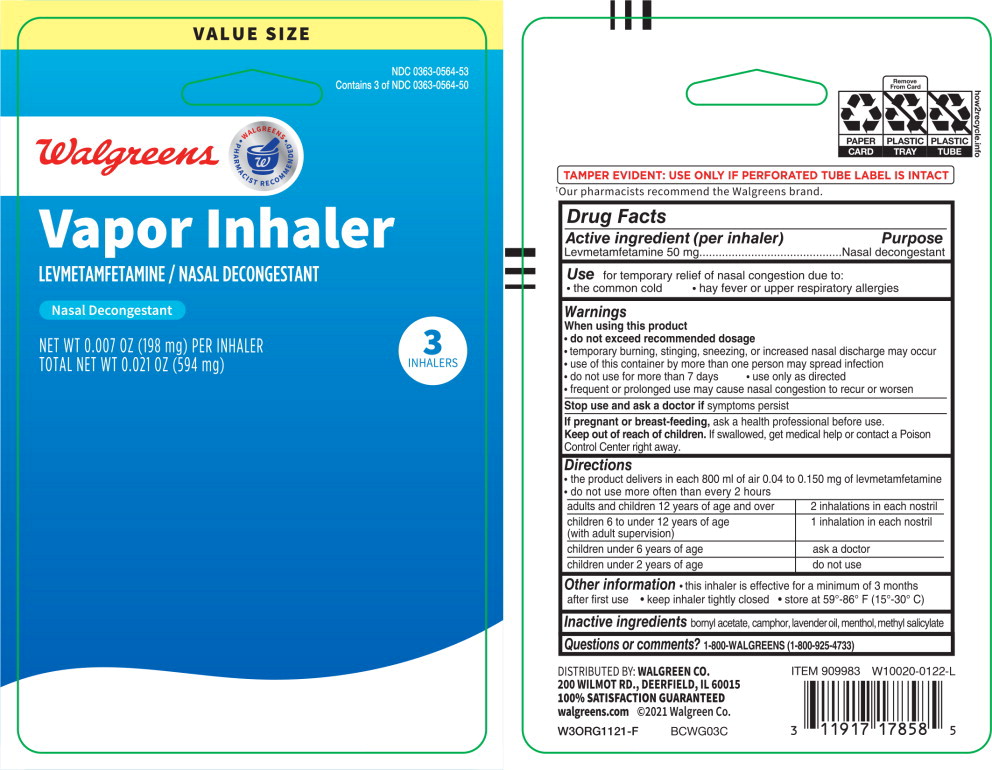
Drug Facts
77924474-ccc7-497b-9168-91490bcfadfc
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Levmetamfetamine 50 mg
Purpose
Nasal decongestant
Medication Information
Purpose
Nasal decongestant
Description
Levmetamfetamine 50 mg
Use
for temporary relief of nasal congestion due to:
- the common cold
- hay fever or upper respiratory allergies
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if symptoms persist
Section 50567-7
When using this product
- do not exceed recommended dosage
- temporary burning, stinging, sneezing, or increased nasal discharge may occur
- use of this container by more than one person may spread infection
Section 50570-1
- do not use for more than 7 days
- use only as directed
- frequent or prolonged use may cause nasal congestion to recur or worsen
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Directions
- the product delivers in each 800 ml of air 0.04 to 0.150 mg of levmetamfetamine
- do not use more often than every 2 hours
| adults and children 12 years of age and over | 2 inhalations in each nostril |
| children 6 to under 12 years of age (with adult supervision) |
1 inhalation in each nostril |
| children under 6 years of age | ask a doctor |
| children under 2 years of age | do not use |
Other Information
- this inhaler is effective for a minimum of 3 months after first use
- keep inhaler tightly closed
- store at 59°-86° F (15°-30° C)
Inactive Ingredients
bornyl acetate, camphor, lavender oil, menthol, methyl salicylate
Questions Or Comments?
1-800-walgreens (1-800-925-4733)
Active Ingredient (per Inhaler)
Levmetamfetamine 50 mg
Principal Display Panel – 3 Inhaler Carton
VALUE SIZE
NDC 0363-0564-53
Contains 3 of NDC 0363-0564-50
Walgreens
WALGREENS PHARMACIST RECOMMENDED
Vapor Inhaler
LEVMETAMFETAMINE / NASAL DECONGESTANT
Nasal Decongestant
NET WT 0.007 OZ (198 mg) PER INHALER
TOTAL NET WT 0.021 0Z (594 mg)
3
INHALERS
Structured Label Content
Use
for temporary relief of nasal congestion due to:
- the common cold
- hay fever or upper respiratory allergies
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if symptoms persist
Section 50567-7 (50567-7)
When using this product
- do not exceed recommended dosage
- temporary burning, stinging, sneezing, or increased nasal discharge may occur
- use of this container by more than one person may spread infection
Section 50570-1 (50570-1)
- do not use for more than 7 days
- use only as directed
- frequent or prolonged use may cause nasal congestion to recur or worsen
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Nasal decongestant
Directions
- the product delivers in each 800 ml of air 0.04 to 0.150 mg of levmetamfetamine
- do not use more often than every 2 hours
| adults and children 12 years of age and over | 2 inhalations in each nostril |
| children 6 to under 12 years of age (with adult supervision) |
1 inhalation in each nostril |
| children under 6 years of age | ask a doctor |
| children under 2 years of age | do not use |
Other Information (Other information)
- this inhaler is effective for a minimum of 3 months after first use
- keep inhaler tightly closed
- store at 59°-86° F (15°-30° C)
Inactive Ingredients (Inactive ingredients)
bornyl acetate, camphor, lavender oil, menthol, methyl salicylate
Questions Or Comments? (Questions or comments?)
1-800-walgreens (1-800-925-4733)
Active Ingredient (per Inhaler) (Active ingredient (per inhaler))
Levmetamfetamine 50 mg
Principal Display Panel – 3 Inhaler Carton
VALUE SIZE
NDC 0363-0564-53
Contains 3 of NDC 0363-0564-50
Walgreens
WALGREENS PHARMACIST RECOMMENDED
Vapor Inhaler
LEVMETAMFETAMINE / NASAL DECONGESTANT
Nasal Decongestant
NET WT 0.007 OZ (198 mg) PER INHALER
TOTAL NET WT 0.021 0Z (594 mg)
3
INHALERS
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:02.241752 · Updated: 2026-03-14T23:06:18.731306