quality plus 44-043
75a9c736-bc7e-4b7f-84ea-9474a37508ec
34390-5
HUMAN OTC DRUG LABEL
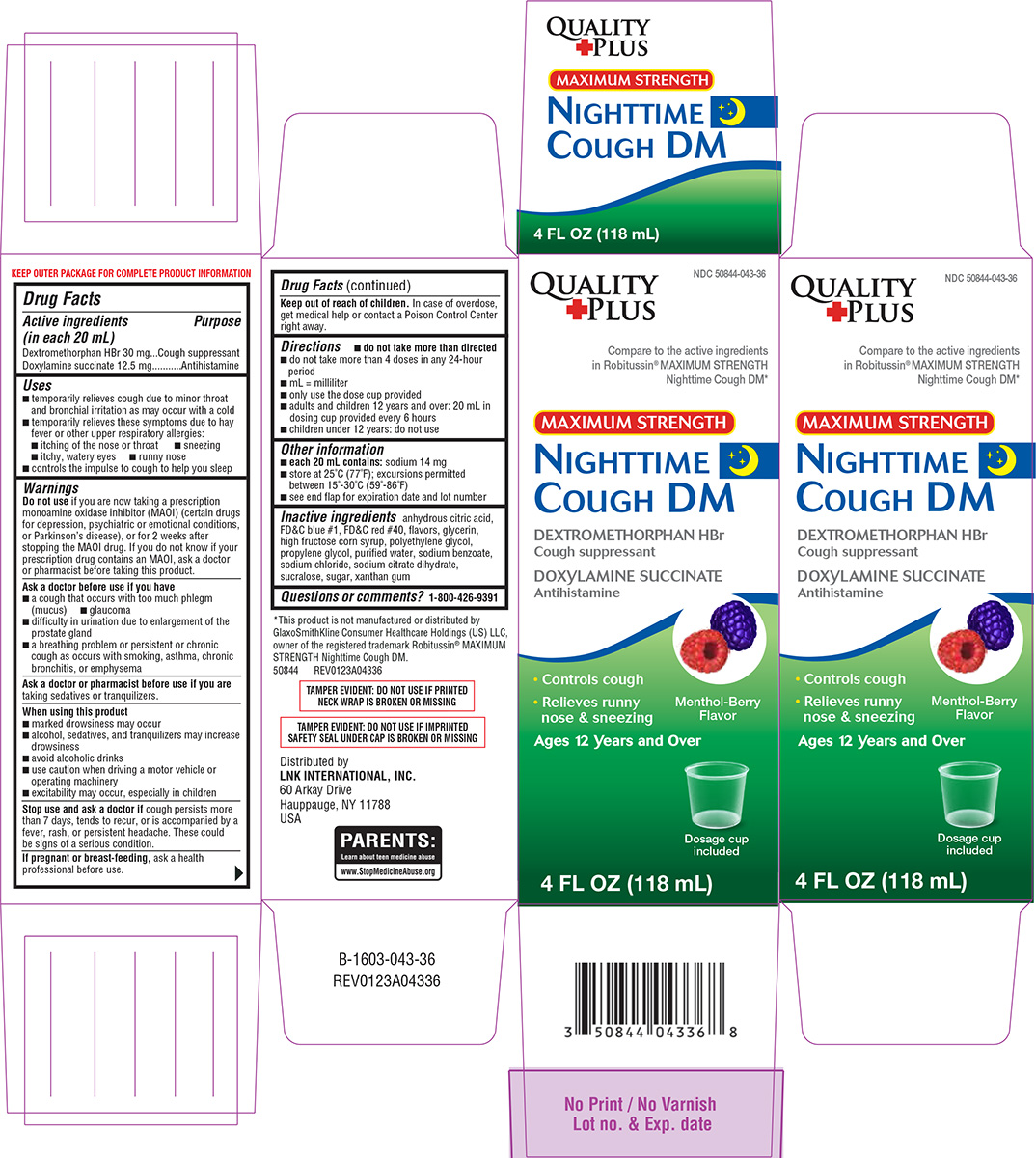
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Cough suppressant Antihistamine
Description
Dextromethorphan HBr 30 mg Doxylamine succinate 12.5 mg
Medication Information
Uses
- temporarily relieves cough due to minor throat and bronchial irritation as may occur with a cold
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- itching of the nose or throat
- sneezing
- itchy, watery eyes
- runny nose
- controls the impulse to cough to help you sleep
Purpose
Cough suppressant
Antihistamine
Directions
- do not take more than directed
- do not take more than 4 doses in any 24-hour period
- mL = milliliter
- only use the dose cup provided
- adults and children 12 years and over: 20 mL in dosing cup provided every 6 hours
- children under 12 years: do not use
Other Information
-
each 20 mL contains: sodium 14 mg
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive Ingredients
anhydrous citric acid, FD&C blue #1, FD&C red #40, flavors, glycerin, high fructose corn syrup, polyethylene glycol, propylene glycol, purified water, sodium benzoate, sodium chloride, sodium citrate dihydrate, sucralose, sugar, xanthan gum
Description
Dextromethorphan HBr 30 mg Doxylamine succinate 12.5 mg
Principal Display Panel
NDC 50844-043-36
Quality
+Plus
Compare to the active ingredients
in Robitussin® MAXIMUM STRENGTH
Nighttime Cough DM*
MAXIMUM STRENGTH
NIGHTTIME
COUGH DM
DEXTROMETHORPHAN HBr
Cough suppressant
DOXYLAMINE SUCCINATE
Antihistamine
• Controls cough
• Relieves runny
nose & sneezing
Menthol-Berry
Flavor
Ages 12 Years and Over
Dosage cup
included
4 FL OZ (118 mL)
TAMPER EVIDENT: DO NOT USE IF PRINTED
NECK WRAP IS BROKEN OR MISSING
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
*This product is not manufactured or distributed by
GlaxoSmithKline Consumer Healthcare Holdings (US) LLC,
owner of the registered trademark Robitussin® MAXIMUM
STRENGTH Nighttime Cough DM.
50844 REV0123A04336
Distributed by
LNK INTERNATIONAL, INC.
60 Arkay Drive
Hauppauge, NY 11788
USA
PARENTS:
Learn about teen medicine abuse
www.StopMedicineAbuse.org
Do Not Use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Questions Or Comments?
1-800-426-9391
When Using This Product
- marked drowsiness may occur
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic drinks
- use caution when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
Stop Use and Ask A Doctor If
cough persists more than 7 days, tends to recur, or is accompanied by a fever, rash, or persistent headache. These could be signs of a serious condition.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredients (in Each 20 Ml)
Dextromethorphan HBr 30 mg
Doxylamine succinate 12.5 mg
Ask A Doctor Before Use If You Have
- a cough that occurs with too much phlegm (mucus)
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
- a breathing problem or persistent or chronic cough as occurs with smoking, asthma, chronic bronchitis, or emphysema
Ask A Doctor Or Pharmacist Before Use If You Are
taking sedatives or tranquilizers.
Structured Label Content
Uses
- temporarily relieves cough due to minor throat and bronchial irritation as may occur with a cold
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- itching of the nose or throat
- sneezing
- itchy, watery eyes
- runny nose
- controls the impulse to cough to help you sleep
Purpose
Cough suppressant
Antihistamine
Directions
- do not take more than directed
- do not take more than 4 doses in any 24-hour period
- mL = milliliter
- only use the dose cup provided
- adults and children 12 years and over: 20 mL in dosing cup provided every 6 hours
- children under 12 years: do not use
Other Information (Other information)
-
each 20 mL contains: sodium 14 mg
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive Ingredients (Inactive ingredients)
anhydrous citric acid, FD&C blue #1, FD&C red #40, flavors, glycerin, high fructose corn syrup, polyethylene glycol, propylene glycol, purified water, sodium benzoate, sodium chloride, sodium citrate dihydrate, sucralose, sugar, xanthan gum
Principal Display Panel (Principal display panel)
NDC 50844-043-36
Quality
+Plus
Compare to the active ingredients
in Robitussin® MAXIMUM STRENGTH
Nighttime Cough DM*
MAXIMUM STRENGTH
NIGHTTIME
COUGH DM
DEXTROMETHORPHAN HBr
Cough suppressant
DOXYLAMINE SUCCINATE
Antihistamine
• Controls cough
• Relieves runny
nose & sneezing
Menthol-Berry
Flavor
Ages 12 Years and Over
Dosage cup
included
4 FL OZ (118 mL)
TAMPER EVIDENT: DO NOT USE IF PRINTED
NECK WRAP IS BROKEN OR MISSING
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
*This product is not manufactured or distributed by
GlaxoSmithKline Consumer Healthcare Holdings (US) LLC,
owner of the registered trademark Robitussin® MAXIMUM
STRENGTH Nighttime Cough DM.
50844 REV0123A04336
Distributed by
LNK INTERNATIONAL, INC.
60 Arkay Drive
Hauppauge, NY 11788
USA
PARENTS:
Learn about teen medicine abuse
www.StopMedicineAbuse.org
Do Not Use (Do not use)
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Questions Or Comments? (Questions or comments?)
1-800-426-9391
When Using This Product (When using this product)
- marked drowsiness may occur
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic drinks
- use caution when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
cough persists more than 7 days, tends to recur, or is accompanied by a fever, rash, or persistent headache. These could be signs of a serious condition.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Active Ingredients (in Each 20 Ml) (Active ingredients (in each 20 mL))
Dextromethorphan HBr 30 mg
Doxylamine succinate 12.5 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- a cough that occurs with too much phlegm (mucus)
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
- a breathing problem or persistent or chronic cough as occurs with smoking, asthma, chronic bronchitis, or emphysema
Ask A Doctor Or Pharmacist Before Use If You Are (Ask a doctor or pharmacist before use if you are)
taking sedatives or tranquilizers.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:17.071703 · Updated: 2026-03-14T23:08:57.795400