities. Micromelia, micrognathia, clubfoot, subcutaneous hematoma, open eyelids, and embryolethality were observed at doses that are approximately 2 times the maximum recommended oral dose for adults on a mg/m2 basis.
In a study with rats dosed from conception through organogenesis, an oral dose of 150 mg/kg/day (approximately 2 times the maximum recommended oral dose for adults on a mg/m2 basis) produced digital abnormalities. Embryolethality was observed with a subcutaneous dose of 200 mg/kg/day (approximately 4 times the maximum recommended oral dose for adults on a mg/m2 basis).
In a study in which pregnant rabbits were dosed throughout organogenesis, an intravenous dose of 60 mg/kg/day (approximately 2 times the maximum recommended oral dose for adults on a mg/m2 basis), which caused the death of one doe and clinical signs in others, produced cleft palate and was embryolethal. Doses at and above 15 mg/kg/day (less than the maximum recommended oral dose for adults on a mg/m2 basis) increased the incidence of skeletal variations.
There are no adequate and well-controlled studies in pregnant women. Theophylline should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Section 34080-2
Nursing Mothers:
Theophylline is excreted into breast milk and may cause irritability or other signs of mild toxicity in nursing human infants. The concentration of theophylline in breast milk is about equivalent to the maternal serum concentration. An infant ingesting a liter of breast milk containing 10 - 20 mcg/mL of theophylline per day is likely to receive 10 - 20 mg of theophylline per day. Serious adverse effects in the infant are unlikely unless the mother has toxic serum theophylline concentrations.
Section 34081-0
Pediatric Use:
Theophylline is safe and effective for the approved indications in pediatric patients (see INDICATIONS AND USAGE). The maintenance dose of theophylline must be selected with caution in pediatric patients since the rate of theophylline clearance is highly variable across the age range of neonates to adolescents (see CLINICAL PHARMACOLOGY, Table I, WARNINGS, and DOSAGE AND ADMINISTRATION, Table V). Due to the immaturity of theophylline metabolic pathways in infants under the age of one year, particular attention to dosage selection and frequent monitoring of serum theophylline concentrations are required when theophylline is prescribed to pediatric patients in this age group.
Section 34082-8
Geriatric Use:
Elderly patients are at a significantly greater risk of experiencing serious toxicity from theophylline than younger patients due to pharmacokinetic and pharmacodynamic changes associated with aging. The clearance of theophylline is decreased by an average of 30% in healthy elderly adults (>60 yrs) compared to healthy young adults. Theophylline clearance may be further reduced by concomitant diseases prevalent in the elderly, which further impair clearance of this drug and have the potential to increase serum levels and potential toxicity. These conditions include impaired renal function, chronic obstructive pulmonary disease, congestive heart failure, hepatic disease and an increased prevalence of use of certain medications (see PRECAUTIONS: Drug Interactions) with the potential for pharmacokinetic and pharmacodynamic interaction. Protein binding may be decreased in the elderly resulting in an increased proportion of the total serum theophylline concentration in the pharmacologically active unbound form. Elderly patients also appear to be more sensitive to the toxic effects of theophylline after chronic overdosage than younger patients. Careful attention to dose reduction and frequent monitoring of serum theophylline concentrations are required in elderly patients (see PRECAUTIONS, Monitoring Serum Theophylline Concentrations, and DOSAGE AND ADMINISTRATION). The maximum daily dose of theophylline in patients greater than 60 years of age ordinarily should not exceed 400 mg/day unless the patient continues to be symptomatic and the peak steady-state serum theophylline concentration is <10 mcg/mL (see DOSAGE AND ADMINISTRATION). Theophylline doses greater than 400 mg/d should be prescribed with caution in elderly patients.
Section 34083-6
Carcinogenesis, Mutagenesis, and Impairment of Fertility:
Long term carcinogenicity studies have been carried out in mice (oral doses 30 - 150 mg/kg) and rats (oral doses 5 - 75 mg/kg). Results are pending.
Theophylline has been studied in Ames salmonella, in vivo and in vitro cytogenetics, micronucleus and Chinese hamster ovary test systems and has not been shown to be genotoxic.
In a 14 week continuous breeding study, theophylline, administered to mating pairs of B6C3F1 mice at oral doses of 120, 270 and 500 mg/kg (approximately 1.0 - 3.0 times the human dose on a mg/m2 basis) impaired fertility, as evidenced by decreases in the number of live pups per litter, decreases in the mean number of litters per fertile pair, and increases in the gestation period at the high dose as well as decreases in the proportion of pups born alive at the mid and high dose. In 13 week toxicity studies, theophylline was administered to F344 rats and B6C3F1 mice at oral doses of 40 - 300 mg/kg (approximately 2.0 times the human dose on a mg/m2 basis). At the high dose, systemic toxicity was observed in both species including decreases in testicular weight.
Section 34092-7
Clinical Studies:
In patients with chronic asthma, including patients with severe asthma requiring inhaled corticosteroids or alternate-day oral corticosteroids, many clinical studies have shown that theophylline decreases the frequency and severity of symptoms, including nocturnal exacerbations, and decreases the "as needed" use of inhaled beta2 agonists. Theophylline has also been shown to reduce the need for short courses of daily oral prednisone to relieve exacerbations of airway obstruction that are unresponsive to bronchodilators in asthmatics.
In patients with chronic obstructive pulmonary disease (COPD), clinical studies have shown that theophylline decreases dyspnea, air trapping, the work of breathing, and improves contractility of diaphragmatic muscles with little or no improvement in pulmonary function measurements.
Section 42229-5
Rx Only
Section 43679-0
Mechanism of Action:
Theophylline has two distinct actions in the airways of patients with reversible obstruction: smooth muscle relaxation (i.e., bronchodilation) and suppression of the response of the airways to stimuli (i.e., non-bronchodilator prophylactic effects). While the mechanisms of action of theophylline are not known with certainty, studies in animals suggest that bronchodilation is mediated by the inhibition of two isozymes of phosphodiesterase (PDE III and, to a lesser extent, PDE IV) while non-bronchodilator prophylactic actions are probably mediated through one or more different molecular mechanisms that do not involve inhibition of PDE III or antagonism of adenosine receptors. Some of the adverse effects associated with theophylline appear to be mediated by inhibition of PDE III (e.g., hypotension, tachycardia, headache, and emesis) and adenosine receptor antagonism (e.g., alterations in cerebral blood flow).
Theophylline increases the force of contraction of diaphragmatic muscles. This action appears to be due to enhancement of calcium uptake through an adenosine-mediated channel.
Storage
Store below 77° F (25° C).
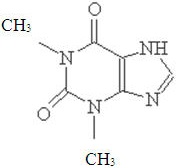
Theophylline
Theophylline is structurally classified as a methylxanthine. It occurs as a white, odorless, crystalline powder with a bitter taste. Anhydrous theophylline has the chemical name 1H-Purine-2, 6-dione, 3, 7-dihydro-1, 3-dimethyl-, and is represented by the following structural formula:
The molecular formula of anhydrous theophylline is C7H8N4O2 with a molecular weight of 180.17.
Theo-24 is available as capsules intended for oral administration, containing 100 mg, 200 mg, 300 mg, or 400 mg of anhydrous theophylline per capsule, in an extended-release formulation which allows a 24-hour dosing interval for appropriate patients.
Inactive ingredients are edible ink (which contains synthetic black iron oxide, FD&C Blue No. 1, FD&C Blue No. 2, FD&C Yellow No. 6, D&C Yellow No. 10, FD&C Red No. 40), ethylcellulose, gelatin, pharmaceutical glaze, colloidal silicon dioxide, starch, sucrose, talc, titanium dioxide, and coloring agents: 100 mg - includes FD&C Yellow No. 6; 200 mg - FD&C Red No. 3 and D&C Yellow No. 10; 300 mg - FD&C Blue No. 1 and FD&C Red No. 40; 400 mg - FD&C Red No. 40 and D&C Red No. 28.
Theo-24 Extended-release capsules meet Drug Release Test 6 as published in the current USP monograph for Theophylline Extended-release Capsules.
Acute Overdose
-
Serum Concentration > 20 < 30 mcg/mL
- Administer a single dose of oral activated charcoal.
- Monitor the patient and obtain a serum theophylline concentration in 2 - 4 hours to insure that the concentration is not increasing.
-
Serum Concentration > 30 < 100 mcg/mL
- Administer multiple dose oral activated charcoal and measures to control emesis.
- Monitor the patient and obtain serial theophylline concentrations every 2 - 4 hours to gauge the effectiveness of therapy and to guide further treatment decisions.
- Institute extracorporeal removal if emesis, seizures, or cardiac arrhythmias cannot be adequately controlled (see OVERDOSAGE, Extracorporeal Removal).
-
Serum Concentration > 100 mcg/mL
- Consider prophylactic anticonvulsant therapy.
- Administer multiple-dose oral activated charcoal and measures to control emesis.
- Consider extracorporeal removal, even if the patient has not experienced a seizure (see OVERDOSAGE, Extracorporeal Removal).
- Monitor the patient and obtain serial theophylline concentrations every 2 - 4 hours to gauge the effectiveness of therapy and to guide further treatment decisions.
Chronic Overdosage
-
Serum Concentration > 20 < 30 mcg/mL (with manifestations of theophylline toxicity)
- Administer a single dose of oral activated charcoal.
- Monitor the patient and obtain a serum theophylline concentration in 2 - 4 hours to insure that the concentration is not increasing.
-
Serum Concentration > 30 mcg/mL in patients < 60 years of age
- Administer multiple-dose oral activated charcoal and measures to control emesis.
- Monitor the patient and obtain serial theophylline concentrations every 2 - 4 hours to gauge the effectiveness of therapy and to guide further treatment decisions.
- Institute extracorporeal removal if emesis, seizures, or cardiac arrhythmias cannot be adequately controlled (see OVERDOSAGE, Extracorporeal Removal).
-
Serum Concentration > 30 mcg/mL in patients ≥ 60 years of age.
- Consider prophylactic anticonvulsant therapy.
- Administer multiple-dose oral activated charcoal and measures to control emesis.
- Consider extracorporeal removal even if the patient has not experienced a seizure (see OVERDOSAGE, Extracorporeal Removal).
- Monitor the patient and obtain serial theophylline concentrations every 2 - 4 hours to gauge the effectiveness of therapy and to guide further treatment decisions.
Extracorporeal Removal:
Increasing the rate of theophylline clearance by extracorporeal methods may rapidly decrease serum concentrations, but the risks of the procedure must be weighed against the potential benefit. Charcoal hemoperfusion is the most effective method of extracorporeal removal, increasing theophylline clearance up to six fold, but serious complications, including hypotension, hypocalcemia, platelet consumption and bleeding diatheses may occur. Hemodialysis is about as efficient as multiple-dose oral activated charcoal and has a lower risk of serious complications than charcoal hemoperfusion. Hemodialysis should be considered as an alternative when charcoal hemoperfusion is not feasible and multiple-dose oral charcoal is ineffective because of intractable emesis. Serum theophylline concentrations may rebound 5 - 10 mcg/mL after discontinuation of charcoal hemoperfusion or hemodialysis due to redistribution of theophylline from the tissue compartment. Peritoneal dialysis is ineffective for theophylline removal; exchange transfusions in neonates have been minimally effective.
For Medical Information
Contact: Endo .
Phone: 1-800-462-3636
Manufactured for:
Endo USA
Malvern, PA 19355
Manufactured by:
Neolpharma, Inc.
Caguas, PR 00725
© 2024 Endo Inc. or one of its affiliates.
Revised 05/2024
General Considerations:
Theo-24, like other extended-release theophylline products, is intended for patients with relatively continuous or recurring symptoms who have a need to maintain therapeutic serum levels of theophylline. It is not intended for patients experiencing an acute episode of bronchospasm (associated with asthma, chronic bronchitis, or emphysema). Such patients require rapid relief of symptoms and should be treated with an immediate-release or intravenous theophylline preparation (or other bronchodilators) and not with extended-release products.
Patients who metabolize theophylline at a normal or slow rate are reasonable candidates for once-daily dosing with Theo-24. Patients who metabolize theophylline rapidly (e.g., the young, smokers, and some nonsmoking adults) and who have symptoms repeatedly at the end of a dosing interval, will require either increased doses given once a day or preferably, are likely to be better controlled by a schedule of twice-daily dosing. Those patients who require increased daily doses are more likely to experience relatively wide peak-trough differences and may be candidates for twice-a-day dosing with Theo-24.
Patients should be instructed to take this medication each morning at approximately the same time and not to exceed the prescribed dose.
Recent studies suggest that dosing of extended-release theophylline products at night (after the evening meal) results in serum concentrations of theophylline which are not identical to those recorded during waking hours and may be characterized by early trough and delayed peak levels. This appears to occur whether the drug is given as an immediate-release, extended-release, or intravenous product. To avoid this phenomenon when two doses per day are prescribed, it is recommended that the second dose be given 10 to 12 hours after the morning dose and before the evening meal.
Food and posture, along with changes associated with circadian rhythm, may influence the rate of absorption and/or clearance rates of theophylline from extended-release dosage forms administered at night. The exact relationship of these and other factors to nighttime serum concentrations and the clinical significance of such findings require additional study. Therefore, it is not recommended that Theo-24 (when used as a once-a-day product) be administered at night.
Patients who require a relatively high dose of theophylline (i.e., a dose equal to or greater than 900 mg or 13 mg/kg, whichever is less) should not take Theo-24 less than 1 hour before a high-fat-content meal since this may result in a significant increase in peak serum level and in the extent of absorption of theophylline as compared to administration in the fasted state (see PRECAUTIONS, Drug/Food Interactions).
The steady-state peak serum theophylline concentration is a function of the dose, the dosing interval, and the rate of theophylline absorption and clearance in the individual patient. Because of marked individual differences in the rate of theophylline clearance, the dose required to achieve a peak serum theophylline concentration in the 10 - 20 mcg/mL range varies fourfold among otherwise similar patients in the absence of factors known to alter theophylline clearance (e.g., 400 - 1600 mg/day in adults <60 years old and 10 - 36 mg/kg/day in children 1 - 9 years old). For a given population there is no single theophylline dose that will provide both safe and effective serum concentrations for all patients. Administration of the median theophylline dose required to achieve a therapeutic serum theophylline concentration in a given population may result in either sub-therapeutic or potentially toxic serum theophylline concentrations in individual patients. For example, at a dose of 900 mg/day in adults <60 years or 22 mg/kg/day in children 1-9 years, the steady-state peak serum theophylline concentration will be <10 mcg/mL in about 30% of patients, 10 - 20 mcg/mL in about 50% and 20 - 30 mcg/mL in about 20% of patients. The dose of theophylline must be individualized on the basis of peak serum theophylline concentration measurements in order to achieve a dose that will provide maximum potential benefit with minimal risk of adverse effects.
Transient caffeine-like adverse effects and excessive serum concentrations in slow metabolizers can be avoided in most patients by starting with a sufficiently low dose and slowly increasing the dose, if judged to be clinically indicated, in small increments (See Table V). Dose increases should only be made if the previous dosage is well tolerated and at intervals of no less than 3 days to allow serum theophylline concentrations to reach the new steady state. Dosage adjustment should be guided by serum theophylline concentration measurement (see PRECAUTIONS, Laboratory Tests and DOSAGE AND ADMINISTRATION, Table VI). Health care providers should instruct patients and care givers to discontinue any dosage that causes adverse effects, to withhold the medication until these symptoms are gone and to then resume therapy at a lower, previously tolerated dosage (see WARNINGS).
If the patient's symptoms are well controlled, there are no apparent adverse effects, and no intervening factors that might alter dosage requirements (see WARNINGS and PRECAUTIONS), serum theophylline concentrations should be monitored at 6 month intervals for rapidly growing children and at yearly intervals for all others. In acutely ill patients, serum theophylline concentrations should be monitored at frequent intervals, e.g., every 24 hours.
Theophylline distributes poorly into body fat, therefore, mg/kg dose should be calculated on the basis of ideal body weight.
Table V contains theophylline dosing titration schema recommended for patients in various age groups and clinical circumstances. Table VI contains recommendations for theophylline dosage adjustment based upon serum theophylline concentrations. Application of these general dosing recommendations to individual patients must take into account the unique clinical characteristics of each patient. In general, these recommendations should serve as the upper limit for dosage adjustments in order to decrease the risk of potentially serious adverse events associated with unexpected large increases in serum theophylline concentration.
| * Patients with more rapid metabolism, clinically identified by higher than average dose
requirements, should receive a smaller dose more frequently to prevent breakthrough symptoms resulting from low trough concentrations before the next dose. A reliably absorbed slow-release formulation will decrease fluctuations and permit longer dosing intervals. |
|||
| A. | Children (12-15 years) and adults (16-60 years) without risk factors for impaired clearance . | ||
| Titration Step | Children <45 kg | Children >45 kg and adults | |
| 1. | Starting Dosage | 12 - 14 mg/kg/day up to a
maximum of 300 mg/day divided Q 24 hrs* |
300 - 400 mg/day1 divided Q
24 hrs* |
| 2. | After 3 days,
if
tolerated , increase dose to: |
16 mg/kg/day up to a
maximum of 400 mg/day divided Q 24 hrs* |
400 - 600 mg/day1 divided Q
24 hrs* |
| 3. | After 3 more days,
if
tolerated and if needed , increase dose to: |
20 mg/kg/day up to a
maximum of 600 mg/day divided Q 24 hrs* |
As with all theophylline
products, doses greater than 600 mg should be titrated according to blood level (see Table VI) |
| 1 | If caffeine-like adverse effects occur, then consideration should be given to a lower
dose and titrating the dose more slowly (see ADVERSE REACTIONS). |
||
| B. |
Patients with risk factors for impaired clearance, the elderly (>60 Years), and
those in whom it is not feasible to monitor serum theophylline concentrations: In children 12-15 years of age, the final theophylline dose should not exceed 16 mg/kg/day up to a maximum of 400 mg/day in the presence of risk factors for reduced theophylline clearance (see WARNINGS) or if it is not feasible to monitor serum theophylline concentrations. In adolescents ≥16 years and adults, including the elderly, the final theophylline dose should not exceed 400 mg/day in the presence of risk factors for reduced theophylline clearance (see WARNINGS) or if it is not feasible to monitor serum theophylline concentrations. |
| Peak Serum
Concentration |
Dosage Adjustment |
|---|---|
| * Dose reduction and/or serum theophylline concentration measurement is indicated whenever adverse effects are present,
physiologic abnormalities that can reduce theophylline clearance occur (e.g., sustained fever), or a drug that interacts with theophylline is added or discontinued (see WARNINGS). |
|
| <9.9 mcg/mL | If symptoms are not controlled and current dosage is tolerated, increase dose about 25%.
Recheck serum concentration after three days for further dosage adjustment. |
| 10 - 14.9 mcg/mL | If symptoms are controlled and current dosage is tolerated, maintain dose and recheck serum
concentration at 6-12 month intervals.* If symptoms are not controlled and current dosage is tolerated consider adding additional medication(s) to treatment regimen. |
| 15 - 19.9 mcg/mL | Consider 10% decrease in dose to provide greater margin of safety even if current dosage is
tolerated. * |
| 20 - 24.9 mcg/mL | Decrease dose by 25% even if no adverse effects are present. Recheck serum concentration after
3 days to guide further dosage adjustment. |
| 25 - 30 mcg/mL | Skip next dose and decrease subsequent doses at least 25% even if no adverse effects are present.
Recheck serum concentration after 3 days to guide further dosage adjustment. If symptomatic, consider whether overdosage treatment is indicated (see recommendations for chronic overdosage). |
| >30 mcg/mL | Treat overdose as indicated (see recommendations for chronic overdosage). If theophylline is
subsequently resumed, decrease dose by at least 50% and recheck serum concentration after 3 days to guide further dosage adjustment. |
Principal Display Panel 100 Mg Bottle Label
Principal Display Panel 200 Mg Bottle Label
Principal Display Panel 300 Mg Bottle Label
Principal Display Panel 400 Mg Bottle Label
Structured Label Content
Section 34072-9 (34072-9)
General:
Careful consideration of the various interacting drugs and physiologic conditions that can alter theophylline clearance and require dosage adjustment should occur prior to initiation of theophylline therapy, prior to increases in theophylline dose, and during follow up (see WARNINGS). The dose of theophylline selected for initiation of therapy should be low and,
if tolerated,
increased slowly over a period of a week or longer with the final dose guided by monitoring serum theophylline concentrations and the patient's clinical response (see DOSAGE AND ADMINISTRATION, Table V).
Section 34075-2 (34075-2)
Effects on Laboratory Tests:
As a result of its pharmacological effects, theophylline at serum concentrations within the 10 - 20 mcg/mL range modestly increases plasma glucose (from a mean of 88 mg% to 98 mg%), uric acid (from a mean of 4 mg/dL to 6 mg/dL), free fatty acids (from a mean of 451 µEq/L to 800 µEq/L, total cholesterol (from a mean of 140 vs 160 mg/dL), HDL (from a mean of 36 to 50 mg/dL), HDL/LDL ratio (from a mean of 0.5 to 0.7), and urinary free cortisol excretion (from a mean of 44 to 63 mcg/24 hr). Theophylline at serum concentrations within the 10 - 20 mcg/mL range may also transiently decrease serum concentrations of tri-iodothyronine (144 before, 131 after one week and 142 ng/dL after 4 weeks of theophylline). The clinical importance of these changes should be weighed against the potential therapeutic benefit of theophylline in individual patients.
Section 34076-0 (34076-0)
Information for Patients:
The patient (or parent/care giver) should be instructed to seek medical advice whenever nausea, vomiting, persistent headache, insomnia or rapid heart beat occurs during treatment with theophylline, even if another cause is suspected. The patient should be instructed to contact their healthcare professional if they develop a new illness, especially if accompanied by a persistent fever, if they experience worsening of a chronic illness, if they start or stop smoking cigarettes or marijuana, or if another healthcare professional adds a new medication or discontinues a previously prescribed medication. Patients should be informed that theophylline interacts with a wide variety of drugs (see Table II). The dietary supplement St. John's Wort (Hypericum perforatum) should not be taken at the same time as theophylline, since it may result in decreased theophylline levels. If patients are already taking St. John's Wort and theophylline together, they should consult their healthcare professional before stopping the St. John's Wort, since their theophylline concentrations may rise when this is done, resulting in toxicity. Patients should be instructed to inform all healthcare professionals involved in their care that they are taking theophylline, especially when a medication is being added or deleted from their treatment. Patients should be instructed to not alter the dose, timing of the dose, or frequency of administration without first consulting their healthcare professional. If a dose is missed, the patient should be instructed to take the next dose at the usually scheduled time and to not attempt to make up for the missed dose.
Patients should be instructed to take this medication each morning at approximately the same time and not to exceed the prescribed dose.
Patients who require a relatively high dose of theophylline should be informed of important considerations relating to time of drug administration and meal content (see PRECAUTIONS, Drug/Food Interactions; and DOSAGE AND ADMINISTRATION).
Section 34077-8 (34077-8)
CATEGORY C:
In studies in which pregnant mice, rats and rabbits were dosed during the period of organogenesis, theophylline produced teratogenic effects.
In studies with mice, a single intraperitoneal dose at and above 100 mg/kg (approximately equal to the maximum recommended oral dose for adults on a mg/m2 basis) during organogenesis produced cleft palate and digital abnormalities. Micromelia, micrognathia, clubfoot, subcutaneous hematoma, open eyelids, and embryolethality were observed at doses that are approximately 2 times the maximum recommended oral dose for adults on a mg/m2 basis.
In a study with rats dosed from conception through organogenesis, an oral dose of 150 mg/kg/day (approximately 2 times the maximum recommended oral dose for adults on a mg/m2 basis) produced digital abnormalities. Embryolethality was observed with a subcutaneous dose of 200 mg/kg/day (approximately 4 times the maximum recommended oral dose for adults on a mg/m2 basis).
In a study in which pregnant rabbits were dosed throughout organogenesis, an intravenous dose of 60 mg/kg/day (approximately 2 times the maximum recommended oral dose for adults on a mg/m2 basis), which caused the death of one doe and clinical signs in others, produced cleft palate and was embryolethal. Doses at and above 15 mg/kg/day (less than the maximum recommended oral dose for adults on a mg/m2 basis) increased the incidence of skeletal variations.
There are no adequate and well-controlled studies in pregnant women. Theophylline should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Section 34080-2 (34080-2)
Nursing Mothers:
Theophylline is excreted into breast milk and may cause irritability or other signs of mild toxicity in nursing human infants. The concentration of theophylline in breast milk is about equivalent to the maternal serum concentration. An infant ingesting a liter of breast milk containing 10 - 20 mcg/mL of theophylline per day is likely to receive 10 - 20 mg of theophylline per day. Serious adverse effects in the infant are unlikely unless the mother has toxic serum theophylline concentrations.
Section 34081-0 (34081-0)
Pediatric Use:
Theophylline is safe and effective for the approved indications in pediatric patients (see INDICATIONS AND USAGE). The maintenance dose of theophylline must be selected with caution in pediatric patients since the rate of theophylline clearance is highly variable across the age range of neonates to adolescents (see CLINICAL PHARMACOLOGY, Table I, WARNINGS, and DOSAGE AND ADMINISTRATION, Table V). Due to the immaturity of theophylline metabolic pathways in infants under the age of one year, particular attention to dosage selection and frequent monitoring of serum theophylline concentrations are required when theophylline is prescribed to pediatric patients in this age group.
Section 34082-8 (34082-8)
Geriatric Use:
Elderly patients are at a significantly greater risk of experiencing serious toxicity from theophylline than younger patients due to pharmacokinetic and pharmacodynamic changes associated with aging. The clearance of theophylline is decreased by an average of 30% in healthy elderly adults (>60 yrs) compared to healthy young adults. Theophylline clearance may be further reduced by concomitant diseases prevalent in the elderly, which further impair clearance of this drug and have the potential to increase serum levels and potential toxicity. These conditions include impaired renal function, chronic obstructive pulmonary disease, congestive heart failure, hepatic disease and an increased prevalence of use of certain medications (see PRECAUTIONS: Drug Interactions) with the potential for pharmacokinetic and pharmacodynamic interaction. Protein binding may be decreased in the elderly resulting in an increased proportion of the total serum theophylline concentration in the pharmacologically active unbound form. Elderly patients also appear to be more sensitive to the toxic effects of theophylline after chronic overdosage than younger patients. Careful attention to dose reduction and frequent monitoring of serum theophylline concentrations are required in elderly patients (see PRECAUTIONS, Monitoring Serum Theophylline Concentrations, and DOSAGE AND ADMINISTRATION). The maximum daily dose of theophylline in patients greater than 60 years of age ordinarily should not exceed 400 mg/day unless the patient continues to be symptomatic and the peak steady-state serum theophylline concentration is <10 mcg/mL (see DOSAGE AND ADMINISTRATION). Theophylline doses greater than 400 mg/d should be prescribed with caution in elderly patients.
Section 34083-6 (34083-6)
Carcinogenesis, Mutagenesis, and Impairment of Fertility:
Long term carcinogenicity studies have been carried out in mice (oral doses 30 - 150 mg/kg) and rats (oral doses 5 - 75 mg/kg). Results are pending.
Theophylline has been studied in Ames salmonella, in vivo and in vitro cytogenetics, micronucleus and Chinese hamster ovary test systems and has not been shown to be genotoxic.
In a 14 week continuous breeding study, theophylline, administered to mating pairs of B6C3F1 mice at oral doses of 120, 270 and 500 mg/kg (approximately 1.0 - 3.0 times the human dose on a mg/m2 basis) impaired fertility, as evidenced by decreases in the number of live pups per litter, decreases in the mean number of litters per fertile pair, and increases in the gestation period at the high dose as well as decreases in the proportion of pups born alive at the mid and high dose. In 13 week toxicity studies, theophylline was administered to F344 rats and B6C3F1 mice at oral doses of 40 - 300 mg/kg (approximately 2.0 times the human dose on a mg/m2 basis). At the high dose, systemic toxicity was observed in both species including decreases in testicular weight.
Section 34092-7 (34092-7)
Clinical Studies:
In patients with chronic asthma, including patients with severe asthma requiring inhaled corticosteroids or alternate-day oral corticosteroids, many clinical studies have shown that theophylline decreases the frequency and severity of symptoms, including nocturnal exacerbations, and decreases the "as needed" use of inhaled beta2 agonists. Theophylline has also been shown to reduce the need for short courses of daily oral prednisone to relieve exacerbations of airway obstruction that are unresponsive to bronchodilators in asthmatics.
In patients with chronic obstructive pulmonary disease (COPD), clinical studies have shown that theophylline decreases dyspnea, air trapping, the work of breathing, and improves contractility of diaphragmatic muscles with little or no improvement in pulmonary function measurements.
Section 42229-5 (42229-5)
Rx Only
Section 43679-0 (43679-0)
Mechanism of Action:
Theophylline has two distinct actions in the airways of patients with reversible obstruction: smooth muscle relaxation (i.e., bronchodilation) and suppression of the response of the airways to stimuli (i.e., non-bronchodilator prophylactic effects). While the mechanisms of action of theophylline are not known with certainty, studies in animals suggest that bronchodilation is mediated by the inhibition of two isozymes of phosphodiesterase (PDE III and, to a lesser extent, PDE IV) while non-bronchodilator prophylactic actions are probably mediated through one or more different molecular mechanisms that do not involve inhibition of PDE III or antagonism of adenosine receptors. Some of the adverse effects associated with theophylline appear to be mediated by inhibition of PDE III (e.g., hypotension, tachycardia, headache, and emesis) and adenosine receptor antagonism (e.g., alterations in cerebral blood flow).
Theophylline increases the force of contraction of diaphragmatic muscles. This action appears to be due to enhancement of calcium uptake through an adenosine-mediated channel.
Storage (STORAGE)
Store below 77° F (25° C).
How Supplied (HOW SUPPLIED)
Theo-24 (theophylline anhydrous) is supplied in extended-release capsules containing 100, 200, 300 or 400 mg of anhydrous theophylline.
Theo-24 100 mg capsules are orange opaque and natural, with markings Theo-24, 100 mg, AP, and 2832, supplied as:
NDC Number
Size
52244-100-10 bottle of 100
Theo-24 200 mg capsules are orange opaque and clear, with markings Theo-24, 200 mg, AP, and 2842, supplied as:
NDC Number
Size
52244-200-10 bottle of 100
Theo-24 300 mg capsules are Swedish orange and natural, with markings Theo-24, 300 mg, AP, and 2852, supplied as:
NDC Number
Size
52244-300-10 bottle of 100
Theo-24 400 mg capsules are pink opaque and natural, with markings Theo-24, 400 mg, AP, and 2902, supplied as:
NDC Number
Size
52244-400-10 bottle of 100
Theophylline
Theophylline is structurally classified as a methylxanthine. It occurs as a white, odorless, crystalline powder with a bitter taste. Anhydrous theophylline has the chemical name 1H-Purine-2, 6-dione, 3, 7-dihydro-1, 3-dimethyl-, and is represented by the following structural formula:
The molecular formula of anhydrous theophylline is C7H8N4O2 with a molecular weight of 180.17.
Theo-24 is available as capsules intended for oral administration, containing 100 mg, 200 mg, 300 mg, or 400 mg of anhydrous theophylline per capsule, in an extended-release formulation which allows a 24-hour dosing interval for appropriate patients.
Inactive ingredients are edible ink (which contains synthetic black iron oxide, FD&C Blue No. 1, FD&C Blue No. 2, FD&C Yellow No. 6, D&C Yellow No. 10, FD&C Red No. 40), ethylcellulose, gelatin, pharmaceutical glaze, colloidal silicon dioxide, starch, sucrose, talc, titanium dioxide, and coloring agents: 100 mg - includes FD&C Yellow No. 6; 200 mg - FD&C Red No. 3 and D&C Yellow No. 10; 300 mg - FD&C Blue No. 1 and FD&C Red No. 40; 400 mg - FD&C Red No. 40 and D&C Red No. 28.
Theo-24 Extended-release capsules meet Drug Release Test 6 as published in the current USP monograph for Theophylline Extended-release Capsules.
Acute Overdose
-
Serum Concentration > 20 < 30 mcg/mL
- Administer a single dose of oral activated charcoal.
- Monitor the patient and obtain a serum theophylline concentration in 2 - 4 hours to insure that the concentration is not increasing.
-
Serum Concentration > 30 < 100 mcg/mL
- Administer multiple dose oral activated charcoal and measures to control emesis.
- Monitor the patient and obtain serial theophylline concentrations every 2 - 4 hours to gauge the effectiveness of therapy and to guide further treatment decisions.
- Institute extracorporeal removal if emesis, seizures, or cardiac arrhythmias cannot be adequately controlled (see OVERDOSAGE, Extracorporeal Removal).
-
Serum Concentration > 100 mcg/mL
- Consider prophylactic anticonvulsant therapy.
- Administer multiple-dose oral activated charcoal and measures to control emesis.
- Consider extracorporeal removal, even if the patient has not experienced a seizure (see OVERDOSAGE, Extracorporeal Removal).
- Monitor the patient and obtain serial theophylline concentrations every 2 - 4 hours to gauge the effectiveness of therapy and to guide further treatment decisions.
Adverse Reactions (ADVERSE REACTIONS)
Adverse reactions associated with theophylline are generally mild when peak serum theophylline concentrations are <20 mcg/mL and mainly consist of transient caffeine-like adverse effects such as nausea, vomiting, headache, and insomnia. When peak serum theophylline concentrations exceed 20 mcg/mL, however, theophylline produces a wide range of adverse reactions including persistent vomiting, cardiac arrhythmias, and intractable seizures which can be lethal (see OVERDOSAGE). The transient caffeine-like adverse reactions occur in about 50% of patients when theophylline therapy is initiated at doses higher than recommended initial doses (e.g., >300 mg/day in adults and >12 mg/kg/day in children beyond 1 year of age). During the initiation of theophylline therapy, caffeine-like adverse effects may transiently alter patient behavior, especially in school age children, but this response rarely persists. Initiation of theophylline therapy at a low dose with subsequent slow titration to a predetermined age-related maximum dose will significantly reduce the frequency of these transient adverse effects (see DOSAGE AND ADMINISTRATION, Table V). In a small percentage of patients (<3% of children and <10% of adults) the caffeine-like adverse effects persist during maintenance therapy, even at peak serum theophylline concentrations within the therapeutic range (i.e., 10 - 20 mcg/mL). Dosage reduction may alleviate the caffeine-like adverse effects in these patients, however, persistent adverse effects should result in a reevaluation of the need for continued theophylline therapy and the potential therapeutic benefit of alternative treatment.
Other adverse reactions that have been reported at serum theophylline concentrations <20 mcg/mL include diarrhea, irritability, restlessness, fine skeletal muscle tremors, and transient diuresis. In patients with hypoxia secondary to COPD, multifocal atrial tachycardia and flutter have been reported at serum theophylline concentrations ≥15 mcg/mL. There have been a few isolated reports of seizures at serum theophylline concentrations <20 mcg/mL in patients with an underlying neurological disease or in elderly patients. The occurrence of seizures in elderly patients with serum theophylline concentrations <20 mcg/mL may be secondary to decreased protein binding resulting in a larger proportion of the total serum theophylline concentration in the pharmacologically active unbound form. The clinical characteristics of the seizures reported in patients with serum theophylline concentrations <20 mcg/mL have generally been milder than seizures associated with excessive serum theophylline concentrations resulting from an overdose (i.e., they have generally been transient, often stopped without anticonvulsant therapy, and did not result in neurological residua).
| Percentage of patients reported
with sign or symptom |
||||
|---|---|---|---|---|
| Acute Overdose
(Large Single Ingestion) |
Chronic Overdosage
(Multiple Excessive Doses) |
|||
| Sign/Symptom | Study 1
(n=157) |
Study 2
(n=14) |
Study 1
(n=92) |
Study 2
(n=102) |
| * These data are derived from two studies in patients with serum theophylline concentrations >30 mcg/mL. In
the first study (Study #1—Shanon, Ann Intern Med 1993;119:1161-67), data were prospectively collected from 249 consecutive cases of theophylline toxicity referred to a regional poison center for consultation. In the second study (Study #2—Sessler, Am J Med 1990;88:567-76), data were retrospectively collected from 116 cases with serum theophylline concentrations >30 mcg/mL among 6000 blood samples obtained for measurement of serum theophylline concentrations in three emergency departments. Differences in the incidence of manifestations of theophylline toxicity between the two studies may reflect sample selection as a result of study design (e.g., in Study #1, 48% of the patients had acute intoxications versus only 10 in Study #2) and different methods of reporting results. |
||||
| † NR =Not reported in a comparable manner. | ||||
| Asymptomatic | NR† | 0 | NR† | 6 |
| Gastrointestinal | ||||
| Vomiting | 73 | 93 | 30 | 61 |
| Abdominal pain | NR† | 21 | NR† | 12 |
| Diarrhea | NR† | 0 | NR† | 14 |
| Hematemesis | NR† | 0 | NR† | 2 |
| Metabolic/Other | ||||
| Hypokalemia | 85 | 79 | 44 | 43 |
| Hyperglycemia | 98 | NR† | 18 | NR† |
| Acid/base disturbance | 34 | 21 | 9 | 5 |
| Rhabdomyolysis | NR† | 7 | NR† | 0 |
| Cardiovascular | ||||
| Sinus tachycardia | 100 | 86 | 100 | 62 |
| Other supraventricular | ||||
| tachycardias | 2 | 21 | 12 | 14 |
| Ventricular premature beats | 3 | 21 | 10 | 19 |
| Atrial fibrillation or flutter | 1 | NR† | 12 | NR† |
| Multifocal atrial tachycardia | 0 | NR† | 2 | NR† |
| Ventricular arrhythmias with | ||||
| hemodynamic instability | 7 | 14 | 40 | 0 |
| Hypotension/shock | NR† | 21 | NR† | 8 |
| Neurologic | ||||
| Nervousness | NR† | 64 | NR† | 21 |
| Tremors | 38 | 29 | 16 | 14 |
| Disorientation | NR† | 7 | NR† | 11 |
| Seizures | 5 | 14 | 14 | 5 |
| Death | 3 | 21 | 10 | 4 |
Contraindications (CONTRAINDICATIONS)
Theo-24 is contraindicated in patients with a history of hypersensitivity to theophylline or other components in the product.
Chronic Overdosage
-
Serum Concentration > 20 < 30 mcg/mL (with manifestations of theophylline toxicity)
- Administer a single dose of oral activated charcoal.
- Monitor the patient and obtain a serum theophylline concentration in 2 - 4 hours to insure that the concentration is not increasing.
-
Serum Concentration > 30 mcg/mL in patients < 60 years of age
- Administer multiple-dose oral activated charcoal and measures to control emesis.
- Monitor the patient and obtain serial theophylline concentrations every 2 - 4 hours to gauge the effectiveness of therapy and to guide further treatment decisions.
- Institute extracorporeal removal if emesis, seizures, or cardiac arrhythmias cannot be adequately controlled (see OVERDOSAGE, Extracorporeal Removal).
-
Serum Concentration > 30 mcg/mL in patients ≥ 60 years of age.
- Consider prophylactic anticonvulsant therapy.
- Administer multiple-dose oral activated charcoal and measures to control emesis.
- Consider extracorporeal removal even if the patient has not experienced a seizure (see OVERDOSAGE, Extracorporeal Removal).
- Monitor the patient and obtain serial theophylline concentrations every 2 - 4 hours to gauge the effectiveness of therapy and to guide further treatment decisions.
Indications and Usage (INDICATIONS AND USAGE)
Theophylline is indicated for the treatment of the symptoms and reversible airflow obstruction associated with chronic asthma and other chronic lung diseases, e.g., emphysema and chronic bronchitis.
Extracorporeal Removal:
Increasing the rate of theophylline clearance by extracorporeal methods may rapidly decrease serum concentrations, but the risks of the procedure must be weighed against the potential benefit. Charcoal hemoperfusion is the most effective method of extracorporeal removal, increasing theophylline clearance up to six fold, but serious complications, including hypotension, hypocalcemia, platelet consumption and bleeding diatheses may occur. Hemodialysis is about as efficient as multiple-dose oral activated charcoal and has a lower risk of serious complications than charcoal hemoperfusion. Hemodialysis should be considered as an alternative when charcoal hemoperfusion is not feasible and multiple-dose oral charcoal is ineffective because of intractable emesis. Serum theophylline concentrations may rebound 5 - 10 mcg/mL after discontinuation of charcoal hemoperfusion or hemodialysis due to redistribution of theophylline from the tissue compartment. Peritoneal dialysis is ineffective for theophylline removal; exchange transfusions in neonates have been minimally effective.
For Medical Information (FOR MEDICAL INFORMATION)
Contact: Endo .
Phone: 1-800-462-3636
Manufactured for:
Endo USA
Malvern, PA 19355
Manufactured by:
Neolpharma, Inc.
Caguas, PR 00725
© 2024 Endo Inc. or one of its affiliates.
Revised 05/2024
General Considerations:
Theo-24, like other extended-release theophylline products, is intended for patients with relatively continuous or recurring symptoms who have a need to maintain therapeutic serum levels of theophylline. It is not intended for patients experiencing an acute episode of bronchospasm (associated with asthma, chronic bronchitis, or emphysema). Such patients require rapid relief of symptoms and should be treated with an immediate-release or intravenous theophylline preparation (or other bronchodilators) and not with extended-release products.
Patients who metabolize theophylline at a normal or slow rate are reasonable candidates for once-daily dosing with Theo-24. Patients who metabolize theophylline rapidly (e.g., the young, smokers, and some nonsmoking adults) and who have symptoms repeatedly at the end of a dosing interval, will require either increased doses given once a day or preferably, are likely to be better controlled by a schedule of twice-daily dosing. Those patients who require increased daily doses are more likely to experience relatively wide peak-trough differences and may be candidates for twice-a-day dosing with Theo-24.
Patients should be instructed to take this medication each morning at approximately the same time and not to exceed the prescribed dose.
Recent studies suggest that dosing of extended-release theophylline products at night (after the evening meal) results in serum concentrations of theophylline which are not identical to those recorded during waking hours and may be characterized by early trough and delayed peak levels. This appears to occur whether the drug is given as an immediate-release, extended-release, or intravenous product. To avoid this phenomenon when two doses per day are prescribed, it is recommended that the second dose be given 10 to 12 hours after the morning dose and before the evening meal.
Food and posture, along with changes associated with circadian rhythm, may influence the rate of absorption and/or clearance rates of theophylline from extended-release dosage forms administered at night. The exact relationship of these and other factors to nighttime serum concentrations and the clinical significance of such findings require additional study. Therefore, it is not recommended that Theo-24 (when used as a once-a-day product) be administered at night.
Patients who require a relatively high dose of theophylline (i.e., a dose equal to or greater than 900 mg or 13 mg/kg, whichever is less) should not take Theo-24 less than 1 hour before a high-fat-content meal since this may result in a significant increase in peak serum level and in the extent of absorption of theophylline as compared to administration in the fasted state (see PRECAUTIONS, Drug/Food Interactions).
The steady-state peak serum theophylline concentration is a function of the dose, the dosing interval, and the rate of theophylline absorption and clearance in the individual patient. Because of marked individual differences in the rate of theophylline clearance, the dose required to achieve a peak serum theophylline concentration in the 10 - 20 mcg/mL range varies fourfold among otherwise similar patients in the absence of factors known to alter theophylline clearance (e.g., 400 - 1600 mg/day in adults <60 years old and 10 - 36 mg/kg/day in children 1 - 9 years old). For a given population there is no single theophylline dose that will provide both safe and effective serum concentrations for all patients. Administration of the median theophylline dose required to achieve a therapeutic serum theophylline concentration in a given population may result in either sub-therapeutic or potentially toxic serum theophylline concentrations in individual patients. For example, at a dose of 900 mg/day in adults <60 years or 22 mg/kg/day in children 1-9 years, the steady-state peak serum theophylline concentration will be <10 mcg/mL in about 30% of patients, 10 - 20 mcg/mL in about 50% and 20 - 30 mcg/mL in about 20% of patients. The dose of theophylline must be individualized on the basis of peak serum theophylline concentration measurements in order to achieve a dose that will provide maximum potential benefit with minimal risk of adverse effects.
Transient caffeine-like adverse effects and excessive serum concentrations in slow metabolizers can be avoided in most patients by starting with a sufficiently low dose and slowly increasing the dose, if judged to be clinically indicated, in small increments (See Table V). Dose increases should only be made if the previous dosage is well tolerated and at intervals of no less than 3 days to allow serum theophylline concentrations to reach the new steady state. Dosage adjustment should be guided by serum theophylline concentration measurement (see PRECAUTIONS, Laboratory Tests and DOSAGE AND ADMINISTRATION, Table VI). Health care providers should instruct patients and care givers to discontinue any dosage that causes adverse effects, to withhold the medication until these symptoms are gone and to then resume therapy at a lower, previously tolerated dosage (see WARNINGS).
If the patient's symptoms are well controlled, there are no apparent adverse effects, and no intervening factors that might alter dosage requirements (see WARNINGS and PRECAUTIONS), serum theophylline concentrations should be monitored at 6 month intervals for rapidly growing children and at yearly intervals for all others. In acutely ill patients, serum theophylline concentrations should be monitored at frequent intervals, e.g., every 24 hours.
Theophylline distributes poorly into body fat, therefore, mg/kg dose should be calculated on the basis of ideal body weight.
Table V contains theophylline dosing titration schema recommended for patients in various age groups and clinical circumstances. Table VI contains recommendations for theophylline dosage adjustment based upon serum theophylline concentrations. Application of these general dosing recommendations to individual patients must take into account the unique clinical characteristics of each patient. In general, these recommendations should serve as the upper limit for dosage adjustments in order to decrease the risk of potentially serious adverse events associated with unexpected large increases in serum theophylline concentration.
| * Patients with more rapid metabolism, clinically identified by higher than average dose
requirements, should receive a smaller dose more frequently to prevent breakthrough symptoms resulting from low trough concentrations before the next dose. A reliably absorbed slow-release formulation will decrease fluctuations and permit longer dosing intervals. |
|||
| A. | Children (12-15 years) and adults (16-60 years) without risk factors for impaired clearance . | ||
| Titration Step | Children <45 kg | Children >45 kg and adults | |
| 1. | Starting Dosage | 12 - 14 mg/kg/day up to a
maximum of 300 mg/day divided Q 24 hrs* |
300 - 400 mg/day1 divided Q
24 hrs* |
| 2. | After 3 days,
if
tolerated , increase dose to: |
16 mg/kg/day up to a
maximum of 400 mg/day divided Q 24 hrs* |
400 - 600 mg/day1 divided Q
24 hrs* |
| 3. | After 3 more days,
if
tolerated and if needed , increase dose to: |
20 mg/kg/day up to a
maximum of 600 mg/day divided Q 24 hrs* |
As with all theophylline
products, doses greater than 600 mg should be titrated according to blood level (see Table VI) |
| 1 | If caffeine-like adverse effects occur, then consideration should be given to a lower
dose and titrating the dose more slowly (see ADVERSE REACTIONS). |
||
| B. |
Patients with risk factors for impaired clearance, the elderly (>60 Years), and
those in whom it is not feasible to monitor serum theophylline concentrations: In children 12-15 years of age, the final theophylline dose should not exceed 16 mg/kg/day up to a maximum of 400 mg/day in the presence of risk factors for reduced theophylline clearance (see WARNINGS) or if it is not feasible to monitor serum theophylline concentrations. In adolescents ≥16 years and adults, including the elderly, the final theophylline dose should not exceed 400 mg/day in the presence of risk factors for reduced theophylline clearance (see WARNINGS) or if it is not feasible to monitor serum theophylline concentrations. |
| Peak Serum
Concentration |
Dosage Adjustment |
|---|---|
| * Dose reduction and/or serum theophylline concentration measurement is indicated whenever adverse effects are present,
physiologic abnormalities that can reduce theophylline clearance occur (e.g., sustained fever), or a drug that interacts with theophylline is added or discontinued (see WARNINGS). |
|
| <9.9 mcg/mL | If symptoms are not controlled and current dosage is tolerated, increase dose about 25%.
Recheck serum concentration after three days for further dosage adjustment. |
| 10 - 14.9 mcg/mL | If symptoms are controlled and current dosage is tolerated, maintain dose and recheck serum
concentration at 6-12 month intervals.* If symptoms are not controlled and current dosage is tolerated consider adding additional medication(s) to treatment regimen. |
| 15 - 19.9 mcg/mL | Consider 10% decrease in dose to provide greater margin of safety even if current dosage is
tolerated. * |
| 20 - 24.9 mcg/mL | Decrease dose by 25% even if no adverse effects are present. Recheck serum concentration after
3 days to guide further dosage adjustment. |
| 25 - 30 mcg/mL | Skip next dose and decrease subsequent doses at least 25% even if no adverse effects are present.
Recheck serum concentration after 3 days to guide further dosage adjustment. If symptomatic, consider whether overdosage treatment is indicated (see recommendations for chronic overdosage). |
| >30 mcg/mL | Treat overdose as indicated (see recommendations for chronic overdosage). If theophylline is
subsequently resumed, decrease dose by at least 50% and recheck serum concentration after 3 days to guide further dosage adjustment. |
Principal Display Panel 100 Mg Bottle Label (PRINCIPAL DISPLAY PANEL - 100 mg Bottle Label)
Principal Display Panel 200 Mg Bottle Label (PRINCIPAL DISPLAY PANEL - 200 mg Bottle Label)
Principal Display Panel 300 Mg Bottle Label (PRINCIPAL DISPLAY PANEL - 300 mg Bottle Label)
Principal Display Panel 400 Mg Bottle Label (PRINCIPAL DISPLAY PANEL - 400 mg Bottle Label)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:45.928504 · Updated: 2026-03-14T22:06:44.115888