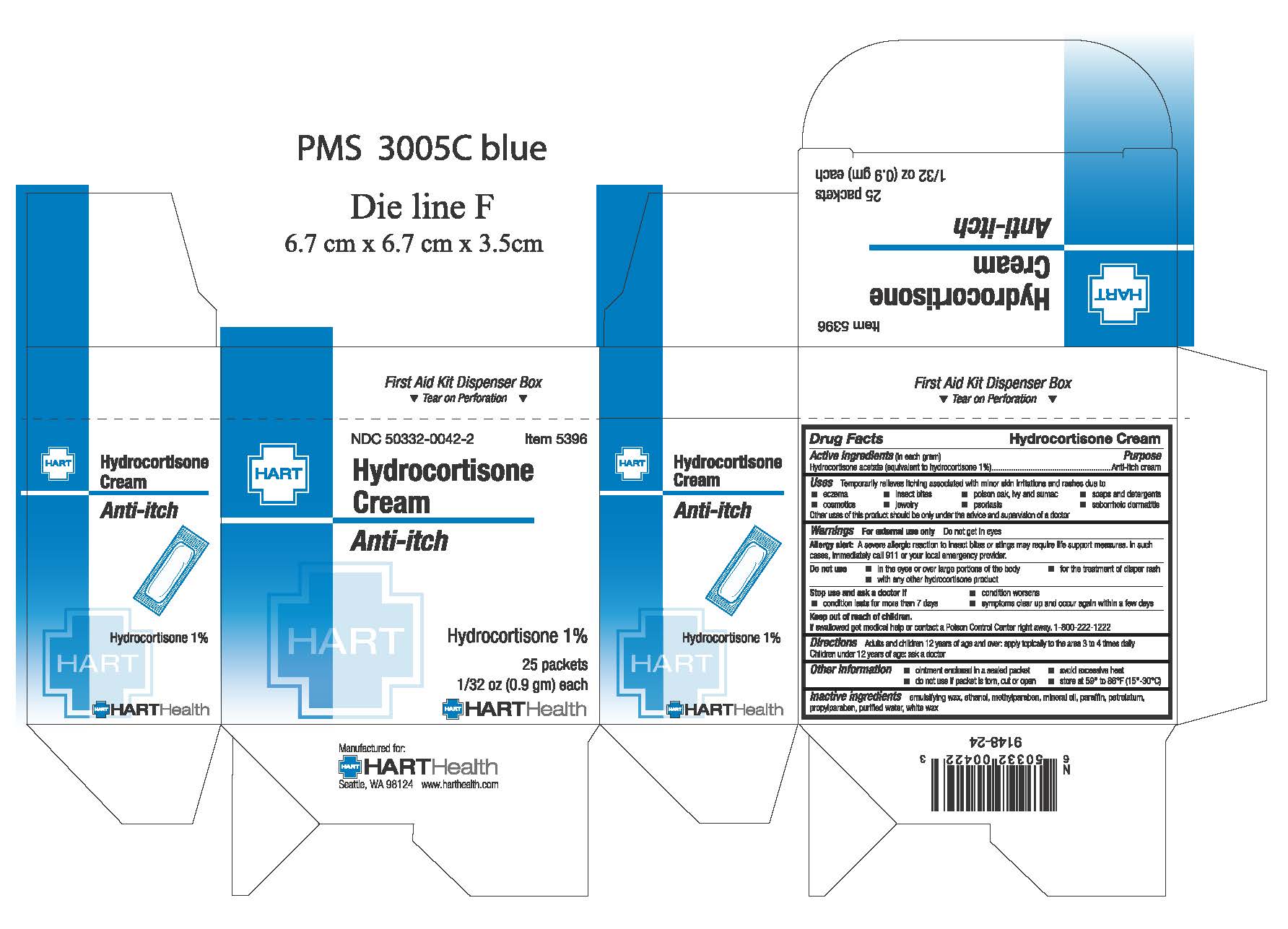
Hydrocortisone Cream
779f945f-ab69-40de-8d86-ab76debc5c7c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredient (in each gram): Hydrocortisone Acetate 10mg (equivalent to Hydrocortisone 1%)
Medication Information
Warnings and Precautions
Warnings: For external use only. Do not get in eyes
Allergy alert: A severe allergic reaction to insect bites or stings may require life support measures. In such cases, immediately clal 911 or your local emergency provider.
Indications and Usage
Dosage and Administration
Directions:
Adults and children 12 years of age and over: apply topically to the area 3 to 4 times daily.
Children under 12 years of age: do not use, ask a doctor
Description
Active Ingredient (in each gram): Hydrocortisone Acetate 10mg (equivalent to Hydrocortisone 1%)
Section 50565-1
Keep out of reach of children. If swallowed get medical help or contact a Poison Control Center right away. 1-800-222-1222
Section 50566-9
Stop use and ask a doctor if:
- condition worsens
- condition lasts for more than 7 days
- symptoms clear up and occur again within a few days
Section 50570-1
Do not use:
- in the eyes or over large portions of the body
- for the treatment of diaper rash
- with any other hydrocortisone product
Section 51727-6
Inactive Ingredients: emulsifying wax, ethanol, methylparaben, mineral oil, paraffin, petrolatum, propylparaben, purified water, white wax
Section 51945-4
Section 55105-1
Purpose: anti-itch cream
Section 55106-9
Active Ingredient (in each gram): Hydrocortisone Acetate 10mg (equivalent to Hydrocortisone 1%)
Structured Label Content
Indications and Usage (34067-9)
Dosage and Administration (34068-7)
Directions:
Adults and children 12 years of age and over: apply topically to the area 3 to 4 times daily.
Children under 12 years of age: do not use, ask a doctor
Warnings and Precautions (34071-1)
Warnings: For external use only. Do not get in eyes
Allergy alert: A severe allergic reaction to insect bites or stings may require life support measures. In such cases, immediately clal 911 or your local emergency provider.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed get medical help or contact a Poison Control Center right away. 1-800-222-1222
Section 50566-9 (50566-9)
Stop use and ask a doctor if:
- condition worsens
- condition lasts for more than 7 days
- symptoms clear up and occur again within a few days
Section 50570-1 (50570-1)
Do not use:
- in the eyes or over large portions of the body
- for the treatment of diaper rash
- with any other hydrocortisone product
Section 51727-6 (51727-6)
Inactive Ingredients: emulsifying wax, ethanol, methylparaben, mineral oil, paraffin, petrolatum, propylparaben, purified water, white wax
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose: anti-itch cream
Section 55106-9 (55106-9)
Active Ingredient (in each gram): Hydrocortisone Acetate 10mg (equivalent to Hydrocortisone 1%)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:47.684098 · Updated: 2026-03-14T23:02:32.213463