Dramamine Chewable Orange
76166156-bce1-492f-833e-6bbd07859c3f
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antiemetic
Medication Information
Purpose
Antiemetic
Description
Drug Facts
I
ask a doctor before use.
Use
for prevention and treatment of these symptoms associated with motion sickness
- nausea
- vomiting
- dizziness
Do Not
children under 2 years of age unless directed by a doctor
Section 42229-5
Drug Facts
Ask A D
if you are taking sedatives or tranquilizers
Directions
to prevent motion sickness, the first dose should be taken ½ to 1 hour before starting activity
to prevent or treat motion sickness, see below:
|
Adults and
children 12 years and over |
|
|
Children 6 to
under 12 years |
|
|
Children 2 to
under 6 years |
|
Active Ingredient
(in each tablet)
Dimenhydrinate 50 mg
Other Information
- Phenylketonurics: contains phenylalanine 0.84 mg per tablet
- store at room temperature 68-77ºF (20-25ºC)
-
do not use if carton is opened or if blister is broken or torn
- see side panel for lot number and expiration date
Inactive Ingredients
anhydrous citric acid, aspartame, FD&C yellow #6 aluminum lake, flavors, magnesium stearate, maltodextrin, methacrylic acid copolymer, modified starch, sorbitol
Questions Or Comments?
Call 1-800-382-7219
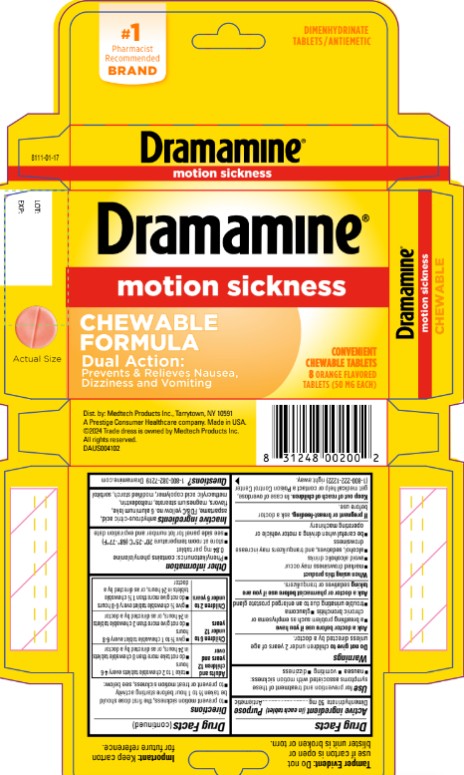
Principal Display Panel
Dimenhydrinate Tablets/Antiemetic
CHEWABLE FORMULA
Dramamine®
MOTION SICKNESS RELIEF
DUAL ACTION
1. TREATS SYMPTOMS ON THE SPOT
2. PREVENTS NAUSEA, DIZZINESS & VOMITING
Convenient Chewable Tablets
8 ORANGE FLAVORED TABLETS (50 mg EACH)
When Using This Product
- marked drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Keep Out of Reach of Children.
In case of accidental overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Ask A Doctor Before Use If You Have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Structured Label Content
I
ask a doctor before use.
Use
for prevention and treatment of these symptoms associated with motion sickness
- nausea
- vomiting
- dizziness
Do Not (Do not)
children under 2 years of age unless directed by a doctor
Section 42229-5 (42229-5)
Drug Facts
Ask A D (Ask a d)
if you are taking sedatives or tranquilizers
Purpose
Antiemetic
Directions
to prevent motion sickness, the first dose should be taken ½ to 1 hour before starting activity
to prevent or treat motion sickness, see below:
|
Adults and
children 12 years and over |
|
|
Children 6 to
under 12 years |
|
|
Children 2 to
under 6 years |
|
Active Ingredient (Active ingredient)
(in each tablet)
Dimenhydrinate 50 mg
Other Information (Other information)
- Phenylketonurics: contains phenylalanine 0.84 mg per tablet
- store at room temperature 68-77ºF (20-25ºC)
-
do not use if carton is opened or if blister is broken or torn
- see side panel for lot number and expiration date
Inactive Ingredients (Inactive ingredients)
anhydrous citric acid, aspartame, FD&C yellow #6 aluminum lake, flavors, magnesium stearate, maltodextrin, methacrylic acid copolymer, modified starch, sorbitol
Questions Or Comments? (Questions or comments?)
Call 1-800-382-7219
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Dimenhydrinate Tablets/Antiemetic
CHEWABLE FORMULA
Dramamine®
MOTION SICKNESS RELIEF
DUAL ACTION
1. TREATS SYMPTOMS ON THE SPOT
2. PREVENTS NAUSEA, DIZZINESS & VOMITING
Convenient Chewable Tablets
8 ORANGE FLAVORED TABLETS (50 mg EACH)
When Using This Product (When using this product)
- marked drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of accidental overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:58.372551 · Updated: 2026-03-14T23:10:05.459246