Drug Facts
798ffdde-1dba-4a3f-a0b6-937f9e958cca
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients Purpose Propylene Glycol 0.06% Lubricant
Medication Information
Description
Active Ingredients Purpose Propylene Glycol 0.06% Lubricant
Uses
- for the temporary relief of burning and irritation due to dryness of the eye
- for the temporary relief of discomfort due to minor irritations of the eye or to exposure to wind or sun
- for use as a protectant against further irritation or to relieve dryness of the eye
- for use as a lubricant to prevent further irritation or to relieve dryness of the eye
Section 50566-9
Stop use and ask a doctor if you experience any of the following:
- eye pain
- changes in vision
- continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Section 55105-1
| Active Ingredients | Purpose |
| Propylene Glycol 0.06% | Lubricant |
Warnings
For external use only
Directions
- shake well before using
- put 1 or 2 drops in the affected eye(s) as needed
- see side of carton for directions for use
Do Not Use
- if this product changes color
- if you are sensitive to any ingredient in this product
Questions?
In the U.S. call 1-800-757-9195
www.systane.com
[email protected]
Directions for Use:
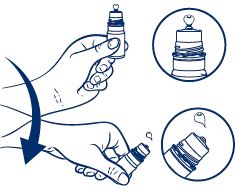
- Hold the bottle just below the cap and twist the cap to open.
- BEFORE FIRST USE, hold the bottle upside down and dispense one drop of product to discard.
- Shake the bottle downward to remove residual product (see picture).
- Place 1 or 2 drops in the affected eye(s) as needed and blink.
- After use, shake the bottle downward to remove residual product that may be left on the tip.
- Replace cap after use.
- Discard 90 days after opening.
TAMPER EVIDENT:
For your protection, this bottle has a tamper evident ring around the bottom of the cap. Do not use if this ring is damaged or missing at the time of purchase.
1Based on a survey of eye care professionals. Data on file.
Other Information
- store at room temperature
Inactive Ingredients
boric acid, dimyristoyl phosphatidylglycerol, hydroxypropyl guar, mineral oil, polyoxyl 40 stearate, sorbitan tristearate, sorbitol and purified water. May contain hydrochloric acid and/or sodium hydroxide to adjust pH.
Principal Display Panel
3 PACK
Preservative-Free
SYSTANE
®
LUBRICANT EYE DROPS
COMPLETE PF
MULTI-SYMPTON
DRY EYE RELIEF
All-in-One Formula
Tear evaporation protection
Preservative-Free Bottles
#1 Doctor Recommended Brands1
Alcon
Sterile
Three 10mL (0.34 FL OZ) Bottles
LOT: EXP:
Side Panel:
Preservative-Free
Multi-Symptom
Relief for
Sensitive eyes
Alcon Laboratories, Inc.
Fort Worth, TX 76134 USA
Country of Origin: United States
U.S. Patent: www.alconpatents.com
Actual Size
300073255-0725
When Using This Product
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
- for the temporary relief of burning and irritation due to dryness of the eye
- for the temporary relief of discomfort due to minor irritations of the eye or to exposure to wind or sun
- for use as a protectant against further irritation or to relieve dryness of the eye
- for use as a lubricant to prevent further irritation or to relieve dryness of the eye
Section 50566-9 (50566-9)
Stop use and ask a doctor if you experience any of the following:
- eye pain
- changes in vision
- continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Section 55105-1 (55105-1)
| Active Ingredients | Purpose |
| Propylene Glycol 0.06% | Lubricant |
Warnings
For external use only
Directions
- shake well before using
- put 1 or 2 drops in the affected eye(s) as needed
- see side of carton for directions for use
Do Not Use (Do not use)
- if this product changes color
- if you are sensitive to any ingredient in this product
Questions?
In the U.S. call 1-800-757-9195
www.systane.com
[email protected]
Directions for Use:
- Hold the bottle just below the cap and twist the cap to open.
- BEFORE FIRST USE, hold the bottle upside down and dispense one drop of product to discard.
- Shake the bottle downward to remove residual product (see picture).
- Place 1 or 2 drops in the affected eye(s) as needed and blink.
- After use, shake the bottle downward to remove residual product that may be left on the tip.
- Replace cap after use.
- Discard 90 days after opening.
TAMPER EVIDENT:
For your protection, this bottle has a tamper evident ring around the bottom of the cap. Do not use if this ring is damaged or missing at the time of purchase.
1Based on a survey of eye care professionals. Data on file.
Other Information (Other information)
- store at room temperature
Inactive Ingredients (Inactive ingredients)
boric acid, dimyristoyl phosphatidylglycerol, hydroxypropyl guar, mineral oil, polyoxyl 40 stearate, sorbitan tristearate, sorbitol and purified water. May contain hydrochloric acid and/or sodium hydroxide to adjust pH.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
3 PACK
Preservative-Free
SYSTANE
®
LUBRICANT EYE DROPS
COMPLETE PF
MULTI-SYMPTON
DRY EYE RELIEF
All-in-One Formula
Tear evaporation protection
Preservative-Free Bottles
#1 Doctor Recommended Brands1
Alcon
Sterile
Three 10mL (0.34 FL OZ) Bottles
LOT: EXP:
Side Panel:
Preservative-Free
Multi-Symptom
Relief for
Sensitive eyes
Alcon Laboratories, Inc.
Fort Worth, TX 76134 USA
Country of Origin: United States
U.S. Patent: www.alconpatents.com
Actual Size
300073255-0725
When Using This Product (When using this product)
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:54.944943 · Updated: 2026-03-14T23:13:23.763125