These Highlights Do Not Include All The Information Needed To Use Sivextro®
75672079-589f-451a-bdbf-eaebcfcc80a9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1.1 , 1.2 ) 4/2025 Dosage and Administration ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 , 2.6 , 2.7 ) 4/2025
Indications and Usage
SIVEXTRO is an oxazolidinone antibacterial indicated for the treatment of acute bacterial skin and skin structure infections (ABSSSI) caused by designated susceptible microorganisms in adult and pediatric patients (at least 26 weeks gestational age and weighing at least 1 kg) ( 1.1 ) Usage to Reduce Development of Drug-Resistant Bacteria To reduce the development of drug-resistant bacteria and maintain the effectiveness of SIVEXTRO and other antibacterial drugs, SIVEXTRO should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria.
Dosage and Administration
Do not administer SIVEXTRO Tablets to pediatric patients weighing less than 35 kg. ( 2.1 ) Adult Patients Intravenous and Oral Dosage : 200 mg administered once daily orally or as an intravenous (IV) infusion over 1 hour for 6 days as specified in Table 1 in the full prescribing information. ( 2.2 ) Pediatric Patients Intravenous Dosage (at least 26 weeks gestational age and weighing at least 1 kg): Weight-based dosing as an intravenous infusion as specified in Table 2 in the full prescribing information. ( 2.3 ) Pediatric Patients Oral Dosage (weighing greater than or equal to 35 kg): Weight-based dosing as an oral tablet administered once daily as specified in Table 3 in the full prescribing information. ( 2.3 )
Warnings and Precautions
Patients with neutropenia: The safety and efficacy of SIVEXTRO in patients with neutropenia (neutrophil counts <1000 cells/mm 3 ) have not been adequately evaluated. In an animal model of infection, the antibacterial activity of SIVEXTRO was reduced in the absence of granulocytes. Consider alternative therapies in neutropenic patients. ( 5.1 ) Clostridioides difficile -associated diarrhea: Evaluate if diarrhea occurs. ( 5.2 )
Contraindications
None.
Adverse Reactions
The most common adverse reactions (≥2%) in adult patients are nausea, headache, diarrhea, infusion- or injection-related adverse reactions, vomiting, and dizziness. ( 6.1 ) The most common adverse reactions (>2%) in pediatric patients (12 years to less than 18 years of age) are phlebitis and increased hepatic transaminases. ( 6.1 ) The most common adverse reactions (>2%) in pediatric patients (less than 12 years of age) are infusion- or injection-related adverse reactions and vomiting. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
Orally administered SIVEXTRO inhibits Breast Cancer Resistance Protein (BCRP) in the intestine, which can increase the plasma concentrations of orally administered BCRP substrates, and the potential for adverse reactions. If possible, an interruption in the treatment of the co-administered BCRP substrate medicinal product should be considered during treatment with SIVEXTRO, especially for BCRP substrates with a narrow therapeutic index (e.g., methotrexate or topotecan). If coadministration cannot be avoided, monitor for adverse reactions related to the concomitantly administered BCRP substrates, including rosuvastatin. [see Clinical Pharmacology (12.3) .]
Storage and Handling
SIVEXTRO Tablets and SIVEXTRO for Injection should be stored at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Discard any unused portion of the single-dose vials.
Medication Information
Warnings and Precautions
Patients with neutropenia: The safety and efficacy of SIVEXTRO in patients with neutropenia (neutrophil counts <1000 cells/mm 3 ) have not been adequately evaluated. In an animal model of infection, the antibacterial activity of SIVEXTRO was reduced in the absence of granulocytes. Consider alternative therapies in neutropenic patients. ( 5.1 ) Clostridioides difficile -associated diarrhea: Evaluate if diarrhea occurs. ( 5.2 )
Indications and Usage
SIVEXTRO is an oxazolidinone antibacterial indicated for the treatment of acute bacterial skin and skin structure infections (ABSSSI) caused by designated susceptible microorganisms in adult and pediatric patients (at least 26 weeks gestational age and weighing at least 1 kg) ( 1.1 ) Usage to Reduce Development of Drug-Resistant Bacteria To reduce the development of drug-resistant bacteria and maintain the effectiveness of SIVEXTRO and other antibacterial drugs, SIVEXTRO should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria.
Dosage and Administration
Do not administer SIVEXTRO Tablets to pediatric patients weighing less than 35 kg. ( 2.1 ) Adult Patients Intravenous and Oral Dosage : 200 mg administered once daily orally or as an intravenous (IV) infusion over 1 hour for 6 days as specified in Table 1 in the full prescribing information. ( 2.2 ) Pediatric Patients Intravenous Dosage (at least 26 weeks gestational age and weighing at least 1 kg): Weight-based dosing as an intravenous infusion as specified in Table 2 in the full prescribing information. ( 2.3 ) Pediatric Patients Oral Dosage (weighing greater than or equal to 35 kg): Weight-based dosing as an oral tablet administered once daily as specified in Table 3 in the full prescribing information. ( 2.3 )
Contraindications
None.
Adverse Reactions
The most common adverse reactions (≥2%) in adult patients are nausea, headache, diarrhea, infusion- or injection-related adverse reactions, vomiting, and dizziness. ( 6.1 ) The most common adverse reactions (>2%) in pediatric patients (12 years to less than 18 years of age) are phlebitis and increased hepatic transaminases. ( 6.1 ) The most common adverse reactions (>2%) in pediatric patients (less than 12 years of age) are infusion- or injection-related adverse reactions and vomiting. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
Orally administered SIVEXTRO inhibits Breast Cancer Resistance Protein (BCRP) in the intestine, which can increase the plasma concentrations of orally administered BCRP substrates, and the potential for adverse reactions. If possible, an interruption in the treatment of the co-administered BCRP substrate medicinal product should be considered during treatment with SIVEXTRO, especially for BCRP substrates with a narrow therapeutic index (e.g., methotrexate or topotecan). If coadministration cannot be avoided, monitor for adverse reactions related to the concomitantly administered BCRP substrates, including rosuvastatin. [see Clinical Pharmacology (12.3) .]
Storage and Handling
SIVEXTRO Tablets and SIVEXTRO for Injection should be stored at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Discard any unused portion of the single-dose vials.
Description
Indications and Usage ( 1.1 , 1.2 ) 4/2025 Dosage and Administration ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 , 2.6 , 2.7 ) 4/2025
Section 42229-5
Preparation of SIVEXTRO for Injection
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
The contents of the vial should be reconstituted using aseptic technique as follows:
Note: To minimize foaming, AVOID vigorous agitation or shaking of the vial during or after reconstitution.
For Adults and Pediatric Patients Weighing at Least 35 kg:
- Reconstitute the SIVEXTRO vial with 4 mL of Sterile Water for Injection to provide a concentration of 50 mg/mL in each vial.
-
Gently swirl the contents and let the vial stand until the cake has completely dissolved and any foam disperses.
Inspect the vial to ensure the solution contains no particulate matter and no cake or powder remains attached to the sides of the vial. If necessary, invert the vial to dissolve any remaining powder and swirl gently to prevent foaming. The reconstituted solution is clear and colorless to pale-yellow in color; the total time from reconstitution to the end of administration should not exceed 24 hours at either room temperature or under refrigeration at 2°C to 8°C (36°F to 46°F).
Tilt the upright vial and insert a syringe with appropriately sized needle into the bottom corner of the vial and remove 4 mL of the reconstituted solution for the 200 mg dose. Do not invert the vial during extraction. - The reconstituted solution must be further diluted in 250 mL of 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP. Slowly inject the required volume of reconstituted solution as determined in Step 2 into a 250 mL bag of 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP. Invert the bag gently to mix. Do NOT shake the bag as this may cause foaming.
For Pediatric Patients Weighing Less than 35 kg:
- Reconstitute the SIVEXTRO vial with 4 mL of Sterile Water for Injection to provide a concentration of 50 mg/mL in each vial.
- Gently swirl the contents and let the vial stand until the cake has completely dissolved and any foam disperses.
-
Inspect the vial to ensure the solution contains no particulate matter and no cake or powder remains attached to the sides of the vial. If necessary, invert the vial to dissolve any remaining powder and swirl gently to prevent foaming. The reconstituted solution is clear and colorless to pale-yellow in color; the total time from reconstitution to the end of administration should not exceed 24 hours at either room temperature or under refrigeration at 2°C to 8°C (36°F to 46°F).
Prepare a stock solution (100 mL of 0.8 mg/mL tedizolid phosphate): Tilt the upright vial and insert a syringe with appropriately sized needle into the bottom corner of the vial and remove 1.6 mL of the reconstituted solution and add it to an infusion bag containing 98.4 mL of 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP. Do not invert the vial during extraction. - Prepare the required volume of stock solution for infusion: Refer to Table 4 to convert the dose in mg to the appropriate volume of stock solution to be administered. Transfer this volume of stock solution to an adequately sized infusion bag or infusion syringe. It may be necessary to round to the nearest graduation mark of an appropriately sized syringe for smaller volumes. The total time from reconstitution to the end of administration should not exceed 24 hours at either room temperature or under refrigeration at 2°C to 8°C (36°F to 46°F).
| Body Weight (kg) |
Amount of SIVEXTRO per dose (given twice daily) |
Volume of stock solution (0.8 mg/mL) to be transferred to an adequately sized infusion bag or infusion syringe |
|---|---|---|
| Pediatric Patients Weighing Less than 2 kg | ||
| 1 to less than 2 | 3 mg/kg | Volume (mL) = Weight (kg) x 3.75 mL/kg |
| Pediatric Patients Weighing at Least 2 kg | ||
| 2 to less than 3 | 6 mg | 7.5 mL |
| 3 to less than 6 | 12 mg | 15 mL |
| 6 to less than 10 | 20 mg | 25 mL |
| 10 to less than 14 | 30 mg | 37.5 mL |
| 14 to less than 20 | 40 mg | 50 mL |
| 20 to less than 35 | 60 mg | 75 mL |
Section 42230-3
|
Patient Information SIVEXTRO® (sih-vex-tro) (tedizolid phosphate) tablets |
What you need to know about SIVEXTRO
|
| What is SIVEXTRO? |
SIVEXTRO tablet is for adults and for children weighing at least 77 pounds (35 kg) who have a skin infection or an infection in the tissue below the skin. SIVEXTRO is an antibiotic that works by stopping the growth of certain bacteria.
| What should I tell my doctor before taking SIVEXTRO? |
Medical Conditions
Tell your doctor if you:
- have diarrhea or have ever had diarrhea while taking antibiotics. Tell your doctor, even if you had diarrhea that occurred up to 2 months after you took the antibiotic.
- are allergic to tedizolid phosphate or any of the ingredients in SIVEXTRO. See the end of this Patient Information for a complete list of ingredients in SIVEXTRO.
- are pregnant or plan to get pregnant, tell your doctor if you become pregnant while taking SIVEXTRO. It is not known if SIVEXTRO will harm your baby while you are pregnant. You and your doctor should decide together if you will take SIVEXTRO.
- are breastfeeding or plan to breastfeed, tell your doctor before you take SIVEXTRO. It is not known if SIVEXTRO passes into your breast milk. You and your doctor should decide together if you will take SIVEXTRO or breastfeed.
| Are you taking other medicines? |
- SIVEXTRO can affect the way other medicines work, and other medicines can affect how SIVEXTRO works. Some medicines cannot be taken with SIVEXTRO at all. Your doctor will tell you if it is safe to take SIVEXTRO with other medicines.
- It is especially important to tell your doctor if you take any of the following medicines:
- Methotrexate (for cancer or rheumatoid arthritis)
- Topotecan (for cancer)
- Rosuvastatin (for cholesterol)
- Tell your doctor about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal and dietary supplements.
- Know the medicines you take. Keep a list of them and show the list to your doctor and pharmacist when you get a new medicine.
| How do I take SIVEXTRO? |
- Do not give SIVEXTRO tablets to children weighing less than 77 pounds (35 kg).
- Take SIVEXTRO exactly how your doctor tells you to take it.
- Take 1 SIVEXTRO tablet 1 time each day.
- Take SIVEXTRO for 6 days, at the same time every day.
- Take SIVEXTRO by mouth, with or without food.
| What if I forget to take SIVEXTRO? |
- If you miss a dose, take the missed dose as soon as you remember.
If it is less than 8 hours until your next dose, skip the missed dose and take the next tablet at the time you usually take it. - Do not take 2 doses of SIVEXTRO at the same time to make up for a missed dose.
- If you are not sure how to take SIVEXTRO, call your doctor or pharmacist.
- Take all 6 tablets to finish your SIVEXTRO, even if you have missed a dose.
If you do not finish your medicine, SIVEXTRO may not work.
You may get sick again and the remaining bacteria may be harder to treat.
| What are the possible side effects of SIVEXTRO? |
SIVEXTRO may cause serious side effects, including diarrhea from C-diff (Clostridioides difficile) infection. Call your healthcare provider right away if you get stomach cramps, fever, watery diarrhea, diarrhea that does not go away, or bloody stools. C-diff infection can happen 2 or more months after you have finished your antibacterial medicine.
C-diff is an infection of your intestines (bowels) that can happen with many antibiotics like SIVEXTRO and may cause mild diarrhea to life-threatening swelling of your intestines (colitis).
Common side effects of SIVEXTRO include:
|
|
Some less common side effects are:
Problems with your skin
- itching, red or itchy rash, hives, acne
- hot flushes or feeling like you are blushing or your face, neck or chest is red
- not able to feel something as well
- a tingling or prickling sensation
Problems with your sleep
- hard time sleeping
Problems with your body
- numbness
- facial paralysis
Problems with infections
- vagina that is infected, inflamed, or itchy
- fungal infections of skin, mouth
Problems with your eyes
- eye strain
- blurred or impaired vision
- seeing dots or spots in your eyes
Problems with your heart
- Your heartbeat does not feel normal. It could feel like your heart is beating too fast or pumping harder than usual.
Problems with your vascular system
- high blood pressure
Problems with your blood work
Your doctor may tell you that you have the following while taking SIVEXTRO:
- a low white blood cell count
- anemia (low red blood cells)
- low platelet count, the small cells involved in clotting your blood
Side effects where the frequency is not known:
- bleeding or bruising easily
If you have any side effect that bothers you or does not go away, tell your doctor.
These are not all the possible side effects of SIVEXTRO. For information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
| How should I store SIVEXTRO? |
- Store SIVEXTRO at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep SIVEXTRO and all medicines out of the reach of children.
| General information about SIVEXTRO. |
- Medicines are sometimes prescribed for purposes that are not mentioned here.
- Do not use SIVEXTRO for a condition for which it was not prescribed.
| What if I have questions? |
- Call your doctor.
- Call the company that makes SIVEXTRO at 1-800-444-2080.
- Go to the website – www.SIVEXTRO.com.
- You can ask your doctor or pharmacist for information about SIVEXTRO that is written for health professionals.
| What are the ingredients in SIVEXTRO? |
- The active ingredient: tedizolid phosphate.
- The inactive ingredients: crospovidone, magnesium stearate, mannitol, microcrystalline cellulose, and povidone. Film coating: polyethylene glycol/macrogol, polyvinyl alcohol, talc, titanium dioxide, and yellow iron oxide.
Manuf. for: Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
Sivextro tablets Manufactured by: Patheon Inc., Whitby, Ontario, L1N 5Z5 Canada
Sivextro for injection Manufactured by: Patheon Italia S.p.A., 03013, Ferentino, FR Italy
For patent information: www.msd.com/research/patent. The trademarks depicted herein are owned by their respective companies.
Copyright © 2017-2025 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates. All rights reserved.
usppi-mk1986-mf-2504r005
This Patient Information has been approved by the U.S. Food and Drug Administration Revised: 4/2025
Section 43683-2
16.1 Tablets
SIVEXTRO Tablets are yellow film-coated oval tablets containing 200 mg of tedizolid phosphate; each tablet is debossed with "TZD" on one side and "200" on the other side.
They are supplied as follows:
HDPE bottles of 30 tablets with child-resistant closure (NDC 67919-041-04)
Unit dose blister packs of 6 tablets (NDC 67919-041-05)
10 Overdosage
In the event of overdosage, SIVEXTRO should be discontinued and general supportive treatment given. Hemodialysis does not result in meaningful removal of tedizolid from systemic circulation.
11 Description
SIVEXTRO (tedizolid phosphate), a phosphate prodrug, is converted to tedizolid in the presence of phosphatases.
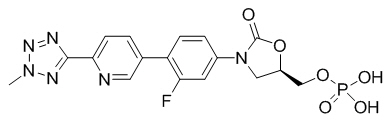
Tedizolid phosphate has the chemical name [(5R)-(3-{3-Fluoro-4-[6-(2-methyl-2H-tetrazol- 5-yl) pyridin-3-yl]phenyl}-2-oxooxazolidin- 5-yl]methyl hydrogen phosphate.
Its empirical formula is C17H16FN6O6P and its molecular weight is 450.32. Its structural formula is:
Tedizolid phosphate is a white to yellow solid and is administered orally or by intravenous infusion.
The pharmacologically active moiety, tedizolid, is an antibacterial agent of the oxazolidinone class.
SIVEXTRO Tablets contain 200 mg of tedizolid phosphate, and the following inactive ingredients: crospovidone, magnesium stearate, mannitol, microcrystalline cellulose, and povidone. In addition, the film coating contains the following inactive ingredients: polyethylene glycol/macrogol, polyvinyl alcohol, talc, titanium dioxide, and yellow iron oxide.
SIVEXTRO for Injection is a sterile, white to off-white sterile lyophilized powder supplied in a clear glass single-dose vial. Each vial contains 200 mg of tedizolid phosphate and the inactive ingredient, mannitol (105 mg). Sodium hydroxide and hydrochloric acid are used as needed for pH adjustment. When reconstituted as directed with 4 mL of Sterile Water for Injection, each mL contains 50 mg of tedizolid phosphate. The pH of the reconstituted solution is 7.4 to 8.1.
8.4 Pediatric Use
The safety and effectiveness of SIVEXTRO for the treatment of ABSSSI have been established in pediatric patients at least 26 weeks gestational age and weighing at least 1 kg. Use of SIVEXTRO for the treatment of ABSSSI is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic and safety data in pediatric patients from birth (includes neonates at least 26 weeks gestational age) to less than 18 years of age [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)].
The safety and effectiveness of SIVEXTRO in pediatric patients less than 26 weeks gestational age and weighing less than 1 kg have not been established.
8.5 Geriatric Use
Clinical studies of SIVEXTRO did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. No overall differences in pharmacokinetics were observed between elderly subjects and younger subjects.
16.2 for Injection
SIVEXTRO is supplied as a sterile, white to off-white lyophilized powder for injection in single-dose vials of 200 mg. Each 200 mg vial must be reconstituted with Sterile Water for Injection and subsequently diluted with 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP.
They are supplied as follows:
Package of ten 200 mg single-dose vials (NDC 67919-040-02)
4 Contraindications
None.
6 Adverse Reactions
- The most common adverse reactions (≥2%) in adult patients are nausea, headache, diarrhea, infusion- or injection-related adverse reactions, vomiting, and dizziness. (6.1)
- The most common adverse reactions (>2%) in pediatric patients (12 years to less than 18 years of age) are phlebitis and increased hepatic transaminases. (6.1)
- The most common adverse reactions (>2%) in pediatric patients (less than 12 years of age) are infusion- or injection-related adverse reactions and vomiting. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
7 Drug Interactions
Orally administered SIVEXTRO inhibits Breast Cancer Resistance Protein (BCRP) in the intestine, which can increase the plasma concentrations of orally administered BCRP substrates, and the potential for adverse reactions. If possible, an interruption in the treatment of the co-administered BCRP substrate medicinal product should be considered during treatment with SIVEXTRO, especially for BCRP substrates with a narrow therapeutic index (e.g., methotrexate or topotecan). If coadministration cannot be avoided, monitor for adverse reactions related to the concomitantly administered BCRP substrates, including rosuvastatin. [see Clinical Pharmacology (12.3).]
12.2 Pharmacodynamics
The AUC/minimum inhibitory concentration (MIC) was shown to best correlate with tedizolid activity in animal infection models.
In the mouse thigh infection model of S. aureus, antistaphylococcal killing activity was impacted by the presence of granulocytes. In granulocytopenic mice (neutrophil count <100 cells/mL), bacterial stasis was achieved at a human-equivalent dose of approximately 2000 mg/day; whereas, in non-granulocytopenic animals, stasis was achieved at a human-equivalent dose of approximately 100 mg/day. The safety and efficacy of SIVEXTRO for the treatment of neutropenic patients (neutrophil counts <1000 cells/mm3) have not been evaluated.
12.3 Pharmacokinetics
Tedizolid phosphate is a prodrug that is converted by phosphatases to tedizolid, the microbiologically active moiety, following oral and intravenous administration. Only the pharmacokinetic profile of tedizolid is discussed further due to negligible systemic exposure of tedizolid phosphate following oral and intravenous administration. Following multiple once-daily oral or intravenous administration, steady-state concentrations are achieved within approximately three days with tedizolid accumulation of approximately 30% (tedizolid half-life of approximately 12 hours). Pharmacokinetic (PK) parameters of tedizolid following oral and intravenous administration of 200 mg once daily tedizolid phosphate in adults are shown in Table 8.
| Pharmacokinetic Parameters of Tedizolid Cmax, maximum concentration; Tmax, time to reach Cmax; AUC, area under the concentration-time curve; CL, systemic clearance; CL/F, apparent oral clearance
|
Oral | Intravenous | ||
|---|---|---|---|---|
| Single Dose | Steady State | Single Dose | Steady State | |
| Cmax (mcg/mL) | 2.0 (0.7) | 2.2 (0.6) | 2.3 (0.6) | 3.0 (0.7) |
| Tmax (hr) Median (range)
|
2.5 (1.0 - 8.0) | 3.5 (1.0 - 6.0) | 1.1 (0.9 - 1.5) | 1.2 (0.9 - 1.5) |
| AUC (mcg∙hr/mL) AUC is AUC0-
∞ (AUC from time 0 to infinity) for single-dose administration and AUC0-24 (AUC from time 0 to 24 hours) for multiple-dose administration
|
23.8 (6.8) | 25.6 (8.5) | 26.6 (5.2) | 29.2 (6.2) |
| CL or CL/F (L/hr) | 7.5 (2.3) | 6.9 (1.7) | 6.4 (1.2) | 5.9 (1.4) |
2.7 Incompatibilities
SIVEXTRO for Injection is incompatible with any solution containing divalent cations (e.g., Ca2+, Mg2+), including Lactated Ringer’s Injection and Hartmann’s Solution.
1 Indications and Usage
SIVEXTRO is an oxazolidinone antibacterial indicated for the treatment of acute bacterial skin and skin structure infections (ABSSSI) caused by designated susceptible microorganisms in adult and pediatric patients (at least 26 weeks gestational age and weighing at least 1 kg) (1.1)
Usage to Reduce Development of Drug-Resistant Bacteria
To reduce the development of drug-resistant bacteria and maintain the effectiveness of SIVEXTRO and other antibacterial drugs, SIVEXTRO should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria.
12.1 Mechanism of Action
Tedizolid is an antibacterial drug [see Microbiology (12.4)].
16.3 Storage and Handling
SIVEXTRO Tablets and SIVEXTRO for Injection should be stored at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Discard any unused portion of the single-dose vials.
5 Warnings and Precautions
- Patients with neutropenia: The safety and efficacy of SIVEXTRO in patients with neutropenia (neutrophil counts <1000 cells/mm3) have not been adequately evaluated. In an animal model of infection, the antibacterial activity of SIVEXTRO was reduced in the absence of granulocytes. Consider alternative therapies in neutropenic patients. (5.1)
- Clostridioides difficile-associated diarrhea: Evaluate if diarrhea occurs. (5.2)
2 Dosage and Administration
- Do not administer SIVEXTRO Tablets to pediatric patients weighing less than 35 kg. (2.1)
- Adult Patients Intravenous and Oral Dosage : 200 mg administered once daily orally or as an intravenous (IV) infusion over 1 hour for 6 days as specified in Table 1 in the full prescribing information. (2.2)
- Pediatric Patients Intravenous Dosage (at least 26 weeks gestational age and weighing at least 1 kg): Weight-based dosing as an intravenous infusion as specified in Table 2 in the full prescribing information. (2.3)
- Pediatric Patients Oral Dosage (weighing greater than or equal to 35 kg): Weight-based dosing as an oral tablet administered once daily as specified in Table 3 in the full prescribing information. (2.3)
3 Dosage Forms and Strengths
SIVEXTRO 200 mg tablet is a yellow film-coated oval tablet; each tablet is debossed with "TZD" on one side and "200" on the other side.
SIVEXTRO for Injection is a sterile, white to off-white lyophilized powder for injection in single-dose vials of 200 mg. Each 200 mg vial must be reconstituted with Sterile Water for Injection and subsequently diluted with 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP.
5.2 Clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported for nearly all systemic antibacterial agents including SIVEXTRO, with severity ranging from mild diarrhea to fatal colitis. Treatment with antibacterial agents can alter the normal flora of the colon and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary because CDAD has been reported to occur more than two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, antibacterial use not directed against C. difficile should be discontinued, if possible. Appropriate measures such as fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of SIVEXTRO. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: thrombocytopenia
5.1 Patients With Neutropenia
The safety and efficacy of SIVEXTRO in patients with neutropenia (neutrophil counts <1000 cells/mm3) have not been adequately evaluated. In an animal model of infection, the antibacterial activity of SIVEXTRO was reduced in the absence of granulocytes [see Clinical Pharmacology (12.2)]. Alternative therapies should be considered when treating patients with neutropenia and ABSSSI.
8 Use in Specific Populations
Pregnancy: Based on animal data, SIVEXTRO may cause fetal harm. Advise pregnant women of the potential risks to a fetus. (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be compared directly to rates from clinical trials of another drug and may not reflect rates observed in practice.
Clinical Trials Experience in Adult Patients
Adverse reactions were evaluated for 1425 adult patients treated with SIVEXTRO in two Phase 2 and four Phase 3 clinical trials (three Phase 3 trials for 6 days of therapy and one Phase 3 trial for 7-21 days of therapy). The median age of adult patients treated with SIVEXTRO in the Phase 2 and Phase 3 trials was 44 years, ranging between 17 and 94 years old. The majority of adult patients treated with SIVEXTRO were male (66%) and White (67%).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.6 Compatible Intravenous Solutions
SIVEXTRO is compatible with 0.9% Sodium Chloride Injection, USP and 5% Dextrose Injection, USP.
Limited data are available on the compatibility of SIVEXTRO for Injection with other intravenous substances, additives or other medications and they should not be added to SIVEXTRO single-dose vials or infused simultaneously. If the same intravenous line is used for sequential infusion of several different drugs, the line should be flushed before and after infusion of SIVEXTRO with 0.9% Sodium Chloride Injection, USP.
2.2 Recommended Dosage for Adult Patients
The recommended dosage of SIVEXTRO is 200 mg administered once daily for six (6) days either as an oral tablet (with or without food) or as an intravenous (IV) infusion in adult patients.
The recommended dosage and administration of SIVEXTRO in adult patients are described in Table 1.
| Infection | Route | Dose | Frequency | Infusion Time | Duration of Treatment |
|---|---|---|---|---|---|
| Acute Bacterial Skin and Skin Structure Infections (ABSSSI) |
Intravenous | 200 mg | Once daily | 1 hour | 6 days |
| Oral | 200 mg | Once daily | Not Applicable |
No dose adjustment is necessary when changing from intravenous to oral SIVEXTRO.
13.2 Animal Toxicology And/or Pharmacology
Repeated-oral and intravenous dosing of tedizolid phosphate in rats in 1 month and 3 month toxicology studies produced dose- and time-dependent bone marrow hypocellularity (myeloid, erythroid, and megakaryocyte), with associated reduction in circulating RBCs, WBCs, and platelets. These effects showed evidence of reversibility and occurred at plasma tedizolid exposure levels (AUC) ≥6-fold greater than the plasma exposure associated with the human therapeutic dose. In a 1-month immunotoxicology study in rats, repeated oral dosing of tedizolid phosphate was shown to significantly reduce splenic B cells and T cells and reduce plasma IgG titers. These effects occurred at plasma tedizolid exposure levels (AUC) ≥3-fold greater than the expected human plasma exposure associated with the therapeutic dose.
5.3 Development of Drug Resistant Bacteria
Prescribing SIVEXTRO in the absence of a proven or strongly suspected bacterial infection or prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Principal Display Panel 200 Mg Vial Carton
NDC 67919-040-02
10 single-dose vials
Sivextro
®
(tedizolid phosphate) for injection
200 mg per vial
For Intravenous Infusion
Must be reconstituted and further
diluted prior to administration
by intravenous infusion.
Sterile
Rx only
2.3 Recommended Dosage for Pediatric Patients
Recommended Dosage of SIVEXTRO for Injection for Pediatric Patients: Intravenous Dosage
The recommended intravenous dosage of SIVEXTRO for Injection for pediatric patients (at least 26 weeks gestational age and weighing at least 1 kg) is presented in Table 2.
| Weight Band (kg) | Dose | Frequency | Infusion Time | Duration of Treatment |
|---|---|---|---|---|
| Pediatric Patients Weighing Less than 2 kg | ||||
| 1 to less than 2
Recommended Dosage for 1 kg to less than 2 kg is based on actual body weight.
|
3 mg/kg | Twice daily | 1 hour | 6 days |
| Pediatric Patients Weighing at Least 2 kg | ||||
| 2 to less than 3 | 6 mg | Twice daily | 1 hour | 6 days |
| 3 to less than 6 | 12 mg | |||
| 6 to less than 10 | 20 mg | |||
| 10 to less than 14 | 30 mg | |||
| 14 to less than 20 | 40 mg | |||
| 20 to less than 35 | 60 mg | |||
| Pediatric Patients Weighing at Least 35 kg | ||||
| Greater than or equal to 35 | 200 mg | Once daily | 1 hour | 6 days |
Recommended Dosage of SIVEXTRO Tablets for Pediatric Patients: Oral Dosage
The recommended oral dosage of SIVEXTRO Tablets for pediatric patients is presented in Table 3. SIVEXTRO Tablets can be administered with or without food. Do not administer SIVEXTRO Tablets to pediatric patients weighing less than 35 kg.
| Weight Band (kg) | Dose | Frequency | Duration of Treatment |
|---|---|---|---|
| Greater than or equal to 35 | 200 mg | Once daily | 6 days |
No dose adjustment is necessary when changing from intravenous to oral SIVEXTRO.
2.4 Recommendations Regarding Missed Doses(s)
For Once Daily Oral Dosing of SIVEXTRO Tablets
If patients miss a dose, administer the dose as soon as possible anytime up to 8 hours prior to their next scheduled dose. If less than 8 hours remain before the next dose, instruct the patients to wait until their next scheduled dose.
Principal Display Panel 200 Mg Tablet Bottle Label
NDC 67919-041-04
30 tablets
Sivextro
®
(tedizolid phosphate) tablets
200 mg per tablet
Rx only
1.1 Acute Bacterial Skin and Skin Structure Infections
SIVEXTRO® is indicated for the treatment of acute bacterial skin and skin structure infections (ABSSSI) caused by susceptible isolates of the following gram-positive microorganisms: Staphylococcus aureus (including methicillin-resistant [MRSA] and methicillin-susceptible [MSSA] isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus anginosus Group (including Streptococcus anginosus, Streptococcus intermedius, and Streptococcus constellatus), and Enterococcus faecalis, in adult and pediatric patients (at least 26 weeks gestational age and weighing at least 1 kg).
14.1 Acute Bacterial Skin and Skin Structure Infections
Adults
A total of 1333 adults with acute bacterial skin and skin structure infections (ABSSSI) were randomized in two multicenter, multinational, double-blind, non-inferiority trials. Both trials compared SIVEXTRO 200 mg once daily for 6 days versus linezolid 600 mg every 12 hours for 10 days. In Trial 1, patients were treated with oral therapy, while in Trial 2, patients could receive oral therapy after a minimum of one day of intravenous therapy. Patients with cellulitis/erysipelas, major cutaneous abscess, or wound infection were enrolled in the trials. Patients with wound infections could have received aztreonam and/or metronidazole as adjunctive therapy for gram-negative bacterial coverage, if needed. The intent-to-treat (ITT) patient population included all randomized patients.
In Trial 1, 332 patients with ABSSSI were randomized to SIVEXTRO and 335 patients were randomized to linezolid. The majority (91%) of patients treated with SIVEXTRO in Trial 1 were less than 65 years old with a median age of 43 years (range: 18 to 86 years). Patients treated with SIVEXTRO were predominantly male (61%) and White (84%); 13% had BMI ≥35 kg/m2, 8% had diabetes mellitus, 35% were current or recent intravenous drug users, and 2% had moderate to severe renal impairment. The overall median surface area of infection was 188 cm2. The types of ABSSSI included were cellulitis/erysipelas (41%), wound infection (29%), and major cutaneous abscess (30%). In addition to local signs and symptoms of infection, patients were also required to have at least one regional or systemic sign of infection at baseline, defined as lymphadenopathy (87% of patients), temperature 38°C or higher (16% of patients), white blood cell count greater than 10,000 cells/mm3 or less than 4000 cells/mm3 (42%), or 10% or more band forms on white blood cell differential (4%).
The primary endpoint in Trial 1 was early clinical response defined as no increase from baseline lesion area at 48-72 hours after the first dose and oral temperature of ≤37.6°C, confirmed by a second temperature measurement within 24 hours in the ITT population.
In Trial 2, 332 patients with ABSSSI were randomized to SIVEXTRO and 334 patients were randomized to linezolid. The majority (87%) of patients treated with SIVEXTRO in Trial 2 were less than 65 years old with a median age of 46 years (range: 17 to 86 years). Patients treated with SIVEXTRO were predominantly male (68%) and White (86%); 16% had BMI ≥35 kg/m2, 10% had diabetes mellitus, 20% were current or recent intravenous drug users, and 4% had moderate to severe renal impairment. The overall median surface area of infection was 231 cm2. The types of ABSSSI included were cellulitis/erysipelas (50%), wound infection (30%), and major cutaneous abscess (20%). In addition to local signs and symptoms of infection, patients were also required to have at least one regional or systemic sign of infection at baseline, defined as lymphadenopathy (71% of patients), temperature 38°C or higher (31% of patients), white blood cell count greater than 10,000 cells/mm3 or less than 4000 cells/mm3 (53%), or 10% or more band forms on white blood cell differential (16%).
The primary endpoint in Trial 2 was early clinical response defined as at least a 20% decrease from baseline lesion area at 48-72 hours after the first dose in the ITT population (Table 10).
| SIVEXTRO (200 mg) |
Linezolid (1200 mg) |
Treatment Difference (2-sided 95% CI) |
|
|---|---|---|---|
| CI=confidence interval | |||
|
No increase in lesion surface area from baseline and oral temperature of ≤37.6°C, confirmed by a second temperature measurement within 24 hours at 48-72 hours Primary endpoint for Trial 1; sensitivity analysis for Trial 2
|
|||
| Trial 1, N | 332 | 335 | |
| Responder, n (%) | 264 (79.5) | 266 (79.4) | 0.1 (-6.1, 6.2) |
| Trial 2, N | 332 | 334 | |
| Responder, n (%) | 286 (86.1) | 281 (84.1) | 2.0 (-3.5, 7.3) |
|
At least a 20% decrease from baseline in lesion area at 48-72 hours Primary endpoint for Trial 2; sensitivity analysis for Trial 1
|
|||
| Trial 1, N | 332 | 335 | |
| Responder, n (%) | 259 (78.0) | 255 (76.1) | 1.9 (-4.5, 8.3) |
| Trial 2, N | 332 | 334 | |
| Responder, n (%) | 283 (85.2) | 276 (82.6) | 2.6 (-3.0, 8.2) |
An investigator assessment of clinical response was made at the post-therapy evaluation (PTE) (7 - 14 days after the end of therapy) in the ITT and CE (Clinically Evaluable) populations. Clinical success was defined as resolution or near resolution of most disease-specific signs and symptoms, absence or near resolution of systemic signs of infection if present at baseline (lymphadenopathy, fever, >10% immature neutrophils, abnormal WBC count), and no new signs, symptoms, or complications attributable to the ABSSSI requiring further treatment of the primary lesion (Table 11).
| SIVEXTRO (200 mg) n/N (%) |
Linezolid (1200 mg) n/N (%) |
Treatment Difference (2-sided 95% CI) |
|
|---|---|---|---|
| CI=confidence interval; ITT=intent-to-treat; CE=clinically evaluable | |||
| Trial 1 | |||
| ITT | 284/332 (85.5) | 288/335 (86.0) | -0.5 (-5.8, 4.9) |
| CE | 264/279 (94.6) | 267/280 (95.4) | -0.8 (-4.6, 3.0) |
| Trial 2 | |||
| ITT | 292/332 (88.0) | 293/334 (87.7) | 0.3 (-4.8, 5.3) |
| CE | 268/290 (92.4) | 269/280 (96.1) | -3.7 (-7.7, 0.2) |
Clinical success by baseline pathogens from the primary infection site or blood cultures for the microbiological intent-to-treat (MITT) patient population for two integrated Phase 3 ABSSSI studies are presented in Table 12 and Table 13.
| Pathogen | No increase in lesion surface area from baseline and oral temperature of ≤37.6°C Primary endpoint of Trial 1
|
At least a 20% decrease from baseline in lesion area Primary endpoint of Trial 2
|
||
|---|---|---|---|---|
| SIVEXTRO (200 mg) n/N (%) |
Linezolid (1200 mg) n/N (%) |
SIVEXTRO (200 mg) n/N (%) |
Linezolid (1200 mg) n/N (%) |
|
| Pooled analysis; n=number of patients in the specific category; N=Number of patients with the specific pathogen isolated from the ABSSSI | ||||
| Staphylococcus aureus | 276/329 (83.9) | 278/342 (81.3) | 280/329 (85.1) | 276/342 (80.7) |
| Methicillin-resistant S. aureus | 112/141 (79.4) | 113/146 (77.4) | 114/141 (80.9) | 111/146 (76.0) |
| Methicillin-susceptible S. aureus | 164/188 (87.2) | 167/198 (84.3) | 166/188 (88.3) | 167/198 (84.3) |
| Streptococcus pyogenes | 27/33 (81.8) | 18/20 (90.0) | 25/33 (75.8) | 16/20 (80.0) |
| Streptococcus anginosus Group | 22/30 (73.3) | 26/28 (92.9) | 22/30 (73.3) | 25/28 (89.3) |
| Streptococcus agalactiae | 6/9 (66.7) | 8/10 (80.0) | 6/9 (66.7) | 7/10 (70.0) |
| Enterococcus faecalis | 7/10 (70.0) | 3/4 (75.0) | 6/10 (60.0) | 1/4 (25.0) |
Baseline bacteremia in the tedizolid arm with relevant pathogens included two subjects with MRSA, four subjects with MSSA, two subjects with S. pyogenes, one subject with S. agalactiae, and one subject with S. constellatus. All of these subjects were Responders at the 48-72 hour evaluation. At the Post-therapy Evaluation (PTE), 8 of 10 subjects were considered clinical successes.
| Pathogen | Clinical Response at PTE | |
|---|---|---|
| SIVEXTRO (200 mg) n/N (%) |
Linezolid (1200 mg) n/N (%) |
|
| Pooled analysis; n=number of patients in the specific category; N=Number of patients with the specific pathogen isolated from the ABSSSI | ||
| Staphylococcus aureus | 291/329 (88.5) | 303/342 (88.6) |
| Methicillin-resistant S. aureus | 118/141 (83.7) | 119/146 (81.5) |
| Methicillin-susceptible S. aureus | 173/188 (92.0) | 186/198 (93.9) |
| Streptococcus pyogenes | 30/33 (90.9) | 19/20 (95.0) |
| Streptococcus anginosus Group | 21/30 (70.0) | 25/28 (89.3) |
| Streptococcus agalactiae | 8/9 (88.9) | 8/10 (80.0) |
| Enterococcus faecalis | 7/10 (70.0) | 4/4 (100.0) |
Baseline bacteremia in the tedizolid arm with relevant pathogens included two subjects with MRSA, four subjects with MSSA, two subjects with S. pyogenes, one subject with S. agalactiae, and one subject with S. constellatus. All of these subjects were Responders at the 48-72 hour evaluation. At the Post-therapy Evaluation (PTE) 8 of 10 subjects were considered clinical successes.
Pediatric Patients
The safety and efficacy of SIVEXTRO were investigated in Pediatric Trials 1 and 2. Pediatric Trial 1 included pediatric patients 12 to < 18 years of age who were investigated in a randomized, single blind, active-controlled trial of 120 patients with clinically documented ABSSSI (91 receiving tedizolid, 29 receiving comparator). Patients were randomized in a 3:1 ratio with stratification by geographic region to receive SIVEXTRO IV and/or oral therapy, dosed 200 mg once daily for 6 days, or comparator IV and/or oral therapy, dosed over 10 days. Comparator therapy was selected by the investigator from a list of 5 IV and 4 oral comparators per local standard of care. The most frequently used comparators were cefazolin (11 patients) and vancomycin (8 patients).
Additionally, Pediatric Trial 2 (NCT03176134) evaluated the safety and efficacy of SIVEXTRO in pediatric patients 4 months to <12 years of age in a randomized, single blind, active-controlled trial of 100 patients with clinically documented ABSSSI (75 receiving tedizolid, 25 receiving comparator). Patients were randomized in a 3:1 ratio to receive SIVEXTRO for 6 to 10 days or comparator for 10 to 14 days. SIVEXTRO (IV and/or oral) was dosed as a weight-based dose of 2 to 2.5 mg/kg every 12 hours or as 200 mg given once daily. Patients who received oral therapy received an oral suspension formulation that is not currently approved for use. Comparator therapy was selected from a pre-specified list by the investigator and dosed per local standard of care.
The primary objective of Pediatric Trials 1 and 2 was to evaluate the safety and tolerability of SIVEXTRO. The trials were not powered for comparative inferential efficacy analysis. Clinical response at the test of cure visit (Day 18-25) was assessed by a blinded investigator in the ITT population (all randomized patients). Clinical successes were required to have resolution or near resolution of all related signs and symptoms such that no further antibacterial therapy was needed. Early clinical response, defined as at least a 20% reduction in lesion size at 48-72 hours after start of treatment, was also assessed in the ITT population.
In Pediatric Trial 1, clinical success at test of cure was 96.7% (88/91) in the tedizolid group and 93.1% (27/29) in the comparator group (difference: 3.6%, 95% CI: -6.3, 13.5). Early clinical response at 48-72 hours was 92.3% (84/91) in the tedizolid group and 96.6% (28/29) in the comparator group (difference: -4.2%, 95% CI: -12.9, 4.4).
In Pediatric Trial 2, clinical success at test of cure was 93.3% (70/75) in the tedizolid group and 92.0% (23/25) in the comparator group (difference: 1.3%, 95% CI: -10.7, 13.4). Early clinical response was not adequately assessed in Pediatric Trial 2 because an in-person outpatient visit at this timepoint was not required during the COVID-19 pandemic.
1.2 Usage to Reduce Development of Drug Resistant Bacteria
To reduce the development of drug-resistant bacteria and maintain the effectiveness of SIVEXTRO and other antibacterial drugs, SIVEXTRO should be used only to treat infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
2.5 Preparation and Administration of Intravenous Solution
SIVEXTRO is supplied as a sterile, lyophilized powder for injection in single-dose vials of 200 mg. Each 200 mg vial must be reconstituted with Sterile Water for Injection and subsequently diluted with 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP.
SIVEXTRO vials contain no antimicrobial preservatives and are intended for single dose only. Discard any unused portion.
Principal Display Panel 200 Mg Tablet Blister Pack Carton
NDC 67919-041-05
6 tablets
Sivextro
®
(tedizolid phosphate) tablets
200 mg per tablet
Rx only
2.1 Important Administration Instructions for Pediatric Patients Weighing Less Than 35 Kg
Do not administer SIVEXTRO Tablets to pediatric patients weighing less than 35 kg [see Dosage and Administration 2.3].
Structured Label Content
Section 42229-5 (42229-5)
Preparation of SIVEXTRO for Injection
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
The contents of the vial should be reconstituted using aseptic technique as follows:
Note: To minimize foaming, AVOID vigorous agitation or shaking of the vial during or after reconstitution.
For Adults and Pediatric Patients Weighing at Least 35 kg:
- Reconstitute the SIVEXTRO vial with 4 mL of Sterile Water for Injection to provide a concentration of 50 mg/mL in each vial.
-
Gently swirl the contents and let the vial stand until the cake has completely dissolved and any foam disperses.
Inspect the vial to ensure the solution contains no particulate matter and no cake or powder remains attached to the sides of the vial. If necessary, invert the vial to dissolve any remaining powder and swirl gently to prevent foaming. The reconstituted solution is clear and colorless to pale-yellow in color; the total time from reconstitution to the end of administration should not exceed 24 hours at either room temperature or under refrigeration at 2°C to 8°C (36°F to 46°F).
Tilt the upright vial and insert a syringe with appropriately sized needle into the bottom corner of the vial and remove 4 mL of the reconstituted solution for the 200 mg dose. Do not invert the vial during extraction. - The reconstituted solution must be further diluted in 250 mL of 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP. Slowly inject the required volume of reconstituted solution as determined in Step 2 into a 250 mL bag of 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP. Invert the bag gently to mix. Do NOT shake the bag as this may cause foaming.
For Pediatric Patients Weighing Less than 35 kg:
- Reconstitute the SIVEXTRO vial with 4 mL of Sterile Water for Injection to provide a concentration of 50 mg/mL in each vial.
- Gently swirl the contents and let the vial stand until the cake has completely dissolved and any foam disperses.
-
Inspect the vial to ensure the solution contains no particulate matter and no cake or powder remains attached to the sides of the vial. If necessary, invert the vial to dissolve any remaining powder and swirl gently to prevent foaming. The reconstituted solution is clear and colorless to pale-yellow in color; the total time from reconstitution to the end of administration should not exceed 24 hours at either room temperature or under refrigeration at 2°C to 8°C (36°F to 46°F).
Prepare a stock solution (100 mL of 0.8 mg/mL tedizolid phosphate): Tilt the upright vial and insert a syringe with appropriately sized needle into the bottom corner of the vial and remove 1.6 mL of the reconstituted solution and add it to an infusion bag containing 98.4 mL of 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP. Do not invert the vial during extraction. - Prepare the required volume of stock solution for infusion: Refer to Table 4 to convert the dose in mg to the appropriate volume of stock solution to be administered. Transfer this volume of stock solution to an adequately sized infusion bag or infusion syringe. It may be necessary to round to the nearest graduation mark of an appropriately sized syringe for smaller volumes. The total time from reconstitution to the end of administration should not exceed 24 hours at either room temperature or under refrigeration at 2°C to 8°C (36°F to 46°F).
| Body Weight (kg) |
Amount of SIVEXTRO per dose (given twice daily) |
Volume of stock solution (0.8 mg/mL) to be transferred to an adequately sized infusion bag or infusion syringe |
|---|---|---|
| Pediatric Patients Weighing Less than 2 kg | ||
| 1 to less than 2 | 3 mg/kg | Volume (mL) = Weight (kg) x 3.75 mL/kg |
| Pediatric Patients Weighing at Least 2 kg | ||
| 2 to less than 3 | 6 mg | 7.5 mL |
| 3 to less than 6 | 12 mg | 15 mL |
| 6 to less than 10 | 20 mg | 25 mL |
| 10 to less than 14 | 30 mg | 37.5 mL |
| 14 to less than 20 | 40 mg | 50 mL |
| 20 to less than 35 | 60 mg | 75 mL |
Section 42230-3 (42230-3)
|
Patient Information SIVEXTRO® (sih-vex-tro) (tedizolid phosphate) tablets |
What you need to know about SIVEXTRO
|
| What is SIVEXTRO? |
SIVEXTRO tablet is for adults and for children weighing at least 77 pounds (35 kg) who have a skin infection or an infection in the tissue below the skin. SIVEXTRO is an antibiotic that works by stopping the growth of certain bacteria.
| What should I tell my doctor before taking SIVEXTRO? |
Medical Conditions
Tell your doctor if you:
- have diarrhea or have ever had diarrhea while taking antibiotics. Tell your doctor, even if you had diarrhea that occurred up to 2 months after you took the antibiotic.
- are allergic to tedizolid phosphate or any of the ingredients in SIVEXTRO. See the end of this Patient Information for a complete list of ingredients in SIVEXTRO.
- are pregnant or plan to get pregnant, tell your doctor if you become pregnant while taking SIVEXTRO. It is not known if SIVEXTRO will harm your baby while you are pregnant. You and your doctor should decide together if you will take SIVEXTRO.
- are breastfeeding or plan to breastfeed, tell your doctor before you take SIVEXTRO. It is not known if SIVEXTRO passes into your breast milk. You and your doctor should decide together if you will take SIVEXTRO or breastfeed.
| Are you taking other medicines? |
- SIVEXTRO can affect the way other medicines work, and other medicines can affect how SIVEXTRO works. Some medicines cannot be taken with SIVEXTRO at all. Your doctor will tell you if it is safe to take SIVEXTRO with other medicines.
- It is especially important to tell your doctor if you take any of the following medicines:
- Methotrexate (for cancer or rheumatoid arthritis)
- Topotecan (for cancer)
- Rosuvastatin (for cholesterol)
- Tell your doctor about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal and dietary supplements.
- Know the medicines you take. Keep a list of them and show the list to your doctor and pharmacist when you get a new medicine.
| How do I take SIVEXTRO? |
- Do not give SIVEXTRO tablets to children weighing less than 77 pounds (35 kg).
- Take SIVEXTRO exactly how your doctor tells you to take it.
- Take 1 SIVEXTRO tablet 1 time each day.
- Take SIVEXTRO for 6 days, at the same time every day.
- Take SIVEXTRO by mouth, with or without food.
| What if I forget to take SIVEXTRO? |
- If you miss a dose, take the missed dose as soon as you remember.
If it is less than 8 hours until your next dose, skip the missed dose and take the next tablet at the time you usually take it. - Do not take 2 doses of SIVEXTRO at the same time to make up for a missed dose.
- If you are not sure how to take SIVEXTRO, call your doctor or pharmacist.
- Take all 6 tablets to finish your SIVEXTRO, even if you have missed a dose.
If you do not finish your medicine, SIVEXTRO may not work.
You may get sick again and the remaining bacteria may be harder to treat.
| What are the possible side effects of SIVEXTRO? |
SIVEXTRO may cause serious side effects, including diarrhea from C-diff (Clostridioides difficile) infection. Call your healthcare provider right away if you get stomach cramps, fever, watery diarrhea, diarrhea that does not go away, or bloody stools. C-diff infection can happen 2 or more months after you have finished your antibacterial medicine.
C-diff is an infection of your intestines (bowels) that can happen with many antibiotics like SIVEXTRO and may cause mild diarrhea to life-threatening swelling of your intestines (colitis).
Common side effects of SIVEXTRO include:
|
|
Some less common side effects are:
Problems with your skin
- itching, red or itchy rash, hives, acne
- hot flushes or feeling like you are blushing or your face, neck or chest is red
- not able to feel something as well
- a tingling or prickling sensation
Problems with your sleep
- hard time sleeping
Problems with your body
- numbness
- facial paralysis
Problems with infections
- vagina that is infected, inflamed, or itchy
- fungal infections of skin, mouth
Problems with your eyes
- eye strain
- blurred or impaired vision
- seeing dots or spots in your eyes
Problems with your heart
- Your heartbeat does not feel normal. It could feel like your heart is beating too fast or pumping harder than usual.
Problems with your vascular system
- high blood pressure
Problems with your blood work
Your doctor may tell you that you have the following while taking SIVEXTRO:
- a low white blood cell count
- anemia (low red blood cells)
- low platelet count, the small cells involved in clotting your blood
Side effects where the frequency is not known:
- bleeding or bruising easily
If you have any side effect that bothers you or does not go away, tell your doctor.
These are not all the possible side effects of SIVEXTRO. For information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
| How should I store SIVEXTRO? |
- Store SIVEXTRO at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep SIVEXTRO and all medicines out of the reach of children.
| General information about SIVEXTRO. |
- Medicines are sometimes prescribed for purposes that are not mentioned here.
- Do not use SIVEXTRO for a condition for which it was not prescribed.
| What if I have questions? |
- Call your doctor.
- Call the company that makes SIVEXTRO at 1-800-444-2080.
- Go to the website – www.SIVEXTRO.com.
- You can ask your doctor or pharmacist for information about SIVEXTRO that is written for health professionals.
| What are the ingredients in SIVEXTRO? |
- The active ingredient: tedizolid phosphate.
- The inactive ingredients: crospovidone, magnesium stearate, mannitol, microcrystalline cellulose, and povidone. Film coating: polyethylene glycol/macrogol, polyvinyl alcohol, talc, titanium dioxide, and yellow iron oxide.
Manuf. for: Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
Sivextro tablets Manufactured by: Patheon Inc., Whitby, Ontario, L1N 5Z5 Canada
Sivextro for injection Manufactured by: Patheon Italia S.p.A., 03013, Ferentino, FR Italy
For patent information: www.msd.com/research/patent. The trademarks depicted herein are owned by their respective companies.
Copyright © 2017-2025 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates. All rights reserved.
usppi-mk1986-mf-2504r005
This Patient Information has been approved by the U.S. Food and Drug Administration Revised: 4/2025
Section 43683-2 (43683-2)
16.1 Tablets
SIVEXTRO Tablets are yellow film-coated oval tablets containing 200 mg of tedizolid phosphate; each tablet is debossed with "TZD" on one side and "200" on the other side.
They are supplied as follows:
HDPE bottles of 30 tablets with child-resistant closure (NDC 67919-041-04)
Unit dose blister packs of 6 tablets (NDC 67919-041-05)
10 Overdosage (10 OVERDOSAGE)
In the event of overdosage, SIVEXTRO should be discontinued and general supportive treatment given. Hemodialysis does not result in meaningful removal of tedizolid from systemic circulation.
11 Description (11 DESCRIPTION)
SIVEXTRO (tedizolid phosphate), a phosphate prodrug, is converted to tedizolid in the presence of phosphatases.
Tedizolid phosphate has the chemical name [(5R)-(3-{3-Fluoro-4-[6-(2-methyl-2H-tetrazol- 5-yl) pyridin-3-yl]phenyl}-2-oxooxazolidin- 5-yl]methyl hydrogen phosphate.
Its empirical formula is C17H16FN6O6P and its molecular weight is 450.32. Its structural formula is:
Tedizolid phosphate is a white to yellow solid and is administered orally or by intravenous infusion.
The pharmacologically active moiety, tedizolid, is an antibacterial agent of the oxazolidinone class.
SIVEXTRO Tablets contain 200 mg of tedizolid phosphate, and the following inactive ingredients: crospovidone, magnesium stearate, mannitol, microcrystalline cellulose, and povidone. In addition, the film coating contains the following inactive ingredients: polyethylene glycol/macrogol, polyvinyl alcohol, talc, titanium dioxide, and yellow iron oxide.
SIVEXTRO for Injection is a sterile, white to off-white sterile lyophilized powder supplied in a clear glass single-dose vial. Each vial contains 200 mg of tedizolid phosphate and the inactive ingredient, mannitol (105 mg). Sodium hydroxide and hydrochloric acid are used as needed for pH adjustment. When reconstituted as directed with 4 mL of Sterile Water for Injection, each mL contains 50 mg of tedizolid phosphate. The pH of the reconstituted solution is 7.4 to 8.1.
8.4 Pediatric Use
The safety and effectiveness of SIVEXTRO for the treatment of ABSSSI have been established in pediatric patients at least 26 weeks gestational age and weighing at least 1 kg. Use of SIVEXTRO for the treatment of ABSSSI is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic and safety data in pediatric patients from birth (includes neonates at least 26 weeks gestational age) to less than 18 years of age [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)].
The safety and effectiveness of SIVEXTRO in pediatric patients less than 26 weeks gestational age and weighing less than 1 kg have not been established.
8.5 Geriatric Use
Clinical studies of SIVEXTRO did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. No overall differences in pharmacokinetics were observed between elderly subjects and younger subjects.
16.2 for Injection (16.2 For Injection)
SIVEXTRO is supplied as a sterile, white to off-white lyophilized powder for injection in single-dose vials of 200 mg. Each 200 mg vial must be reconstituted with Sterile Water for Injection and subsequently diluted with 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP.
They are supplied as follows:
Package of ten 200 mg single-dose vials (NDC 67919-040-02)
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
- The most common adverse reactions (≥2%) in adult patients are nausea, headache, diarrhea, infusion- or injection-related adverse reactions, vomiting, and dizziness. (6.1)
- The most common adverse reactions (>2%) in pediatric patients (12 years to less than 18 years of age) are phlebitis and increased hepatic transaminases. (6.1)
- The most common adverse reactions (>2%) in pediatric patients (less than 12 years of age) are infusion- or injection-related adverse reactions and vomiting. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
7 Drug Interactions (7 DRUG INTERACTIONS)
Orally administered SIVEXTRO inhibits Breast Cancer Resistance Protein (BCRP) in the intestine, which can increase the plasma concentrations of orally administered BCRP substrates, and the potential for adverse reactions. If possible, an interruption in the treatment of the co-administered BCRP substrate medicinal product should be considered during treatment with SIVEXTRO, especially for BCRP substrates with a narrow therapeutic index (e.g., methotrexate or topotecan). If coadministration cannot be avoided, monitor for adverse reactions related to the concomitantly administered BCRP substrates, including rosuvastatin. [see Clinical Pharmacology (12.3).]
12.2 Pharmacodynamics
The AUC/minimum inhibitory concentration (MIC) was shown to best correlate with tedizolid activity in animal infection models.
In the mouse thigh infection model of S. aureus, antistaphylococcal killing activity was impacted by the presence of granulocytes. In granulocytopenic mice (neutrophil count <100 cells/mL), bacterial stasis was achieved at a human-equivalent dose of approximately 2000 mg/day; whereas, in non-granulocytopenic animals, stasis was achieved at a human-equivalent dose of approximately 100 mg/day. The safety and efficacy of SIVEXTRO for the treatment of neutropenic patients (neutrophil counts <1000 cells/mm3) have not been evaluated.
12.3 Pharmacokinetics
Tedizolid phosphate is a prodrug that is converted by phosphatases to tedizolid, the microbiologically active moiety, following oral and intravenous administration. Only the pharmacokinetic profile of tedizolid is discussed further due to negligible systemic exposure of tedizolid phosphate following oral and intravenous administration. Following multiple once-daily oral or intravenous administration, steady-state concentrations are achieved within approximately three days with tedizolid accumulation of approximately 30% (tedizolid half-life of approximately 12 hours). Pharmacokinetic (PK) parameters of tedizolid following oral and intravenous administration of 200 mg once daily tedizolid phosphate in adults are shown in Table 8.
| Pharmacokinetic Parameters of Tedizolid Cmax, maximum concentration; Tmax, time to reach Cmax; AUC, area under the concentration-time curve; CL, systemic clearance; CL/F, apparent oral clearance
|
Oral | Intravenous | ||
|---|---|---|---|---|
| Single Dose | Steady State | Single Dose | Steady State | |
| Cmax (mcg/mL) | 2.0 (0.7) | 2.2 (0.6) | 2.3 (0.6) | 3.0 (0.7) |
| Tmax (hr) Median (range)
|
2.5 (1.0 - 8.0) | 3.5 (1.0 - 6.0) | 1.1 (0.9 - 1.5) | 1.2 (0.9 - 1.5) |
| AUC (mcg∙hr/mL) AUC is AUC0-
∞ (AUC from time 0 to infinity) for single-dose administration and AUC0-24 (AUC from time 0 to 24 hours) for multiple-dose administration
|
23.8 (6.8) | 25.6 (8.5) | 26.6 (5.2) | 29.2 (6.2) |
| CL or CL/F (L/hr) | 7.5 (2.3) | 6.9 (1.7) | 6.4 (1.2) | 5.9 (1.4) |
2.7 Incompatibilities
SIVEXTRO for Injection is incompatible with any solution containing divalent cations (e.g., Ca2+, Mg2+), including Lactated Ringer’s Injection and Hartmann’s Solution.
1 Indications and Usage (1 INDICATIONS AND USAGE)
SIVEXTRO is an oxazolidinone antibacterial indicated for the treatment of acute bacterial skin and skin structure infections (ABSSSI) caused by designated susceptible microorganisms in adult and pediatric patients (at least 26 weeks gestational age and weighing at least 1 kg) (1.1)
Usage to Reduce Development of Drug-Resistant Bacteria
To reduce the development of drug-resistant bacteria and maintain the effectiveness of SIVEXTRO and other antibacterial drugs, SIVEXTRO should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria.
12.1 Mechanism of Action
Tedizolid is an antibacterial drug [see Microbiology (12.4)].
16.3 Storage and Handling
SIVEXTRO Tablets and SIVEXTRO for Injection should be stored at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Discard any unused portion of the single-dose vials.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Patients with neutropenia: The safety and efficacy of SIVEXTRO in patients with neutropenia (neutrophil counts <1000 cells/mm3) have not been adequately evaluated. In an animal model of infection, the antibacterial activity of SIVEXTRO was reduced in the absence of granulocytes. Consider alternative therapies in neutropenic patients. (5.1)
- Clostridioides difficile-associated diarrhea: Evaluate if diarrhea occurs. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Do not administer SIVEXTRO Tablets to pediatric patients weighing less than 35 kg. (2.1)
- Adult Patients Intravenous and Oral Dosage : 200 mg administered once daily orally or as an intravenous (IV) infusion over 1 hour for 6 days as specified in Table 1 in the full prescribing information. (2.2)
- Pediatric Patients Intravenous Dosage (at least 26 weeks gestational age and weighing at least 1 kg): Weight-based dosing as an intravenous infusion as specified in Table 2 in the full prescribing information. (2.3)
- Pediatric Patients Oral Dosage (weighing greater than or equal to 35 kg): Weight-based dosing as an oral tablet administered once daily as specified in Table 3 in the full prescribing information. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
SIVEXTRO 200 mg tablet is a yellow film-coated oval tablet; each tablet is debossed with "TZD" on one side and "200" on the other side.
SIVEXTRO for Injection is a sterile, white to off-white lyophilized powder for injection in single-dose vials of 200 mg. Each 200 mg vial must be reconstituted with Sterile Water for Injection and subsequently diluted with 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP.
5.2 Clostridioides Difficile (5.2 Clostridioides difficile)
Clostridioides difficile-associated diarrhea (CDAD) has been reported for nearly all systemic antibacterial agents including SIVEXTRO, with severity ranging from mild diarrhea to fatal colitis. Treatment with antibacterial agents can alter the normal flora of the colon and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary because CDAD has been reported to occur more than two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, antibacterial use not directed against C. difficile should be discontinued, if possible. Appropriate measures such as fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of SIVEXTRO. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: thrombocytopenia
5.1 Patients With Neutropenia (5.1 Patients with Neutropenia)
The safety and efficacy of SIVEXTRO in patients with neutropenia (neutrophil counts <1000 cells/mm3) have not been adequately evaluated. In an animal model of infection, the antibacterial activity of SIVEXTRO was reduced in the absence of granulocytes [see Clinical Pharmacology (12.2)]. Alternative therapies should be considered when treating patients with neutropenia and ABSSSI.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Based on animal data, SIVEXTRO may cause fetal harm. Advise pregnant women of the potential risks to a fetus. (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be compared directly to rates from clinical trials of another drug and may not reflect rates observed in practice.
Clinical Trials Experience in Adult Patients
Adverse reactions were evaluated for 1425 adult patients treated with SIVEXTRO in two Phase 2 and four Phase 3 clinical trials (three Phase 3 trials for 6 days of therapy and one Phase 3 trial for 7-21 days of therapy). The median age of adult patients treated with SIVEXTRO in the Phase 2 and Phase 3 trials was 44 years, ranging between 17 and 94 years old. The majority of adult patients treated with SIVEXTRO were male (66%) and White (67%).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.6 Compatible Intravenous Solutions
SIVEXTRO is compatible with 0.9% Sodium Chloride Injection, USP and 5% Dextrose Injection, USP.
Limited data are available on the compatibility of SIVEXTRO for Injection with other intravenous substances, additives or other medications and they should not be added to SIVEXTRO single-dose vials or infused simultaneously. If the same intravenous line is used for sequential infusion of several different drugs, the line should be flushed before and after infusion of SIVEXTRO with 0.9% Sodium Chloride Injection, USP.
2.2 Recommended Dosage for Adult Patients
The recommended dosage of SIVEXTRO is 200 mg administered once daily for six (6) days either as an oral tablet (with or without food) or as an intravenous (IV) infusion in adult patients.
The recommended dosage and administration of SIVEXTRO in adult patients are described in Table 1.
| Infection | Route | Dose | Frequency | Infusion Time | Duration of Treatment |
|---|---|---|---|---|---|
| Acute Bacterial Skin and Skin Structure Infections (ABSSSI) |
Intravenous | 200 mg | Once daily | 1 hour | 6 days |
| Oral | 200 mg | Once daily | Not Applicable |
No dose adjustment is necessary when changing from intravenous to oral SIVEXTRO.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Repeated-oral and intravenous dosing of tedizolid phosphate in rats in 1 month and 3 month toxicology studies produced dose- and time-dependent bone marrow hypocellularity (myeloid, erythroid, and megakaryocyte), with associated reduction in circulating RBCs, WBCs, and platelets. These effects showed evidence of reversibility and occurred at plasma tedizolid exposure levels (AUC) ≥6-fold greater than the plasma exposure associated with the human therapeutic dose. In a 1-month immunotoxicology study in rats, repeated oral dosing of tedizolid phosphate was shown to significantly reduce splenic B cells and T cells and reduce plasma IgG titers. These effects occurred at plasma tedizolid exposure levels (AUC) ≥3-fold greater than the expected human plasma exposure associated with the therapeutic dose.
5.3 Development of Drug Resistant Bacteria (5.3 Development of Drug-Resistant Bacteria)
Prescribing SIVEXTRO in the absence of a proven or strongly suspected bacterial infection or prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Principal Display Panel 200 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 200 mg Vial Carton)
NDC 67919-040-02
10 single-dose vials
Sivextro
®
(tedizolid phosphate) for injection
200 mg per vial
For Intravenous Infusion
Must be reconstituted and further
diluted prior to administration
by intravenous infusion.
Sterile
Rx only
2.3 Recommended Dosage for Pediatric Patients
Recommended Dosage of SIVEXTRO for Injection for Pediatric Patients: Intravenous Dosage
The recommended intravenous dosage of SIVEXTRO for Injection for pediatric patients (at least 26 weeks gestational age and weighing at least 1 kg) is presented in Table 2.
| Weight Band (kg) | Dose | Frequency | Infusion Time | Duration of Treatment |
|---|---|---|---|---|
| Pediatric Patients Weighing Less than 2 kg | ||||
| 1 to less than 2
Recommended Dosage for 1 kg to less than 2 kg is based on actual body weight.
|
3 mg/kg | Twice daily | 1 hour | 6 days |
| Pediatric Patients Weighing at Least 2 kg | ||||
| 2 to less than 3 | 6 mg | Twice daily | 1 hour | 6 days |
| 3 to less than 6 | 12 mg | |||
| 6 to less than 10 | 20 mg | |||
| 10 to less than 14 | 30 mg | |||
| 14 to less than 20 | 40 mg | |||
| 20 to less than 35 | 60 mg | |||
| Pediatric Patients Weighing at Least 35 kg | ||||
| Greater than or equal to 35 | 200 mg | Once daily | 1 hour | 6 days |
Recommended Dosage of SIVEXTRO Tablets for Pediatric Patients: Oral Dosage
The recommended oral dosage of SIVEXTRO Tablets for pediatric patients is presented in Table 3. SIVEXTRO Tablets can be administered with or without food. Do not administer SIVEXTRO Tablets to pediatric patients weighing less than 35 kg.
| Weight Band (kg) | Dose | Frequency | Duration of Treatment |
|---|---|---|---|
| Greater than or equal to 35 | 200 mg | Once daily | 6 days |
No dose adjustment is necessary when changing from intravenous to oral SIVEXTRO.
2.4 Recommendations Regarding Missed Doses(s)
For Once Daily Oral Dosing of SIVEXTRO Tablets
If patients miss a dose, administer the dose as soon as possible anytime up to 8 hours prior to their next scheduled dose. If less than 8 hours remain before the next dose, instruct the patients to wait until their next scheduled dose.
Principal Display Panel 200 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 200 mg Tablet Bottle Label)
NDC 67919-041-04
30 tablets
Sivextro
®
(tedizolid phosphate) tablets
200 mg per tablet
Rx only
1.1 Acute Bacterial Skin and Skin Structure Infections
SIVEXTRO® is indicated for the treatment of acute bacterial skin and skin structure infections (ABSSSI) caused by susceptible isolates of the following gram-positive microorganisms: Staphylococcus aureus (including methicillin-resistant [MRSA] and methicillin-susceptible [MSSA] isolates), Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus anginosus Group (including Streptococcus anginosus, Streptococcus intermedius, and Streptococcus constellatus), and Enterococcus faecalis, in adult and pediatric patients (at least 26 weeks gestational age and weighing at least 1 kg).
14.1 Acute Bacterial Skin and Skin Structure Infections
Adults
A total of 1333 adults with acute bacterial skin and skin structure infections (ABSSSI) were randomized in two multicenter, multinational, double-blind, non-inferiority trials. Both trials compared SIVEXTRO 200 mg once daily for 6 days versus linezolid 600 mg every 12 hours for 10 days. In Trial 1, patients were treated with oral therapy, while in Trial 2, patients could receive oral therapy after a minimum of one day of intravenous therapy. Patients with cellulitis/erysipelas, major cutaneous abscess, or wound infection were enrolled in the trials. Patients with wound infections could have received aztreonam and/or metronidazole as adjunctive therapy for gram-negative bacterial coverage, if needed. The intent-to-treat (ITT) patient population included all randomized patients.
In Trial 1, 332 patients with ABSSSI were randomized to SIVEXTRO and 335 patients were randomized to linezolid. The majority (91%) of patients treated with SIVEXTRO in Trial 1 were less than 65 years old with a median age of 43 years (range: 18 to 86 years). Patients treated with SIVEXTRO were predominantly male (61%) and White (84%); 13% had BMI ≥35 kg/m2, 8% had diabetes mellitus, 35% were current or recent intravenous drug users, and 2% had moderate to severe renal impairment. The overall median surface area of infection was 188 cm2. The types of ABSSSI included were cellulitis/erysipelas (41%), wound infection (29%), and major cutaneous abscess (30%). In addition to local signs and symptoms of infection, patients were also required to have at least one regional or systemic sign of infection at baseline, defined as lymphadenopathy (87% of patients), temperature 38°C or higher (16% of patients), white blood cell count greater than 10,000 cells/mm3 or less than 4000 cells/mm3 (42%), or 10% or more band forms on white blood cell differential (4%).
The primary endpoint in Trial 1 was early clinical response defined as no increase from baseline lesion area at 48-72 hours after the first dose and oral temperature of ≤37.6°C, confirmed by a second temperature measurement within 24 hours in the ITT population.
In Trial 2, 332 patients with ABSSSI were randomized to SIVEXTRO and 334 patients were randomized to linezolid. The majority (87%) of patients treated with SIVEXTRO in Trial 2 were less than 65 years old with a median age of 46 years (range: 17 to 86 years). Patients treated with SIVEXTRO were predominantly male (68%) and White (86%); 16% had BMI ≥35 kg/m2, 10% had diabetes mellitus, 20% were current or recent intravenous drug users, and 4% had moderate to severe renal impairment. The overall median surface area of infection was 231 cm2. The types of ABSSSI included were cellulitis/erysipelas (50%), wound infection (30%), and major cutaneous abscess (20%). In addition to local signs and symptoms of infection, patients were also required to have at least one regional or systemic sign of infection at baseline, defined as lymphadenopathy (71% of patients), temperature 38°C or higher (31% of patients), white blood cell count greater than 10,000 cells/mm3 or less than 4000 cells/mm3 (53%), or 10% or more band forms on white blood cell differential (16%).
The primary endpoint in Trial 2 was early clinical response defined as at least a 20% decrease from baseline lesion area at 48-72 hours after the first dose in the ITT population (Table 10).
| SIVEXTRO (200 mg) |
Linezolid (1200 mg) |
Treatment Difference (2-sided 95% CI) |
|
|---|---|---|---|
| CI=confidence interval | |||
|
No increase in lesion surface area from baseline and oral temperature of ≤37.6°C, confirmed by a second temperature measurement within 24 hours at 48-72 hours Primary endpoint for Trial 1; sensitivity analysis for Trial 2
|
|||
| Trial 1, N | 332 | 335 | |
| Responder, n (%) | 264 (79.5) | 266 (79.4) | 0.1 (-6.1, 6.2) |
| Trial 2, N | 332 | 334 | |
| Responder, n (%) | 286 (86.1) | 281 (84.1) | 2.0 (-3.5, 7.3) |
|
At least a 20% decrease from baseline in lesion area at 48-72 hours Primary endpoint for Trial 2; sensitivity analysis for Trial 1
|
|||
| Trial 1, N | 332 | 335 | |
| Responder, n (%) | 259 (78.0) | 255 (76.1) | 1.9 (-4.5, 8.3) |
| Trial 2, N | 332 | 334 | |
| Responder, n (%) | 283 (85.2) | 276 (82.6) | 2.6 (-3.0, 8.2) |
An investigator assessment of clinical response was made at the post-therapy evaluation (PTE) (7 - 14 days after the end of therapy) in the ITT and CE (Clinically Evaluable) populations. Clinical success was defined as resolution or near resolution of most disease-specific signs and symptoms, absence or near resolution of systemic signs of infection if present at baseline (lymphadenopathy, fever, >10% immature neutrophils, abnormal WBC count), and no new signs, symptoms, or complications attributable to the ABSSSI requiring further treatment of the primary lesion (Table 11).
| SIVEXTRO (200 mg) n/N (%) |
Linezolid (1200 mg) n/N (%) |
Treatment Difference (2-sided 95% CI) |
|
|---|---|---|---|
| CI=confidence interval; ITT=intent-to-treat; CE=clinically evaluable | |||
| Trial 1 | |||
| ITT | 284/332 (85.5) | 288/335 (86.0) | -0.5 (-5.8, 4.9) |
| CE | 264/279 (94.6) | 267/280 (95.4) | -0.8 (-4.6, 3.0) |
| Trial 2 | |||
| ITT | 292/332 (88.0) | 293/334 (87.7) | 0.3 (-4.8, 5.3) |
| CE | 268/290 (92.4) | 269/280 (96.1) | -3.7 (-7.7, 0.2) |
Clinical success by baseline pathogens from the primary infection site or blood cultures for the microbiological intent-to-treat (MITT) patient population for two integrated Phase 3 ABSSSI studies are presented in Table 12 and Table 13.
| Pathogen | No increase in lesion surface area from baseline and oral temperature of ≤37.6°C Primary endpoint of Trial 1
|
At least a 20% decrease from baseline in lesion area Primary endpoint of Trial 2
|
||
|---|---|---|---|---|
| SIVEXTRO (200 mg) n/N (%) |
Linezolid (1200 mg) n/N (%) |
SIVEXTRO (200 mg) n/N (%) |
Linezolid (1200 mg) n/N (%) |
|
| Pooled analysis; n=number of patients in the specific category; N=Number of patients with the specific pathogen isolated from the ABSSSI | ||||
| Staphylococcus aureus | 276/329 (83.9) | 278/342 (81.3) | 280/329 (85.1) | 276/342 (80.7) |
| Methicillin-resistant S. aureus | 112/141 (79.4) | 113/146 (77.4) | 114/141 (80.9) | 111/146 (76.0) |
| Methicillin-susceptible S. aureus | 164/188 (87.2) | 167/198 (84.3) | 166/188 (88.3) | 167/198 (84.3) |
| Streptococcus pyogenes | 27/33 (81.8) | 18/20 (90.0) | 25/33 (75.8) | 16/20 (80.0) |
| Streptococcus anginosus Group | 22/30 (73.3) | 26/28 (92.9) | 22/30 (73.3) | 25/28 (89.3) |
| Streptococcus agalactiae | 6/9 (66.7) | 8/10 (80.0) | 6/9 (66.7) | 7/10 (70.0) |
| Enterococcus faecalis | 7/10 (70.0) | 3/4 (75.0) | 6/10 (60.0) | 1/4 (25.0) |
Baseline bacteremia in the tedizolid arm with relevant pathogens included two subjects with MRSA, four subjects with MSSA, two subjects with S. pyogenes, one subject with S. agalactiae, and one subject with S. constellatus. All of these subjects were Responders at the 48-72 hour evaluation. At the Post-therapy Evaluation (PTE), 8 of 10 subjects were considered clinical successes.
| Pathogen | Clinical Response at PTE | |
|---|---|---|
| SIVEXTRO (200 mg) n/N (%) |
Linezolid (1200 mg) n/N (%) |
|
| Pooled analysis; n=number of patients in the specific category; N=Number of patients with the specific pathogen isolated from the ABSSSI | ||
| Staphylococcus aureus | 291/329 (88.5) | 303/342 (88.6) |
| Methicillin-resistant S. aureus | 118/141 (83.7) | 119/146 (81.5) |
| Methicillin-susceptible S. aureus | 173/188 (92.0) | 186/198 (93.9) |
| Streptococcus pyogenes | 30/33 (90.9) | 19/20 (95.0) |
| Streptococcus anginosus Group | 21/30 (70.0) | 25/28 (89.3) |
| Streptococcus agalactiae | 8/9 (88.9) | 8/10 (80.0) |
| Enterococcus faecalis | 7/10 (70.0) | 4/4 (100.0) |
Baseline bacteremia in the tedizolid arm with relevant pathogens included two subjects with MRSA, four subjects with MSSA, two subjects with S. pyogenes, one subject with S. agalactiae, and one subject with S. constellatus. All of these subjects were Responders at the 48-72 hour evaluation. At the Post-therapy Evaluation (PTE) 8 of 10 subjects were considered clinical successes.
Pediatric Patients
The safety and efficacy of SIVEXTRO were investigated in Pediatric Trials 1 and 2. Pediatric Trial 1 included pediatric patients 12 to < 18 years of age who were investigated in a randomized, single blind, active-controlled trial of 120 patients with clinically documented ABSSSI (91 receiving tedizolid, 29 receiving comparator). Patients were randomized in a 3:1 ratio with stratification by geographic region to receive SIVEXTRO IV and/or oral therapy, dosed 200 mg once daily for 6 days, or comparator IV and/or oral therapy, dosed over 10 days. Comparator therapy was selected by the investigator from a list of 5 IV and 4 oral comparators per local standard of care. The most frequently used comparators were cefazolin (11 patients) and vancomycin (8 patients).
Additionally, Pediatric Trial 2 (NCT03176134) evaluated the safety and efficacy of SIVEXTRO in pediatric patients 4 months to <12 years of age in a randomized, single blind, active-controlled trial of 100 patients with clinically documented ABSSSI (75 receiving tedizolid, 25 receiving comparator). Patients were randomized in a 3:1 ratio to receive SIVEXTRO for 6 to 10 days or comparator for 10 to 14 days. SIVEXTRO (IV and/or oral) was dosed as a weight-based dose of 2 to 2.5 mg/kg every 12 hours or as 200 mg given once daily. Patients who received oral therapy received an oral suspension formulation that is not currently approved for use. Comparator therapy was selected from a pre-specified list by the investigator and dosed per local standard of care.
The primary objective of Pediatric Trials 1 and 2 was to evaluate the safety and tolerability of SIVEXTRO. The trials were not powered for comparative inferential efficacy analysis. Clinical response at the test of cure visit (Day 18-25) was assessed by a blinded investigator in the ITT population (all randomized patients). Clinical successes were required to have resolution or near resolution of all related signs and symptoms such that no further antibacterial therapy was needed. Early clinical response, defined as at least a 20% reduction in lesion size at 48-72 hours after start of treatment, was also assessed in the ITT population.
In Pediatric Trial 1, clinical success at test of cure was 96.7% (88/91) in the tedizolid group and 93.1% (27/29) in the comparator group (difference: 3.6%, 95% CI: -6.3, 13.5). Early clinical response at 48-72 hours was 92.3% (84/91) in the tedizolid group and 96.6% (28/29) in the comparator group (difference: -4.2%, 95% CI: -12.9, 4.4).
In Pediatric Trial 2, clinical success at test of cure was 93.3% (70/75) in the tedizolid group and 92.0% (23/25) in the comparator group (difference: 1.3%, 95% CI: -10.7, 13.4). Early clinical response was not adequately assessed in Pediatric Trial 2 because an in-person outpatient visit at this timepoint was not required during the COVID-19 pandemic.
1.2 Usage to Reduce Development of Drug Resistant Bacteria (1.2 Usage to Reduce Development of Drug-Resistant Bacteria)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of SIVEXTRO and other antibacterial drugs, SIVEXTRO should be used only to treat infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
2.5 Preparation and Administration of Intravenous Solution
SIVEXTRO is supplied as a sterile, lyophilized powder for injection in single-dose vials of 200 mg. Each 200 mg vial must be reconstituted with Sterile Water for Injection and subsequently diluted with 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP.
SIVEXTRO vials contain no antimicrobial preservatives and are intended for single dose only. Discard any unused portion.
Principal Display Panel 200 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 200 mg Tablet Blister Pack Carton)
NDC 67919-041-05
6 tablets
Sivextro
®
(tedizolid phosphate) tablets
200 mg per tablet
Rx only
2.1 Important Administration Instructions for Pediatric Patients Weighing Less Than 35 Kg (2.1 Important Administration Instructions for Pediatric Patients Weighing Less than 35 kg)
Do not administer SIVEXTRO Tablets to pediatric patients weighing less than 35 kg [see Dosage and Administration 2.3].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:44.289161 · Updated: 2026-03-14T22:25:35.146821