These Highlights Do Not Include All The Information Needed To Use Norepinephrine In Sodium Chloride Injection Safely And Effectively. See Full Prescribing Information For Norepinephrine In Sodium Chloride Injection.
759807dd-61a8-4f6f-8bf5-f6ee773c81bc
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Norepinephrine in Sodium Chloride Injection is indicated to raise blood pressure in adult patients with severe, acute hypotension.
Indications and Usage
Norepinephrine in Sodium Chloride Injection is indicated to raise blood pressure in adult patients with severe, acute hypotension.
Dosage and Administration
No further dilution prior to infusion is required ( 2.1 ) Initial intravenous infusion rate of 8 to 12 mcg per minute, adjust the rate of flow to establish and maintain a low to normal blood pressure (usually 80 to 100 mm Hg) sufficient to maintain the circulation of vital organs ( 2.2 ) The average maintenance dose ranges from 2 to 4 mcg per minute. ( 2.2 )
Warnings and Precautions
Tissue Ischemia : Avoid extravasation into tissues, which can cause local necrosis ( 5.1 ) Hypotension After Abrupt Discontinuation : Sudden cessation of the infusion rate may result in marked hypotension. Reduce the Norepinephrine in Sodium Chloride Injection infusion rate gradually. ( 5.2 ) Cardiac Arrhythmias : Norepinephrine in Sodium Chloride Injection may cause arrhythmias. Monitor cardiac function in patients with underlying heart disease. ( 5.3 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are described in greater detail in other sections: Tissue Ischemia [see Warnings and Precautions ( 5.1 )] Hypotension [see Warnings and Precautions ( 5.2 )] Cardiac Arrhythmias [see Warnings and Precautions ( 5.3 )] The most common adverse reactions are hypertension and bradycardia. The following adverse reactions can occur: Nervous system disorders: Anxiety, headache Respiratory disorders: Respiratory difficulty, pulmonary edema
Drug Interactions
Monoamine oxidase inhibitors (MAOI) or antidepressants of the triptyline or imipramine types may result in hypertension. ( 7.1 ) Cyclopropane and halothane anesthetics increase cardiac autonomic irritability. ( 7.4 )
Storage and Handling
Norepinephrine in Sodium Chloride Injection (norepinephrine bitartrate) is supplied as a clear, colorless sterile solution in a 250 mL non-PVC infusion bag with single function connector system consisting of a port and cap, packaged individually in an aluminum foil pouch with an oxygen scavenger. Supplied as: Unit of Sale Concentration Package Size NDC 81298-9659-3 4 mg/250 mL (16 mcg/mL) 10 bags NDC 81298-9655-3 8 mg/250 mL (32 mcg/mL) 10 bags NDC 81298-9658-3 16 mg/250 mL (64 mcg/mL) 10 bags Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature.] Protect from light. Once the overwrap is removed, the bag can be stored at room temperature for up to 45 days.
How Supplied
Norepinephrine in Sodium Chloride Injection (norepinephrine bitartrate) is supplied as a clear, colorless sterile solution in a 250 mL non-PVC infusion bag with single function connector system consisting of a port and cap, packaged individually in an aluminum foil pouch with an oxygen scavenger. Supplied as: Unit of Sale Concentration Package Size NDC 81298-9659-3 4 mg/250 mL (16 mcg/mL) 10 bags NDC 81298-9655-3 8 mg/250 mL (32 mcg/mL) 10 bags NDC 81298-9658-3 16 mg/250 mL (64 mcg/mL) 10 bags Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature.] Protect from light. Once the overwrap is removed, the bag can be stored at room temperature for up to 45 days.
Medication Information
Warnings and Precautions
Tissue Ischemia : Avoid extravasation into tissues, which can cause local necrosis ( 5.1 ) Hypotension After Abrupt Discontinuation : Sudden cessation of the infusion rate may result in marked hypotension. Reduce the Norepinephrine in Sodium Chloride Injection infusion rate gradually. ( 5.2 ) Cardiac Arrhythmias : Norepinephrine in Sodium Chloride Injection may cause arrhythmias. Monitor cardiac function in patients with underlying heart disease. ( 5.3 )
Indications and Usage
Norepinephrine in Sodium Chloride Injection is indicated to raise blood pressure in adult patients with severe, acute hypotension.
Dosage and Administration
No further dilution prior to infusion is required ( 2.1 ) Initial intravenous infusion rate of 8 to 12 mcg per minute, adjust the rate of flow to establish and maintain a low to normal blood pressure (usually 80 to 100 mm Hg) sufficient to maintain the circulation of vital organs ( 2.2 ) The average maintenance dose ranges from 2 to 4 mcg per minute. ( 2.2 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are described in greater detail in other sections: Tissue Ischemia [see Warnings and Precautions ( 5.1 )] Hypotension [see Warnings and Precautions ( 5.2 )] Cardiac Arrhythmias [see Warnings and Precautions ( 5.3 )] The most common adverse reactions are hypertension and bradycardia. The following adverse reactions can occur: Nervous system disorders: Anxiety, headache Respiratory disorders: Respiratory difficulty, pulmonary edema
Drug Interactions
Monoamine oxidase inhibitors (MAOI) or antidepressants of the triptyline or imipramine types may result in hypertension. ( 7.1 ) Cyclopropane and halothane anesthetics increase cardiac autonomic irritability. ( 7.4 )
Storage and Handling
Norepinephrine in Sodium Chloride Injection (norepinephrine bitartrate) is supplied as a clear, colorless sterile solution in a 250 mL non-PVC infusion bag with single function connector system consisting of a port and cap, packaged individually in an aluminum foil pouch with an oxygen scavenger. Supplied as: Unit of Sale Concentration Package Size NDC 81298-9659-3 4 mg/250 mL (16 mcg/mL) 10 bags NDC 81298-9655-3 8 mg/250 mL (32 mcg/mL) 10 bags NDC 81298-9658-3 16 mg/250 mL (64 mcg/mL) 10 bags Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature.] Protect from light. Once the overwrap is removed, the bag can be stored at room temperature for up to 45 days.
How Supplied
Norepinephrine in Sodium Chloride Injection (norepinephrine bitartrate) is supplied as a clear, colorless sterile solution in a 250 mL non-PVC infusion bag with single function connector system consisting of a port and cap, packaged individually in an aluminum foil pouch with an oxygen scavenger. Supplied as: Unit of Sale Concentration Package Size NDC 81298-9659-3 4 mg/250 mL (16 mcg/mL) 10 bags NDC 81298-9655-3 8 mg/250 mL (32 mcg/mL) 10 bags NDC 81298-9658-3 16 mg/250 mL (64 mcg/mL) 10 bags Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature.] Protect from light. Once the overwrap is removed, the bag can be stored at room temperature for up to 45 days.
Description
Norepinephrine in Sodium Chloride Injection is indicated to raise blood pressure in adult patients with severe, acute hypotension.
Section 42229-5
Correct Hypovolemia
Address hypovolemia before initiation of Norepinephrine in Sodium Chloride Injection therapy. If the patient does not respond to therapy, suspect occult hypovolemia [see Warnings and Precautions (5.1)].
Section 51945-4
Principal Display Panel – 4 mg Pouch Label
NDC: 81298-9659-1
Rx only
1 x 250 mL
Norepinephrine
in 0.9% Sodium Chloride injection
4 mg per 250 mL
(16 mcg/mL)
Keep in sealed overwrap until time of use.
LONG GROVE
PHARMACEUTICALS™
2.2 Dosage
After an initial dose of 8 to 12 mcg per minute via intravenous infusion, assess patient response and adjust dosage to maintain desired hemodynamic effect. Monitor blood pressure every two minutes or continuously until the desired hemodynamic effect is achieved, and then monitor blood pressure every five minutes for the duration of the infusion.
Recommended Average Maintenance Dosage:
Typical maintenance intravenous dosage is 2 to 4 mcg per minute.
10 Overdosage
Overdosage with Norepinephrine in Sodium Chloride Injection may result in headache, severe hypertension, reflex bradycardia, marked increase in peripheral resistance, and decreased cardiac output.
In case of accidental overdosage, discontinue Norepinephrine in Sodium Chloride Injection until the condition of the patient stabilizes.
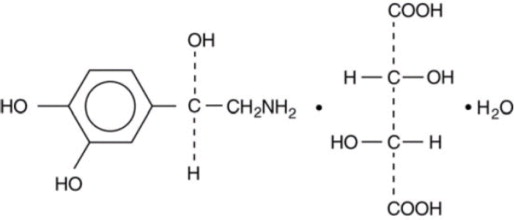
11 Description
Norepinephrine (sometimes referred to as l-arterenol/Levarterenol or l-norepinephrine) is a catecholamine which differs from epinephrine by the absence of a methyl group on the nitrogen atom.
Chemically, Norepinephrine Bitartrate is (-)-α-(aminomethyl)-3,4-dihydroxybenzyl alcohol tartrate (1:1) (salt) monohydrate (molecular weight 337.3 g/mol) and has the following structural formula:
Norepinephrine in Sodium Chloride Injection is a clear, colorless, single dose sterile solution supplied as a ready-to-use intravenous infusion bag for intravenous use and does not require further dilution. Each mL contains the equivalent of 16 or 32 or 64 micrograms of norepinephrine base supplied as 31.90 or 63.80 or 127.6 micrograms per mL of norepinephrine bitartrate monohydrate. In addition, each mL of solution contains 0.01 mg edetate disodium dihydrate as a metal chelator and 9.0 mg sodium chloride for isotonicity. It has a pH of 3.5 to 4.5.
7.3 Antidiabetics
Norepinephrine in Sodium Chloride Injection can decrease insulin sensitivity and raise blood glucose. Monitor glucose and consider dosage adjustment of antidiabetic drugs.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of Norepinephrine in Sodium Chloride Injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Avoid administration of Norepinephrine in Sodium Chloride Injection into the veins in the leg in elderly patients [see Warnings and Precautions (5.1)].
4 Contraindications
None.
5.1 Tissue Ischemia
Administration of Norepinephrine in Sodium Chloride Injection to patients who are hypotensive from hypovolemia can result in severe peripheral and visceral vasoconstriction, decreased renal perfusion and reduced urine output, tissue hypoxia, lactic acidosis, and reduced systemic blood flow despite “normal” blood pressure. Address hypovolemia prior to initiating Norepinephrine in Sodium Chloride Injection [see Dosage and Administration (2.1)]. Avoid Norepinephrine in Sodium Chloride Injection in patients with mesenteric or peripheral vascular thrombosis, as this may increase ischemia and extend the area of infarction.
Gangrene of the extremities has occurred in patients with occlusive or thrombotic vascular disease or who received prolonged or high dose infusions. Monitor for changes to the skin of the extremities in susceptible patients.
Extravasation of Norepinephrine in Sodium Chloride Injection may cause necrosis and sloughing of surrounding tissue. To reduce the risk of extravasation, infuse into a large vein, check the infusion site frequently for free flow, and monitor for signs of extravasation [see Dosage and Administration (2.1)].
6 Adverse Reactions
The following adverse reactions are described in greater detail in other sections:
- Tissue Ischemia [see Warnings and Precautions (5.1)]
- Hypotension [see Warnings and Precautions (5.2)]
- Cardiac Arrhythmias [see Warnings and Precautions (5.3)]
The most common adverse reactions are hypertension and bradycardia.
The following adverse reactions can occur:
Nervous system disorders: Anxiety, headache
Respiratory disorders: Respiratory difficulty, pulmonary edema
7 Drug Interactions
12.2 Pharmacodynamics
The primary pharmacodynamic effects of norepinephrine are cardiac stimulation and vasoconstriction. Cardiac output is generally unaffected, although it can be decreased, and total peripheral resistance is also elevated. The elevation in resistance and pressure result in reflex vagal activity, which slows the heart rate and increases stroke volume. The elevation in vascular tone or resistance reduces blood flow to the major abdominal organs as well as to skeletal muscle. Coronary blood flow is substantially increased secondary to the indirect effects of alpha stimulation. After intravenous administration, a pressor response occurs rapidly and reaches steady state within 5 minutes. The pharmacologic actions of norepinephrine are terminated primarily by uptake and metabolism in sympathetic nerve endings. The pressor action stops within 1-2 minutes after the infusion is discontinued.
1 Indications and Usage
Norepinephrine in Sodium Chloride Injection is indicated to raise blood pressure in adult patients with severe, acute hypotension.
5.3 Cardiac Arrhythmias
Norepinephrine in Sodium Chloride Injection elevates intracellular calcium concentrations and may cause arrhythmias, particularly in the setting of hypoxia or hypercarbia. Perform continuous cardiac monitoring of patients with arrhythmias.
12.1 Mechanism of Action
Norepinephrine is a peripheral vasoconstrictor (alpha-adrenergic action) and an inotropic stimulator of the heart and dilator of coronary arteries (beta-adrenergic action).
7.1 Mao Inhibiting Drugs
Co-administration of Norepinephrine in Sodium Chloride Injection with monoamine oxidase (MAO) inhibitors or other drugs with MAO-inhibiting properties (e.g., linezolid) can cause severe, prolonged hypertension.
If administration of Norepinephrine in Sodium Chloride Injection cannot be avoided in patients who recently have received any of these drugs and in whom, after discontinuation, MAO activity has not yet sufficiently recovered, monitor for hypertension.
2.3 Drug Incompatibilities
Avoid contact with iron salts and alkalizing and oxidizing agents.
Whole blood or plasma, if indicated to increase blood volume, should be administered separately.
5 Warnings and Precautions
- Tissue Ischemia: Avoid extravasation into tissues, which can cause local necrosis (5.1)
- Hypotension After Abrupt Discontinuation: Sudden cessation of the infusion rate may result in marked hypotension. Reduce the Norepinephrine in Sodium Chloride Injection infusion rate gradually. (5.2)
- Cardiac Arrhythmias: Norepinephrine in Sodium Chloride Injection may cause arrhythmias. Monitor cardiac function in patients with underlying heart disease. (5.3)
2 Dosage and Administration
- No further dilution prior to infusion is required (2.1)
- Initial intravenous infusion rate of 8 to 12 mcg per minute, adjust the rate of flow to establish and maintain a low to normal blood pressure (usually 80 to 100 mm Hg) sufficient to maintain the circulation of vital organs (2.2)
- The average maintenance dose ranges from 2 to 4 mcg per minute. (2.2)
7.4 Halogenated Anesthetics
Concomitant use of Norepinephrine in Sodium Chloride Injection with halogenated anesthetics (e.g., cyclopropane, desflurane, enflurane, isoflurane, and sevoflurane) may lead to ventricular tachycardia or ventricular fibrillation. Monitor cardiac rhythm in patients receiving concomitant halogenated anesthetics.
3 Dosage Forms and Strengths
Injection: Norepinephrine is a clear, colorless sterile solution in the premixed, ready-to-use single dose intravenous infusion bags available as:
- 4 mg/250 mL (16 mcg/mL) of norepinephrine in 0.9% Sodium Chloride
- 8 mg/250 mL (32 mcg/mL) of norepinephrine in 0.9% Sodium Chloride
- 16 mg/250 mL (64 mcg/mL) of norepinephrine in 0.9% Sodium Chloride
7.2 Tricyclic Antidepressants
Co-administration of Norepinephrine in Sodium Chloride Injection with tricyclic antidepressants (including amitriptyline, nortriptyline, protriptyline, clomipramine, desipramine, imipramine) can cause severe, prolonged hypertension. If administration of Norepinephrine in Sodium Chloride Injection cannot be avoided in these patients, monitor for hypertension.
8 Use in Specific Populations
- Elderly patients may be at greater risk of developing adverse reactions. (8.5)
16 How Supplied/storage and Handling
Norepinephrine in Sodium Chloride Injection (norepinephrine bitartrate) is supplied as a clear, colorless sterile solution in a 250 mL non-PVC infusion bag with single function connector system consisting of a port and cap, packaged individually in an aluminum foil pouch with an oxygen scavenger. Supplied as:
| Unit of Sale | Concentration | Package Size |
|---|---|---|
| NDC 81298-9659-3 | 4 mg/250 mL (16 mcg/mL) |
10 bags |
| NDC 81298-9655-3 | 8 mg/250 mL (32 mcg/mL) |
10 bags |
| NDC 81298-9658-3 | 16 mg/250 mL (64 mcg/mL) |
10 bags |
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature.] Protect from light. Once the overwrap is removed, the bag can be stored at room temperature for up to 45 days.
5.2 Hypotension After Abrupt Discontinuation
Sudden cessation of the infusion rate may result in marked hypotension. When discontinuing the infusion, gradually reduce the Norepinephrine in Sodium Chloride Injection infusion rate while expanding blood volume with intravenous fluids.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis, mutagenesis, and fertility studies have not been performed.
Structured Label Content
Section 42229-5 (42229-5)
Correct Hypovolemia
Address hypovolemia before initiation of Norepinephrine in Sodium Chloride Injection therapy. If the patient does not respond to therapy, suspect occult hypovolemia [see Warnings and Precautions (5.1)].
Section 51945-4 (51945-4)
Principal Display Panel – 4 mg Pouch Label
NDC: 81298-9659-1
Rx only
1 x 250 mL
Norepinephrine
in 0.9% Sodium Chloride injection
4 mg per 250 mL
(16 mcg/mL)
Keep in sealed overwrap until time of use.
LONG GROVE
PHARMACEUTICALS™
2.2 Dosage
After an initial dose of 8 to 12 mcg per minute via intravenous infusion, assess patient response and adjust dosage to maintain desired hemodynamic effect. Monitor blood pressure every two minutes or continuously until the desired hemodynamic effect is achieved, and then monitor blood pressure every five minutes for the duration of the infusion.
Recommended Average Maintenance Dosage:
Typical maintenance intravenous dosage is 2 to 4 mcg per minute.
10 Overdosage (10 OVERDOSAGE)
Overdosage with Norepinephrine in Sodium Chloride Injection may result in headache, severe hypertension, reflex bradycardia, marked increase in peripheral resistance, and decreased cardiac output.
In case of accidental overdosage, discontinue Norepinephrine in Sodium Chloride Injection until the condition of the patient stabilizes.
11 Description (11 DESCRIPTION)
Norepinephrine (sometimes referred to as l-arterenol/Levarterenol or l-norepinephrine) is a catecholamine which differs from epinephrine by the absence of a methyl group on the nitrogen atom.
Chemically, Norepinephrine Bitartrate is (-)-α-(aminomethyl)-3,4-dihydroxybenzyl alcohol tartrate (1:1) (salt) monohydrate (molecular weight 337.3 g/mol) and has the following structural formula:
Norepinephrine in Sodium Chloride Injection is a clear, colorless, single dose sterile solution supplied as a ready-to-use intravenous infusion bag for intravenous use and does not require further dilution. Each mL contains the equivalent of 16 or 32 or 64 micrograms of norepinephrine base supplied as 31.90 or 63.80 or 127.6 micrograms per mL of norepinephrine bitartrate monohydrate. In addition, each mL of solution contains 0.01 mg edetate disodium dihydrate as a metal chelator and 9.0 mg sodium chloride for isotonicity. It has a pH of 3.5 to 4.5.
7.3 Antidiabetics
Norepinephrine in Sodium Chloride Injection can decrease insulin sensitivity and raise blood glucose. Monitor glucose and consider dosage adjustment of antidiabetic drugs.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of Norepinephrine in Sodium Chloride Injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Avoid administration of Norepinephrine in Sodium Chloride Injection into the veins in the leg in elderly patients [see Warnings and Precautions (5.1)].
4 Contraindications (4 CONTRAINDICATIONS)
None.
5.1 Tissue Ischemia
Administration of Norepinephrine in Sodium Chloride Injection to patients who are hypotensive from hypovolemia can result in severe peripheral and visceral vasoconstriction, decreased renal perfusion and reduced urine output, tissue hypoxia, lactic acidosis, and reduced systemic blood flow despite “normal” blood pressure. Address hypovolemia prior to initiating Norepinephrine in Sodium Chloride Injection [see Dosage and Administration (2.1)]. Avoid Norepinephrine in Sodium Chloride Injection in patients with mesenteric or peripheral vascular thrombosis, as this may increase ischemia and extend the area of infarction.
Gangrene of the extremities has occurred in patients with occlusive or thrombotic vascular disease or who received prolonged or high dose infusions. Monitor for changes to the skin of the extremities in susceptible patients.
Extravasation of Norepinephrine in Sodium Chloride Injection may cause necrosis and sloughing of surrounding tissue. To reduce the risk of extravasation, infuse into a large vein, check the infusion site frequently for free flow, and monitor for signs of extravasation [see Dosage and Administration (2.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described in greater detail in other sections:
- Tissue Ischemia [see Warnings and Precautions (5.1)]
- Hypotension [see Warnings and Precautions (5.2)]
- Cardiac Arrhythmias [see Warnings and Precautions (5.3)]
The most common adverse reactions are hypertension and bradycardia.
The following adverse reactions can occur:
Nervous system disorders: Anxiety, headache
Respiratory disorders: Respiratory difficulty, pulmonary edema
7 Drug Interactions (7 DRUG INTERACTIONS)
12.2 Pharmacodynamics
The primary pharmacodynamic effects of norepinephrine are cardiac stimulation and vasoconstriction. Cardiac output is generally unaffected, although it can be decreased, and total peripheral resistance is also elevated. The elevation in resistance and pressure result in reflex vagal activity, which slows the heart rate and increases stroke volume. The elevation in vascular tone or resistance reduces blood flow to the major abdominal organs as well as to skeletal muscle. Coronary blood flow is substantially increased secondary to the indirect effects of alpha stimulation. After intravenous administration, a pressor response occurs rapidly and reaches steady state within 5 minutes. The pharmacologic actions of norepinephrine are terminated primarily by uptake and metabolism in sympathetic nerve endings. The pressor action stops within 1-2 minutes after the infusion is discontinued.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Norepinephrine in Sodium Chloride Injection is indicated to raise blood pressure in adult patients with severe, acute hypotension.
5.3 Cardiac Arrhythmias
Norepinephrine in Sodium Chloride Injection elevates intracellular calcium concentrations and may cause arrhythmias, particularly in the setting of hypoxia or hypercarbia. Perform continuous cardiac monitoring of patients with arrhythmias.
12.1 Mechanism of Action
Norepinephrine is a peripheral vasoconstrictor (alpha-adrenergic action) and an inotropic stimulator of the heart and dilator of coronary arteries (beta-adrenergic action).
7.1 Mao Inhibiting Drugs (7.1 MAO-Inhibiting Drugs)
Co-administration of Norepinephrine in Sodium Chloride Injection with monoamine oxidase (MAO) inhibitors or other drugs with MAO-inhibiting properties (e.g., linezolid) can cause severe, prolonged hypertension.
If administration of Norepinephrine in Sodium Chloride Injection cannot be avoided in patients who recently have received any of these drugs and in whom, after discontinuation, MAO activity has not yet sufficiently recovered, monitor for hypertension.
2.3 Drug Incompatibilities
Avoid contact with iron salts and alkalizing and oxidizing agents.
Whole blood or plasma, if indicated to increase blood volume, should be administered separately.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Tissue Ischemia: Avoid extravasation into tissues, which can cause local necrosis (5.1)
- Hypotension After Abrupt Discontinuation: Sudden cessation of the infusion rate may result in marked hypotension. Reduce the Norepinephrine in Sodium Chloride Injection infusion rate gradually. (5.2)
- Cardiac Arrhythmias: Norepinephrine in Sodium Chloride Injection may cause arrhythmias. Monitor cardiac function in patients with underlying heart disease. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- No further dilution prior to infusion is required (2.1)
- Initial intravenous infusion rate of 8 to 12 mcg per minute, adjust the rate of flow to establish and maintain a low to normal blood pressure (usually 80 to 100 mm Hg) sufficient to maintain the circulation of vital organs (2.2)
- The average maintenance dose ranges from 2 to 4 mcg per minute. (2.2)
7.4 Halogenated Anesthetics
Concomitant use of Norepinephrine in Sodium Chloride Injection with halogenated anesthetics (e.g., cyclopropane, desflurane, enflurane, isoflurane, and sevoflurane) may lead to ventricular tachycardia or ventricular fibrillation. Monitor cardiac rhythm in patients receiving concomitant halogenated anesthetics.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: Norepinephrine is a clear, colorless sterile solution in the premixed, ready-to-use single dose intravenous infusion bags available as:
- 4 mg/250 mL (16 mcg/mL) of norepinephrine in 0.9% Sodium Chloride
- 8 mg/250 mL (32 mcg/mL) of norepinephrine in 0.9% Sodium Chloride
- 16 mg/250 mL (64 mcg/mL) of norepinephrine in 0.9% Sodium Chloride
7.2 Tricyclic Antidepressants
Co-administration of Norepinephrine in Sodium Chloride Injection with tricyclic antidepressants (including amitriptyline, nortriptyline, protriptyline, clomipramine, desipramine, imipramine) can cause severe, prolonged hypertension. If administration of Norepinephrine in Sodium Chloride Injection cannot be avoided in these patients, monitor for hypertension.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Elderly patients may be at greater risk of developing adverse reactions. (8.5)
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Norepinephrine in Sodium Chloride Injection (norepinephrine bitartrate) is supplied as a clear, colorless sterile solution in a 250 mL non-PVC infusion bag with single function connector system consisting of a port and cap, packaged individually in an aluminum foil pouch with an oxygen scavenger. Supplied as:
| Unit of Sale | Concentration | Package Size |
|---|---|---|
| NDC 81298-9659-3 | 4 mg/250 mL (16 mcg/mL) |
10 bags |
| NDC 81298-9655-3 | 8 mg/250 mL (32 mcg/mL) |
10 bags |
| NDC 81298-9658-3 | 16 mg/250 mL (64 mcg/mL) |
10 bags |
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature.] Protect from light. Once the overwrap is removed, the bag can be stored at room temperature for up to 45 days.
5.2 Hypotension After Abrupt Discontinuation (5.2 Hypotension after Abrupt Discontinuation)
Sudden cessation of the infusion rate may result in marked hypotension. When discontinuing the infusion, gradually reduce the Norepinephrine in Sodium Chloride Injection infusion rate while expanding blood volume with intravenous fluids.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis, mutagenesis, and fertility studies have not been performed.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:58.575695 · Updated: 2026-03-14T22:39:05.457483