guaifenesin and pseudoephedrine hcl extended-release tablets, 600 mg/60 mg and 1200 mg/120 mg
75f4ff44-02e7-4a23-a394-85fe8b442e1a
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Expectorant Nasal Decongestant
Description
(For 600 mg/60 mg) Guaifenesin USP, 600 mg Pseudoephedrine HCL USP, 60 mg (For 1200 mg/120 mg) Guaifenesin USP, 1200 mg Pseudoephedrine HCL USP, 120 mg
Medication Information
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Uses
• helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
• temporarily relieves nasal congestion due to:
• common cold
• hay fever
• upper respiratory allergies
• temporarily restores freer breathing through the nose
• promotes nasal and/or sinus drainage
• temporarily relieves sinus congestion and pressure
Purpose
Expectorant Nasal Decongestant
Directions
• do not crush, chew, or break extended-release tablet
• take with a full glass of water
• this product can be administered without regard for timing of meals
• adults and children 12 years and older:
For 600 mg/60 mg: 2 extended-release tablets every 12 hours; not more than 4 extended-release tablets in 24 hours
For 1200 mg/120 mg: 1 extended-release tablet every 12 hours; not more than 2 extended-release tablets in 24 hours
• children under 12 years of age: do not use
Other Information
• Tamper evident: do not use if carton is open or if printed seal on blister is broken or missing.
• store between 20-25°C (68-77°F)
Inactive Ingredients
carbomer homopolymer Type B, cellulose microcrystalline, hypromellose, lake of FD&C yellow 6, magnesium stearate, sodium starch glycolate Type A
Description
(For 600 mg/60 mg) Guaifenesin USP, 600 mg Pseudoephedrine HCL USP, 60 mg (For 1200 mg/120 mg) Guaifenesin USP, 1200 mg Pseudoephedrine HCL USP, 120 mg
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Centre right away (1-800 222-1222).
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Purposes
Expectorant
Nasal Decongestant
Questions Or Comments?
Contact 1-877-770-3183 Mon-Fri 8:00 AM EST to 5:00 PM PST.
You may also report side effects to this phone number.
Keep the carton.
It contains important information. See end panel for expiration date.
Manufactured By:
Granules India Limited
Hyderabad – 500 081, INDIA
MADE IN INDIA
Distributed By:
Granules USA, Inc.
Parsippany, NJ 07054
When Using This Product
• do not use more than directed
Stop Use and Ask A Doctor If
• you get nervous, dizzy, or sleepless
• symptoms do not get better within 7 days, come back or occur with a fever, rash, or persistent headache. These could be signs of a serious illness.
Ask A Doctor Before Use If You Have
• heart disease
• high blood pressure
• thyroid disease
• diabetes
• trouble urinating due to an enlarged prostate gland
• persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
• cough accompanied by too much phlegm (mucus)
Active Ingredients (in Each Extended Release Tablet)
(For 600 mg/60 mg)
Guaifenesin USP, 600 mg
Pseudoephedrine HCL USP, 60 mg
(For 1200 mg/120 mg)
Guaifenesin USP, 1200 mg
Pseudoephedrine HCL USP, 120 mg
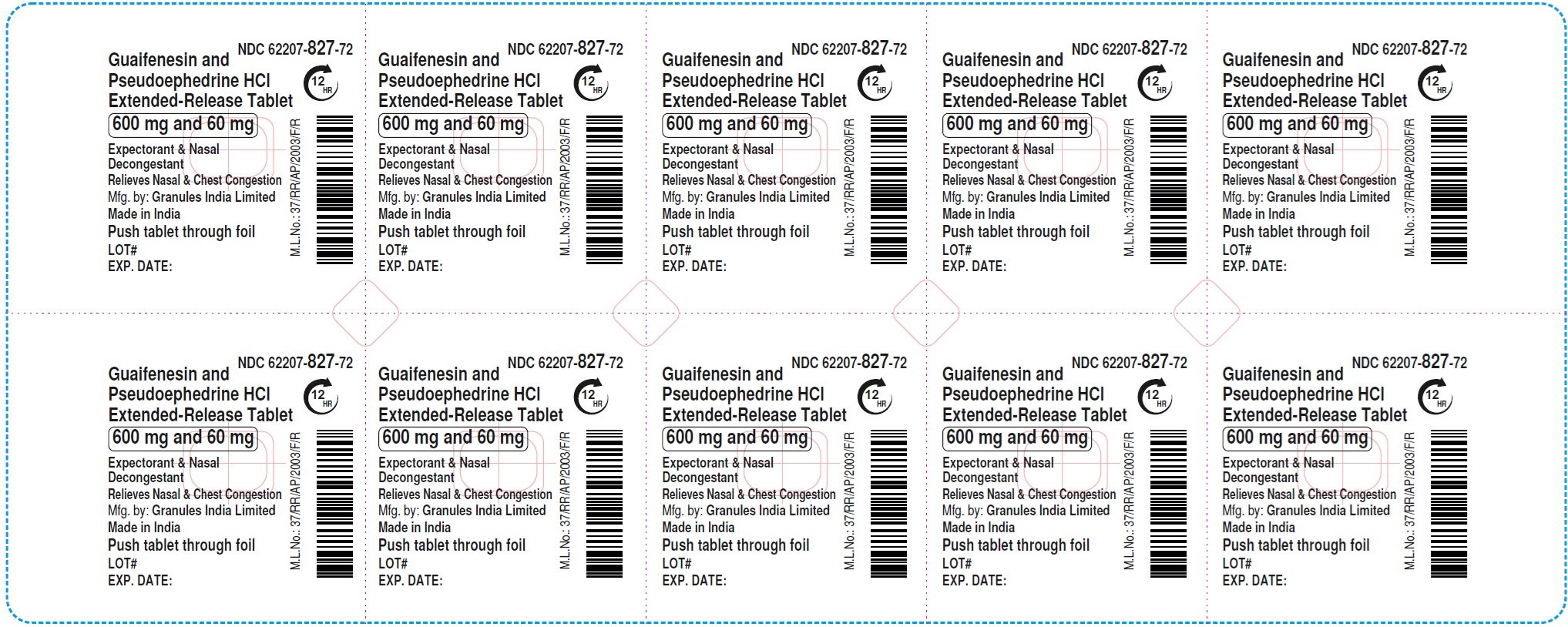
Principal Display Panel 600 Mg and 60 Mg Blister Foil Label
Principal Display Panel 1200 Mg and 120 Mg Blister Foil Label
Principal Display Panel 600 Mg and 60 Mg Blister Carton Label
NDC 62207-827-72
†Compare To
the active ingredients of
Mucinex
® D
Guaifenesin and
Pseudoephedrine Hydrochloride
FOR DAY OR NIGHT
Extended-Release Tablets 12HR
600 mg and 60 mg
Guaifenesin 600 mg - Expectorant •
Clears Nasal & Sinus Congestion
Pseudoephedrine HCl
60 mg - Nasal Decongestant •
Thins & Loosens Mucus
• Immediate & Extended Release
RELIEVES NASAL & CHEST CONGESTION
90 (9x10) Extended-Release Tablets
Principal Display Panel 1200 Mg and 120 Mg Blister Carton Label
NDC 62207-828-71
†Compare To
the active ingredients of
Mucinex
® D
Guaifenesin and
Pseudoephedrine Hydrochloride
FOR DAY OR NIGHT
Extended-Release Tablets
12HR
1200 mg and 120 mg
Guaifenesin 1200 mg - Expectorant
• Clears Nasal & Sinus Congestion
Pseudoephedrine HCl 120 mg - Nasal Decongestant
• Thins & Loosens Mucus
• Immediate & Extended Release
RELIEVES NASAL & CHEST CONGESTION
70 (7x10) Extended-Release Tablets
Structured Label Content
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Uses
• helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
• temporarily relieves nasal congestion due to:
• common cold
• hay fever
• upper respiratory allergies
• temporarily restores freer breathing through the nose
• promotes nasal and/or sinus drainage
• temporarily relieves sinus congestion and pressure
Directions
• do not crush, chew, or break extended-release tablet
• take with a full glass of water
• this product can be administered without regard for timing of meals
• adults and children 12 years and older:
For 600 mg/60 mg: 2 extended-release tablets every 12 hours; not more than 4 extended-release tablets in 24 hours
For 1200 mg/120 mg: 1 extended-release tablet every 12 hours; not more than 2 extended-release tablets in 24 hours
• children under 12 years of age: do not use
Other Information (Other information)
• Tamper evident: do not use if carton is open or if printed seal on blister is broken or missing.
• store between 20-25°C (68-77°F)
Inactive Ingredients (Inactive ingredients)
carbomer homopolymer Type B, cellulose microcrystalline, hypromellose, lake of FD&C yellow 6, magnesium stearate, sodium starch glycolate Type A
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Centre right away (1-800 222-1222).
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purposes
Expectorant
Nasal Decongestant
Questions Or Comments? (Questions or comments?)
Contact 1-877-770-3183 Mon-Fri 8:00 AM EST to 5:00 PM PST.
You may also report side effects to this phone number.
Keep the carton.
It contains important information. See end panel for expiration date.
Manufactured By:
Granules India Limited
Hyderabad – 500 081, INDIA
MADE IN INDIA
Distributed By:
Granules USA, Inc.
Parsippany, NJ 07054
When Using This Product (When using this product)
• do not use more than directed
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
• you get nervous, dizzy, or sleepless
• symptoms do not get better within 7 days, come back or occur with a fever, rash, or persistent headache. These could be signs of a serious illness.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
• heart disease
• high blood pressure
• thyroid disease
• diabetes
• trouble urinating due to an enlarged prostate gland
• persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
• cough accompanied by too much phlegm (mucus)
Active Ingredients (in Each Extended Release Tablet) (Active ingredients (in each extended-release tablet))
(For 600 mg/60 mg)
Guaifenesin USP, 600 mg
Pseudoephedrine HCL USP, 60 mg
(For 1200 mg/120 mg)
Guaifenesin USP, 1200 mg
Pseudoephedrine HCL USP, 120 mg
Principal Display Panel 600 Mg and 60 Mg Blister Foil Label (PRINCIPAL DISPLAY PANEL 600 mg and 60 mg - Blister Foil Label)
Principal Display Panel 1200 Mg and 120 Mg Blister Foil Label (PRINCIPAL DISPLAY PANEL 1200 mg and 120 mg - Blister Foil Label)
Principal Display Panel 600 Mg and 60 Mg Blister Carton Label (PRINCIPAL DISPLAY PANEL 600 mg and 60 mg - Blister Carton Label)
NDC 62207-827-72
†Compare To
the active ingredients of
Mucinex
® D
Guaifenesin and
Pseudoephedrine Hydrochloride
FOR DAY OR NIGHT
Extended-Release Tablets 12HR
600 mg and 60 mg
Guaifenesin 600 mg - Expectorant •
Clears Nasal & Sinus Congestion
Pseudoephedrine HCl
60 mg - Nasal Decongestant •
Thins & Loosens Mucus
• Immediate & Extended Release
RELIEVES NASAL & CHEST CONGESTION
90 (9x10) Extended-Release Tablets
Principal Display Panel 1200 Mg and 120 Mg Blister Carton Label (PRINCIPAL DISPLAY PANEL 1200 mg and 120 mg - Blister Carton Label)
NDC 62207-828-71
†Compare To
the active ingredients of
Mucinex
® D
Guaifenesin and
Pseudoephedrine Hydrochloride
FOR DAY OR NIGHT
Extended-Release Tablets
12HR
1200 mg and 120 mg
Guaifenesin 1200 mg - Expectorant
• Clears Nasal & Sinus Congestion
Pseudoephedrine HCl 120 mg - Nasal Decongestant
• Thins & Loosens Mucus
• Immediate & Extended Release
RELIEVES NASAL & CHEST CONGESTION
70 (7x10) Extended-Release Tablets
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:25.153223 · Updated: 2026-03-14T23:10:50.302579