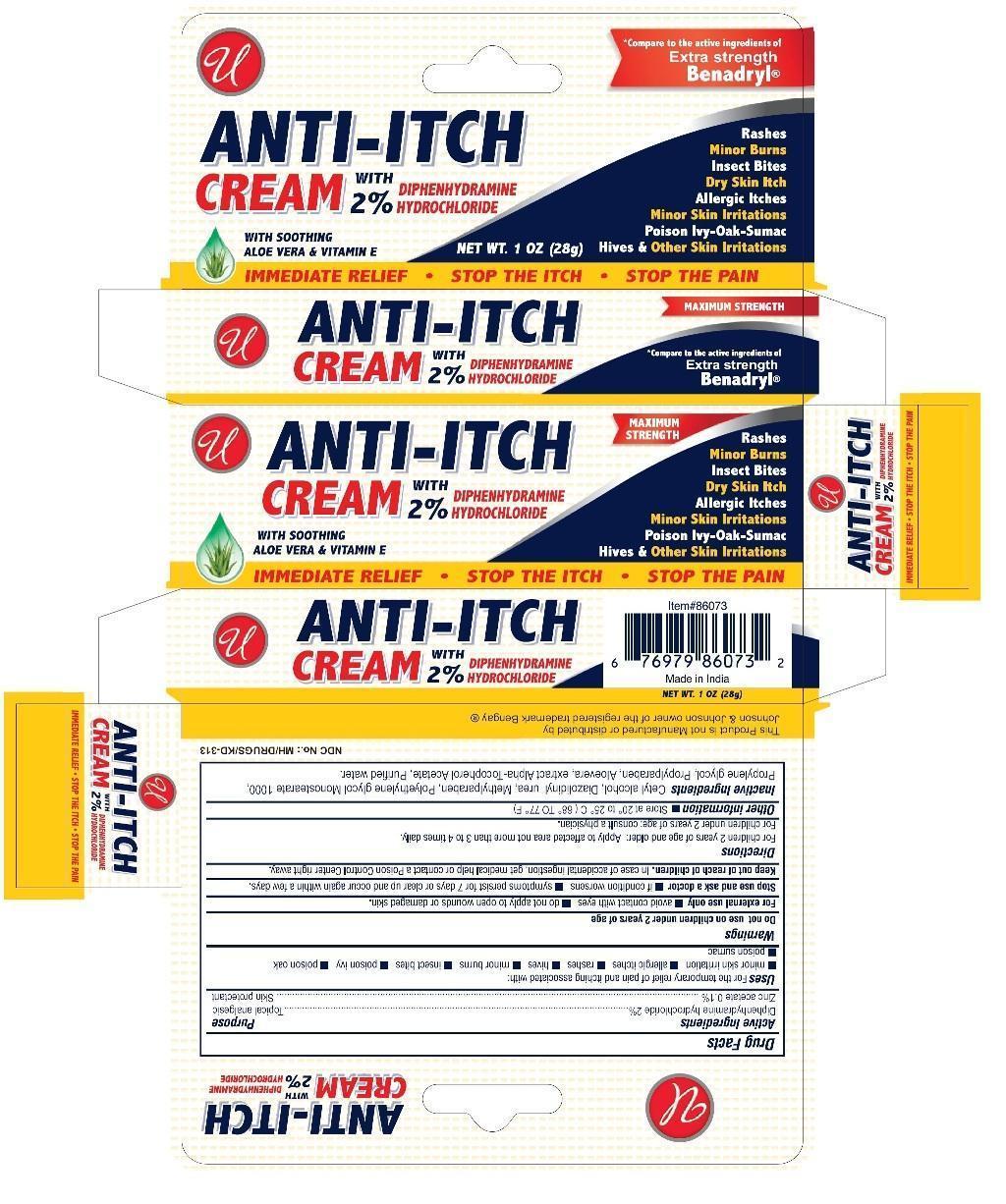
Anti-itch Cream
79fd6b63-5fd4-4ab0-81df-e9d582984ace
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients Diphenhydramine Hydrochloride 2% Zinc Acetate 0.1%
Medication Information
Warnings and Precautions
Warnings
Indications and Usage
Uses
For the temporary relief of pain and itching associated with:
- minor skin irritation
- allergic itches
- rashes
- hives
- minor burns
- insect bites
- poison ivy
- poison oak
- poison sumac
Dosage and Administration
Directions
-
For children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily.
-
For children under 2 years of age: consult a physician.
Description
Active Ingredients Diphenhydramine Hydrochloride 2% Zinc Acetate 0.1%
Section 42229-5
Other information
- Store at 20ºC to 25ºC (68ºF to 77ºF).
Section 50565-1
Keep out of reach of children.
In case of accidental ingestion, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor
- if condition worsens
- symptoms persist for 7 days or clear up and occur again within a few days.
Section 50570-1
Do not use on children under 2 years of age.
For external use only
- avoid contact with eyes
- do not apply to open wound or damaged skin.
Section 51727-6
Inactive ingredients
Cetyl Alcohol, Diazolidinly urea, Methylparaben, Polyethylene Glycol Monostearate 1000, Propylene Gylcol, Propylparaben, Aloevera extract, Alpha-Tocopherol Acetate, Purified Water.
Section 51945-4
PRINCIPAL DISPLAY PANEL
Anti-Itch Cream
NET WT 1 OZ (28 g)
Section 55105-1
Purpose
Topical Analgesic
Skin Protectant
Section 55106-9
Active Ingredients
Diphenhydramine Hydrochloride 2%
Zinc Acetate 0.1%
Structured Label Content
Indications and Usage (34067-9)
Uses
For the temporary relief of pain and itching associated with:
- minor skin irritation
- allergic itches
- rashes
- hives
- minor burns
- insect bites
- poison ivy
- poison oak
- poison sumac
Dosage and Administration (34068-7)
Directions
-
For children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily.
-
For children under 2 years of age: consult a physician.
Warnings and Precautions (34071-1)
Warnings
Section 42229-5 (42229-5)
Other information
- Store at 20ºC to 25ºC (68ºF to 77ºF).
Section 50565-1 (50565-1)
Keep out of reach of children.
In case of accidental ingestion, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor
- if condition worsens
- symptoms persist for 7 days or clear up and occur again within a few days.
Section 50570-1 (50570-1)
Do not use on children under 2 years of age.
For external use only
- avoid contact with eyes
- do not apply to open wound or damaged skin.
Section 51727-6 (51727-6)
Inactive ingredients
Cetyl Alcohol, Diazolidinly urea, Methylparaben, Polyethylene Glycol Monostearate 1000, Propylene Gylcol, Propylparaben, Aloevera extract, Alpha-Tocopherol Acetate, Purified Water.
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL
Anti-Itch Cream
NET WT 1 OZ (28 g)
Section 55105-1 (55105-1)
Purpose
Topical Analgesic
Skin Protectant
Section 55106-9 (55106-9)
Active Ingredients
Diphenhydramine Hydrochloride 2%
Zinc Acetate 0.1%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:44.422042 · Updated: 2026-03-14T23:05:38.431426