These Highlights Do Not Include All The Information Needed To Use Taclonex Topical Suspension Safely And Effectively. See Full Prescribing Information For Taclonex Topical Suspension.
757c914f-dd8d-43cc-a973-212d591af4af
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Taclonex ® Topical Suspension is indicated for the topical treatment of plaque psoriasis of the scalp and body in patients 12 years and older.
Indications and Usage
Taclonex ® Topical Suspension is indicated for the topical treatment of plaque psoriasis of the scalp and body in patients 12 years and older.
Dosage and Administration
Instruct patients to shake bottle prior to using Taclonex Topical Suspension. Apply Taclonex Topical Suspension to affected areas on the scalp and body once daily for up to 8 weeks. Taclonex Topical Suspension should be discontinued when control is achieved. Instruct patients to wash their hands after applying the product. Inform patients that they should not take a bath or shower or wash their hair right after application of Taclonex Topical Suspension. Patients 12 to 17 years should not use more than 60 grams per week and patients 18 years and older should not use more than 100 grams per week. Taclonex Topical Suspension should not be: Used with occlusive dressings unless directed by a healthcare provider. Used on the face, groin, or axillae, or if skin atrophy is present at the treatment site. Applied to the scalp in the 12 hours before or after any chemical treatments to the hair. Taclonex Topical Suspension is not for oral, ophthalmic, or intravaginal use.
Warnings and Precautions
Hypercalcemia and Hypercalciuria: Hypercalcemia and hypercalciuria have been reported. If either occurs, discontinue until parameters of calcium metabolism normalize. ( 5.1 ) Effects on Endocrine System: Can cause reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency during and after withdrawal of treatment. Risk factors include the use of high-potency topical corticosteroid, use over a large surface area or to areas under occlusion, prolonged use, altered skin barrier, liver failure, and use in pediatric patients. Modify use should HPA axis suppression develop. ( 5.2 , 8.4 ) Ophthalmic Adverse Reactions: May increase the risk of cataracts and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist. ( 5.5 )
Contraindications
None.
Adverse Reactions
The most common adverse reactions (≥ 1%) are folliculitis and burning sensation of skin. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact LEO Pharma Inc. at 1-877-494-4536 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
Taclonex (calcipotriene and betamethasone dipropionate) Topical Suspension, 0.005%/0.064% is a viscous, nearly odorless, almost clear, colorless to slightly off-white suspension. It is available as: 60 gram bottle (NDC 50222-501-06) 120 gram (2 bottles of 60 gram) (NDC 50222-501-66)
How Supplied
Taclonex (calcipotriene and betamethasone dipropionate) Topical Suspension, 0.005%/0.064% is a viscous, nearly odorless, almost clear, colorless to slightly off-white suspension. It is available as: 60 gram bottle (NDC 50222-501-06) 120 gram (2 bottles of 60 gram) (NDC 50222-501-66)
Medication Information
Warnings and Precautions
Hypercalcemia and Hypercalciuria: Hypercalcemia and hypercalciuria have been reported. If either occurs, discontinue until parameters of calcium metabolism normalize. ( 5.1 ) Effects on Endocrine System: Can cause reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency during and after withdrawal of treatment. Risk factors include the use of high-potency topical corticosteroid, use over a large surface area or to areas under occlusion, prolonged use, altered skin barrier, liver failure, and use in pediatric patients. Modify use should HPA axis suppression develop. ( 5.2 , 8.4 ) Ophthalmic Adverse Reactions: May increase the risk of cataracts and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist. ( 5.5 )
Indications and Usage
Taclonex ® Topical Suspension is indicated for the topical treatment of plaque psoriasis of the scalp and body in patients 12 years and older.
Dosage and Administration
Instruct patients to shake bottle prior to using Taclonex Topical Suspension. Apply Taclonex Topical Suspension to affected areas on the scalp and body once daily for up to 8 weeks. Taclonex Topical Suspension should be discontinued when control is achieved. Instruct patients to wash their hands after applying the product. Inform patients that they should not take a bath or shower or wash their hair right after application of Taclonex Topical Suspension. Patients 12 to 17 years should not use more than 60 grams per week and patients 18 years and older should not use more than 100 grams per week. Taclonex Topical Suspension should not be: Used with occlusive dressings unless directed by a healthcare provider. Used on the face, groin, or axillae, or if skin atrophy is present at the treatment site. Applied to the scalp in the 12 hours before or after any chemical treatments to the hair. Taclonex Topical Suspension is not for oral, ophthalmic, or intravaginal use.
Contraindications
None.
Adverse Reactions
The most common adverse reactions (≥ 1%) are folliculitis and burning sensation of skin. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact LEO Pharma Inc. at 1-877-494-4536 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
Taclonex (calcipotriene and betamethasone dipropionate) Topical Suspension, 0.005%/0.064% is a viscous, nearly odorless, almost clear, colorless to slightly off-white suspension. It is available as: 60 gram bottle (NDC 50222-501-06) 120 gram (2 bottles of 60 gram) (NDC 50222-501-66)
How Supplied
Taclonex (calcipotriene and betamethasone dipropionate) Topical Suspension, 0.005%/0.064% is a viscous, nearly odorless, almost clear, colorless to slightly off-white suspension. It is available as: 60 gram bottle (NDC 50222-501-06) 120 gram (2 bottles of 60 gram) (NDC 50222-501-66)
Description
Taclonex ® Topical Suspension is indicated for the topical treatment of plaque psoriasis of the scalp and body in patients 12 years and older.
Section 42229-5
Taclonex Topical Suspension can cause reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for clinical glucocorticosteroid insufficiency. This may occur during treatment or upon withdrawal of treatment. Factors that predispose a patient to HPA axis suppression include the use of high-potency steroids, large treatment surface areas, prolonged use, use of occlusive dressings, altered skin barrier, liver failure, and young age.
Evaluation for HPA axis suppression may be done by using the adrenocorticotropic hormone (ACTH) stimulation test. If HPA axis suppression is documented, gradually withdraw Taclonex Topical Suspension, reduce the frequency of application, or substitute with a less potent corticosteroid.
The following trials evaluated the effects of Taclonex Topical Suspension on HPA axis suppression:
- In a trial evaluating the effects of Taclonex Topical Suspension and Taclonex Ointment on the HPA axis, 32 adult subjects applied both Taclonex Topical Suspension on the scalp and Taclonex Ointment on the body. Adrenal suppression was identified in 5 of 32 subjects (16%) after 4 weeks of treatment and in 2 of 11 subjects (18%) who continued treatment for 8 weeks. In another trial, 36 adult subjects applied Taclonex Topical Suspension on the body and scalp and 7 subjects applied Taclonex Topical Suspension on the body. Adrenal suppression occurred in 3 out of 43 subjects (7%) after 4 weeks of treatment and in none of the 36 subjects who continued treatment for 8 weeks [see Clinical Pharmacology (12.2) ].
- In two trials, the effects of Taclonex Topical Suspension on the HPA axis were evaluated in 31 and 30 pediatric subjects aged 12 to 17 years old who applied Taclonex Topical suspension on the scalp and the scalp/body, respectively. Adrenal suppression occurred in 1 of 30 evaluable subjects (3%) after 4 weeks of treatment (scalp) and 5 of 31 evaluable subjects (16%) after up to 8 weeks of treatment (scalp and body) [see Use in Specific Populations (8.4) and Clinical Pharmacology (12.2)].
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 7/2019 | |
|
PATIENT INFORMATION TACLONEX® (TAK-lo-NEKS) (calcipotriene and betamethasone dipropionate) Topical Suspension |
||
| Important: Taclonex Topical Suspension is for use on skin only (topical). Do not get Taclonex Topical Suspension near or in your mouth, eyes, or vagina. | ||
| There are other medicines that contain the same medicine that is in Taclonex Topical Suspension and are used to treat plaque psoriasis. Do not use other products containing calcipotriene or a corticosteroid medicine with Taclonex Topical Suspension without talking to your healthcare provider first. | ||
|
What is Taclonex Topical Suspension?
Taclonex Topical Suspension is a prescription medicine used on the skin (topical) to treat plaque psoriasis of the scalp and body in people 12 years and older. It is not known if Taclonex Topical Suspension is safe and effective in children under 12 years of age. |
||
Before you use Taclonex Topical Suspension, tell your healthcare provider about all of your medical conditions, including if you:
|
||
|
How should I use Taclonex Topical Suspension?
See the "Instructions for Use" for detailed information about the right way to use Taclonex Topical Suspension.
|
||
|
What are the possible side effects of Taclonex® Topical Suspension?
Taclonex Topical Suspension may cause serious side effects, including:
|
||
|
|
|
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store Taclonex Topical Suspension?
|
||
|
General information about Taclonex Topical Suspension.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Taclonex Topical Suspension for a condition for which it was not prescribed. Do not give Taclonex Topical Suspension to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Taclonex Topical Suspension that is written for health professionals. |
||
|
What are the ingredients in Taclonex Topical Suspension?
Active ingredients: calcipotriene and betamethasone dipropionate. Inactive ingredients: hydrogenated castor oil, polyoxypropylene stearyl ether, all-rac-alpha-tocopherol, butylhydroxytoluene and mineral oil. Manufactured by: LEO Laboratories Ltd., 285 Cashel Road, Dublin 12, Ireland Distributed by: LEO Pharma Inc., Madison, NJ 07940, USA For more information, go to www.taclonex.com or call 1-877-494-4536. |
Section 44425-7
Store between 20°C - 25°C (68°F - 77°F); excursions permitted between 15°C - 30°C (59°F - 86°F). [See USP controlled room temperature]. Do not refrigerate.
Keep the bottle in the carton when not in use. Unused product should be discarded six months after the bottle has been opened.
Shake before use. Keep out of reach of children.
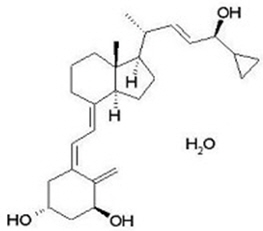
11 Description
Taclonex Topical Suspension contains calcipotriene hydrate and betamethasone dipropionate. It is for topical use only. Calcipotriene hydrate is a synthetic vitamin D3 analog.
8.4 Pediatric Use
The safety and effectiveness of Taclonex Topical Suspension for the treatment of plaque psoriasis of the scalp and body have been established in pediatric patients age 12 to 17 years. The use of Taclonex Topical Suspension for this indication is supported by evidence from adequate and well-controlled trials in adults and from three uncontrolled trials in pediatric subjects that enrolled 109 adolescents with moderate psoriasis of the scalp and 107 adolescents with psoriasis of the scalp and body. After 4 weeks of once daily treatment with Taclonex Topical Suspension, HPA axis suppression was observed in 3% of adolescents with psoriasis of the scalp and 16% of adolescents with psoriasis of the scalp and body. Calcium metabolism was evaluated in 107 adolescents with psoriasis of the scalp and body treated with Taclonex Topical Suspension and no cases of hypercalcemia or clinically relevant changes in urinary calcium were reported [see Warnings and Precautions (5.2), Adverse Reactions (6.1), and Clinical Pharmacology (12.2)].
Because of a higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of systemic toxicity when treated with topical corticosteroids. Pediatric patients are, therefore, also at greater risk of HPA axis suppression and adrenal insufficiency with the use of topical corticosteroids including Taclonex® Topical Suspension [see Clinical Pharmacology (12.2)].
Rare systemic toxicities such as Cushing's syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in pediatric patients, especially those with prolonged exposure to large doses of high potency topical corticosteroids. Local adverse reactions including striae have also been reported with use of topical corticosteroids in pediatric patients.
The safety and effectiveness of Taclonex Topical Suspension in pediatric patients less than 12 years of age have not been established.
8.5 Geriatric Use
Clinical studies of Taclonex Topical Suspension in plaque psoriasis on non-scalp areas included 124 subjects who were 65 years of age or older, and 36 were 75 years of age or older. Clinical studies of Taclonex Topical Suspension in subjects with psoriasis of the scalp included 334 subjects who were 65 years or older and 84 subjects who were 75 years or older.
No overall differences in safety or effectiveness of Taclonex Topical Suspension were observed between these subjects and younger subjects, and other reported clinical experience has not identified any differences in responses between the elderly and younger subjects, but greater sensitivity of some older individuals cannot be ruled out.
4 Contraindications
None.
6 Adverse Reactions
The most common adverse reactions (≥ 1%) are folliculitis and burning sensation of skin. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact LEO Pharma Inc. at 1-877-494-4536 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Instructions for Use
TACLONEX® (TAK-lo-NEKS)
(calcipotriene and betamethasone dipropionate)
Topical Suspension
Important: Taclonex Topical Suspension is for use on skin only (topical). Do not get Taclonex Topical Suspension near or in your mouth, eyes, or vagina.
Read this Instructions for Use before you start using Taclonex Topical Suspension and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment.
How to apply Taclonex Topical Suspension to your body:
Follow your healthcare provider's instructions of how much Taclonex Topical Suspension to use and where to use it. Apply Taclonex Topical Suspension directly to areas affected by plaque psoriasis and gently rub in. Wash your hands after applying Taclonex Topical Suspension, unless you are treating areas on your hands.
How to apply Taclonex Topical Suspension to your scalp:
You do not need to wash your hair before you apply Taclonex Topical Suspension.
|
|
Step 1: Shake the bottle before use. Remove the cap from the bottle. (See Figure A). |
| Step 2: Locate the area to treat using your fingers and part your hair. (See Figure B). | |
| Step 3: Squeeze a drop of Taclonex Topical Suspension to your fingertip. (See Figure C). | |
| Step 4: Use your fingers to apply the drop of Taclonex Topical Suspension directly to scalp affected by plaque psoriasis. Gently rub in. (See Figure D). | |
|
Step 5: After applying Taclonex Topical Suspension, put the cap back on the bottle. Step 6: Wash your hands after applying Taclonex Topical Suspension. Do not wash your hair right after you apply Taclonex Topical Suspension to your scalp. |
How should I store Taclonex® Topical Suspension?
- Store the Taclonex Topical Suspension at room temperature between 68°F to 77°F (20°C to 25°C).
- Do not refrigerate Taclonex Topical Suspension.
- Keep bottle in the carton when not in use.
- Discard unused Taclonex Topical Suspension 6 months after it has been opened.
Keep Taclonex Topical Suspension and all medicines out of reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
LEO Laboratories Ltd.
285 Cashel Road
Dublin 12, Ireland
Distributed by:
LEO Pharma Inc.
Madison, NJ 07940, USA
Revised: 7/2019
1 Indications and Usage
Taclonex® Topical Suspension is indicated for the topical treatment of plaque psoriasis of the scalp and body in patients 12 years and older.
12.1 Mechanism of Action
Taclonex Topical Suspension combines the pharmacological effects of calcipotriene hydrate as a synthetic vitamin D3 analog and betamethasone dipropionate as a synthetic corticosteroid. However, while their pharmacologic and clinical effects are known, the exact mechanisms of their actions in the treatment of plaque psoriasis of the scalp and body are unknown.
5 Warnings and Precautions
- Hypercalcemia and Hypercalciuria: Hypercalcemia and hypercalciuria have been reported. If either occurs, discontinue until parameters of calcium metabolism normalize. (5.1)
- Effects on Endocrine System: Can cause reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency during and after withdrawal of treatment. Risk factors include the use of high-potency topical corticosteroid, use over a large surface area or to areas under occlusion, prolonged use, altered skin barrier, liver failure, and use in pediatric patients. Modify use should HPA axis suppression develop. (5.2, 8.4)
- Ophthalmic Adverse Reactions: May increase the risk of cataracts and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist. (5.5)
2 Dosage and Administration
Instruct patients to shake bottle prior to using Taclonex Topical Suspension. Apply Taclonex Topical Suspension to affected areas on the scalp and body once daily for up to 8 weeks. Taclonex Topical Suspension should be discontinued when control is achieved. Instruct patients to wash their hands after applying the product. Inform patients that they should not take a bath or shower or wash their hair right after application of Taclonex Topical Suspension.
Patients 12 to 17 years should not use more than 60 grams per week and patients 18 years and older should not use more than 100 grams per week.
Taclonex Topical Suspension should not be:
- Used with occlusive dressings unless directed by a healthcare provider.
- Used on the face, groin, or axillae, or if skin atrophy is present at the treatment site.
- Applied to the scalp in the 12 hours before or after any chemical treatments to the hair.
Taclonex Topical Suspension is not for oral, ophthalmic, or intravaginal use.
3 Dosage Forms and Strengths
Topical Suspension: 0.005%/0.064% - each gram contains 50 mcg of calcipotriene and 0.643 mg of betamethasone dipropionate in a viscous, nearly odorless, almost clear, colorless to slightly off-white suspension.
6.2 Postmarketing Experience
Because adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Postmarketing reports for local adverse reactions to topical corticosteroids included atrophy, striae, telangiectasias, itching, dryness, hypopigmentation, perioral dermatitis, secondary infection, and miliaria.
Ophthalmic adverse reactions of cataracts, glaucoma, increased intraocular pressure, and central serous chorioretinopathy have been reported during use of topical corticosteroids, including topical betamethasone products.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.5 Ophthalmic Adverse Reactions
Use of topical corticosteroids, including Taclonex® Topical Suspension, may increase the risk of posterior subcapsular cataracts and glaucoma. Cataracts and glaucoma have been reported with the postmarketing use of topical corticosteroid products [see Adverse Reactions (6.2)]. Avoid contact of Taclonex Topical Suspension with eyes. Taclonex Topical Suspension may cause eye irritation. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
17 Patient Counseling Information
See FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied/storage and Handling
Taclonex (calcipotriene and betamethasone dipropionate) Topical Suspension, 0.005%/0.064% is a viscous, nearly odorless, almost clear, colorless to slightly off-white suspension. It is available as:
- 60 gram bottle (NDC 50222-501-06)
- 120 gram (2 bottles of 60 gram) (NDC 50222-501-66)
5.1 Hypercalcemia and Hypercalciuria
Hypercalcemia and hypercalciuria have been observed with use of Taclonex Topical Suspension. If hypercalcemia or hypercalciuria develop, discontinue treatment until parameters of calcium metabolism have normalized. The incidence of hypercalcemia and hypercalciuria following Taclonex Topical Suspension treatment of more than 8 weeks has not been evaluated [see Clinical Pharmacology (12.2)].
Principal Display Panel 60 G Carton Ndc 50222 501 06
NDC 50222-501-06
Rx only
Taclonex®
(calcipotriene and
betamethasone
dipropionate)
Topical Suspension,
0.005%/0.064%
Shake before use
For Topical Use Only
Net Wt. 60 gram
LEO®
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
When calcipotriene was applied topically to mice for up to 24 months at dosages of 3, 10, and 30 mcg/kg/day (9, 30, and 90 mcg/m2/day, respectively), no significant changes in tumor incidence were observed when compared to control.
A 104-week oral carcinogenicity study was conducted with calcipotriene in male and female rats at doses of 1, 5 and 15 mcg/kg/day (6, 30, and 90 mcg/m2/day, respectively). Beginning week 71, the dosage for high-dose animals of both genders was reduced to 10 mcg/kg/day (60 mcg/m2/day). A treatment-related increase in benign C-cell adenomas was observed in the thyroid of females that received 15 mcg/kg/day. A treatment-related increase in benign pheochromocytomas was observed in the adrenal glands of males that received 15 mcg/kg/day. No other statistically significant differences in tumor incidence were observed when compared to control. The relevance of these findings to patients is unknown.
When betamethasone dipropionate was applied topically to CD-1 mice for up to 24 months at dosages approximating 1.3, 4.2, and 8.5 mcg/kg/day in females, and 1.3, 4.2, and 12.9 mcg/kg/day in males (up to 26 mcg/m2/day and 39 mcg/m2/day, in females and males, respectively), no significant changes in tumor incidence were observed when compared to control.
When betamethasone dipropionate was administered via oral gavage to male and female Sprague Dawley rats for up to 24 months at dosages of 20, 60, and 200 mcg/kg/day (120, 360, and 1200 mcg/m2/day, respectively), no significant changes in tumor incidence were observed when compared to control.
Calcipotriene did not elicit any genotoxic effects in the Ames mutagenicity assay, the mouse lymphoma TK locus assay, the human lymphocyte chromosome aberration test, or the mouse micronucleus test. Betamethasone dipropionate did not elicit any genotoxic effects in the Ames mutagenicity assay, the mouse lymphoma TK locus assay, or in the rat micronucleus test.
Studies in rats with oral doses of up to 54 mcg/kg/day (324 mcg/m2/day) of calcipotriene indicated no impairment of fertility or general reproductive performance. Studies in male rats at oral doses of up to 200 mcg/kg/day (1200 mcg/m2/day), and in female rats at oral doses of up to 1000 mcg/kg/day (6000 mcg/m2/day), of betamethasone dipropionate indicated no impairment of fertility.
Principal Display Panel 120 G Carton Ndc 50222 501 66
NDC 50222-501-66
Rx only
Taclonex®
(calcipotriene and
betamethasone
dipropionate)
Topical Suspension,
0.005%/0.064%
Shake before use
For Topical Use Only
Net Wt. 120 gram
(2 bottles of 60 gram)
Individual bottles (60 gram)
not for individual retail sale
LEO®
5.4 Allergic Contact Dermatitis With Topical Calcipotriene
Allergic contact dermatitis has been observed with use of topical calcipotriene. Such an observation should be corroborated with appropriate diagnostic patch testing.
5.3 Allergic Contact Dermatitis With Topical Corticosteroids
Allergic contact dermatitis to a topical corticosteroid is usually diagnosed by observing a failure to heal rather than a clinical exacerbation. Such an observation should be corroborated with appropriate diagnostic patch testing.
Structured Label Content
Section 42229-5 (42229-5)
Taclonex Topical Suspension can cause reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for clinical glucocorticosteroid insufficiency. This may occur during treatment or upon withdrawal of treatment. Factors that predispose a patient to HPA axis suppression include the use of high-potency steroids, large treatment surface areas, prolonged use, use of occlusive dressings, altered skin barrier, liver failure, and young age.
Evaluation for HPA axis suppression may be done by using the adrenocorticotropic hormone (ACTH) stimulation test. If HPA axis suppression is documented, gradually withdraw Taclonex Topical Suspension, reduce the frequency of application, or substitute with a less potent corticosteroid.
The following trials evaluated the effects of Taclonex Topical Suspension on HPA axis suppression:
- In a trial evaluating the effects of Taclonex Topical Suspension and Taclonex Ointment on the HPA axis, 32 adult subjects applied both Taclonex Topical Suspension on the scalp and Taclonex Ointment on the body. Adrenal suppression was identified in 5 of 32 subjects (16%) after 4 weeks of treatment and in 2 of 11 subjects (18%) who continued treatment for 8 weeks. In another trial, 36 adult subjects applied Taclonex Topical Suspension on the body and scalp and 7 subjects applied Taclonex Topical Suspension on the body. Adrenal suppression occurred in 3 out of 43 subjects (7%) after 4 weeks of treatment and in none of the 36 subjects who continued treatment for 8 weeks [see Clinical Pharmacology (12.2) ].
- In two trials, the effects of Taclonex Topical Suspension on the HPA axis were evaluated in 31 and 30 pediatric subjects aged 12 to 17 years old who applied Taclonex Topical suspension on the scalp and the scalp/body, respectively. Adrenal suppression occurred in 1 of 30 evaluable subjects (3%) after 4 weeks of treatment (scalp) and 5 of 31 evaluable subjects (16%) after up to 8 weeks of treatment (scalp and body) [see Use in Specific Populations (8.4) and Clinical Pharmacology (12.2)].
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 7/2019 | |
|
PATIENT INFORMATION TACLONEX® (TAK-lo-NEKS) (calcipotriene and betamethasone dipropionate) Topical Suspension |
||
| Important: Taclonex Topical Suspension is for use on skin only (topical). Do not get Taclonex Topical Suspension near or in your mouth, eyes, or vagina. | ||
| There are other medicines that contain the same medicine that is in Taclonex Topical Suspension and are used to treat plaque psoriasis. Do not use other products containing calcipotriene or a corticosteroid medicine with Taclonex Topical Suspension without talking to your healthcare provider first. | ||
|
What is Taclonex Topical Suspension?
Taclonex Topical Suspension is a prescription medicine used on the skin (topical) to treat plaque psoriasis of the scalp and body in people 12 years and older. It is not known if Taclonex Topical Suspension is safe and effective in children under 12 years of age. |
||
Before you use Taclonex Topical Suspension, tell your healthcare provider about all of your medical conditions, including if you:
|
||
|
How should I use Taclonex Topical Suspension?
See the "Instructions for Use" for detailed information about the right way to use Taclonex Topical Suspension.
|
||
|
What are the possible side effects of Taclonex® Topical Suspension?
Taclonex Topical Suspension may cause serious side effects, including:
|
||
|
|
|
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store Taclonex Topical Suspension?
|
||
|
General information about Taclonex Topical Suspension.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Taclonex Topical Suspension for a condition for which it was not prescribed. Do not give Taclonex Topical Suspension to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Taclonex Topical Suspension that is written for health professionals. |
||
|
What are the ingredients in Taclonex Topical Suspension?
Active ingredients: calcipotriene and betamethasone dipropionate. Inactive ingredients: hydrogenated castor oil, polyoxypropylene stearyl ether, all-rac-alpha-tocopherol, butylhydroxytoluene and mineral oil. Manufactured by: LEO Laboratories Ltd., 285 Cashel Road, Dublin 12, Ireland Distributed by: LEO Pharma Inc., Madison, NJ 07940, USA For more information, go to www.taclonex.com or call 1-877-494-4536. |
Section 44425-7 (44425-7)
Store between 20°C - 25°C (68°F - 77°F); excursions permitted between 15°C - 30°C (59°F - 86°F). [See USP controlled room temperature]. Do not refrigerate.
Keep the bottle in the carton when not in use. Unused product should be discarded six months after the bottle has been opened.
Shake before use. Keep out of reach of children.
11 Description (11 DESCRIPTION)
Taclonex Topical Suspension contains calcipotriene hydrate and betamethasone dipropionate. It is for topical use only. Calcipotriene hydrate is a synthetic vitamin D3 analog.
8.4 Pediatric Use
The safety and effectiveness of Taclonex Topical Suspension for the treatment of plaque psoriasis of the scalp and body have been established in pediatric patients age 12 to 17 years. The use of Taclonex Topical Suspension for this indication is supported by evidence from adequate and well-controlled trials in adults and from three uncontrolled trials in pediatric subjects that enrolled 109 adolescents with moderate psoriasis of the scalp and 107 adolescents with psoriasis of the scalp and body. After 4 weeks of once daily treatment with Taclonex Topical Suspension, HPA axis suppression was observed in 3% of adolescents with psoriasis of the scalp and 16% of adolescents with psoriasis of the scalp and body. Calcium metabolism was evaluated in 107 adolescents with psoriasis of the scalp and body treated with Taclonex Topical Suspension and no cases of hypercalcemia or clinically relevant changes in urinary calcium were reported [see Warnings and Precautions (5.2), Adverse Reactions (6.1), and Clinical Pharmacology (12.2)].
Because of a higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of systemic toxicity when treated with topical corticosteroids. Pediatric patients are, therefore, also at greater risk of HPA axis suppression and adrenal insufficiency with the use of topical corticosteroids including Taclonex® Topical Suspension [see Clinical Pharmacology (12.2)].
Rare systemic toxicities such as Cushing's syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in pediatric patients, especially those with prolonged exposure to large doses of high potency topical corticosteroids. Local adverse reactions including striae have also been reported with use of topical corticosteroids in pediatric patients.
The safety and effectiveness of Taclonex Topical Suspension in pediatric patients less than 12 years of age have not been established.
8.5 Geriatric Use
Clinical studies of Taclonex Topical Suspension in plaque psoriasis on non-scalp areas included 124 subjects who were 65 years of age or older, and 36 were 75 years of age or older. Clinical studies of Taclonex Topical Suspension in subjects with psoriasis of the scalp included 334 subjects who were 65 years or older and 84 subjects who were 75 years or older.
No overall differences in safety or effectiveness of Taclonex Topical Suspension were observed between these subjects and younger subjects, and other reported clinical experience has not identified any differences in responses between the elderly and younger subjects, but greater sensitivity of some older individuals cannot be ruled out.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions (≥ 1%) are folliculitis and burning sensation of skin. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact LEO Pharma Inc. at 1-877-494-4536 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Instructions for Use (INSTRUCTIONS FOR USE)
TACLONEX® (TAK-lo-NEKS)
(calcipotriene and betamethasone dipropionate)
Topical Suspension
Important: Taclonex Topical Suspension is for use on skin only (topical). Do not get Taclonex Topical Suspension near or in your mouth, eyes, or vagina.
Read this Instructions for Use before you start using Taclonex Topical Suspension and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment.
How to apply Taclonex Topical Suspension to your body:
Follow your healthcare provider's instructions of how much Taclonex Topical Suspension to use and where to use it. Apply Taclonex Topical Suspension directly to areas affected by plaque psoriasis and gently rub in. Wash your hands after applying Taclonex Topical Suspension, unless you are treating areas on your hands.
How to apply Taclonex Topical Suspension to your scalp:
You do not need to wash your hair before you apply Taclonex Topical Suspension.
|
|
Step 1: Shake the bottle before use. Remove the cap from the bottle. (See Figure A). |
| Step 2: Locate the area to treat using your fingers and part your hair. (See Figure B). | |
| Step 3: Squeeze a drop of Taclonex Topical Suspension to your fingertip. (See Figure C). | |
| Step 4: Use your fingers to apply the drop of Taclonex Topical Suspension directly to scalp affected by plaque psoriasis. Gently rub in. (See Figure D). | |
|
Step 5: After applying Taclonex Topical Suspension, put the cap back on the bottle. Step 6: Wash your hands after applying Taclonex Topical Suspension. Do not wash your hair right after you apply Taclonex Topical Suspension to your scalp. |
How should I store Taclonex® Topical Suspension?
- Store the Taclonex Topical Suspension at room temperature between 68°F to 77°F (20°C to 25°C).
- Do not refrigerate Taclonex Topical Suspension.
- Keep bottle in the carton when not in use.
- Discard unused Taclonex Topical Suspension 6 months after it has been opened.
Keep Taclonex Topical Suspension and all medicines out of reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
LEO Laboratories Ltd.
285 Cashel Road
Dublin 12, Ireland
Distributed by:
LEO Pharma Inc.
Madison, NJ 07940, USA
Revised: 7/2019
1 Indications and Usage (1 INDICATIONS AND USAGE)
Taclonex® Topical Suspension is indicated for the topical treatment of plaque psoriasis of the scalp and body in patients 12 years and older.
12.1 Mechanism of Action
Taclonex Topical Suspension combines the pharmacological effects of calcipotriene hydrate as a synthetic vitamin D3 analog and betamethasone dipropionate as a synthetic corticosteroid. However, while their pharmacologic and clinical effects are known, the exact mechanisms of their actions in the treatment of plaque psoriasis of the scalp and body are unknown.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypercalcemia and Hypercalciuria: Hypercalcemia and hypercalciuria have been reported. If either occurs, discontinue until parameters of calcium metabolism normalize. (5.1)
- Effects on Endocrine System: Can cause reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency during and after withdrawal of treatment. Risk factors include the use of high-potency topical corticosteroid, use over a large surface area or to areas under occlusion, prolonged use, altered skin barrier, liver failure, and use in pediatric patients. Modify use should HPA axis suppression develop. (5.2, 8.4)
- Ophthalmic Adverse Reactions: May increase the risk of cataracts and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist. (5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Instruct patients to shake bottle prior to using Taclonex Topical Suspension. Apply Taclonex Topical Suspension to affected areas on the scalp and body once daily for up to 8 weeks. Taclonex Topical Suspension should be discontinued when control is achieved. Instruct patients to wash their hands after applying the product. Inform patients that they should not take a bath or shower or wash their hair right after application of Taclonex Topical Suspension.
Patients 12 to 17 years should not use more than 60 grams per week and patients 18 years and older should not use more than 100 grams per week.
Taclonex Topical Suspension should not be:
- Used with occlusive dressings unless directed by a healthcare provider.
- Used on the face, groin, or axillae, or if skin atrophy is present at the treatment site.
- Applied to the scalp in the 12 hours before or after any chemical treatments to the hair.
Taclonex Topical Suspension is not for oral, ophthalmic, or intravaginal use.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Topical Suspension: 0.005%/0.064% - each gram contains 50 mcg of calcipotriene and 0.643 mg of betamethasone dipropionate in a viscous, nearly odorless, almost clear, colorless to slightly off-white suspension.
6.2 Postmarketing Experience
Because adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Postmarketing reports for local adverse reactions to topical corticosteroids included atrophy, striae, telangiectasias, itching, dryness, hypopigmentation, perioral dermatitis, secondary infection, and miliaria.
Ophthalmic adverse reactions of cataracts, glaucoma, increased intraocular pressure, and central serous chorioretinopathy have been reported during use of topical corticosteroids, including topical betamethasone products.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.5 Ophthalmic Adverse Reactions
Use of topical corticosteroids, including Taclonex® Topical Suspension, may increase the risk of posterior subcapsular cataracts and glaucoma. Cataracts and glaucoma have been reported with the postmarketing use of topical corticosteroid products [see Adverse Reactions (6.2)]. Avoid contact of Taclonex Topical Suspension with eyes. Taclonex Topical Suspension may cause eye irritation. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
See FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Taclonex (calcipotriene and betamethasone dipropionate) Topical Suspension, 0.005%/0.064% is a viscous, nearly odorless, almost clear, colorless to slightly off-white suspension. It is available as:
- 60 gram bottle (NDC 50222-501-06)
- 120 gram (2 bottles of 60 gram) (NDC 50222-501-66)
5.1 Hypercalcemia and Hypercalciuria
Hypercalcemia and hypercalciuria have been observed with use of Taclonex Topical Suspension. If hypercalcemia or hypercalciuria develop, discontinue treatment until parameters of calcium metabolism have normalized. The incidence of hypercalcemia and hypercalciuria following Taclonex Topical Suspension treatment of more than 8 weeks has not been evaluated [see Clinical Pharmacology (12.2)].
Principal Display Panel 60 G Carton Ndc 50222 501 06 (PRINCIPAL DISPLAY PANEL - 60 g Carton - NDC 50222-501-06)
NDC 50222-501-06
Rx only
Taclonex®
(calcipotriene and
betamethasone
dipropionate)
Topical Suspension,
0.005%/0.064%
Shake before use
For Topical Use Only
Net Wt. 60 gram
LEO®
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
When calcipotriene was applied topically to mice for up to 24 months at dosages of 3, 10, and 30 mcg/kg/day (9, 30, and 90 mcg/m2/day, respectively), no significant changes in tumor incidence were observed when compared to control.
A 104-week oral carcinogenicity study was conducted with calcipotriene in male and female rats at doses of 1, 5 and 15 mcg/kg/day (6, 30, and 90 mcg/m2/day, respectively). Beginning week 71, the dosage for high-dose animals of both genders was reduced to 10 mcg/kg/day (60 mcg/m2/day). A treatment-related increase in benign C-cell adenomas was observed in the thyroid of females that received 15 mcg/kg/day. A treatment-related increase in benign pheochromocytomas was observed in the adrenal glands of males that received 15 mcg/kg/day. No other statistically significant differences in tumor incidence were observed when compared to control. The relevance of these findings to patients is unknown.
When betamethasone dipropionate was applied topically to CD-1 mice for up to 24 months at dosages approximating 1.3, 4.2, and 8.5 mcg/kg/day in females, and 1.3, 4.2, and 12.9 mcg/kg/day in males (up to 26 mcg/m2/day and 39 mcg/m2/day, in females and males, respectively), no significant changes in tumor incidence were observed when compared to control.
When betamethasone dipropionate was administered via oral gavage to male and female Sprague Dawley rats for up to 24 months at dosages of 20, 60, and 200 mcg/kg/day (120, 360, and 1200 mcg/m2/day, respectively), no significant changes in tumor incidence were observed when compared to control.
Calcipotriene did not elicit any genotoxic effects in the Ames mutagenicity assay, the mouse lymphoma TK locus assay, the human lymphocyte chromosome aberration test, or the mouse micronucleus test. Betamethasone dipropionate did not elicit any genotoxic effects in the Ames mutagenicity assay, the mouse lymphoma TK locus assay, or in the rat micronucleus test.
Studies in rats with oral doses of up to 54 mcg/kg/day (324 mcg/m2/day) of calcipotriene indicated no impairment of fertility or general reproductive performance. Studies in male rats at oral doses of up to 200 mcg/kg/day (1200 mcg/m2/day), and in female rats at oral doses of up to 1000 mcg/kg/day (6000 mcg/m2/day), of betamethasone dipropionate indicated no impairment of fertility.
Principal Display Panel 120 G Carton Ndc 50222 501 66 (PRINCIPAL DISPLAY PANEL - 120 g Carton - NDC 50222-501-66)
NDC 50222-501-66
Rx only
Taclonex®
(calcipotriene and
betamethasone
dipropionate)
Topical Suspension,
0.005%/0.064%
Shake before use
For Topical Use Only
Net Wt. 120 gram
(2 bottles of 60 gram)
Individual bottles (60 gram)
not for individual retail sale
LEO®
5.4 Allergic Contact Dermatitis With Topical Calcipotriene (5.4 Allergic Contact Dermatitis with Topical Calcipotriene)
Allergic contact dermatitis has been observed with use of topical calcipotriene. Such an observation should be corroborated with appropriate diagnostic patch testing.
5.3 Allergic Contact Dermatitis With Topical Corticosteroids (5.3 Allergic Contact Dermatitis with Topical Corticosteroids)
Allergic contact dermatitis to a topical corticosteroid is usually diagnosed by observing a failure to heal rather than a clinical exacerbation. Such an observation should be corroborated with appropriate diagnostic patch testing.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:12.801598 · Updated: 2026-03-14T21:45:34.753203