These Highlights Do Not Include All The Information Needed To Use Miglustat Safely And Effectively. See Full Prescribing Information For Miglustat.
76eb7ac8-e0ee-4409-a4e7-e326c6725836
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Miglustat is a glucosylceramide synthase inhibitor indicated as monotherapy for treatment of adult patients with mild/moderate type 1 Gaucher disease for whom enzyme replacement therapy is not a therapeutic option ( 1.1 ).
Indications and Usage
Miglustat is a glucosylceramide synthase inhibitor indicated as monotherapy for treatment of adult patients with mild/moderate type 1 Gaucher disease for whom enzyme replacement therapy is not a therapeutic option ( 1.1 ).
Dosage and Administration
Recommended dosage is 100 mg administered orally three times a day at regular intervals ( 2.1 ). May reduce dosage to 100 mg once or twice a day in some patients due to tremor or diarrhea ( 2.1 ). Adjust in patients with renal impairment ( 2.2 ): Renal Impairment Adjusted Creatinine Clearance (in mL/min/1.73 m 2 ) Recommendations Mild 50 – 70 Start dose at 100 mg twice a day Moderate 30 – 50 Start dose at 100 mg once a day Severe <30 Use is not recommended
Warnings and Precautions
Peripheral neuropathy : Perform baseline and follow-up neurological evaluations at 6- month intervals in all patients ( 5.1 ). Tremor : Reduce dose to ameliorate tremor or discontinue treatment if tremor does not resolve within days of dose reduction ( 5.2 ). Diarrhea and Weight Loss : Evaluate for underlying gastrointestinal disease in patients who do not respond to usual interventions (e.g. diet modification) ( 5.3 ). Reductions in Platelet Count : Mild reductions in platelet counts without association with bleeding were observed in some patients. Monitoring of platelet counts is recommended ( 5.4 ).
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling: Peripheral Neuropathy [see Warnings and Precautions (5.1) ] Tremor [see Warnings and Precautions (5.2) ] Diarrhea and Weight Loss [see Warnings and Precautions (5.3) ] Reductions in Platelet Count [see Warnings and Precautions (5.4) ]
Drug Interactions
While co-administration of miglustat appeared to increase the clearance of imiglucerase by 70%, these results are not conclusive because of the small number of patients studied and because patients took variable doses of imiglucerase [see Clinical Pharmacology (12.3) ].
Storage and Handling
Miglustat is supplied in hard gelatin capsules containing 100 mg miglustat. Miglustat 100 mg capsules are white opaque with "OGT 918" printed in black on the cap and "100" printed in black on the body. Miglustat 100 mg capsules are packed in blister cards. Six blister cards of 15 capsules are supplied in each carton. NDC 10148-201-90: carton containing 90 capsules NDC 10148-201-15: blister card containing 15 capsules
How Supplied
Miglustat is supplied in hard gelatin capsules containing 100 mg miglustat. Miglustat 100 mg capsules are white opaque with "OGT 918" printed in black on the cap and "100" printed in black on the body. Miglustat 100 mg capsules are packed in blister cards. Six blister cards of 15 capsules are supplied in each carton. NDC 10148-201-90: carton containing 90 capsules NDC 10148-201-15: blister card containing 15 capsules
Medication Information
Warnings and Precautions
Peripheral neuropathy : Perform baseline and follow-up neurological evaluations at 6- month intervals in all patients ( 5.1 ). Tremor : Reduce dose to ameliorate tremor or discontinue treatment if tremor does not resolve within days of dose reduction ( 5.2 ). Diarrhea and Weight Loss : Evaluate for underlying gastrointestinal disease in patients who do not respond to usual interventions (e.g. diet modification) ( 5.3 ). Reductions in Platelet Count : Mild reductions in platelet counts without association with bleeding were observed in some patients. Monitoring of platelet counts is recommended ( 5.4 ).
Indications and Usage
Miglustat is a glucosylceramide synthase inhibitor indicated as monotherapy for treatment of adult patients with mild/moderate type 1 Gaucher disease for whom enzyme replacement therapy is not a therapeutic option ( 1.1 ).
Dosage and Administration
Recommended dosage is 100 mg administered orally three times a day at regular intervals ( 2.1 ). May reduce dosage to 100 mg once or twice a day in some patients due to tremor or diarrhea ( 2.1 ). Adjust in patients with renal impairment ( 2.2 ): Renal Impairment Adjusted Creatinine Clearance (in mL/min/1.73 m 2 ) Recommendations Mild 50 – 70 Start dose at 100 mg twice a day Moderate 30 – 50 Start dose at 100 mg once a day Severe <30 Use is not recommended
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling: Peripheral Neuropathy [see Warnings and Precautions (5.1) ] Tremor [see Warnings and Precautions (5.2) ] Diarrhea and Weight Loss [see Warnings and Precautions (5.3) ] Reductions in Platelet Count [see Warnings and Precautions (5.4) ]
Drug Interactions
While co-administration of miglustat appeared to increase the clearance of imiglucerase by 70%, these results are not conclusive because of the small number of patients studied and because patients took variable doses of imiglucerase [see Clinical Pharmacology (12.3) ].
Storage and Handling
Miglustat is supplied in hard gelatin capsules containing 100 mg miglustat. Miglustat 100 mg capsules are white opaque with "OGT 918" printed in black on the cap and "100" printed in black on the body. Miglustat 100 mg capsules are packed in blister cards. Six blister cards of 15 capsules are supplied in each carton. NDC 10148-201-90: carton containing 90 capsules NDC 10148-201-15: blister card containing 15 capsules
How Supplied
Miglustat is supplied in hard gelatin capsules containing 100 mg miglustat. Miglustat 100 mg capsules are white opaque with "OGT 918" printed in black on the cap and "100" printed in black on the body. Miglustat 100 mg capsules are packed in blister cards. Six blister cards of 15 capsules are supplied in each carton. NDC 10148-201-90: carton containing 90 capsules NDC 10148-201-15: blister card containing 15 capsules
Description
Miglustat is a glucosylceramide synthase inhibitor indicated as monotherapy for treatment of adult patients with mild/moderate type 1 Gaucher disease for whom enzyme replacement therapy is not a therapeutic option ( 1.1 ).
Section 42229-5
Risk Summary
Based on findings from animal reproduction studies, miglustat may cause fetal harm when administered to a pregnant woman. Available data from postmarketing case reports with miglustat use in pregnancy are insufficient to assess a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. There are risks associated with symptomatic Type I Gaucher disease in pregnancy, including hepatosplenomegaly and thrombocytopenia (see Clinical Considerations). Advise pregnant women of the potential risks to the fetus.
In animal reproduction studies, miglustat was maternally toxic in rabbits at exposures near the expected human therapeutic dose and caused embryo-fetal toxicities in rats at doses twice the recommended human dose. No adverse developmental outcomes were observed with administration of miglustat to pregnant rats at dose levels 6 times the recommended human dose. (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2–4% and 15–20%, respectively.
Section 42230-3
| PATIENT INFORMATION
MIGLUSTAT (MIG-loo-stat) (miglustat) Capsules |
||
|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 12/2022 | |
| Read this Patient Information before you start taking miglustat and each time you get a refill. There may be new information. | ||
|
What is MIGLUSTAT?
Miglustat is a prescription medicine used alone to treat adults with mild to moderate type 1 Gaucher disease. Miglustat is used only in people who cannot be treated with enzyme replacement therapy. It is not known if miglustat is safe and effective in children under 18 years of age. |
||
| Before taking MIGLUSTAT, tell your healthcare provider about all of your medical conditions, including if you: | ||
|
||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Miglustat may affect how other medicines work. | ||
| How should I take MIGLUSTAT? | ||
|
||
|
What are the possible side effects of MIGLUSTAT?
Miglustat may cause serious side effects including: |
||
|
||
| The most common side effects of miglustat include: | ||
|
|
|
| These are not all of the possible side effects of miglustat.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store MIGLUSTAT?
|
||
| Keep miglustat and all medicines out of the reach of children. | ||
|
General information about the safe and effective use of MIGLUSTAT.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use miglustat for a condition for which it was not prescribed. Do not give miglustat to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about miglustat that is written for health professionals. |
||
|
What are the ingredients in MIGLUSTAT?
Active ingredient: miglustat Inactive ingredients: magnesium stearate, povidone (K30), and sodium starch glycolate. The capsule shell contains: gelatin and titanium dioxide; the edible printing ink contains black iron oxide and shellac. Manufactured for: CoTherix, Inc. Titusville, NJ 08560, USA JN20221206 |
Section 44425-7
Storage
Store at 20°C to 25°C (68°F to 77°F). Excursions are permitted between 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature].
Keep out of reach of children.
5.2 Tremor
Approximately 30% of patients have reported tremor or exacerbation of existing tremor on treatment. These tremors were described as an exaggerated physiological tremor of the hands. Tremor usually began within the first month of therapy and in many cases resolved between 1 to 3 months during treatment. Reduce dose to ameliorate tremor or discontinue treatment if tremor does not resolve within days of dose reduction.
11 Description
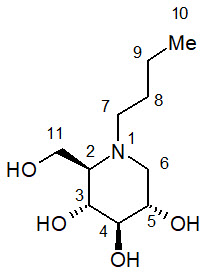
Miglustat (miglustat capsules, 100 mg) is a glucosylceramide synthase inhibitor, which is a glucosyl transferase enzyme responsible for the first step in the synthesis of most glycosphingolipids. miglustat is an N-alkylated imino sugar, a synthetic analog of D-glucose.
The chemical name for miglustat is 1,5-(butylimino)-1,5-dideoxy-D-glucitol with the chemical formula C 10H 21NO 4 and a molecular weight of 219.28.
Miglustat is a white to off-white crystalline solid and has a bitter taste. It is highly soluble in water (>1000 mg/mL as a free base).
Miglustat is supplied in hard gelatin capsules each containing 100 mg miglustat for oral administration. Each miglustat 100 mg capsule also contains magnesium stearate, povidone (K30), and sodium starch glycolate. Ingredients in the capsule shell include gelatin and titanium dioxide, and the shells are printed with edible ink consisting of black iron oxide and shellac.
8.4 Pediatric Use
The safety and effectiveness of miglustat in pediatric patients have not been established.
In a combined clinical trial safety data set of 45 patients less than 18 years of age exposed to miglustat in indications other than type 1 Gaucher disease, the median weight and height percentiles adjusted for age and gender decreased during the first year of treatment but then stabilized. The mean length of exposure in these studies ranged from 2 to 2.6 years; some pediatric patients were exposed for up to 4 years. However, the effect of miglustat on long-term gain in weight and height in pediatric patients is unclear.
8.5 Geriatric Use
Clinical studies of miglustat did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently than younger patients. Other reported clinical experience has not identified differences in responses between elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, and cardiac function and of concomitant disease or other drug therapy.
14 Clinical Studies
The efficacy of miglustat in type 1 Gaucher disease has been investigated in two open-label, uncontrolled trials and one randomized, open-label, active-controlled trial with enzyme replacement given as imiglucerase. Patients who received miglustat were treated with doses ranging from 100 to 600 mg a day, although the majority of patients were maintained on doses between 200 to 300 mg a day. Efficacy parameters included the evaluation of liver and spleen organ volume, hemoglobin concentration, and platelet count. A total of 80 patients were exposed to miglustat during the three trials and their extension period.
4 Contraindications
None.
6 Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling:
- Peripheral Neuropathy [see Warnings and Precautions (5.1)]
- Tremor [see Warnings and Precautions (5.2)]
- Diarrhea and Weight Loss [see Warnings and Precautions (5.3)]
- Reductions in Platelet Count [see Warnings and Precautions (5.4)]
7 Drug Interactions
While co-administration of miglustat appeared to increase the clearance of imiglucerase by 70%, these results are not conclusive because of the small number of patients studied and because patients took variable doses of imiglucerase [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Miglustat is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function [see Clinical Pharmacology (12.3)].
In patients with mild renal impairment (adjusted creatinine clearance 50–70 mL/min/1.73 m 2), miglustat administration should commence at a dose of 100 mg twice per day.
In patients with moderate renal impairment (adjusted creatinine clearance of 30–50 mL/min/1.73 m 2), miglustat administration should commence at a dose of 100 mg once a day.
Use of miglustat in patients with severe renal impairment (creatinine clearance <30 mL/min/1.73 m 2) is not recommended.
Since elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function. The impact of hemodialysis on the disposition of miglustat has not been investigated.
1 Indications and Usage
Miglustat is a glucosylceramide synthase inhibitor indicated as monotherapy for treatment of adult patients with mild/moderate type 1 Gaucher disease for whom enzyme replacement therapy is not a therapeutic option ( 1.1).
12.1 Mechanism of Action
Type 1 Gaucher disease is caused by a functional deficiency of glucocerebrosidase, the enzyme that mediates the degradation of the glycosphingolipid glucosylceramide.
Miglustat functions as a competitive and reversible inhibitor of the enzyme glucosylceramide synthase, the initial enzyme in a series of reactions which results in the synthesis of most glycosphingolipids.
Miglustat helps reduce the rate of glycosphingolipid biosynthesis so that the amount of glycosphingolipid substrate is reduced to a level which allows the residual activity of the deficient glucocerebrosidase enzyme to be more effective (substrate reduction therapy). In vitro and in vivo studies have shown that miglustat can reduce the synthesis of glucosylceramide-based glycosphingolipids.
5.1 Peripheral Neuropathy
In clinical trials, cases of peripheral neuropathy have been reported in 3% of Gaucher's patients treated with miglustat. All patients receiving miglustat treatment should undergo baseline and repeat neurological evaluations at approximately 6-month intervals. Patients who develop symptoms of peripheral neuropathy such as pain, weakness, numbness and tingling should have a careful re-assessment of the risk/benefit of miglustat therapy, and cessation of treatment may be considered.
1.1 Type 1 Gaucher Disease
Miglustat is indicated as monotherapy for the treatment of adult patients with mild to moderate type 1 Gaucher disease for whom enzyme replacement therapy is not a therapeutic option (e.g. due to allergy, hypersensitivity, or poor venous access).
5 Warnings and Precautions
- Peripheral neuropathy: Perform baseline and follow-up neurological evaluations at 6- month intervals in all patients ( 5.1).
- Tremor: Reduce dose to ameliorate tremor or discontinue treatment if tremor does not resolve within days of dose reduction ( 5.2).
- Diarrhea and Weight Loss: Evaluate for underlying gastrointestinal disease in patients who do not respond to usual interventions (e.g. diet modification) ( 5.3).
- Reductions in Platelet Count: Mild reductions in platelet counts without association with bleeding were observed in some patients. Monitoring of platelet counts is recommended ( 5.4).
2 Dosage and Administration
- Recommended dosage is 100 mg administered orally three times a day at regular intervals ( 2.1).
- May reduce dosage to 100 mg once or twice a day in some patients due to tremor or diarrhea ( 2.1).
- Adjust in patients with renal impairment ( 2.2):
| Renal Impairment | Adjusted Creatinine Clearance
(in mL/min/1.73 m 2) |
Recommendations |
|---|---|---|
| Mild | 50 – 70 | Start dose at 100 mg twice a day |
| Moderate | 30 – 50 | Start dose at 100 mg once a day |
| Severe | <30 | Use is not recommended |
3 Dosage Forms and Strengths
Capsules: 100 mg of miglustat, white opaque hard gelatin capsules with "OGT 918" printed in black on the cap and "100" printed in black on the body.
5.3 Diarrhea and Weight Loss
Diarrhea and weight loss were common in clinical studies of patients treated with miglustat, occurring in approximately 85% and up to 65% of treated patients, respectively. Diarrhea appears to be the result of the inhibitory activity of miglustat on intestinal disaccharidases such as sucrase-isomaltase in the gastrointestinal tract leading to reduced absorption of dietary disaccharides in the small intestine, with a resultant osmotic diarrhea. It is unclear if weight loss results from the diarrhea and associated gastrointestinal complaints, a decrease in food intake, or a combination of these or other factors. The incidence of weight loss was most evident in the first 12 months of treatment. Diarrhea decreased over time with continued miglustat treatment, and may respond to individualized diet modification (e.g., reduction of sucrose, lactose and other carbohydrate intake), to taking miglustat between meals, and/or to anti-diarrheal medications, most commonly loperamide. Patients may be instructed to avoid high carbohydrate content foods during treatment with miglustat if they present with diarrhea.
Patients with persistent gastrointestinal events that continue during treatment with miglustat, and who do not respond to usual interventions (e.g. diet modification), should be evaluated to determine whether significant underlying gastrointestinal disease is present. The safety of treatment with miglustat has not been evaluated in patients with significant gastrointestinal disease, such as inflammatory bowel disease, and continued treatment of these patients with miglustat should occur only after consideration of the risks and benefits of continued treatment.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure of 80 patients with type 1 Gaucher disease in two open-label, uncontrolled, monotherapy trials, one open-label, active-controlled trial, and two extensions, who received miglustat at doses ranging from 50 mg to 200 mg three times daily. Patients were aged 18 to 69 years at first treatment. The population was evenly distributed by gender.
The most common serious adverse reaction reported with miglustat treatment in clinical trials was peripheral neuropathy [see Warnings and Precautions (5.1)].
The most commonly reported adverse reactions in patients treated with miglustat (occurring in ≥5%) that were considered related to miglustat are shown in Tables 1 and 2 [see Warnings and Precautions (5.2, 5.3)].
The most common adverse reactions requiring intervention were diarrhea and tremor [see Warnings and Precautions (5.2, 5.3)].
In two open-label, uncontrolled monotherapy trials, adult type 1 Gaucher disease patients were treated with miglustat at a starting dose of 100 mg three times daily (dose range 100 to 200 mg three times daily) for up to 12 months in 28 patients [Study 1], or at a dose of 50 mg three times daily for up to 6 months in 18 patients [Study 2]. Table 1 below lists adverse reactions that occurred during the trials in ≥5% of patients.
| Incidence of adverse reactions | ||
|---|---|---|
| Study 1
(starting dose 100 mg three times daily) |
Study 2
(50 mg three times daily) |
|
| Patients entered in Study (n) | 28 | 18 |
| Body System - Preferred Term | % of patients reporting | % of patients reporting |
| Gastrointestinal System | ||
| Diarrhea | 89 | 89 |
| Flatulence | 29 | 44 |
| Abdominal Pain | 18 | 50 |
| Nausea | 14 | 22 |
| Vomiting | 4 | 11 |
| Bloating | 0 | 6 |
| Anorexia | 7 | 0 |
| Dyspepsia | 7 | 0 |
| Epigastric pain not food-related | 0 | 6 |
| Metabolic and Nutritional Disorders | ||
| Weight Decrease | 39 | 67 |
| Central and Peripheral Nervous System | ||
| Headache | 21 | 22 |
| Tremor | 11 | 11 |
| Dizziness | 0 | 11 |
| Leg cramps | 4 | 11 |
| Paresthesia | 7 | 0 |
| Migraine | 0 | 6 |
| Vision Disorders | ||
| Visual Disturbance | 0 | 17 |
| Musculoskeletal Disorders | ||
| Cramps | 0 | 11 |
| Platelet, Bleeding, and Clotting Disorders | ||
| Thrombocytopenia | 7 | 6 |
| Reproductive disorders, female | ||
| Menstrual disorder | 0 | 6 |
In an open-label, active-controlled study, 36 adult type 1 Gaucher disease patients were treated with miglustat, imiglucerase, or miglustat plus imiglucerase [Study 3] for up to 12 months. Table 2 lists adverse reactions that occurred during the trial in ≥5% of patients.
| Incidence of adverse reactions | ||
|---|---|---|
| Miglustat alone | Imiglucerase alone | |
| Patients entered in Study (n) | 12 | 12 |
| Body System - Preferred Term | % of patients reporting | % of patients reporting |
| Gastrointestinal System | ||
| Diarrhea | 100 | 0 |
| Abdominal Pain | 67 | 0 |
| Flatulence | 50 | 0 |
| Constipation | 8 | 0 |
| Nausea | 8 | 0 |
| Dry Mouth | 8 | 0 |
| Body as a Whole | ||
| Pain | 0 | 8 |
| Generalized weakness | 17 | 0 |
| Abdominal distension | 8 | 0 |
| Back pain | 8 | 0 |
| Heaviness in limbs | 8 | 0 |
| Metabolic and Nutritional Disorders | ||
| Weight Decrease | 67 | 0 |
| Central and Peripheral Nervous System | ||
| Tremor | 17 | 0 |
| Dizziness | 8 | 0 |
| Leg cramps | 8 | 0 |
| Unsteady gait | 8 | 0 |
| Psychiatric disorders | ||
| Memory loss | 8 | 0 |
5.4 Reductions in Platelet Count
In clinical trials evaluating the use of miglustat for treatment of indications other than type 1 Gaucher disease, mild reductions in platelet counts without association with bleeding were observed in some patients; approximately 40% of patients in this trial had low platelet counts (defined as below 150×10 9/L) before starting treatment with miglustat. Monitoring of platelet counts is recommended in patients with type 1 Gaucher disease. Mild reductions in platelet counts without association with bleeding were observed in patients with type 1 Gaucher disease who were switched from enzyme replacement therapy (ERT) to miglustat.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.1 Instructions for Administration
Therapy should be directed by physicians who are knowledgeable in the management of Gaucher disease.
The recommended dose for the treatment of adult patients with type 1 Gaucher disease is one 100 mg capsule administered orally three times a day at regular intervals. If a dose is missed, the next miglustat capsule should be taken at the next scheduled time.
It may be necessary to reduce the dose to one 100 mg capsule once or twice a day in some patients due to adverse reactions, such as tremor or diarrhea.
16 How Supplied/storage and Handling
Miglustat is supplied in hard gelatin capsules containing 100 mg miglustat. Miglustat 100 mg capsules are white opaque with "OGT 918" printed in black on the cap and "100" printed in black on the body.
Miglustat 100 mg capsules are packed in blister cards. Six blister cards of 15 capsules are supplied in each carton.
NDC 10148-201-90: carton containing 90 capsules
NDC 10148-201-15: blister card containing 15 capsules
2.2 Patients With Renal Insufficiency
In patients with mild renal impairment (adjusted creatinine clearance 50–70 mL/min/1.73 m 2), initiate miglustat treatment at a dose of 100 mg twice per day. In patients with moderate renal impairment (adjusted creatinine clearance of 30–50 mL/min/1.73 m 2), initiate miglustat treatment at a dose of one 100 mg capsule per day. Miglustat is not recommended for use in patients with severe renal impairment (creatinine clearance <30 mL/min/1.73 m 2) [see Use in Specific Populations (8.6)].
13.2 Animal Toxicology And/or Pharmacology
Histopathology findings in the absence of clinical signs in the central nervous system of the monkey (brain, spine) that included vascular mineralization, in addition to mineralization and necrosis of white matter were observed at >750 mg/kg/day (4 times the human therapeutic systemic exposure based on area-under-the-plasma-concentration curve [AUC] comparisons) in a 52-week oral toxicity study using doses of 750 and 2000 mg/kg/d. Vacuolization of white matter was observed in rats dosed orally by gavage at ≥180 mg/kg/d (6 times the human therapeutic exposure based on surface area comparisons, mg/m 2) in a 4-week study using doses of 180, 840, and 4200 mg/kg/d. Vacuolization can sometimes occur as an artifact of tissue processing. Findings in dogs included tremor and absent corneal reflexes at 105 mg/kg/day (10 times the human therapeutic systemic exposure, based on body surface area comparisons, mg/m 2) after a 4-week oral gavage toxicity study using doses of 35, 70, 105, and 140 mg/kg/d. Ataxia, diminished/absent pupillary, palpebral, or patellar reflexes were observed in a dog at ≥495 mg/kg/day (50 times the human therapeutic systemic exposure based on body surface area comparisons, mg/m 2), in a 2-week oral gavage toxicity study using doses of 85, 165, 495, and 825 mg/kg/d.
Cataracts were observed in rats at ≥180 mg/kg/day (4 times the human therapeutic systemic exposure, based on AUC) in a 52-week oral gavage toxicity study using doses of 180, 420, 840, and 1680 mg/kg/d.
Gastrointestinal necrosis, inflammation, and hemorrhage were observed in dogs at ≥85 mg/kg/day (9 times the human therapeutic systemic exposure based on body surface area comparisons, mg/m 2) after a 2-week oral (capsule) toxicity study using doses of 85, 165, 495, and 825 mg/kg/d. Similar GI toxicity occurred in rats at 1200 mg/kg/day (7 times the human therapeutic systemic exposure, based on AUC) in a 26-week oral gavage toxicity study using doses of 300, 600, and 1200 mg/kg/d. In monkeys, similar GI toxicity occurred at ≥750 mg/kg/day (6 times the human therapeutic systemic exposure based on AUC) following a 52-week oral gavage toxicity study using doses of 750 and 2000 mg/kg/d.
Principal Display Panel 100 Mg Capsule Blister Card Carton
migLUstat
capsules
NDC 10148-201-90
100 mg
This package is not child resistant.
Contents:
90 Capsules (six blister cards of 15 capsules each)
Oral use
Rx only
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
Based on findings from animal reproduction studies, miglustat may cause fetal harm when administered to a pregnant woman. Available data from postmarketing case reports with miglustat use in pregnancy are insufficient to assess a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. There are risks associated with symptomatic Type I Gaucher disease in pregnancy, including hepatosplenomegaly and thrombocytopenia (see Clinical Considerations). Advise pregnant women of the potential risks to the fetus.
In animal reproduction studies, miglustat was maternally toxic in rabbits at exposures near the expected human therapeutic dose and caused embryo-fetal toxicities in rats at doses twice the recommended human dose. No adverse developmental outcomes were observed with administration of miglustat to pregnant rats at dose levels 6 times the recommended human dose. (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2–4% and 15–20%, respectively.
Section 42230-3 (42230-3)
| PATIENT INFORMATION
MIGLUSTAT (MIG-loo-stat) (miglustat) Capsules |
||
|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 12/2022 | |
| Read this Patient Information before you start taking miglustat and each time you get a refill. There may be new information. | ||
|
What is MIGLUSTAT?
Miglustat is a prescription medicine used alone to treat adults with mild to moderate type 1 Gaucher disease. Miglustat is used only in people who cannot be treated with enzyme replacement therapy. It is not known if miglustat is safe and effective in children under 18 years of age. |
||
| Before taking MIGLUSTAT, tell your healthcare provider about all of your medical conditions, including if you: | ||
|
||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Miglustat may affect how other medicines work. | ||
| How should I take MIGLUSTAT? | ||
|
||
|
What are the possible side effects of MIGLUSTAT?
Miglustat may cause serious side effects including: |
||
|
||
| The most common side effects of miglustat include: | ||
|
|
|
| These are not all of the possible side effects of miglustat.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store MIGLUSTAT?
|
||
| Keep miglustat and all medicines out of the reach of children. | ||
|
General information about the safe and effective use of MIGLUSTAT.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use miglustat for a condition for which it was not prescribed. Do not give miglustat to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about miglustat that is written for health professionals. |
||
|
What are the ingredients in MIGLUSTAT?
Active ingredient: miglustat Inactive ingredients: magnesium stearate, povidone (K30), and sodium starch glycolate. The capsule shell contains: gelatin and titanium dioxide; the edible printing ink contains black iron oxide and shellac. Manufactured for: CoTherix, Inc. Titusville, NJ 08560, USA JN20221206 |
Section 44425-7 (44425-7)
Storage
Store at 20°C to 25°C (68°F to 77°F). Excursions are permitted between 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature].
Keep out of reach of children.
5.2 Tremor
Approximately 30% of patients have reported tremor or exacerbation of existing tremor on treatment. These tremors were described as an exaggerated physiological tremor of the hands. Tremor usually began within the first month of therapy and in many cases resolved between 1 to 3 months during treatment. Reduce dose to ameliorate tremor or discontinue treatment if tremor does not resolve within days of dose reduction.
11 Description (11 DESCRIPTION)
Miglustat (miglustat capsules, 100 mg) is a glucosylceramide synthase inhibitor, which is a glucosyl transferase enzyme responsible for the first step in the synthesis of most glycosphingolipids. miglustat is an N-alkylated imino sugar, a synthetic analog of D-glucose.
The chemical name for miglustat is 1,5-(butylimino)-1,5-dideoxy-D-glucitol with the chemical formula C 10H 21NO 4 and a molecular weight of 219.28.
Miglustat is a white to off-white crystalline solid and has a bitter taste. It is highly soluble in water (>1000 mg/mL as a free base).
Miglustat is supplied in hard gelatin capsules each containing 100 mg miglustat for oral administration. Each miglustat 100 mg capsule also contains magnesium stearate, povidone (K30), and sodium starch glycolate. Ingredients in the capsule shell include gelatin and titanium dioxide, and the shells are printed with edible ink consisting of black iron oxide and shellac.
8.4 Pediatric Use
The safety and effectiveness of miglustat in pediatric patients have not been established.
In a combined clinical trial safety data set of 45 patients less than 18 years of age exposed to miglustat in indications other than type 1 Gaucher disease, the median weight and height percentiles adjusted for age and gender decreased during the first year of treatment but then stabilized. The mean length of exposure in these studies ranged from 2 to 2.6 years; some pediatric patients were exposed for up to 4 years. However, the effect of miglustat on long-term gain in weight and height in pediatric patients is unclear.
8.5 Geriatric Use
Clinical studies of miglustat did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently than younger patients. Other reported clinical experience has not identified differences in responses between elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, and cardiac function and of concomitant disease or other drug therapy.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of miglustat in type 1 Gaucher disease has been investigated in two open-label, uncontrolled trials and one randomized, open-label, active-controlled trial with enzyme replacement given as imiglucerase. Patients who received miglustat were treated with doses ranging from 100 to 600 mg a day, although the majority of patients were maintained on doses between 200 to 300 mg a day. Efficacy parameters included the evaluation of liver and spleen organ volume, hemoglobin concentration, and platelet count. A total of 80 patients were exposed to miglustat during the three trials and their extension period.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described below and elsewhere in the labeling:
- Peripheral Neuropathy [see Warnings and Precautions (5.1)]
- Tremor [see Warnings and Precautions (5.2)]
- Diarrhea and Weight Loss [see Warnings and Precautions (5.3)]
- Reductions in Platelet Count [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
While co-administration of miglustat appeared to increase the clearance of imiglucerase by 70%, these results are not conclusive because of the small number of patients studied and because patients took variable doses of imiglucerase [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Miglustat is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function [see Clinical Pharmacology (12.3)].
In patients with mild renal impairment (adjusted creatinine clearance 50–70 mL/min/1.73 m 2), miglustat administration should commence at a dose of 100 mg twice per day.
In patients with moderate renal impairment (adjusted creatinine clearance of 30–50 mL/min/1.73 m 2), miglustat administration should commence at a dose of 100 mg once a day.
Use of miglustat in patients with severe renal impairment (creatinine clearance <30 mL/min/1.73 m 2) is not recommended.
Since elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function. The impact of hemodialysis on the disposition of miglustat has not been investigated.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Miglustat is a glucosylceramide synthase inhibitor indicated as monotherapy for treatment of adult patients with mild/moderate type 1 Gaucher disease for whom enzyme replacement therapy is not a therapeutic option ( 1.1).
12.1 Mechanism of Action
Type 1 Gaucher disease is caused by a functional deficiency of glucocerebrosidase, the enzyme that mediates the degradation of the glycosphingolipid glucosylceramide.
Miglustat functions as a competitive and reversible inhibitor of the enzyme glucosylceramide synthase, the initial enzyme in a series of reactions which results in the synthesis of most glycosphingolipids.
Miglustat helps reduce the rate of glycosphingolipid biosynthesis so that the amount of glycosphingolipid substrate is reduced to a level which allows the residual activity of the deficient glucocerebrosidase enzyme to be more effective (substrate reduction therapy). In vitro and in vivo studies have shown that miglustat can reduce the synthesis of glucosylceramide-based glycosphingolipids.
5.1 Peripheral Neuropathy
In clinical trials, cases of peripheral neuropathy have been reported in 3% of Gaucher's patients treated with miglustat. All patients receiving miglustat treatment should undergo baseline and repeat neurological evaluations at approximately 6-month intervals. Patients who develop symptoms of peripheral neuropathy such as pain, weakness, numbness and tingling should have a careful re-assessment of the risk/benefit of miglustat therapy, and cessation of treatment may be considered.
1.1 Type 1 Gaucher Disease
Miglustat is indicated as monotherapy for the treatment of adult patients with mild to moderate type 1 Gaucher disease for whom enzyme replacement therapy is not a therapeutic option (e.g. due to allergy, hypersensitivity, or poor venous access).
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Peripheral neuropathy: Perform baseline and follow-up neurological evaluations at 6- month intervals in all patients ( 5.1).
- Tremor: Reduce dose to ameliorate tremor or discontinue treatment if tremor does not resolve within days of dose reduction ( 5.2).
- Diarrhea and Weight Loss: Evaluate for underlying gastrointestinal disease in patients who do not respond to usual interventions (e.g. diet modification) ( 5.3).
- Reductions in Platelet Count: Mild reductions in platelet counts without association with bleeding were observed in some patients. Monitoring of platelet counts is recommended ( 5.4).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended dosage is 100 mg administered orally three times a day at regular intervals ( 2.1).
- May reduce dosage to 100 mg once or twice a day in some patients due to tremor or diarrhea ( 2.1).
- Adjust in patients with renal impairment ( 2.2):
| Renal Impairment | Adjusted Creatinine Clearance
(in mL/min/1.73 m 2) |
Recommendations |
|---|---|---|
| Mild | 50 – 70 | Start dose at 100 mg twice a day |
| Moderate | 30 – 50 | Start dose at 100 mg once a day |
| Severe | <30 | Use is not recommended |
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Capsules: 100 mg of miglustat, white opaque hard gelatin capsules with "OGT 918" printed in black on the cap and "100" printed in black on the body.
5.3 Diarrhea and Weight Loss
Diarrhea and weight loss were common in clinical studies of patients treated with miglustat, occurring in approximately 85% and up to 65% of treated patients, respectively. Diarrhea appears to be the result of the inhibitory activity of miglustat on intestinal disaccharidases such as sucrase-isomaltase in the gastrointestinal tract leading to reduced absorption of dietary disaccharides in the small intestine, with a resultant osmotic diarrhea. It is unclear if weight loss results from the diarrhea and associated gastrointestinal complaints, a decrease in food intake, or a combination of these or other factors. The incidence of weight loss was most evident in the first 12 months of treatment. Diarrhea decreased over time with continued miglustat treatment, and may respond to individualized diet modification (e.g., reduction of sucrose, lactose and other carbohydrate intake), to taking miglustat between meals, and/or to anti-diarrheal medications, most commonly loperamide. Patients may be instructed to avoid high carbohydrate content foods during treatment with miglustat if they present with diarrhea.
Patients with persistent gastrointestinal events that continue during treatment with miglustat, and who do not respond to usual interventions (e.g. diet modification), should be evaluated to determine whether significant underlying gastrointestinal disease is present. The safety of treatment with miglustat has not been evaluated in patients with significant gastrointestinal disease, such as inflammatory bowel disease, and continued treatment of these patients with miglustat should occur only after consideration of the risks and benefits of continued treatment.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure of 80 patients with type 1 Gaucher disease in two open-label, uncontrolled, monotherapy trials, one open-label, active-controlled trial, and two extensions, who received miglustat at doses ranging from 50 mg to 200 mg three times daily. Patients were aged 18 to 69 years at first treatment. The population was evenly distributed by gender.
The most common serious adverse reaction reported with miglustat treatment in clinical trials was peripheral neuropathy [see Warnings and Precautions (5.1)].
The most commonly reported adverse reactions in patients treated with miglustat (occurring in ≥5%) that were considered related to miglustat are shown in Tables 1 and 2 [see Warnings and Precautions (5.2, 5.3)].
The most common adverse reactions requiring intervention were diarrhea and tremor [see Warnings and Precautions (5.2, 5.3)].
In two open-label, uncontrolled monotherapy trials, adult type 1 Gaucher disease patients were treated with miglustat at a starting dose of 100 mg three times daily (dose range 100 to 200 mg three times daily) for up to 12 months in 28 patients [Study 1], or at a dose of 50 mg three times daily for up to 6 months in 18 patients [Study 2]. Table 1 below lists adverse reactions that occurred during the trials in ≥5% of patients.
| Incidence of adverse reactions | ||
|---|---|---|
| Study 1
(starting dose 100 mg three times daily) |
Study 2
(50 mg three times daily) |
|
| Patients entered in Study (n) | 28 | 18 |
| Body System - Preferred Term | % of patients reporting | % of patients reporting |
| Gastrointestinal System | ||
| Diarrhea | 89 | 89 |
| Flatulence | 29 | 44 |
| Abdominal Pain | 18 | 50 |
| Nausea | 14 | 22 |
| Vomiting | 4 | 11 |
| Bloating | 0 | 6 |
| Anorexia | 7 | 0 |
| Dyspepsia | 7 | 0 |
| Epigastric pain not food-related | 0 | 6 |
| Metabolic and Nutritional Disorders | ||
| Weight Decrease | 39 | 67 |
| Central and Peripheral Nervous System | ||
| Headache | 21 | 22 |
| Tremor | 11 | 11 |
| Dizziness | 0 | 11 |
| Leg cramps | 4 | 11 |
| Paresthesia | 7 | 0 |
| Migraine | 0 | 6 |
| Vision Disorders | ||
| Visual Disturbance | 0 | 17 |
| Musculoskeletal Disorders | ||
| Cramps | 0 | 11 |
| Platelet, Bleeding, and Clotting Disorders | ||
| Thrombocytopenia | 7 | 6 |
| Reproductive disorders, female | ||
| Menstrual disorder | 0 | 6 |
In an open-label, active-controlled study, 36 adult type 1 Gaucher disease patients were treated with miglustat, imiglucerase, or miglustat plus imiglucerase [Study 3] for up to 12 months. Table 2 lists adverse reactions that occurred during the trial in ≥5% of patients.
| Incidence of adverse reactions | ||
|---|---|---|
| Miglustat alone | Imiglucerase alone | |
| Patients entered in Study (n) | 12 | 12 |
| Body System - Preferred Term | % of patients reporting | % of patients reporting |
| Gastrointestinal System | ||
| Diarrhea | 100 | 0 |
| Abdominal Pain | 67 | 0 |
| Flatulence | 50 | 0 |
| Constipation | 8 | 0 |
| Nausea | 8 | 0 |
| Dry Mouth | 8 | 0 |
| Body as a Whole | ||
| Pain | 0 | 8 |
| Generalized weakness | 17 | 0 |
| Abdominal distension | 8 | 0 |
| Back pain | 8 | 0 |
| Heaviness in limbs | 8 | 0 |
| Metabolic and Nutritional Disorders | ||
| Weight Decrease | 67 | 0 |
| Central and Peripheral Nervous System | ||
| Tremor | 17 | 0 |
| Dizziness | 8 | 0 |
| Leg cramps | 8 | 0 |
| Unsteady gait | 8 | 0 |
| Psychiatric disorders | ||
| Memory loss | 8 | 0 |
5.4 Reductions in Platelet Count
In clinical trials evaluating the use of miglustat for treatment of indications other than type 1 Gaucher disease, mild reductions in platelet counts without association with bleeding were observed in some patients; approximately 40% of patients in this trial had low platelet counts (defined as below 150×10 9/L) before starting treatment with miglustat. Monitoring of platelet counts is recommended in patients with type 1 Gaucher disease. Mild reductions in platelet counts without association with bleeding were observed in patients with type 1 Gaucher disease who were switched from enzyme replacement therapy (ERT) to miglustat.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.1 Instructions for Administration
Therapy should be directed by physicians who are knowledgeable in the management of Gaucher disease.
The recommended dose for the treatment of adult patients with type 1 Gaucher disease is one 100 mg capsule administered orally three times a day at regular intervals. If a dose is missed, the next miglustat capsule should be taken at the next scheduled time.
It may be necessary to reduce the dose to one 100 mg capsule once or twice a day in some patients due to adverse reactions, such as tremor or diarrhea.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Miglustat is supplied in hard gelatin capsules containing 100 mg miglustat. Miglustat 100 mg capsules are white opaque with "OGT 918" printed in black on the cap and "100" printed in black on the body.
Miglustat 100 mg capsules are packed in blister cards. Six blister cards of 15 capsules are supplied in each carton.
NDC 10148-201-90: carton containing 90 capsules
NDC 10148-201-15: blister card containing 15 capsules
2.2 Patients With Renal Insufficiency (2.2 Patients with Renal Insufficiency)
In patients with mild renal impairment (adjusted creatinine clearance 50–70 mL/min/1.73 m 2), initiate miglustat treatment at a dose of 100 mg twice per day. In patients with moderate renal impairment (adjusted creatinine clearance of 30–50 mL/min/1.73 m 2), initiate miglustat treatment at a dose of one 100 mg capsule per day. Miglustat is not recommended for use in patients with severe renal impairment (creatinine clearance <30 mL/min/1.73 m 2) [see Use in Specific Populations (8.6)].
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Histopathology findings in the absence of clinical signs in the central nervous system of the monkey (brain, spine) that included vascular mineralization, in addition to mineralization and necrosis of white matter were observed at >750 mg/kg/day (4 times the human therapeutic systemic exposure based on area-under-the-plasma-concentration curve [AUC] comparisons) in a 52-week oral toxicity study using doses of 750 and 2000 mg/kg/d. Vacuolization of white matter was observed in rats dosed orally by gavage at ≥180 mg/kg/d (6 times the human therapeutic exposure based on surface area comparisons, mg/m 2) in a 4-week study using doses of 180, 840, and 4200 mg/kg/d. Vacuolization can sometimes occur as an artifact of tissue processing. Findings in dogs included tremor and absent corneal reflexes at 105 mg/kg/day (10 times the human therapeutic systemic exposure, based on body surface area comparisons, mg/m 2) after a 4-week oral gavage toxicity study using doses of 35, 70, 105, and 140 mg/kg/d. Ataxia, diminished/absent pupillary, palpebral, or patellar reflexes were observed in a dog at ≥495 mg/kg/day (50 times the human therapeutic systemic exposure based on body surface area comparisons, mg/m 2), in a 2-week oral gavage toxicity study using doses of 85, 165, 495, and 825 mg/kg/d.
Cataracts were observed in rats at ≥180 mg/kg/day (4 times the human therapeutic systemic exposure, based on AUC) in a 52-week oral gavage toxicity study using doses of 180, 420, 840, and 1680 mg/kg/d.
Gastrointestinal necrosis, inflammation, and hemorrhage were observed in dogs at ≥85 mg/kg/day (9 times the human therapeutic systemic exposure based on body surface area comparisons, mg/m 2) after a 2-week oral (capsule) toxicity study using doses of 85, 165, 495, and 825 mg/kg/d. Similar GI toxicity occurred in rats at 1200 mg/kg/day (7 times the human therapeutic systemic exposure, based on AUC) in a 26-week oral gavage toxicity study using doses of 300, 600, and 1200 mg/kg/d. In monkeys, similar GI toxicity occurred at ≥750 mg/kg/day (6 times the human therapeutic systemic exposure based on AUC) following a 52-week oral gavage toxicity study using doses of 750 and 2000 mg/kg/d.
Principal Display Panel 100 Mg Capsule Blister Card Carton (PRINCIPAL DISPLAY PANEL - 100 mg Capsule Blister Card Carton)
migLUstat
capsules
NDC 10148-201-90
100 mg
This package is not child resistant.
Contents:
90 Capsules (six blister cards of 15 capsules each)
Oral use
Rx only
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:38:09.865901 · Updated: 2026-03-14T21:53:26.799717