These Highlights Do Not Include All The Information Needed To Use Gallium Ga 68 Gozetotide §
7744bafb-755e-29a2-e053-2991aa0a86a9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Gallium Ga 68 Gozetotide Injection is indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer: with suspected metastasis who are candidates for initial definitive therapy. with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
Indications and Usage
Gallium Ga 68 Gozetotide Injection is indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer: with suspected metastasis who are candidates for initial definitive therapy. with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
Dosage and Administration
Use appropriate aseptic technique and radiation safety handling measures to maintain sterility during all operations involved in the manipulation and administration of Gallium Ga 68 Gozetotide Injection. ( 2.1 ) The recommended adult dose is 111 MBq to 259 MBq (3 mCi to 7 mCi) as a bolus intravenous injection. ( 2.2 ) A diuretic expected to act within the uptake time period may be administered at the time of radiotracer injection. ( 2.2 ) Initiate imaging 50 to 100 minutes after administration. The patient should void immediately prior to initiation of imaging. Scan should begin caudally and proceed cranially. ( 2.4 ) See full prescribing information for additional preparation, administration, imaging, and radiation dosimetry information. ( 2 )
Warnings and Precautions
Risk for misdiagnosis: Gallium Ga 68 gozetotide uptake can be seen in a variety of tumor types and in non-malignant processes. Image interpretation errors can occur with gallium Ga 68 gozetotide PET. ( 5.1 ) Radiation risk: Ensure safe handling to protect patients and health care workers from unintentional radiation exposure. ( 2.1 , 5.2 )
Contraindications
None
Adverse Reactions
The most commonly reported adverse reactions include nausea, diarrhea, and dizziness. ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact UCLA Nuclear Medicine at 1-844-963-1855 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Medication Information
Warnings and Precautions
Risk for misdiagnosis: Gallium Ga 68 gozetotide uptake can be seen in a variety of tumor types and in non-malignant processes. Image interpretation errors can occur with gallium Ga 68 gozetotide PET. ( 5.1 ) Radiation risk: Ensure safe handling to protect patients and health care workers from unintentional radiation exposure. ( 2.1 , 5.2 )
Indications and Usage
Gallium Ga 68 Gozetotide Injection is indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer: with suspected metastasis who are candidates for initial definitive therapy. with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
Dosage and Administration
Use appropriate aseptic technique and radiation safety handling measures to maintain sterility during all operations involved in the manipulation and administration of Gallium Ga 68 Gozetotide Injection. ( 2.1 ) The recommended adult dose is 111 MBq to 259 MBq (3 mCi to 7 mCi) as a bolus intravenous injection. ( 2.2 ) A diuretic expected to act within the uptake time period may be administered at the time of radiotracer injection. ( 2.2 ) Initiate imaging 50 to 100 minutes after administration. The patient should void immediately prior to initiation of imaging. Scan should begin caudally and proceed cranially. ( 2.4 ) See full prescribing information for additional preparation, administration, imaging, and radiation dosimetry information. ( 2 )
Contraindications
None
Adverse Reactions
The most commonly reported adverse reactions include nausea, diarrhea, and dizziness. ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact UCLA Nuclear Medicine at 1-844-963-1855 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Description
Gallium Ga 68 Gozetotide Injection is indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer: with suspected metastasis who are candidates for initial definitive therapy. with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
Section 42229-5
Recommended Dosage
In adults, the recommended amount of radioactivity to be administered for PET is 111 MBq to 259 MBq (3 mCi to 7 mCi) administered as an intravenous bolus injection.
Section 90374-0
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of Gallium Ga 68 Gozetotide Injection was evaluated in 960 patients, each receiving one dose of Gallium Ga 68 Gozetotide Injection. The average injected activity was 188.7 ± 40.7 MBq (5.1 ± 1.1 mCi).
The most commonly reported adverse reactions were nausea, diarrhea, and dizziness, occurring at a rate of < 1%.
10 Overdosage
In the event of an overdose of Gallium Ga 68 Gozetotide Injection, reduce the radiation absorbed dose to the patient where possible by increasing the elimination of the drug from the body using hydration and frequent bladder voiding. A diuretic might also be considered. If possible, an estimate of the radiation effective dose given to the patient should be made.
8.4 Pediatric Use
The safety and effectiveness of Gallium Ga 68 Gozetotide Injection have not been established in pediatric patients.
8.5 Geriatric Use
The efficacy of Gallium Ga 68 Gozetotide Injection in geriatric patients with prostate cancer is based on data from two prospective studies [see Clinical Studies (14)] . Of the total number of subjects in these studies of Gallium Ga 68 Gozetotide Injection, 691 of 960 (72%) were 65 years of age and older (72%), While 195 (20%) were 75 years of age and older.
The efficacy and safety profiles of Gallium Ga 68 Gozetotide Injection appear similar in younger adult and geriatric patients with prostate cancer, although the number of younger adult patients in the trials was not large enough to allow definitive comparison.
14 Clinical Studies
The safety and efficacy of Gallium Ga 68 Gozetotide Injection were established in two prospective, open-label studies, PSMA-PreRP (NCT03368547 and NCT02919111) and PSMA-BCR (NCT02940262 and NCT02918357) in men with prostate cancer.
4 Contraindications
None
5.2 Radiation Risks
Gallium Ga 68 Gozetotide Injection contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk for cancer. Ensure safe handling to minimize radiation exposure to the patient and health care workers. Advise patients to hydrate before and after administration and to void frequently after administration [see Dosage and Administration (2.1, 2.3)] .
6 Adverse Reactions
The most commonly reported adverse reactions include nausea, diarrhea, and dizziness. ( 6)
To report SUSPECTED ADVERSE REACTIONS, contact UCLA Nuclear Medicine at 1-844-963-1855 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
12.2 Pharmacodynamics
The relationship between gallium Ga 68 gozetotide plasma concentrations and successful imaging was not explored in clinical trials.
2.4 Image Acquisition
Position the patient supine with arms above the head. Begin PET scanning 50 to 100 minutes after the intravenous administration of Gallium Ga 68 Gozetotide Injection. Patients should void immediately prior to image acquisition and that image acquisition should begin at the proximal thighs and proceed cranially to the skull base or skull vertex. Adapt imaging technique according to the equipment used and patient characteristics in order to obtain the best image quality possible.
1 Indications and Usage
Gallium Ga 68 Gozetotide Injection is indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy.
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
11.3 External Radiation
Table 4 displays the radiation attenuation by lead shielding of Ga 68.
| Shield Thickness (Pb) mm | Coefficient of Attenuation |
|---|---|
| 6 | 0.5 |
| 12 | 0.25 |
| 17 | 0.1 |
| 34 | 0.01 |
| 51 | 0.001 |
2.6 Radiation Dosimetry
Estimated radiation absorbed doses per injected activity for organs and tissues of adult male patients following an intravenous bolus of Gallium Ga 68 Gozetotide Injection are shown in Table 1.
The effective radiation dose resulting from the administration of 259 MBq (7 mCi) is about 4.4 mSv. The radiation doses for this administered dose to the critical organs, which are the kidneys, urinary bladder, and spleen, are 96.2 mGy, 25.4 mGy, and 16.8 mGy, respectively.
These radiation doses are for Gallium Ga 68 Gozetotide Injection alone. If CT or a transmission source are used for attenuation correction, the radiation dose will increase by an amount that varies by technique.
| Organ | Absorbed dose (mGy/MBq) | |
|---|---|---|
| Mean | SD | |
| Adrenals | 0.0156 | 0.0014 |
| Brain | 0.0104 | 0.0011 |
| Breasts | 0.0103 | 0.0011 |
| Gallbladder | 0.0157 | 0.0012 |
| Lower Colon | 0.0134 | 0.0009 |
| Small Intestine | 0.0140 | 0.0020 |
| Stomach | 0.0129 | 0.0008 |
| Heart | 0.0120 | 0.0009 |
| Kidneys | 0.3714 | 0.0922 |
| Liver | 0.0409 | 0.0076 |
| Lungs | 0.0111 | 0.0007 |
| Muscle | 0.0103 | 0.0003 |
| Pancreas | 0.0147 | 0.0009 |
| Red Marrow | 0.0114 | 0.0016 |
| Skin | 0.0091 | 0.0003 |
| Spleen | 0.0650 | 0.0180 |
| Testes | 0.0111 | 0.0006 |
| Thymus | 0.0105 | 0.0006 |
| Thyroid | 0.0104 | 0.0006 |
| Urinary Bladder | 0.0982 | 0.0286 |
| Total Body | 0.0143 | 0.0013 |
| Effective Dose (mSv/MBq) | 0.0169 | 0.0015 |
12.1 Mechanism of Action
Gallium Ga 68 gozetotide binds to prostate-specific membrane antigen (PSMA). It binds to cells that express PSMA, including malignant prostate cancer cells, which usually overexpress PSMA. Gallium-68 (Ga 68) is a β+ emitting radionuclide that allows positron emission tomography (PET).
2.5 Image Interpretation
Gallium Ga 68 Gozetotide binds to prostate-specific membrane antigen (PSMA). Based on the intensity of the signals, PET images obtained using Gallium Ga 68 Gozetotide Injection indicate the presence of PSMA in tissues. Lesions should be considered suspicious if uptake is greater than physiologic uptake in that tissue or greater than adjacent background if no physiologic uptake is expected. Tumors that do not bear PSMA will not be visualized. Increased uptake in tumors is not specific for prostate cancer [see Warnings and Precautions (5.1)] .
5.1 Risk for Misdiagnosis
Image interpretation errors can occur with gallium Ga 68 gozetotide PET. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of Gallium Ga 68 Gozetotide Injection for imaging of biochemically recurrent prostate cancer seems to be affected by serum PSA levels and by site of disease [See Clinical Studies (14)] . The performance of Gallium Ga 68 Gozetotide Injection for imaging of metastatic pelvic lymph nodes prior to initial definitive therapy seems to be affected by Gleason score [See Clinical Studies (14)] . Gallium Ga 68 gozetotide uptake is not specific for prostate cancer and may occur with other types of cancer as well as non-malignant processes such as Paget's disease, fibrous dysplasia, and osteophytosis. Clinical correlation, which may include histopathological evaluation of the suspected prostate cancer site, is recommended.
5 Warnings and Precautions
- Risk for misdiagnosis: Gallium Ga 68 gozetotide uptake can be seen in a variety of tumor types and in non-malignant processes. Image interpretation errors can occur with gallium Ga 68 gozetotide PET. ( 5.1)
- Radiation risk: Ensure safe handling to protect patients and health care workers from unintentional radiation exposure. ( 2.1, 5.2)
2 Dosage and Administration
- Use appropriate aseptic technique and radiation safety handling measures to maintain sterility during all operations involved in the manipulation and administration of Gallium Ga 68 Gozetotide Injection. ( 2.1)
- The recommended adult dose is 111 MBq to 259 MBq (3 mCi to 7 mCi) as a bolus intravenous injection. ( 2.2)
- A diuretic expected to act within the uptake time period may be administered at the time of radiotracer injection. ( 2.2)
- Initiate imaging 50 to 100 minutes after administration. The patient should void immediately prior to initiation of imaging. Scan should begin caudally and proceed cranially. ( 2.4)
- See full prescribing information for additional preparation, administration, imaging, and radiation dosimetry information. ( 2)
3 Dosage Forms and Strengths
Injection: supplied as a clear, colorless solution in a multiple-dose vial containing 18.5 MBq/mL to 185 MBq/mL (0.5 mCi/mL to 5 mCi/mL) of gallium Ga 68 gozetotide in approximately 12 mL at calibration time.
11.1 Chemical Characteristics
Gallium Ga 68 Gozetotide Injection is a radioactive diagnostic agent for intravenous administration. It contains 5 mcg gozetotide, 18.5 MBq/mL to 185 MBq/mL (0.5 mCi/mL to 5 mCi/mL) of gallium Ga 68 gozetotide at calibration time, 1 mL ethanol, 1 mL water for injection, and 10 mL of 0.9% sodium chloride solution (approximately 12 mL total volume). Gallium Ga 68 Gozetotide Injection is provided as a sterile, pyrogen free, clear, colorless solution for intravenous use, with a pH between 4.0 and 7.0. Gozetotide is also known as PSMA-11.
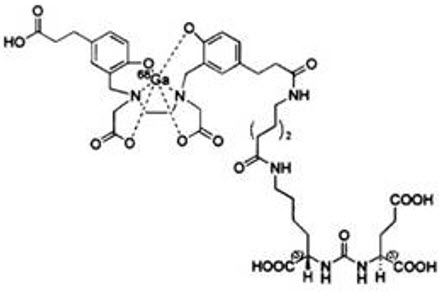
Gallium Ga 68 gozetotide is a urea based peptidomimetic that has a covalently bound chelator (HBED-CC). The peptide has the amino acid sequence Glu-NH-CO-NH-Lys(Ahx)-HBED-CC. Gallium Ga 68 gozetotide has a molecular weight of 1011.91 g/mol and its chemical structure is shown in Figure 1.
Figure 1: Chemical Structure of Gallium Ga 68 Gozetotide
11.2 Physical Characteristics
Gallium-68 (Ga 68) decays with a half-life of 68 minutes to stable zinc-68. Table 2 and Table 3 display the principal radiation emission data and physical decay of Ga 68.
| Radiation/ Emission | % Disintegration | Mean Energy
(MeV) |
|---|---|---|
| beta+ | 88% | 0.8360 |
| beta+ | 1.1% | 0.3526 |
| gamma | 178% | 0.5110 |
| gamma | 3.0% | 1.0770 |
| X-ray | 2.8% | 0.0086 |
| X-ray | 1.4% | 0.0086 |
| Minutes | Fraction Remaining |
|---|---|
| 0 | 1 |
| 15 | 0.858 |
| 30 | 0.736 |
| 60 | 0.541 |
| 90 | 0.398 |
| 120 | 0.293 |
| 180 | 0.158 |
| 240 | 0.086 |
| 360 | 0.025 |
2.1 Radiation Safety – Drug Handling
Handle Gallium Ga 68 Gozetotide Injection with appropriate safety measures to minimize radiation exposure [see Warnings and Precautions (5.2)] . Use waterproof gloves, effective radiation shielding, and other appropriate safety measures when preparing and handling Gallium Ga 68 Gozetotide Injection.
Radiopharmaceuticals should be used by or under the control of physicians who are qualified by specific training and experience in the safe use and handling of radionuclides, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radionuclides.
Principal Display Panel 12 Ml Vial Label
NDC # 76394-2642-3
Gallium Ga 68 Gozetotide Injection
Multiple-Dose Vial
18.5 MBq/mL to 185 MBq/mL (0.5 mCi/mL to 5 mCi/mL) @ EOS*
Activity @EOS*: Total ____ MBq ( _____ mCi) Volume ____ mL
Concentration: ____ MBq/mL ( ____ mCi/mL)
Expiration Date/Time: _______, _____ AM/PM
Sterile, Non-pyrogenic
Calibration (EOS*) Time: _____ AM/PM
Calibration Date: _____
Each mL contains 18.5 MBq to 185 MBq (0.5 mCi to
5 mCi) of gallium Ga 68 gozetotide @ EOS* and 8.3 mg of
sodium chloride.
Do not use if cloudy or if it contains particulate matter.
Recommended Dosage: See Prescribing Information.
*EOS = End of synthesis.
CAUTION: RADIOACTIVE MATERIAL
Diagnostic – For Intravenous Use Only.
Lot #: ________________
(Expires 3 hours after EOS*)
Store at 20° to 25°C (68° to 77°F);
excursions permitted to 15° to 30°C (59° to 86°F).
Store upright in a shielded container.
68Ga Half-life = 68 minutes.
Calculate correct dosage from date and time of
calibration.
Rx ONLY
Manufactured by: UCLA Biomedical Cyclotron, Los Angeles, CA 90095
2.3 Patient Preparation Prior to Pet Imaging
Instruct patients to drink a sufficient amount of water to ensure adequate hydration prior to administration of Gallium Ga 68 Gozetotide Injection and to continue to drink and void frequently following administration to reduce radiation exposure, particularly during the first hour after administration [see Warnings and Precautions (5.2)] .
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies were performed to evaluate the carcinogenicity potential of gallium Ga 68 gozetotide.
Structured Label Content
Section 42229-5 (42229-5)
Recommended Dosage
In adults, the recommended amount of radioactivity to be administered for PET is 111 MBq to 259 MBq (3 mCi to 7 mCi) administered as an intravenous bolus injection.
Section 90374-0 (90374-0)
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of Gallium Ga 68 Gozetotide Injection was evaluated in 960 patients, each receiving one dose of Gallium Ga 68 Gozetotide Injection. The average injected activity was 188.7 ± 40.7 MBq (5.1 ± 1.1 mCi).
The most commonly reported adverse reactions were nausea, diarrhea, and dizziness, occurring at a rate of < 1%.
10 Overdosage (10 OVERDOSAGE)
In the event of an overdose of Gallium Ga 68 Gozetotide Injection, reduce the radiation absorbed dose to the patient where possible by increasing the elimination of the drug from the body using hydration and frequent bladder voiding. A diuretic might also be considered. If possible, an estimate of the radiation effective dose given to the patient should be made.
8.4 Pediatric Use
The safety and effectiveness of Gallium Ga 68 Gozetotide Injection have not been established in pediatric patients.
8.5 Geriatric Use
The efficacy of Gallium Ga 68 Gozetotide Injection in geriatric patients with prostate cancer is based on data from two prospective studies [see Clinical Studies (14)] . Of the total number of subjects in these studies of Gallium Ga 68 Gozetotide Injection, 691 of 960 (72%) were 65 years of age and older (72%), While 195 (20%) were 75 years of age and older.
The efficacy and safety profiles of Gallium Ga 68 Gozetotide Injection appear similar in younger adult and geriatric patients with prostate cancer, although the number of younger adult patients in the trials was not large enough to allow definitive comparison.
14 Clinical Studies (14 CLINICAL STUDIES)
The safety and efficacy of Gallium Ga 68 Gozetotide Injection were established in two prospective, open-label studies, PSMA-PreRP (NCT03368547 and NCT02919111) and PSMA-BCR (NCT02940262 and NCT02918357) in men with prostate cancer.
4 Contraindications (4 CONTRAINDICATIONS)
None
5.2 Radiation Risks
Gallium Ga 68 Gozetotide Injection contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk for cancer. Ensure safe handling to minimize radiation exposure to the patient and health care workers. Advise patients to hydrate before and after administration and to void frequently after administration [see Dosage and Administration (2.1, 2.3)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most commonly reported adverse reactions include nausea, diarrhea, and dizziness. ( 6)
To report SUSPECTED ADVERSE REACTIONS, contact UCLA Nuclear Medicine at 1-844-963-1855 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
12.2 Pharmacodynamics
The relationship between gallium Ga 68 gozetotide plasma concentrations and successful imaging was not explored in clinical trials.
2.4 Image Acquisition
Position the patient supine with arms above the head. Begin PET scanning 50 to 100 minutes after the intravenous administration of Gallium Ga 68 Gozetotide Injection. Patients should void immediately prior to image acquisition and that image acquisition should begin at the proximal thighs and proceed cranially to the skull base or skull vertex. Adapt imaging technique according to the equipment used and patient characteristics in order to obtain the best image quality possible.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Gallium Ga 68 Gozetotide Injection is indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy.
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
11.3 External Radiation
Table 4 displays the radiation attenuation by lead shielding of Ga 68.
| Shield Thickness (Pb) mm | Coefficient of Attenuation |
|---|---|
| 6 | 0.5 |
| 12 | 0.25 |
| 17 | 0.1 |
| 34 | 0.01 |
| 51 | 0.001 |
2.6 Radiation Dosimetry
Estimated radiation absorbed doses per injected activity for organs and tissues of adult male patients following an intravenous bolus of Gallium Ga 68 Gozetotide Injection are shown in Table 1.
The effective radiation dose resulting from the administration of 259 MBq (7 mCi) is about 4.4 mSv. The radiation doses for this administered dose to the critical organs, which are the kidneys, urinary bladder, and spleen, are 96.2 mGy, 25.4 mGy, and 16.8 mGy, respectively.
These radiation doses are for Gallium Ga 68 Gozetotide Injection alone. If CT or a transmission source are used for attenuation correction, the radiation dose will increase by an amount that varies by technique.
| Organ | Absorbed dose (mGy/MBq) | |
|---|---|---|
| Mean | SD | |
| Adrenals | 0.0156 | 0.0014 |
| Brain | 0.0104 | 0.0011 |
| Breasts | 0.0103 | 0.0011 |
| Gallbladder | 0.0157 | 0.0012 |
| Lower Colon | 0.0134 | 0.0009 |
| Small Intestine | 0.0140 | 0.0020 |
| Stomach | 0.0129 | 0.0008 |
| Heart | 0.0120 | 0.0009 |
| Kidneys | 0.3714 | 0.0922 |
| Liver | 0.0409 | 0.0076 |
| Lungs | 0.0111 | 0.0007 |
| Muscle | 0.0103 | 0.0003 |
| Pancreas | 0.0147 | 0.0009 |
| Red Marrow | 0.0114 | 0.0016 |
| Skin | 0.0091 | 0.0003 |
| Spleen | 0.0650 | 0.0180 |
| Testes | 0.0111 | 0.0006 |
| Thymus | 0.0105 | 0.0006 |
| Thyroid | 0.0104 | 0.0006 |
| Urinary Bladder | 0.0982 | 0.0286 |
| Total Body | 0.0143 | 0.0013 |
| Effective Dose (mSv/MBq) | 0.0169 | 0.0015 |
12.1 Mechanism of Action
Gallium Ga 68 gozetotide binds to prostate-specific membrane antigen (PSMA). It binds to cells that express PSMA, including malignant prostate cancer cells, which usually overexpress PSMA. Gallium-68 (Ga 68) is a β+ emitting radionuclide that allows positron emission tomography (PET).
2.5 Image Interpretation
Gallium Ga 68 Gozetotide binds to prostate-specific membrane antigen (PSMA). Based on the intensity of the signals, PET images obtained using Gallium Ga 68 Gozetotide Injection indicate the presence of PSMA in tissues. Lesions should be considered suspicious if uptake is greater than physiologic uptake in that tissue or greater than adjacent background if no physiologic uptake is expected. Tumors that do not bear PSMA will not be visualized. Increased uptake in tumors is not specific for prostate cancer [see Warnings and Precautions (5.1)] .
5.1 Risk for Misdiagnosis
Image interpretation errors can occur with gallium Ga 68 gozetotide PET. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of Gallium Ga 68 Gozetotide Injection for imaging of biochemically recurrent prostate cancer seems to be affected by serum PSA levels and by site of disease [See Clinical Studies (14)] . The performance of Gallium Ga 68 Gozetotide Injection for imaging of metastatic pelvic lymph nodes prior to initial definitive therapy seems to be affected by Gleason score [See Clinical Studies (14)] . Gallium Ga 68 gozetotide uptake is not specific for prostate cancer and may occur with other types of cancer as well as non-malignant processes such as Paget's disease, fibrous dysplasia, and osteophytosis. Clinical correlation, which may include histopathological evaluation of the suspected prostate cancer site, is recommended.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risk for misdiagnosis: Gallium Ga 68 gozetotide uptake can be seen in a variety of tumor types and in non-malignant processes. Image interpretation errors can occur with gallium Ga 68 gozetotide PET. ( 5.1)
- Radiation risk: Ensure safe handling to protect patients and health care workers from unintentional radiation exposure. ( 2.1, 5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Use appropriate aseptic technique and radiation safety handling measures to maintain sterility during all operations involved in the manipulation and administration of Gallium Ga 68 Gozetotide Injection. ( 2.1)
- The recommended adult dose is 111 MBq to 259 MBq (3 mCi to 7 mCi) as a bolus intravenous injection. ( 2.2)
- A diuretic expected to act within the uptake time period may be administered at the time of radiotracer injection. ( 2.2)
- Initiate imaging 50 to 100 minutes after administration. The patient should void immediately prior to initiation of imaging. Scan should begin caudally and proceed cranially. ( 2.4)
- See full prescribing information for additional preparation, administration, imaging, and radiation dosimetry information. ( 2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: supplied as a clear, colorless solution in a multiple-dose vial containing 18.5 MBq/mL to 185 MBq/mL (0.5 mCi/mL to 5 mCi/mL) of gallium Ga 68 gozetotide in approximately 12 mL at calibration time.
11.1 Chemical Characteristics
Gallium Ga 68 Gozetotide Injection is a radioactive diagnostic agent for intravenous administration. It contains 5 mcg gozetotide, 18.5 MBq/mL to 185 MBq/mL (0.5 mCi/mL to 5 mCi/mL) of gallium Ga 68 gozetotide at calibration time, 1 mL ethanol, 1 mL water for injection, and 10 mL of 0.9% sodium chloride solution (approximately 12 mL total volume). Gallium Ga 68 Gozetotide Injection is provided as a sterile, pyrogen free, clear, colorless solution for intravenous use, with a pH between 4.0 and 7.0. Gozetotide is also known as PSMA-11.
Gallium Ga 68 gozetotide is a urea based peptidomimetic that has a covalently bound chelator (HBED-CC). The peptide has the amino acid sequence Glu-NH-CO-NH-Lys(Ahx)-HBED-CC. Gallium Ga 68 gozetotide has a molecular weight of 1011.91 g/mol and its chemical structure is shown in Figure 1.
Figure 1: Chemical Structure of Gallium Ga 68 Gozetotide
11.2 Physical Characteristics
Gallium-68 (Ga 68) decays with a half-life of 68 minutes to stable zinc-68. Table 2 and Table 3 display the principal radiation emission data and physical decay of Ga 68.
| Radiation/ Emission | % Disintegration | Mean Energy
(MeV) |
|---|---|---|
| beta+ | 88% | 0.8360 |
| beta+ | 1.1% | 0.3526 |
| gamma | 178% | 0.5110 |
| gamma | 3.0% | 1.0770 |
| X-ray | 2.8% | 0.0086 |
| X-ray | 1.4% | 0.0086 |
| Minutes | Fraction Remaining |
|---|---|
| 0 | 1 |
| 15 | 0.858 |
| 30 | 0.736 |
| 60 | 0.541 |
| 90 | 0.398 |
| 120 | 0.293 |
| 180 | 0.158 |
| 240 | 0.086 |
| 360 | 0.025 |
2.1 Radiation Safety – Drug Handling
Handle Gallium Ga 68 Gozetotide Injection with appropriate safety measures to minimize radiation exposure [see Warnings and Precautions (5.2)] . Use waterproof gloves, effective radiation shielding, and other appropriate safety measures when preparing and handling Gallium Ga 68 Gozetotide Injection.
Radiopharmaceuticals should be used by or under the control of physicians who are qualified by specific training and experience in the safe use and handling of radionuclides, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radionuclides.
Principal Display Panel 12 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 12 mL Vial Label)
NDC # 76394-2642-3
Gallium Ga 68 Gozetotide Injection
Multiple-Dose Vial
18.5 MBq/mL to 185 MBq/mL (0.5 mCi/mL to 5 mCi/mL) @ EOS*
Activity @EOS*: Total ____ MBq ( _____ mCi) Volume ____ mL
Concentration: ____ MBq/mL ( ____ mCi/mL)
Expiration Date/Time: _______, _____ AM/PM
Sterile, Non-pyrogenic
Calibration (EOS*) Time: _____ AM/PM
Calibration Date: _____
Each mL contains 18.5 MBq to 185 MBq (0.5 mCi to
5 mCi) of gallium Ga 68 gozetotide @ EOS* and 8.3 mg of
sodium chloride.
Do not use if cloudy or if it contains particulate matter.
Recommended Dosage: See Prescribing Information.
*EOS = End of synthesis.
CAUTION: RADIOACTIVE MATERIAL
Diagnostic – For Intravenous Use Only.
Lot #: ________________
(Expires 3 hours after EOS*)
Store at 20° to 25°C (68° to 77°F);
excursions permitted to 15° to 30°C (59° to 86°F).
Store upright in a shielded container.
68Ga Half-life = 68 minutes.
Calculate correct dosage from date and time of
calibration.
Rx ONLY
Manufactured by: UCLA Biomedical Cyclotron, Los Angeles, CA 90095
2.3 Patient Preparation Prior to Pet Imaging (2.3 Patient Preparation Prior to PET Imaging)
Instruct patients to drink a sufficient amount of water to ensure adequate hydration prior to administration of Gallium Ga 68 Gozetotide Injection and to continue to drink and void frequently following administration to reduce radiation exposure, particularly during the first hour after administration [see Warnings and Precautions (5.2)] .
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies were performed to evaluate the carcinogenicity potential of gallium Ga 68 gozetotide.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:55.436690 · Updated: 2026-03-14T22:18:01.967589