These Highlights Do Not Include All The Information Needed To Use Lofexidine Tablets Safely And Effectively. See Full Prescribing Information For Lofexidine Tablets.
77367dfa-434f-4d61-95fe-f0f1628457d1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lofexidine tablets are indicated for mitigation of opioid withdrawal symptoms to facilitate abrupt opioid discontinuation in adults.
Indications and Usage
Lofexidine tablets are indicated for mitigation of opioid withdrawal symptoms to facilitate abrupt opioid discontinuation in adults.
Dosage and Administration
The usual Lofexidine tablet dosage is three 0.18 mg tablets taken orally 4 times daily at 5- to 6-hour intervals. Lofexidine tablet treatment may be continued for up to 14 days with dosing guided by symptoms. ( 2.1 ) Discontinue Lofexidine tablets with a gradual dose reduction over 2 to 4 days. ( 2.1 ) Hepatic or Renal Impairment: Dosage adjustments are recommended based on degree of impairment. ( 2.2 , 2.3 )
Warnings and Precautions
Risk of Hypotension, Bradycardia, and Syncope : May cause a decrease in blood pressure, a decrease in pulse, and syncope. Monitor vital signs before dosing and advise patients on how to minimize the risk of these cardiovascular effects and manage symptoms, should they occur. Monitor symptoms related to bradycardia and orthostasis. When using in outpatients, ensure that patients are capable of self-monitoring for signs and symptoms. Avoid use in patients with severe coronary insufficiency, recent myocardial infarction, cerebrovascular disease, or chronic renal failure, as well as in patients with marked bradycardia. ( 5.1 ) Risk of QT Prolongation : Lofexidine tablets prolong the QT interval. Avoid use in patients with congenital long QT syndrome. Monitor ECG in patients with electrolyte abnormalities, congestive heart failure, bradyarrhythmias, hepatic or renal impairment, or in patients taking other medicinal products that lead to QT prolongation. ( 5.2 ) Increased Risk of CNS Depression with Concomitant use of CNS Depressant Drugs : Lofexidine tablets potentiate the CNS depressant effects of benzodiazepines and may potentiate the CNS depressant effects of alcohol, barbiturates, and other sedating drugs. ( 5.3 ) Increased Risk of Opioid Overdose after Opioid Discontinuation : Patients who complete opioid discontinuation are at an increased risk of fatal overdose should they resume opioid use. Use in conjunction with a comprehensive management program for treatment of opioid use disorder and inform patients and caregivers of increased risk of overdose. ( 5.4 ) Risk of Discontinuation Symptoms : Instruct patients not to discontinue therapy without consulting their healthcare provider. When discontinuing therapy, reduce dose gradually. ( 5.5 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described elsewhere in labeling: Hypotension, Bradycardia, and Syncope [see Warnings and Precautions (5.1) ] QT Prolongation [see Warnings and Precautions (5.2) ] Central Nervous System Depression [see Warnings and Precautions (5.3) ] Opioid Overdose [see Warnings and Precautions (5.4) ] Discontinuation Symptoms [see Warnings and Precautions (5.5) ]
Drug Interactions
Methadone : Methadone and Lofexidine tablets both prolong the QT interval. ECG monitoring is recommended when used concomitantly. ( 7.1 ) Oral Naltrexone : Concomitant use may reduce efficacy of oral naltrexone. ( 7.2 ) CYP2D6 Inhibitors : Concomitant use of paroxetine resulted in increased plasma levels of Lofexidine. Monitor for symptoms of orthostasis and bradycardia with concomitant use of a CYP2D6 inhibitor. ( 7.4 )
Medication Information
Warnings and Precautions
Risk of Hypotension, Bradycardia, and Syncope : May cause a decrease in blood pressure, a decrease in pulse, and syncope. Monitor vital signs before dosing and advise patients on how to minimize the risk of these cardiovascular effects and manage symptoms, should they occur. Monitor symptoms related to bradycardia and orthostasis. When using in outpatients, ensure that patients are capable of self-monitoring for signs and symptoms. Avoid use in patients with severe coronary insufficiency, recent myocardial infarction, cerebrovascular disease, or chronic renal failure, as well as in patients with marked bradycardia. ( 5.1 ) Risk of QT Prolongation : Lofexidine tablets prolong the QT interval. Avoid use in patients with congenital long QT syndrome. Monitor ECG in patients with electrolyte abnormalities, congestive heart failure, bradyarrhythmias, hepatic or renal impairment, or in patients taking other medicinal products that lead to QT prolongation. ( 5.2 ) Increased Risk of CNS Depression with Concomitant use of CNS Depressant Drugs : Lofexidine tablets potentiate the CNS depressant effects of benzodiazepines and may potentiate the CNS depressant effects of alcohol, barbiturates, and other sedating drugs. ( 5.3 ) Increased Risk of Opioid Overdose after Opioid Discontinuation : Patients who complete opioid discontinuation are at an increased risk of fatal overdose should they resume opioid use. Use in conjunction with a comprehensive management program for treatment of opioid use disorder and inform patients and caregivers of increased risk of overdose. ( 5.4 ) Risk of Discontinuation Symptoms : Instruct patients not to discontinue therapy without consulting their healthcare provider. When discontinuing therapy, reduce dose gradually. ( 5.5 )
Indications and Usage
Lofexidine tablets are indicated for mitigation of opioid withdrawal symptoms to facilitate abrupt opioid discontinuation in adults.
Dosage and Administration
The usual Lofexidine tablet dosage is three 0.18 mg tablets taken orally 4 times daily at 5- to 6-hour intervals. Lofexidine tablet treatment may be continued for up to 14 days with dosing guided by symptoms. ( 2.1 ) Discontinue Lofexidine tablets with a gradual dose reduction over 2 to 4 days. ( 2.1 ) Hepatic or Renal Impairment: Dosage adjustments are recommended based on degree of impairment. ( 2.2 , 2.3 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described elsewhere in labeling: Hypotension, Bradycardia, and Syncope [see Warnings and Precautions (5.1) ] QT Prolongation [see Warnings and Precautions (5.2) ] Central Nervous System Depression [see Warnings and Precautions (5.3) ] Opioid Overdose [see Warnings and Precautions (5.4) ] Discontinuation Symptoms [see Warnings and Precautions (5.5) ]
Drug Interactions
Methadone : Methadone and Lofexidine tablets both prolong the QT interval. ECG monitoring is recommended when used concomitantly. ( 7.1 ) Oral Naltrexone : Concomitant use may reduce efficacy of oral naltrexone. ( 7.2 ) CYP2D6 Inhibitors : Concomitant use of paroxetine resulted in increased plasma levels of Lofexidine. Monitor for symptoms of orthostasis and bradycardia with concomitant use of a CYP2D6 inhibitor. ( 7.4 )
Description
Lofexidine tablets are indicated for mitigation of opioid withdrawal symptoms to facilitate abrupt opioid discontinuation in adults.
Section 42229-5
Blood pressure changes and adverse reactions after Lofexidine tablets cessation
Elevations in blood pressure above normal values (≥140 mmHg systolic) and above a subject's pre-treatment baseline are associated with discontinuing Lofexidine tablets, and peaked on the second day after discontinuation, as shown in Table 4. Blood pressure values were evaluated for 3 days following the last dose of a 5-day course of Lofexidine tablets 2.88 mg/day.
| Abrupt Lofexidine Tablet Discontinuation 2.88 mg (N = 134) |
Placebo (N = 129) |
|||
|---|---|---|---|---|
| N at risk | n (%) | N at risk | n (%) | |
| Systolic Blood Pressure on Day 2 after Discontinuation | ||||
| ≥ 140 mmHg and ≥ 20 mmHg increase from baseline | 58 | 23 (39.7) | 37 | 6 (16.2) |
| ≥ 170 mmHg and ≥ 20 mmHg increase from baseline | 58 | 5 (8.6) | 37 | 0 |
Blood pressure elevations of a similar magnitude and incidence were observed in a small number of patients (N=10) that had a one-day, 50% dose reduction prior to discontinuation.
After stopping treatment, subjects who were taking Lofexidine tablets also had a higher incidence of diarrhea, insomnia, anxiety, chills, hyperhidrosis, and extremity pain compared to subjects who were taking placebo.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. |
Issued: 08/2023 |
|
PATIENT INFORMATION
LOFEXIDINE TABLETS (loe fex' I deen) |
|
| What is the most important information I should know about Lofexidine Tablets and discontinuing opioid drugs? | |
| Lofexidine Tablets can cause serious side effects, including low blood pressure (hypotension), slow heart rate (bradycardia), and fainting. | |
| If you have any of the following signs or symptoms, tell your healthcare provider right away: | |
|
|
| If you take Lofexidine tablets at home and have any of these signs and symptoms, do not take your next dose of Lofexidine tablets until you have talked to your healthcare provider. You should avoid becoming dehydrated or overheated during treatment with Lofexidine tablets, which may increase your risk of low blood pressure and fainting. You should also be careful not to stand up too suddenly from lying down or sitting. | |
| When your treatment is complete you will need to stop taking Lofexidine tablets gradually or your blood pressure could increase. For more information about side effects, see "What are the possible side effects of Lofexidine Tablets?" | |
| Increased risk of opioid overdose. After a period of time of not using opioid drugs, you can become more sensitive to the effects of opioids if you start using opioids again. This may increase your risk of overdose and death. | |
| What are Lofexidine Tablets? | |
| Lofexidine tablets are a non-opioid prescription medicine used in adults to help with the symptoms of opioid withdrawal that may happen when you stop taking an opioid suddenly. | |
| Lofexidine tablets will not completely prevent the symptoms of opioid withdrawal, which may include feeling sick, stomach cramps, muscle spasms or twitching, feeling of cold, heart pounding, muscular tension, aches and pains, yawning, runny eyes and sleep problems (insomnia). | |
| Lofexidine tablets are not a treatment for opioid use disorder. If you have been diagnosed with opioid use disorder (opioid addiction), your healthcare provider may prescribe Lofexidine tablets as part of a complete treatment program for your opioid use disorder (opioid addiction). | |
| It is not known if Lofexidine tablets are safe and effective in children. | |
Before taking Lofexidine Tablets, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, herbal supplements, and any medications you may take for the individual symptoms of opioid withdrawal (such as pain relievers or medications for upset stomach). | |
| Especially tell your healthcare provider if you take benzodiazepines, barbiturates, tranquilizers, or sleeping pills. Taking Lofexidine tablets with these medicines can cause serious side effects. Ask your healthcare provider or pharmacist if you are not sure if you are taking any of these medicines. | |
How should I take Lofexidine Tablets?
|
|
| What should I avoid while taking Lofexidine Tablets? | |
| Do not drive, operate heavy machinery, or perform any other dangerous activities until you know how Lofexidine tablets affect you. | |
| What are the possible side effects of Lofexidine Tablets? | |
| The most common side effects of Lofexidine tablets include: | |
|
|
| These are not all the possible side effects of Lofexidine tablets. | |
| Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to US WorldMeds at 1-888-900-8796. | |
How should I store Lofexidine Tablets?
|
|
| Keep Lofexidine tablets and all medicines out of the reach of children. | |
| General information about the safe and effective use of Lofexidine Tablets. | |
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Lofexidine tablets for a condition for which it was not prescribed. Do not give Lofexidine tablets to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Lofexidine tablets that is written for health professionals. | |
| What are the ingredients of Lofexidine Tablets? | |
| Active ingredient: lofexidine. | |
| Inactive ingredients: lactose, citric acid, povidone, microcrystalline cellulose, calcium stearate, sodium lauryl sulphate, and Opadry OY S 9480 (contains indigo carmine and sunset yellow). | |
| Manufactured for: Prasco Laboratories Mason, OH 45040 USA |
|
| For more information, call 1-888-900-8796 Under Sublicense from USWM, LLC. |
640113 |
Section 44425-7
Storage
Store in original container at controlled room temperature, 25°C (77°F); with excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Keep Lofexidine tablets away from excess heat and moisture both in the pharmacy and after dispensing. Do not remove desiccant packs from bottles until all tablets are used. Keep Lofexidine tablets and all medicines out of the reach of children.
10 Overdosage
Overdose with Lofexidine tablets may manifest as hypotension, bradycardia, and sedation. In the event of acute overdose, perform gastric lavage where appropriate. Dialysis will not remove a substantial portion of the drug. Initiate general symptomatic and supportive measures in cases of overdosage.
7.1 Methadone
Lofexidine tablets and methadone both prolong the QT interval. ECG monitoring is recommended in patients receiving methadone and Lofexidine tablets concomitantly [see Warnings and Precautions (5.2), Clinical Pharmacology (12.3)].
11 Description
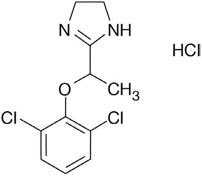
Lofexidine tablets contain lofexidine, a central alpha-2 adrenergic agonist, as the hydrochloride salt. Lofexidine hydrochloride is chemically designated as 2-[1-(2,6-dichlorophenoxy)ethyl]-4,5 dihydro-1H- imidazole monohydrochloride with a molecular formula of C11H12Cl2N2O∙HCl. Its molecular weight is 295.6 g/mole and its structural formula is:
Lofexidine hydrochloride is a white to off-white crystalline powder freely soluble in water, methanol, and ethanol. It is slightly soluble in chloroform and practically insoluble in n-hexane and benzene.
Lofexidine tablets are available as round, convex-shaped, peach-colored, film-coated tablets for oral administration. Each tablet contains 0.18 lofexidine, equivalent to 0.2 mg of lofexidine hydrochloride, and the following inactive ingredients: 92.6 mg lactose, 12.3 mg citric acid, 1.1 mg povidone, 5.7 mg microcrystalline cellulose, 1.4 mg calcium stearate, 0.7 mg sodium lauryl sulphate, and Opadry OY S 9480 (contains indigo carmine and sunset yellow).
8.4 Pediatric Use
The safety and effectiveness of Lofexidine tablets have not been established in pediatric patients.
8.5 Geriatric Use
No studies have been performed to characterize the pharmacokinetics of Lofexidine tablets or to establish its safety and effectiveness in geriatric patients. Caution should be exercised when Lofexidine tablets are administered to patients over 65 years of age. Dosing adjustments similar to those recommended in patients with renal impairment should be considered [see Dosage and Administration (2.3), Use in Specific Populations (8.7)].
14 Clinical Studies
Two randomized, double-blind, placebo-controlled trials supported the efficacy of lofexidine.
4 Contraindications
None.
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in labeling:
- Hypotension, Bradycardia, and Syncope [see Warnings and Precautions (5.1)]
- QT Prolongation [see Warnings and Precautions (5.2)]
- Central Nervous System Depression [see Warnings and Precautions (5.3)]
- Opioid Overdose [see Warnings and Precautions (5.4)]
- Discontinuation Symptoms [see Warnings and Precautions (5.5)]
7 Drug Interactions
- Methadone: Methadone and Lofexidine tablets both prolong the QT interval. ECG monitoring is recommended when used concomitantly. (7.1)
- Oral Naltrexone: Concomitant use may reduce efficacy of oral naltrexone. (7.2)
- CYP2D6 Inhibitors: Concomitant use of paroxetine resulted in increased plasma levels of Lofexidine. Monitor for symptoms of orthostasis and bradycardia with concomitant use of a CYP2D6 inhibitor. (7.4)
7.2 Oral Naltrexone
Coadministration of Lofexidine tablets and oral naltrexone resulted in statistically significant differences in the steady-state pharmacokinetics of naltrexone. It is possible that oral naltrexone efficacy may be reduced if used concomitantly within 2 hours of Lofexidine tablets. This interaction is not expected if naltrexone is administered by non-oral routes [see Clinical Pharmacology (12.3)].
8.7 Renal Impairment
Renal impairment slows the elimination of lofexidine but exhibits less effect on the peak plasma concentration than on AUC values following a single dose. Dosage adjustments are recommended based on the degree of renal impairment [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)].
Only a negligible fraction of the Lofexidine tablet dose is removed during a typical dialysis session, so no additional dose needs to be administered after a dialysis session; Lofexidine tablets may be administered without regard to the timing of dialysis [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)].
Clinically relevant QT prolongation may occur in subjects with renal impairment [see Warnings and Precautions (5.2), Clinical Pharmacology (12.2)].
2.1 Dosing Information
The usual Lofexidine tablet starting dosage is three 0.18 mg tablets taken orally 4 times daily during the period of peak withdrawal symptoms (generally the first 5 to 7 days following last use of opioid) with dosing guided by symptoms and side effects. There should be 5 to 6 hours between each dose. The total daily dosage of Lofexidine tablets should not exceed 2.88 mg (16 tablets) and no single dose should exceed 0.72 mg (4 tablets).
Lofexidine tablet treatment may be continued for up to 14 days with dosing guided by symptoms.
Discontinue Lofexidine tablets with a gradual dose reduction over a 2- to 4-day period to mitigate Lofexidine tablet withdrawal symptoms (e.g., reducing by 1 tablet per dose every 1 to 2 days) [see Warnings & Precautions (5.5)]. The Lofexidine tablet dose should be reduced, held, or discontinued for individuals who demonstrate a greater sensitivity to Lofexidine tablets side effects [see Warnings and Precautions (5.1), Adverse Reactions (6.1)]. Lower doses may be appropriate as opioid withdrawal symptoms wane.
Lofexidine tablets can be administered in the presence or absence of food.
8.6 Hepatic Impairment
Hepatic impairment slows the elimination of lofexidine but exhibits less effect on the peak plasma concentration than on AUC values following a single dose. Dosage adjustments are recommended based on the degree of hepatic impairment. [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
Clinically relevant QT prolongation may occur in subjects with hepatic impairment [see Warnings and Precautions (5.2), Clinical Pharmacology (12.2)].
1 Indications and Usage
Lofexidine tablets are indicated for mitigation of opioid withdrawal symptoms to facilitate abrupt opioid discontinuation in adults.
12.1 Mechanism of Action
Lofexidine is a central alpha-2 adrenergic agonist that binds to receptors on adrenergic neurons. This reduces the release of norepinephrine and decreases sympathetic tone.
7.3 Cns Depressant Drugs
Lofexidine tablets potentiate the CNS depressant effects of benzodiazepines and may potentiate the CNS depressant effects of alcohol, barbiturates, and other sedating drugs. Advise patients to inform their healthcare provider of other medications they are taking, including alcohol [see Warnings and Precautions (5.3)].
5 Warnings and Precautions
- Risk of Hypotension, Bradycardia, and Syncope: May cause a decrease in blood pressure, a decrease in pulse, and syncope. Monitor vital signs before dosing and advise patients on how to minimize the risk of these cardiovascular effects and manage symptoms, should they occur. Monitor symptoms related to bradycardia and orthostasis. When using in outpatients, ensure that patients are capable of self-monitoring for signs and symptoms. Avoid use in patients with severe coronary insufficiency, recent myocardial infarction, cerebrovascular disease, or chronic renal failure, as well as in patients with marked bradycardia. (5.1)
- Risk of QT Prolongation: Lofexidine tablets prolong the QT interval. Avoid use in patients with congenital long QT syndrome. Monitor ECG in patients with electrolyte abnormalities, congestive heart failure, bradyarrhythmias, hepatic or renal impairment, or in patients taking other medicinal products that lead to QT prolongation. (5.2)
- Increased Risk of CNS Depression with Concomitant use of CNS Depressant Drugs: Lofexidine tablets potentiate the CNS depressant effects of benzodiazepines and may potentiate the CNS depressant effects of alcohol, barbiturates, and other sedating drugs. (5.3)
- Increased Risk of Opioid Overdose after Opioid Discontinuation: Patients who complete opioid discontinuation are at an increased risk of fatal overdose should they resume opioid use. Use in conjunction with a comprehensive management program for treatment of opioid use disorder and inform patients and caregivers of increased risk of overdose. (5.4)
- Risk of Discontinuation Symptoms: Instruct patients not to discontinue therapy without consulting their healthcare provider. When discontinuing therapy, reduce dose gradually. (5.5)
2 Dosage and Administration
- The usual Lofexidine tablet dosage is three 0.18 mg tablets taken orally 4 times daily at 5- to 6-hour intervals. Lofexidine tablet treatment may be continued for up to 14 days with dosing guided by symptoms. (2.1)
- Discontinue Lofexidine tablets with a gradual dose reduction over 2 to 4 days. (2.1)
- Hepatic or Renal Impairment: Dosage adjustments are recommended based on degree of impairment. (2.2, 2.3)
5.2 Risk of Qt Prolongation
Lofexidine tablets prolong the QT interval.
Avoid using Lofexidine tablets in patients with congenital long QT syndrome.
Monitor ECG in patients with congestive heart failure, bradyarrhythmias, hepatic impairment, renal impairment, or patients taking other medicinal products that lead to QT prolongation (e.g., methadone). In patients with electrolyte abnormalities (e.g., hypokalemia or hypomagnesemia), correct these abnormalities first, and monitor ECG upon initiation of Lofexidine tablets [see Dosing and Administration (2.1), Adverse Reactions (6.1), Special Populations (8.6, 8.7), Clinical Pharmacology (12.2)].
3 Dosage Forms and Strengths
Lofexidine tablets are available as round, peach-colored, film-coated tablets, imprinted with "LFX" on one side and "18" on the other side. Each tablet contains 0.18 mg lofexidine (equivalent to 0.2 mg of lofexidine hydrochloride).
6.2 Postmarketing Experience
Lofexidine is marketed in other countries for relief of opioid withdrawal symptoms. The following events have been identified during postmarketing use of lofexidine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Since lofexidine's initial market introduction in 1992, the most frequently reported postmarketing adverse event with lofexidine has been hypotension [see Warnings and Precautions (5.1)]. There has been one report of QT prolongation, bradycardia, torsades de pointes, and cardiac arrest with successful resuscitation in a patient who received lofexidine, and three reports of clinically significant QT prolongation in subjects concurrently receiving methadone with lofexidine.
8.8 Cyp2d6 Poor Metabolizers
Although the pharmacokinetics of Lofexidine tablets have not been systematically evaluated in patients who do not express the drug metabolizing enzyme CYP2D6, it is likely that the exposure to Lofexidine tablets would be increased similarly to taking strong CYP2D6 inhibitors (approximately 28%). Monitor adverse events such as orthostatic hypotension and bradycardia in known CYP2D6 poor metabolizers. Approximately 8% of Caucasians and 3 to 8% of Black/African Americans cannot metabolize CYP2D6 substrates and are classified as poor metabolizers (PM) [see Clinical Pharmacology (12.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to adverse reaction rates observed for another drug and may not reflect the rates observed in practice.
The safety of Lofexidine tablets was supported by three randomized, double-blind, placebo-controlled clinical trials, an open-label study, and clinical pharmacology studies with concomitant administration of either methadone, buprenorphine, or naltrexone.
The three randomized, double-blind, placebo-controlled clinical trials enrolled 935 subjects dependent on short-acting opioids undergoing abrupt opioid withdrawal. Patients were monitored before each dose in an inpatient setting.
Table 3 presents the incidence, rounded to the nearest percent, of adverse events that occurred in at least 10% of subjects treated with Lofexidine tablets and for which the incidence in patients treated with Lofexidine tablets were greater than the incidence in subjects treated with placebo in a study that tested two doses of Lofexidine tablets, 2.16 mg per day and 2.88 mg per day, and placebo. The overall safety profile in the combined dataset was similar.
Orthostatic hypotension, bradycardia, hypotension, dizziness, somnolence, sedation, and dry mouth were notably more common in subjects treated with Lofexidine tablets than subjects treated with placebo.
| Adverse Reaction | Lofexidine Tablets 2.16 mg Assigned dose; mean average daily dose received was 79% of assigned dose due to dose-holds for out-of-range vital signs. (%)N=229 |
Lofexidine Tablets 2.88 mg (%) N=222 |
Placebo (%) N=151 |
|---|---|---|---|
| Insomnia | 51 | 55 | 48 |
| Orthostatic Hypotension | 29 | 42 | 5 |
| Bradycardia | 24 | 32 | 5 |
| Hypotension | 30 | 30 | 1 |
| Dizziness | 19 | 23 | 3 |
| Somnolence | 11 | 13 | 5 |
| Sedation | 13 | 12 | 5 |
| Dry Mouth | 10 | 11 | 0 |
Other notable adverse reactions associated with the use of Lofexidine tablets but reported in <10% of patients in the Lofexidine tablets group included:
- Syncope: 0.9%, 1.4% and 0% for Lofexidine tablets 2.16 mg/day and 2.88 mg/day and placebo, respectively
- Tinnitus: 0.9%, 3.2% and 0% for Lofexidine tablets 2.16 mg/day and 2.88 mg/day and placebo, respectively
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Patient Information).
Lofexidine tablets may mitigate, but not completely prevent, the symptoms associated with opioid withdrawal syndrome, which may include feeling sick, stomach cramps, muscle spasms or twitching, feeling of cold, heart pounding, muscular tension, aches and pains, yawning, runny eyes and sleep problems (insomnia). Patients should be advised that withdrawal will not be easy. Additional supportive measures should be clearly advised, as needed.
7.4 Cyp2d6 Inhibitor Paroxetine
Coadministration of Lofexidine tablets and paroxetine resulted in a 28% increase in the extent of absorption of Lofexidine. Monitor for orthostatic hypotension and bradycardia when an inhibitor of CYP2D6 is used concomitantly with Lofexidine tablets [see Clinical Pharmacology (12.3)].
5.5 Risk of Discontinuation Symptoms
Stopping Lofexidine tablets abruptly can cause a marked rise in blood pressure. Symptoms including diarrhea, insomnia, anxiety, chills, hyperhidrosis, and extremity pain have also been observed with Lofexidine tablets discontinuation. Instruct patients not to discontinue therapy without consulting their healthcare provider. When discontinuing therapy with Lofexidine tablets, gradually reduce the dose [see Dosing and Administration (2.1)].
Symptoms related to discontinuation can be managed by administration of the previous Lofexidine tablet dose and subsequent taper.
8.3 Females and Males of Reproductive Potential
In animal studies that included some fertility endpoints, lofexidine decreased breeding rate and increased resorptions at exposures below human exposures. The impact of lofexidine on male fertility has not been adequately characterized in animal studies [see Impairment of Fertility (13.1)].
5.1 Risk of Hypotension, Bradycardia, and Syncope
Lofexidine tablets can cause a decrease in blood pressure, a decrease in pulse, and syncope [see Adverse Reactions (6.1), Clinical Pharmacology (12.2)]. Monitor vital signs before dosing. Monitor symptoms related to bradycardia and orthostasis.
Patients being given Lofexidine tablets in an outpatient setting should be capable of and instructed on self-monitoring for hypotension, orthostasis, bradycardia, and associated symptoms. If clinically significant or symptomatic hypotension and/or bradycardia occur, the next dose of Lofexidine tablets should be reduced in amount, delayed, or skipped.
Inform patients that Lofexidine tablets may cause hypotension and that patients moving from a supine to an upright position may be at increased risk for hypotension and orthostatic effects. Instruct patients to stay hydrated, on how to recognize symptoms of low blood pressure, and on how to reduce the risk of serious consequences should hypotension occur (e.g., sit or lie down, carefully rise from a sitting or lying position). Instruct outpatients to withhold Lofexidine tablets doses when experiencing symptoms of hypotension or bradycardia and to contact their healthcare provider for guidance on how to adjust dosing.
Avoid using Lofexidine tablets in patients with severe coronary insufficiency, recent myocardial infarction, cerebrovascular disease, chronic renal failure, and in patients with marked bradycardia.
Avoid using Lofexidine tablets in combination with medications that decrease pulse or blood pressure to avoid the risk of excessive bradycardia and hypotension.
Principal Display Panel 0.18 Mg Tablet Carton 36 Count
NDC 66993-345-37
36 Tablets
Lofexidine Tablets
0.18 mg
Store and dispense
in
original container.
Protect from heat and moisture.
Do not remove desiccants.
Keep the bottle tightly closed.
Rx Only
Keep out of reach of children.
Principal Display Panel 0.18 Mg Tablet Carton 96 Count
NDC 66993-345-76
96 Tablets
Lofexidine Tablets
0.18 mg
Store and dispense
in
original container.
Protect from heat and moisture.
Do not remove desiccants.
Keep the bottle tightly closed.
Rx Only
Keep out of reach of children.
2.3 Dosage Recommendations for Patients With Renal Impairment
Recommended dosage adjustments based on the degree of renal impairment are shown in Table 2. Lofexidine tablets may be administered without regard to the timing of dialysis [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
| Moderate Impairment | Severe Impairment, End-Stage Renal Disease, or on Dialysis | |
|---|---|---|
| Estimated GFR, mL/min/1.73 m2 | 30-89.9 | < 30 |
| Recommended dose | 2 tablets 4 times daily (1.44 mg per day) |
1 tablet 4 times daily (0.72 mg per day) |
2.2 Dosage Recommendations for Patients With Hepatic Impairment
Recommended dosage adjustments based on the degree of hepatic impairment are shown in Table 1. [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
| Mild Impairment | Moderate Impairment | Severe Impairment | |
|---|---|---|---|
| Child-Pugh score | 5-6 | 7-9 | > 9 |
| Recommended dose | 3 tablets 4 times daily (2.16 mg per day) |
2 tablets 4 times daily (1.44 mg per day) |
1 tablet 4 times daily (0.72 mg per day) |
5.4 Increased Risk of Opioid Overdose After Opioid Discontinuation
Lofexidine tablets are not a treatment for opioid use disorder. Patients who complete opioid discontinuation are likely to have a reduced tolerance to opioids and are at increased risk of fatal overdose should they resume opioid use. Use Lofexidine tablets in patients with opioid use disorder only in conjunction with a comprehensive management program for the treatment of opioid use disorder and inform patients and caregivers of this increased risk of overdose.
5.3 Increased Risk of Central Nervous System Depression With Concomitant Use of Cns Depressant Drugs
Lofexidine tablets potentiate the CNS depressive effects of benzodiazepines and can also be expected to potentiate the CNS depressive effects of alcohol, barbiturates, and other sedating drugs. Advise patients to inform their healthcare provider of other medications they are taking, including alcohol.
Advise patients using Lofexidine tablets in an outpatient setting that, until they learn how they respond to Lofexidine tablets, they should be careful or avoid doing activities such as driving or operating heavy machinery.
Structured Label Content
Section 42229-5 (42229-5)
Blood pressure changes and adverse reactions after Lofexidine tablets cessation
Elevations in blood pressure above normal values (≥140 mmHg systolic) and above a subject's pre-treatment baseline are associated with discontinuing Lofexidine tablets, and peaked on the second day after discontinuation, as shown in Table 4. Blood pressure values were evaluated for 3 days following the last dose of a 5-day course of Lofexidine tablets 2.88 mg/day.
| Abrupt Lofexidine Tablet Discontinuation 2.88 mg (N = 134) |
Placebo (N = 129) |
|||
|---|---|---|---|---|
| N at risk | n (%) | N at risk | n (%) | |
| Systolic Blood Pressure on Day 2 after Discontinuation | ||||
| ≥ 140 mmHg and ≥ 20 mmHg increase from baseline | 58 | 23 (39.7) | 37 | 6 (16.2) |
| ≥ 170 mmHg and ≥ 20 mmHg increase from baseline | 58 | 5 (8.6) | 37 | 0 |
Blood pressure elevations of a similar magnitude and incidence were observed in a small number of patients (N=10) that had a one-day, 50% dose reduction prior to discontinuation.
After stopping treatment, subjects who were taking Lofexidine tablets also had a higher incidence of diarrhea, insomnia, anxiety, chills, hyperhidrosis, and extremity pain compared to subjects who were taking placebo.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. |
Issued: 08/2023 |
|
PATIENT INFORMATION
LOFEXIDINE TABLETS (loe fex' I deen) |
|
| What is the most important information I should know about Lofexidine Tablets and discontinuing opioid drugs? | |
| Lofexidine Tablets can cause serious side effects, including low blood pressure (hypotension), slow heart rate (bradycardia), and fainting. | |
| If you have any of the following signs or symptoms, tell your healthcare provider right away: | |
|
|
| If you take Lofexidine tablets at home and have any of these signs and symptoms, do not take your next dose of Lofexidine tablets until you have talked to your healthcare provider. You should avoid becoming dehydrated or overheated during treatment with Lofexidine tablets, which may increase your risk of low blood pressure and fainting. You should also be careful not to stand up too suddenly from lying down or sitting. | |
| When your treatment is complete you will need to stop taking Lofexidine tablets gradually or your blood pressure could increase. For more information about side effects, see "What are the possible side effects of Lofexidine Tablets?" | |
| Increased risk of opioid overdose. After a period of time of not using opioid drugs, you can become more sensitive to the effects of opioids if you start using opioids again. This may increase your risk of overdose and death. | |
| What are Lofexidine Tablets? | |
| Lofexidine tablets are a non-opioid prescription medicine used in adults to help with the symptoms of opioid withdrawal that may happen when you stop taking an opioid suddenly. | |
| Lofexidine tablets will not completely prevent the symptoms of opioid withdrawal, which may include feeling sick, stomach cramps, muscle spasms or twitching, feeling of cold, heart pounding, muscular tension, aches and pains, yawning, runny eyes and sleep problems (insomnia). | |
| Lofexidine tablets are not a treatment for opioid use disorder. If you have been diagnosed with opioid use disorder (opioid addiction), your healthcare provider may prescribe Lofexidine tablets as part of a complete treatment program for your opioid use disorder (opioid addiction). | |
| It is not known if Lofexidine tablets are safe and effective in children. | |
Before taking Lofexidine Tablets, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, herbal supplements, and any medications you may take for the individual symptoms of opioid withdrawal (such as pain relievers or medications for upset stomach). | |
| Especially tell your healthcare provider if you take benzodiazepines, barbiturates, tranquilizers, or sleeping pills. Taking Lofexidine tablets with these medicines can cause serious side effects. Ask your healthcare provider or pharmacist if you are not sure if you are taking any of these medicines. | |
How should I take Lofexidine Tablets?
|
|
| What should I avoid while taking Lofexidine Tablets? | |
| Do not drive, operate heavy machinery, or perform any other dangerous activities until you know how Lofexidine tablets affect you. | |
| What are the possible side effects of Lofexidine Tablets? | |
| The most common side effects of Lofexidine tablets include: | |
|
|
| These are not all the possible side effects of Lofexidine tablets. | |
| Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to US WorldMeds at 1-888-900-8796. | |
How should I store Lofexidine Tablets?
|
|
| Keep Lofexidine tablets and all medicines out of the reach of children. | |
| General information about the safe and effective use of Lofexidine Tablets. | |
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Lofexidine tablets for a condition for which it was not prescribed. Do not give Lofexidine tablets to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Lofexidine tablets that is written for health professionals. | |
| What are the ingredients of Lofexidine Tablets? | |
| Active ingredient: lofexidine. | |
| Inactive ingredients: lactose, citric acid, povidone, microcrystalline cellulose, calcium stearate, sodium lauryl sulphate, and Opadry OY S 9480 (contains indigo carmine and sunset yellow). | |
| Manufactured for: Prasco Laboratories Mason, OH 45040 USA |
|
| For more information, call 1-888-900-8796 Under Sublicense from USWM, LLC. |
640113 |
Section 44425-7 (44425-7)
Storage
Store in original container at controlled room temperature, 25°C (77°F); with excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Keep Lofexidine tablets away from excess heat and moisture both in the pharmacy and after dispensing. Do not remove desiccant packs from bottles until all tablets are used. Keep Lofexidine tablets and all medicines out of the reach of children.
10 Overdosage (10 OVERDOSAGE)
Overdose with Lofexidine tablets may manifest as hypotension, bradycardia, and sedation. In the event of acute overdose, perform gastric lavage where appropriate. Dialysis will not remove a substantial portion of the drug. Initiate general symptomatic and supportive measures in cases of overdosage.
7.1 Methadone
Lofexidine tablets and methadone both prolong the QT interval. ECG monitoring is recommended in patients receiving methadone and Lofexidine tablets concomitantly [see Warnings and Precautions (5.2), Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
Lofexidine tablets contain lofexidine, a central alpha-2 adrenergic agonist, as the hydrochloride salt. Lofexidine hydrochloride is chemically designated as 2-[1-(2,6-dichlorophenoxy)ethyl]-4,5 dihydro-1H- imidazole monohydrochloride with a molecular formula of C11H12Cl2N2O∙HCl. Its molecular weight is 295.6 g/mole and its structural formula is:
Lofexidine hydrochloride is a white to off-white crystalline powder freely soluble in water, methanol, and ethanol. It is slightly soluble in chloroform and practically insoluble in n-hexane and benzene.
Lofexidine tablets are available as round, convex-shaped, peach-colored, film-coated tablets for oral administration. Each tablet contains 0.18 lofexidine, equivalent to 0.2 mg of lofexidine hydrochloride, and the following inactive ingredients: 92.6 mg lactose, 12.3 mg citric acid, 1.1 mg povidone, 5.7 mg microcrystalline cellulose, 1.4 mg calcium stearate, 0.7 mg sodium lauryl sulphate, and Opadry OY S 9480 (contains indigo carmine and sunset yellow).
8.4 Pediatric Use
The safety and effectiveness of Lofexidine tablets have not been established in pediatric patients.
8.5 Geriatric Use
No studies have been performed to characterize the pharmacokinetics of Lofexidine tablets or to establish its safety and effectiveness in geriatric patients. Caution should be exercised when Lofexidine tablets are administered to patients over 65 years of age. Dosing adjustments similar to those recommended in patients with renal impairment should be considered [see Dosage and Administration (2.3), Use in Specific Populations (8.7)].
14 Clinical Studies (14 CLINICAL STUDIES)
Two randomized, double-blind, placebo-controlled trials supported the efficacy of lofexidine.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in labeling:
- Hypotension, Bradycardia, and Syncope [see Warnings and Precautions (5.1)]
- QT Prolongation [see Warnings and Precautions (5.2)]
- Central Nervous System Depression [see Warnings and Precautions (5.3)]
- Opioid Overdose [see Warnings and Precautions (5.4)]
- Discontinuation Symptoms [see Warnings and Precautions (5.5)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Methadone: Methadone and Lofexidine tablets both prolong the QT interval. ECG monitoring is recommended when used concomitantly. (7.1)
- Oral Naltrexone: Concomitant use may reduce efficacy of oral naltrexone. (7.2)
- CYP2D6 Inhibitors: Concomitant use of paroxetine resulted in increased plasma levels of Lofexidine. Monitor for symptoms of orthostasis and bradycardia with concomitant use of a CYP2D6 inhibitor. (7.4)
7.2 Oral Naltrexone
Coadministration of Lofexidine tablets and oral naltrexone resulted in statistically significant differences in the steady-state pharmacokinetics of naltrexone. It is possible that oral naltrexone efficacy may be reduced if used concomitantly within 2 hours of Lofexidine tablets. This interaction is not expected if naltrexone is administered by non-oral routes [see Clinical Pharmacology (12.3)].
8.7 Renal Impairment
Renal impairment slows the elimination of lofexidine but exhibits less effect on the peak plasma concentration than on AUC values following a single dose. Dosage adjustments are recommended based on the degree of renal impairment [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)].
Only a negligible fraction of the Lofexidine tablet dose is removed during a typical dialysis session, so no additional dose needs to be administered after a dialysis session; Lofexidine tablets may be administered without regard to the timing of dialysis [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)].
Clinically relevant QT prolongation may occur in subjects with renal impairment [see Warnings and Precautions (5.2), Clinical Pharmacology (12.2)].
2.1 Dosing Information
The usual Lofexidine tablet starting dosage is three 0.18 mg tablets taken orally 4 times daily during the period of peak withdrawal symptoms (generally the first 5 to 7 days following last use of opioid) with dosing guided by symptoms and side effects. There should be 5 to 6 hours between each dose. The total daily dosage of Lofexidine tablets should not exceed 2.88 mg (16 tablets) and no single dose should exceed 0.72 mg (4 tablets).
Lofexidine tablet treatment may be continued for up to 14 days with dosing guided by symptoms.
Discontinue Lofexidine tablets with a gradual dose reduction over a 2- to 4-day period to mitigate Lofexidine tablet withdrawal symptoms (e.g., reducing by 1 tablet per dose every 1 to 2 days) [see Warnings & Precautions (5.5)]. The Lofexidine tablet dose should be reduced, held, or discontinued for individuals who demonstrate a greater sensitivity to Lofexidine tablets side effects [see Warnings and Precautions (5.1), Adverse Reactions (6.1)]. Lower doses may be appropriate as opioid withdrawal symptoms wane.
Lofexidine tablets can be administered in the presence or absence of food.
8.6 Hepatic Impairment
Hepatic impairment slows the elimination of lofexidine but exhibits less effect on the peak plasma concentration than on AUC values following a single dose. Dosage adjustments are recommended based on the degree of hepatic impairment. [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
Clinically relevant QT prolongation may occur in subjects with hepatic impairment [see Warnings and Precautions (5.2), Clinical Pharmacology (12.2)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Lofexidine tablets are indicated for mitigation of opioid withdrawal symptoms to facilitate abrupt opioid discontinuation in adults.
12.1 Mechanism of Action
Lofexidine is a central alpha-2 adrenergic agonist that binds to receptors on adrenergic neurons. This reduces the release of norepinephrine and decreases sympathetic tone.
7.3 Cns Depressant Drugs (7.3 CNS Depressant Drugs)
Lofexidine tablets potentiate the CNS depressant effects of benzodiazepines and may potentiate the CNS depressant effects of alcohol, barbiturates, and other sedating drugs. Advise patients to inform their healthcare provider of other medications they are taking, including alcohol [see Warnings and Precautions (5.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risk of Hypotension, Bradycardia, and Syncope: May cause a decrease in blood pressure, a decrease in pulse, and syncope. Monitor vital signs before dosing and advise patients on how to minimize the risk of these cardiovascular effects and manage symptoms, should they occur. Monitor symptoms related to bradycardia and orthostasis. When using in outpatients, ensure that patients are capable of self-monitoring for signs and symptoms. Avoid use in patients with severe coronary insufficiency, recent myocardial infarction, cerebrovascular disease, or chronic renal failure, as well as in patients with marked bradycardia. (5.1)
- Risk of QT Prolongation: Lofexidine tablets prolong the QT interval. Avoid use in patients with congenital long QT syndrome. Monitor ECG in patients with electrolyte abnormalities, congestive heart failure, bradyarrhythmias, hepatic or renal impairment, or in patients taking other medicinal products that lead to QT prolongation. (5.2)
- Increased Risk of CNS Depression with Concomitant use of CNS Depressant Drugs: Lofexidine tablets potentiate the CNS depressant effects of benzodiazepines and may potentiate the CNS depressant effects of alcohol, barbiturates, and other sedating drugs. (5.3)
- Increased Risk of Opioid Overdose after Opioid Discontinuation: Patients who complete opioid discontinuation are at an increased risk of fatal overdose should they resume opioid use. Use in conjunction with a comprehensive management program for treatment of opioid use disorder and inform patients and caregivers of increased risk of overdose. (5.4)
- Risk of Discontinuation Symptoms: Instruct patients not to discontinue therapy without consulting their healthcare provider. When discontinuing therapy, reduce dose gradually. (5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The usual Lofexidine tablet dosage is three 0.18 mg tablets taken orally 4 times daily at 5- to 6-hour intervals. Lofexidine tablet treatment may be continued for up to 14 days with dosing guided by symptoms. (2.1)
- Discontinue Lofexidine tablets with a gradual dose reduction over 2 to 4 days. (2.1)
- Hepatic or Renal Impairment: Dosage adjustments are recommended based on degree of impairment. (2.2, 2.3)
5.2 Risk of Qt Prolongation (5.2 Risk of QT Prolongation)
Lofexidine tablets prolong the QT interval.
Avoid using Lofexidine tablets in patients with congenital long QT syndrome.
Monitor ECG in patients with congestive heart failure, bradyarrhythmias, hepatic impairment, renal impairment, or patients taking other medicinal products that lead to QT prolongation (e.g., methadone). In patients with electrolyte abnormalities (e.g., hypokalemia or hypomagnesemia), correct these abnormalities first, and monitor ECG upon initiation of Lofexidine tablets [see Dosing and Administration (2.1), Adverse Reactions (6.1), Special Populations (8.6, 8.7), Clinical Pharmacology (12.2)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Lofexidine tablets are available as round, peach-colored, film-coated tablets, imprinted with "LFX" on one side and "18" on the other side. Each tablet contains 0.18 mg lofexidine (equivalent to 0.2 mg of lofexidine hydrochloride).
6.2 Postmarketing Experience
Lofexidine is marketed in other countries for relief of opioid withdrawal symptoms. The following events have been identified during postmarketing use of lofexidine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Since lofexidine's initial market introduction in 1992, the most frequently reported postmarketing adverse event with lofexidine has been hypotension [see Warnings and Precautions (5.1)]. There has been one report of QT prolongation, bradycardia, torsades de pointes, and cardiac arrest with successful resuscitation in a patient who received lofexidine, and three reports of clinically significant QT prolongation in subjects concurrently receiving methadone with lofexidine.
8.8 Cyp2d6 Poor Metabolizers (8.8 CYP2D6 Poor Metabolizers)
Although the pharmacokinetics of Lofexidine tablets have not been systematically evaluated in patients who do not express the drug metabolizing enzyme CYP2D6, it is likely that the exposure to Lofexidine tablets would be increased similarly to taking strong CYP2D6 inhibitors (approximately 28%). Monitor adverse events such as orthostatic hypotension and bradycardia in known CYP2D6 poor metabolizers. Approximately 8% of Caucasians and 3 to 8% of Black/African Americans cannot metabolize CYP2D6 substrates and are classified as poor metabolizers (PM) [see Clinical Pharmacology (12.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to adverse reaction rates observed for another drug and may not reflect the rates observed in practice.
The safety of Lofexidine tablets was supported by three randomized, double-blind, placebo-controlled clinical trials, an open-label study, and clinical pharmacology studies with concomitant administration of either methadone, buprenorphine, or naltrexone.
The three randomized, double-blind, placebo-controlled clinical trials enrolled 935 subjects dependent on short-acting opioids undergoing abrupt opioid withdrawal. Patients were monitored before each dose in an inpatient setting.
Table 3 presents the incidence, rounded to the nearest percent, of adverse events that occurred in at least 10% of subjects treated with Lofexidine tablets and for which the incidence in patients treated with Lofexidine tablets were greater than the incidence in subjects treated with placebo in a study that tested two doses of Lofexidine tablets, 2.16 mg per day and 2.88 mg per day, and placebo. The overall safety profile in the combined dataset was similar.
Orthostatic hypotension, bradycardia, hypotension, dizziness, somnolence, sedation, and dry mouth were notably more common in subjects treated with Lofexidine tablets than subjects treated with placebo.
| Adverse Reaction | Lofexidine Tablets 2.16 mg Assigned dose; mean average daily dose received was 79% of assigned dose due to dose-holds for out-of-range vital signs. (%)N=229 |
Lofexidine Tablets 2.88 mg (%) N=222 |
Placebo (%) N=151 |
|---|---|---|---|
| Insomnia | 51 | 55 | 48 |
| Orthostatic Hypotension | 29 | 42 | 5 |
| Bradycardia | 24 | 32 | 5 |
| Hypotension | 30 | 30 | 1 |
| Dizziness | 19 | 23 | 3 |
| Somnolence | 11 | 13 | 5 |
| Sedation | 13 | 12 | 5 |
| Dry Mouth | 10 | 11 | 0 |
Other notable adverse reactions associated with the use of Lofexidine tablets but reported in <10% of patients in the Lofexidine tablets group included:
- Syncope: 0.9%, 1.4% and 0% for Lofexidine tablets 2.16 mg/day and 2.88 mg/day and placebo, respectively
- Tinnitus: 0.9%, 3.2% and 0% for Lofexidine tablets 2.16 mg/day and 2.88 mg/day and placebo, respectively
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Patient Information).
Lofexidine tablets may mitigate, but not completely prevent, the symptoms associated with opioid withdrawal syndrome, which may include feeling sick, stomach cramps, muscle spasms or twitching, feeling of cold, heart pounding, muscular tension, aches and pains, yawning, runny eyes and sleep problems (insomnia). Patients should be advised that withdrawal will not be easy. Additional supportive measures should be clearly advised, as needed.
7.4 Cyp2d6 Inhibitor Paroxetine (7.4 CYP2D6 Inhibitor - Paroxetine)
Coadministration of Lofexidine tablets and paroxetine resulted in a 28% increase in the extent of absorption of Lofexidine. Monitor for orthostatic hypotension and bradycardia when an inhibitor of CYP2D6 is used concomitantly with Lofexidine tablets [see Clinical Pharmacology (12.3)].
5.5 Risk of Discontinuation Symptoms
Stopping Lofexidine tablets abruptly can cause a marked rise in blood pressure. Symptoms including diarrhea, insomnia, anxiety, chills, hyperhidrosis, and extremity pain have also been observed with Lofexidine tablets discontinuation. Instruct patients not to discontinue therapy without consulting their healthcare provider. When discontinuing therapy with Lofexidine tablets, gradually reduce the dose [see Dosing and Administration (2.1)].
Symptoms related to discontinuation can be managed by administration of the previous Lofexidine tablet dose and subsequent taper.
8.3 Females and Males of Reproductive Potential
In animal studies that included some fertility endpoints, lofexidine decreased breeding rate and increased resorptions at exposures below human exposures. The impact of lofexidine on male fertility has not been adequately characterized in animal studies [see Impairment of Fertility (13.1)].
5.1 Risk of Hypotension, Bradycardia, and Syncope
Lofexidine tablets can cause a decrease in blood pressure, a decrease in pulse, and syncope [see Adverse Reactions (6.1), Clinical Pharmacology (12.2)]. Monitor vital signs before dosing. Monitor symptoms related to bradycardia and orthostasis.
Patients being given Lofexidine tablets in an outpatient setting should be capable of and instructed on self-monitoring for hypotension, orthostasis, bradycardia, and associated symptoms. If clinically significant or symptomatic hypotension and/or bradycardia occur, the next dose of Lofexidine tablets should be reduced in amount, delayed, or skipped.
Inform patients that Lofexidine tablets may cause hypotension and that patients moving from a supine to an upright position may be at increased risk for hypotension and orthostatic effects. Instruct patients to stay hydrated, on how to recognize symptoms of low blood pressure, and on how to reduce the risk of serious consequences should hypotension occur (e.g., sit or lie down, carefully rise from a sitting or lying position). Instruct outpatients to withhold Lofexidine tablets doses when experiencing symptoms of hypotension or bradycardia and to contact their healthcare provider for guidance on how to adjust dosing.
Avoid using Lofexidine tablets in patients with severe coronary insufficiency, recent myocardial infarction, cerebrovascular disease, chronic renal failure, and in patients with marked bradycardia.
Avoid using Lofexidine tablets in combination with medications that decrease pulse or blood pressure to avoid the risk of excessive bradycardia and hypotension.
Principal Display Panel 0.18 Mg Tablet Carton 36 Count (PRINCIPAL DISPLAY PANEL - 0.18 mg Tablet Carton 36 count)
NDC 66993-345-37
36 Tablets
Lofexidine Tablets
0.18 mg
Store and dispense
in
original container.
Protect from heat and moisture.
Do not remove desiccants.
Keep the bottle tightly closed.
Rx Only
Keep out of reach of children.
Principal Display Panel 0.18 Mg Tablet Carton 96 Count (PRINCIPAL DISPLAY PANEL - 0.18 mg Tablet Carton 96 count)
NDC 66993-345-76
96 Tablets
Lofexidine Tablets
0.18 mg
Store and dispense
in
original container.
Protect from heat and moisture.
Do not remove desiccants.
Keep the bottle tightly closed.
Rx Only
Keep out of reach of children.
2.3 Dosage Recommendations for Patients With Renal Impairment (2.3 Dosage Recommendations for Patients with Renal Impairment)
Recommended dosage adjustments based on the degree of renal impairment are shown in Table 2. Lofexidine tablets may be administered without regard to the timing of dialysis [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)].
| Moderate Impairment | Severe Impairment, End-Stage Renal Disease, or on Dialysis | |
|---|---|---|
| Estimated GFR, mL/min/1.73 m2 | 30-89.9 | < 30 |
| Recommended dose | 2 tablets 4 times daily (1.44 mg per day) |
1 tablet 4 times daily (0.72 mg per day) |
2.2 Dosage Recommendations for Patients With Hepatic Impairment (2.2 Dosage Recommendations for Patients with Hepatic Impairment)
Recommended dosage adjustments based on the degree of hepatic impairment are shown in Table 1. [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
| Mild Impairment | Moderate Impairment | Severe Impairment | |
|---|---|---|---|
| Child-Pugh score | 5-6 | 7-9 | > 9 |
| Recommended dose | 3 tablets 4 times daily (2.16 mg per day) |
2 tablets 4 times daily (1.44 mg per day) |
1 tablet 4 times daily (0.72 mg per day) |
5.4 Increased Risk of Opioid Overdose After Opioid Discontinuation (5.4 Increased Risk of Opioid Overdose after Opioid Discontinuation)
Lofexidine tablets are not a treatment for opioid use disorder. Patients who complete opioid discontinuation are likely to have a reduced tolerance to opioids and are at increased risk of fatal overdose should they resume opioid use. Use Lofexidine tablets in patients with opioid use disorder only in conjunction with a comprehensive management program for the treatment of opioid use disorder and inform patients and caregivers of this increased risk of overdose.
5.3 Increased Risk of Central Nervous System Depression With Concomitant Use of Cns Depressant Drugs (5.3 Increased Risk of Central Nervous System Depression with Concomitant use of CNS Depressant Drugs)
Lofexidine tablets potentiate the CNS depressive effects of benzodiazepines and can also be expected to potentiate the CNS depressive effects of alcohol, barbiturates, and other sedating drugs. Advise patients to inform their healthcare provider of other medications they are taking, including alcohol.
Advise patients using Lofexidine tablets in an outpatient setting that, until they learn how they respond to Lofexidine tablets, they should be careful or avoid doing activities such as driving or operating heavy machinery.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:42:58.376369 · Updated: 2026-03-14T22:12:08.234791