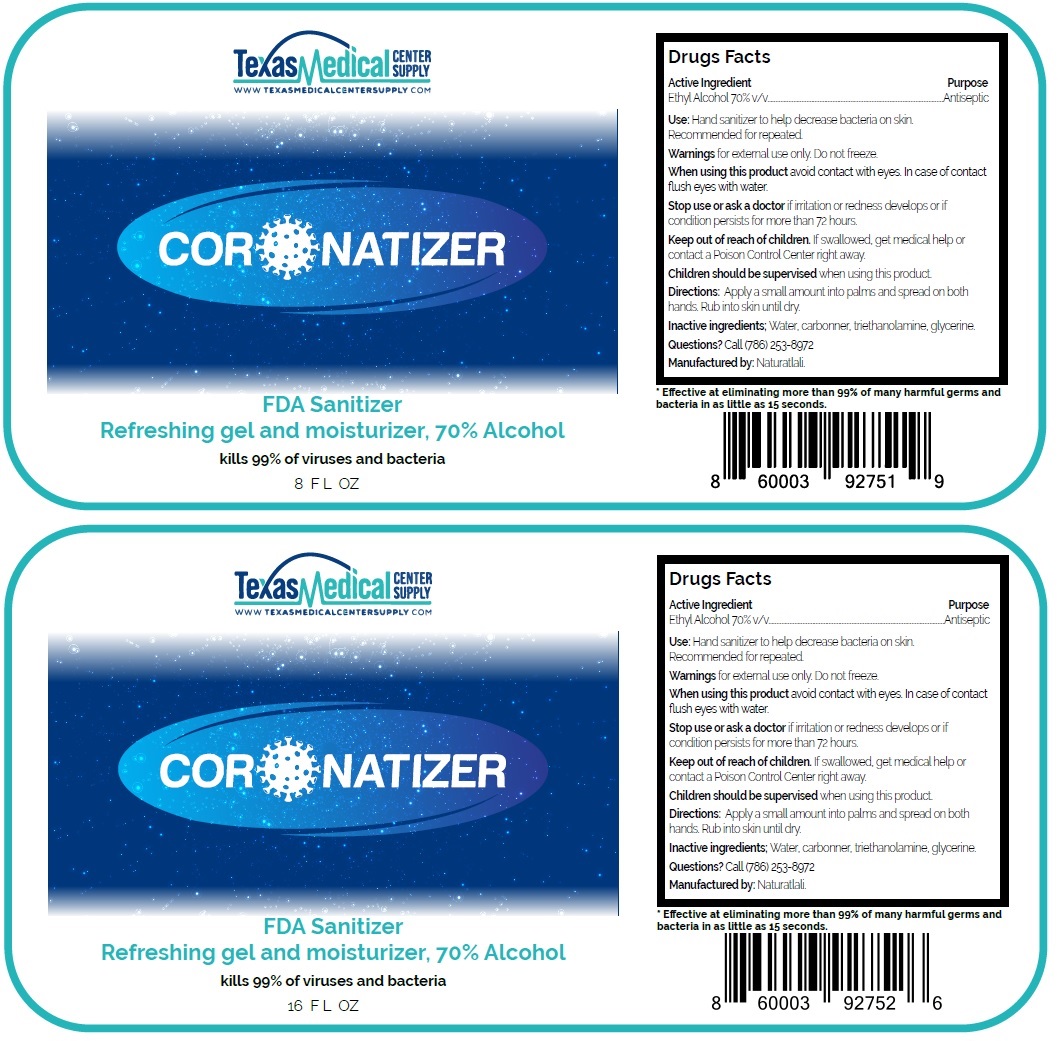
Coronatizer
793aa3f4-0b1c-4881-9976-3a7babd83e69
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyl Alcohol 70% v/v
Purpose
Antiseptic
Medication Information
Warnings and Precautions
Warnings for external use only. Do not freeze.
When using this product avoid contact with eyes. In case of contact flush eyes with water.
Stop use or ask a doctor if irritation or redness develops or if condition persists for more than 72 hours.
Indications and Usage
Use: Hand sanitizer to help decrease bacteria on skin
Recommended for repeated use
Purpose
Antiseptic
Dosage and Administration
Directions: Apply a small amount into palms and spread on both hands. Rub into skin until dry.
Description
Ethyl Alcohol 70% v/v
Section 42229-5
WWW.TEXASMEDICALCENTERSUPPLY.COM
FDA Sanitizer
Refreshing gel and moisturizer, 70% Alcohol
kills 99% of viruses and bacteria
Manufactured by: Naturatlali.
* Effective at eliminating more than 99% of many harmful germs and bacteria in as little as 15 seconds.
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Children should be supervised when using this product.
Section 51727-6
Inactive ingredients; Water, carbomer, triethanolamine, glycerine.
Section 53413-1
Questions? Call (786) 253-8972
Packaging
Drugs Facts
Active Ingredient
Ethyl Alcohol 70% v/v
Structured Label Content
Indications and Usage (34067-9)
Use: Hand sanitizer to help decrease bacteria on skin
Recommended for repeated use
Dosage and Administration (34068-7)
Directions: Apply a small amount into palms and spread on both hands. Rub into skin until dry.
Warnings and Precautions (34071-1)
Warnings for external use only. Do not freeze.
When using this product avoid contact with eyes. In case of contact flush eyes with water.
Stop use or ask a doctor if irritation or redness develops or if condition persists for more than 72 hours.
Section 42229-5 (42229-5)
WWW.TEXASMEDICALCENTERSUPPLY.COM
FDA Sanitizer
Refreshing gel and moisturizer, 70% Alcohol
kills 99% of viruses and bacteria
Manufactured by: Naturatlali.
* Effective at eliminating more than 99% of many harmful germs and bacteria in as little as 15 seconds.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Children should be supervised when using this product.
Section 51727-6 (51727-6)
Inactive ingredients; Water, carbomer, triethanolamine, glycerine.
Section 53413-1 (53413-1)
Questions? Call (786) 253-8972
Purpose
Antiseptic
Packaging
Drugs Facts
Active Ingredient
Ethyl Alcohol 70% v/v
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:57:45.311578 · Updated: 2026-03-14T22:57:01.531445