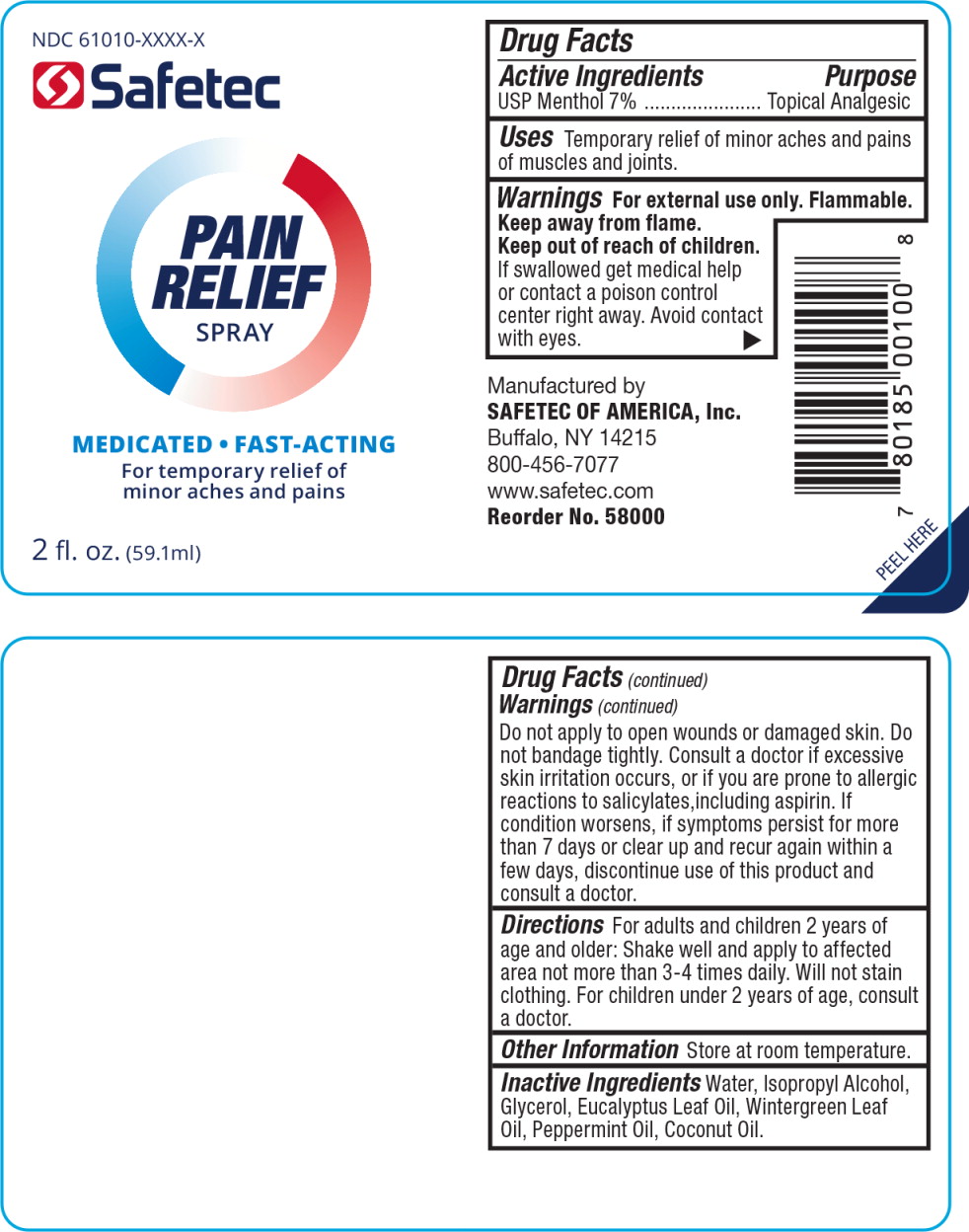
61010-8201. Pain Relief Spray
79e3b0e7-4ba5-4b9d-a980-5b6f18dba802
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
USP Menthol 7%
Purpose
Topical Analgesic
Medication Information
Purpose
Topical Analgesic
Description
USP Menthol 7%
Uses
Temporary relief of minor aches and pains of muscles and joints.
Section 50569-3
Consult a doctor if excessive skin irritation occurs, or if you are prone to allergic reactions to salicylates,including aspirin. If condition worsens, if symptoms persist for more than 7 days or clear up and recur again within a few days, discontinue use of this product and consult a doctor.
Section 50570-1
Do not apply to open wounds or damaged skin. Do not bandage tightly.
Warnings
For external use only.
Flammable.
Keep away from flame.
Directions
For adults and children 2 years of age and older: Shake well and apply to affected area not more than 3-4 times daily. Will not stain clothing. For children under 2 years of age, consult a doctor.
Other Information
Store at room temperature.
Active Ingredients
USP Menthol 7%
Inactive Ingredients
Water, Isopropyl Alcohol, Glycerol, Eucalyptus Leaf Oil, Wintergreen Leaf Oil, Peppermint Oil, Coconut Oil.
Keep Out of Reach of Children.
If swallowed get medical help or contact a poison control center right away. Avoid contact with eyes.
Principal Display Panel Pain Relief Spray Bottle Label
NDC 61010-XXXX-X
Safetec
PAIN
RELIEF
SPRAY
MEDICATED FAST-ACTING
For temporary relief of minor aches and pains
2 fl. oz. (59.1 ml)
Structured Label Content
Uses
Temporary relief of minor aches and pains of muscles and joints.
Section 50569-3 (50569-3)
Consult a doctor if excessive skin irritation occurs, or if you are prone to allergic reactions to salicylates,including aspirin. If condition worsens, if symptoms persist for more than 7 days or clear up and recur again within a few days, discontinue use of this product and consult a doctor.
Section 50570-1 (50570-1)
Do not apply to open wounds or damaged skin. Do not bandage tightly.
Purpose
Topical Analgesic
Warnings
For external use only.
Flammable.
Keep away from flame.
Directions
For adults and children 2 years of age and older: Shake well and apply to affected area not more than 3-4 times daily. Will not stain clothing. For children under 2 years of age, consult a doctor.
Other Information
Store at room temperature.
Active Ingredients
USP Menthol 7%
Inactive Ingredients
Water, Isopropyl Alcohol, Glycerol, Eucalyptus Leaf Oil, Wintergreen Leaf Oil, Peppermint Oil, Coconut Oil.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed get medical help or contact a poison control center right away. Avoid contact with eyes.
Principal Display Panel Pain Relief Spray Bottle Label (Principal Display Panel - Pain Relief Spray Bottle Label)
NDC 61010-XXXX-X
Safetec
PAIN
RELIEF
SPRAY
MEDICATED FAST-ACTING
For temporary relief of minor aches and pains
2 fl. oz. (59.1 ml)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:47.050991 · Updated: 2026-03-14T23:02:30.868416