Actidom®
7797c22e-ffeb-4798-9fb6-aa64cf077ba7
34390-5
HUMAN OTC DRUG LABEL
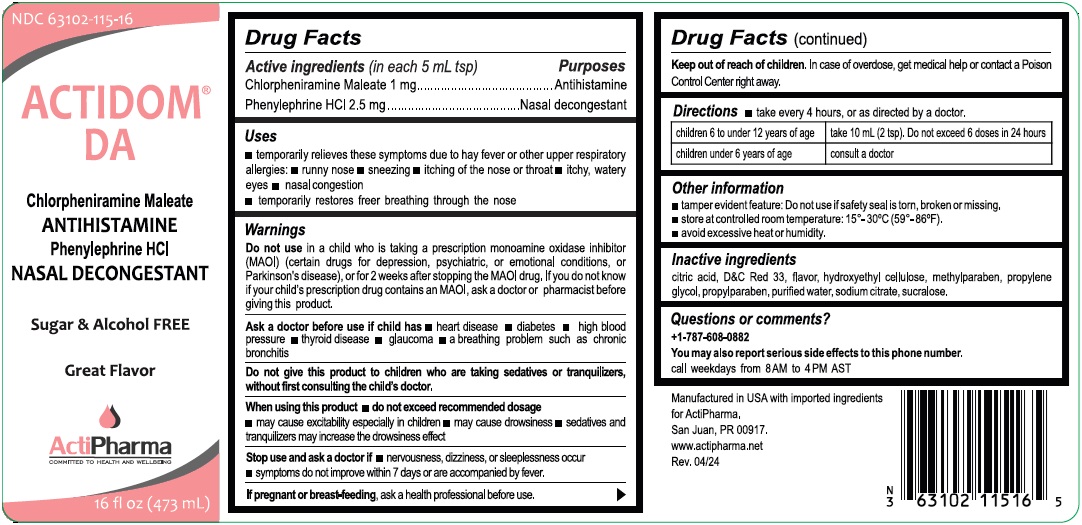
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Chlorpheniramine Maleate 1 mg Phenylephrine HCL 2.5 mg
Purpose
Antihistamine Nasal decongestant
Medication Information
Purpose
Antihistamine Nasal decongestant
Description
Chlorpheniramine Maleate 1 mg Phenylephrine HCL 2.5 mg
Uses
• temporarily relieves these symptoms due to hay fever or other upper respiratory allergies: • runny nose • sneezing • itching of the nose or throat
• itchy, watery eyes • nasal congestion
• temporarily restores free breathing through the nose
Section 42229-5
Sugar & Alcohol FREE
Great Flavor
Manufactured in USA with imported ingredients
for ActiPharma.
San Juan, PR 00917.
www.actipharma.net
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Purposes
Antihistamine
Nasal decongestant
Packaging
Directions
take every 4 hours, or as directed by a doctor.
| children 6 to under 12 years of age | take 10 mL (2 tsp). Do not exceed 6 doses in 24 hours |
| children under 6 years of age | consult a doctor |
Drug Facts
warnings
Do not use in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your child's prescription drug contains an MAOI, ask a doctor or pharmacist before giving this product.
Ask a doctor before use if child has • heart disease • diabetes • high blood pressure • thyroid disease • glaucoma • a breathing problem such as chronic bronchitis
Do not give this product to children who are taking sedatives or tranquilizers, without first consulting the child's doctor.
When using this product • do not exceed recommended dosage
• may cause excitability especially in children • may cause drowsiness • sedatives and tranquilizers may increase the drowsiness effect
Stop use and ask a doctor if • nervousness, dizziness, or sleeplessness occur
• symptoms do not improve within 7 days or are accompanied by fever.
If pregnant or breast-feeding, ask a health professional before use.
Other Information
• tamper evident feature: Do not use if safety seal is torn, broken or missing.
• store at controlled room temperature: 15°-30°C (59°-86°F).
• avoid excessive heat or humidity.
Inactive Ingredients
citric acid, D&C Red 33, flavor, hydroxyethyl cellulose, methylparaben, propylene glycol, propylparaben, purified water, sodium citrate, sucralose.
active Ingredients
Chlorpheniramine Maleate 1 mg
Phenylephrine HCL 2.5 mg
Questions Or Comments?
+1-787-608-0882
You may also report serious side effects to this phone number.
call weekdays from 8AM to 4 PM AST
Structured Label Content
Uses
• temporarily relieves these symptoms due to hay fever or other upper respiratory allergies: • runny nose • sneezing • itching of the nose or throat
• itchy, watery eyes • nasal congestion
• temporarily restores free breathing through the nose
Section 42229-5 (42229-5)
Sugar & Alcohol FREE
Great Flavor
Manufactured in USA with imported ingredients
for ActiPharma.
San Juan, PR 00917.
www.actipharma.net
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Purposes
Antihistamine
Nasal decongestant
Packaging
Directions
take every 4 hours, or as directed by a doctor.
| children 6 to under 12 years of age | take 10 mL (2 tsp). Do not exceed 6 doses in 24 hours |
| children under 6 years of age | consult a doctor |
Drug Facts
warnings (Warnings)
Do not use in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your child's prescription drug contains an MAOI, ask a doctor or pharmacist before giving this product.
Ask a doctor before use if child has • heart disease • diabetes • high blood pressure • thyroid disease • glaucoma • a breathing problem such as chronic bronchitis
Do not give this product to children who are taking sedatives or tranquilizers, without first consulting the child's doctor.
When using this product • do not exceed recommended dosage
• may cause excitability especially in children • may cause drowsiness • sedatives and tranquilizers may increase the drowsiness effect
Stop use and ask a doctor if • nervousness, dizziness, or sleeplessness occur
• symptoms do not improve within 7 days or are accompanied by fever.
If pregnant or breast-feeding, ask a health professional before use.
Other Information (Other information)
• tamper evident feature: Do not use if safety seal is torn, broken or missing.
• store at controlled room temperature: 15°-30°C (59°-86°F).
• avoid excessive heat or humidity.
Inactive Ingredients (Inactive ingredients)
citric acid, D&C Red 33, flavor, hydroxyethyl cellulose, methylparaben, propylene glycol, propylparaben, purified water, sodium citrate, sucralose.
active Ingredients (Active ingredients)
Chlorpheniramine Maleate 1 mg
Phenylephrine HCL 2.5 mg
Questions Or Comments? (Questions or comments?)
+1-787-608-0882
You may also report serious side effects to this phone number.
call weekdays from 8AM to 4 PM AST
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:24.099081 · Updated: 2026-03-14T23:03:27.182760