Theratears Pf 58790-000
797b0394-2f0f-4733-8d20-ec5bdf3e085d
34390-5
HUMAN OTC DRUG LABEL
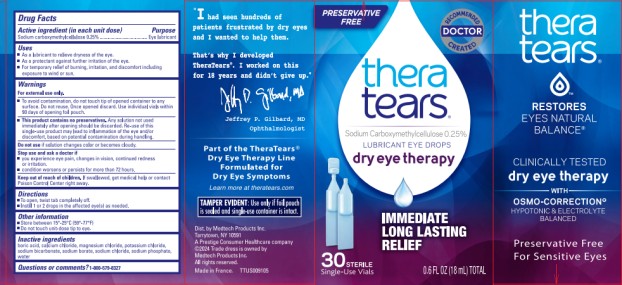
Drug Facts
Composition & Product
Identifiers & Packaging
Description
THERATEARS LUBRICANT- carboxymethylcellulose sodium solution/ drops Medtech Products Inc., a Prestige Consumer Healthcare company Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ---------- Drug Facts
Purpose
Eye lubricant
Medication Information
Purpose
Eye lubricant
Description
THERATEARS LUBRICANT- carboxymethylcellulose sodium solution/ drops Medtech Products Inc., a Prestige Consumer Healthcare company Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ---------- Drug Facts
Uses
- As a lubricant to relieve dryness of the eye.
- As a protectant against further irritation of the eye.
- For temporary relief of burning, irritation, and discomfort including exposure to wind or sun.
Section 42229-5
THERATEARS LUBRICANT- carboxymethylcellulose sodium solution/ drops
Medtech Products Inc., a Prestige Consumer Healthcare company
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Drug Facts
Warnings
For external use only
- To avoid contamination do not touch tip of opened container to any surface. Do not reuse. Once opened discard. Use individual vials within 90 days of opening foil pouch.
- This product contains no preservatives. Any solution not used immediately after opening should be discarded. Re-use of this single-use product may lead to inflammation of the eye and/or discomfort, based on potential contamination during handling.
Directions
- To open, twist tab completely off.
- Instill 1 or 2 drops in the affected eye(s) as needed.
Do Not Use
- If solution changes color or becomes cloudy.
Other Information
- Store between 15-25 C (59-77F)
- Do not touch unit-dose tip to eye.
Inactive Ingredients
boric acid , calcium chloride, magnesium chloride, potassium chloride, sodium bicarbonate, sodium borate, sodium chloride, sodium phosphate, water
Principal Display Panel
PRESERVATIVE FREE
thera
tears®
THERAPY FOR YOUR EYES TM
dry eye therapy
LUBRICANT
EYE DROPS
IMMEDIATE
LONG LASTING
RELIEF
30 STERILE Single-Use Vials
0.60 FL OZ (18.0mL) TOTAL
Questions and Comments?
1-800-579-8327
Stop Use and Ask A Doctor If
- You experience eye pain, changes in vision, continued redness or irritation.
- Condition worsens or persists for more than 72 hours.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center immediately.
Active Ingredient (in Each Unit Dose)
Sodium Carboxymethylcellulose 0.25%
Structured Label Content
Uses
- As a lubricant to relieve dryness of the eye.
- As a protectant against further irritation of the eye.
- For temporary relief of burning, irritation, and discomfort including exposure to wind or sun.
Section 42229-5 (42229-5)
THERATEARS LUBRICANT- carboxymethylcellulose sodium solution/ drops
Medtech Products Inc., a Prestige Consumer Healthcare company
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Drug Facts
Purpose
Eye lubricant
Warnings
For external use only
- To avoid contamination do not touch tip of opened container to any surface. Do not reuse. Once opened discard. Use individual vials within 90 days of opening foil pouch.
- This product contains no preservatives. Any solution not used immediately after opening should be discarded. Re-use of this single-use product may lead to inflammation of the eye and/or discomfort, based on potential contamination during handling.
Directions
- To open, twist tab completely off.
- Instill 1 or 2 drops in the affected eye(s) as needed.
Do Not Use (Do not use)
- If solution changes color or becomes cloudy.
Other Information (Other information)
- Store between 15-25 C (59-77F)
- Do not touch unit-dose tip to eye.
Inactive Ingredients (Inactive ingredients)
boric acid , calcium chloride, magnesium chloride, potassium chloride, sodium bicarbonate, sodium borate, sodium chloride, sodium phosphate, water
Principal Display Panel
PRESERVATIVE FREE
thera
tears®
THERAPY FOR YOUR EYES TM
dry eye therapy
LUBRICANT
EYE DROPS
IMMEDIATE
LONG LASTING
RELIEF
30 STERILE Single-Use Vials
0.60 FL OZ (18.0mL) TOTAL
Questions and Comments? (Questions and comments?)
1-800-579-8327
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- You experience eye pain, changes in vision, continued redness or irritation.
- Condition worsens or persists for more than 72 hours.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center immediately.
Active Ingredient (in Each Unit Dose) (Active ingredient (In each unit dose))
Sodium Carboxymethylcellulose 0.25%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:49.041760 · Updated: 2026-03-14T23:13:10.446567