drug facts
77040348-f3c6-4aa7-b70a-dafbc0447687
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
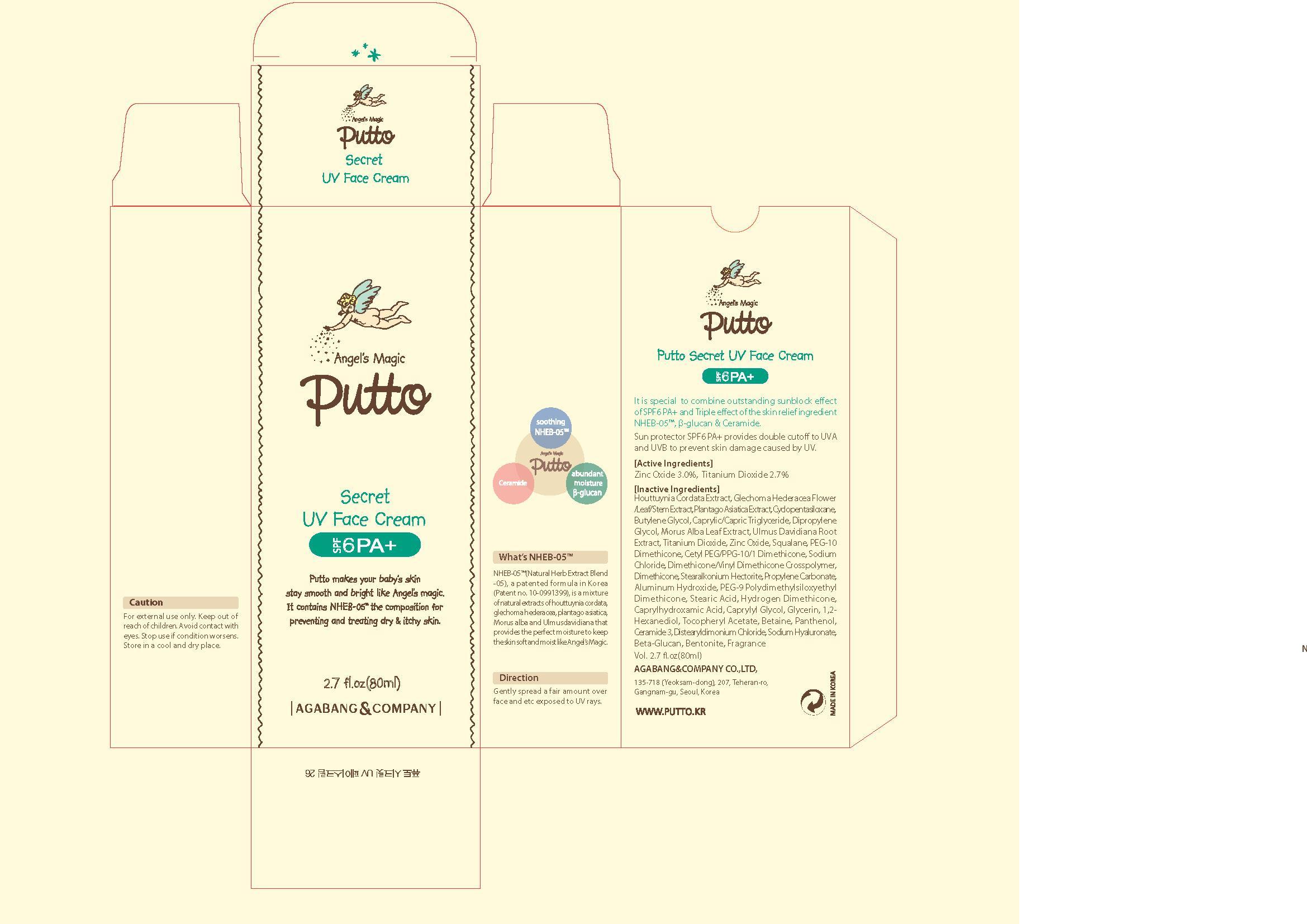
Description
titanium dioxide
Medication Information
Warnings and Precautions
do not apply to sensitive skin
store in a cool, dry place
Active Ingredient
titanium dioxide
Indications and Usage
gently spread a fair amount over face, neck, arm and etc esposed to UV rays
Dosage and Administration
for external use only
Description
titanium dioxide
Section 50565-1
keep out of reach of the children
Section 51727-6
houttuynia cordata ext, glechoma hederacea flower/leaf/stem ext, plantago asiatica ext, cyclopentasiloxane, butylene glycol, crprylic/capric triglyceride, dipropylene glycol, morus alba leaf ext, ulmus davidiana root ext, zinc oxide, squalane, peg-10 dimethicone, cetyl peg/ppg-10/1 dimethicone, sodium chloride, dimethicone/vinyl dimethicone crosspolymer, dimethicone, stearalkonium hectorite, propylene carbonate, aluminum hydroxide, peg-9 polydimethylsiloxyethyl dimethicone, stearic acid,hydrogen dimethicone, caprylhydroxamic acid, caprylyl glycol, glycerin, 1,2-hexanediol, tocopheryl acetate betaine, panthenol, ceramide 3, distearyldimonium chloride, sodium hyaluronate, beta-glucan, bentonite, fragrance
Section 51945-4
Section 55105-1
sun screen
Structured Label Content
Warnings and Precautions (34071-1)
do not apply to sensitive skin
store in a cool, dry place
Active Ingredient (55106-9)
titanium dioxide
Indications and Usage (34067-9)
gently spread a fair amount over face, neck, arm and etc esposed to UV rays
Dosage and Administration (34068-7)
for external use only
Section 50565-1 (50565-1)
keep out of reach of the children
Section 51727-6 (51727-6)
houttuynia cordata ext, glechoma hederacea flower/leaf/stem ext, plantago asiatica ext, cyclopentasiloxane, butylene glycol, crprylic/capric triglyceride, dipropylene glycol, morus alba leaf ext, ulmus davidiana root ext, zinc oxide, squalane, peg-10 dimethicone, cetyl peg/ppg-10/1 dimethicone, sodium chloride, dimethicone/vinyl dimethicone crosspolymer, dimethicone, stearalkonium hectorite, propylene carbonate, aluminum hydroxide, peg-9 polydimethylsiloxyethyl dimethicone, stearic acid,hydrogen dimethicone, caprylhydroxamic acid, caprylyl glycol, glycerin, 1,2-hexanediol, tocopheryl acetate betaine, panthenol, ceramide 3, distearyldimonium chloride, sodium hyaluronate, beta-glucan, bentonite, fragrance
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
sun screen
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:41.566586 · Updated: 2026-03-14T22:53:35.209708