drug facts
7546770d-bc12-44b8-9fe0-fc3f5e1d12d1
34390-5
HUMAN OTC DRUG LABEL
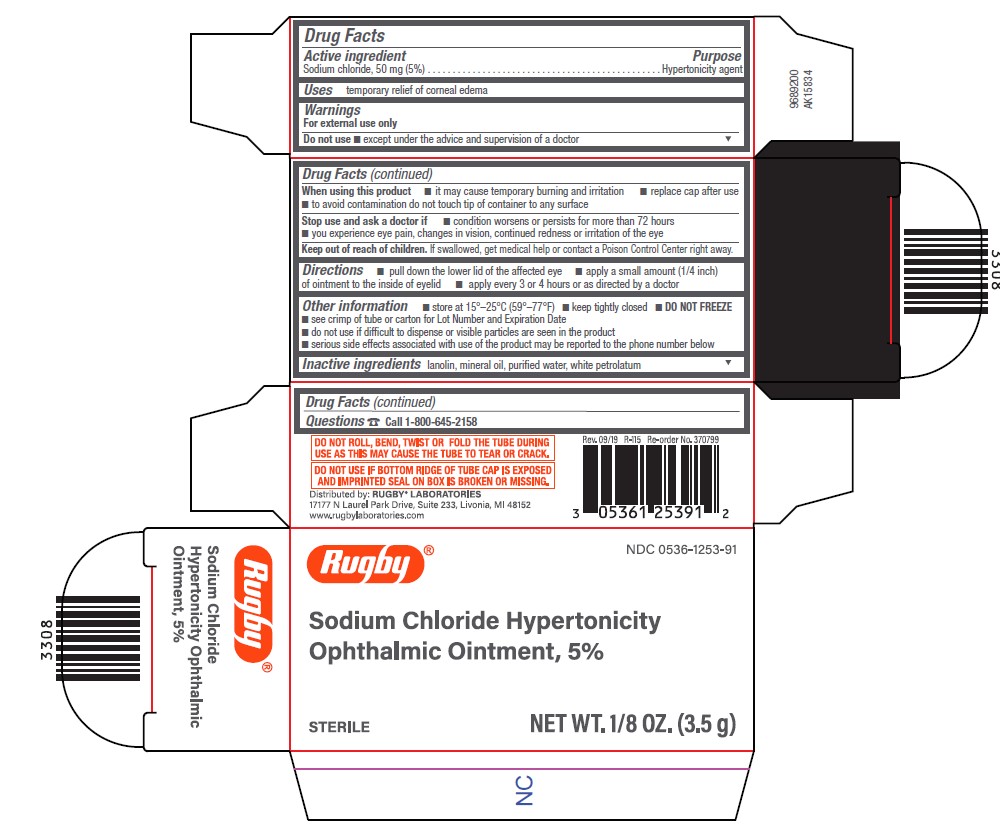
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Hypertonicity agent
Description
Sodium chloride 50 mg (5%)
Medication Information
Warnings
For external use only
Do not useexcept under the advice and supervision of a doctor
When using this product
- it may cause temporary burning and irritation
- replace cap after use
- to avoid contamination do not touch tip of container to any surface
Stop use and ask a doctor if
- condition worsens or persists for more than 72 hours
- you experience eye pain, changes in vision, continued redness or irritation of the eye
Uses
temporary relief of corneal edema
Purpose
Hypertonicity agent
Directions
- pull down the lower lid of the affected eye
- apply a small amount (1/4 inch) of ointment to the inside of eyelid
- apply every 3 or 4 hours or as directed by a doctor
Other Information
- store at 15° - 25°C (59° - 77°F)
- keep tightly closed
- DO NOT FREEZE
- see crimp of tube or carton for Lot Number and Expiration Date
- do not use if difficult to dispense or visible particles are seen in the product
- serious side effects associated with use of the product may be reported to the phone number below
Inactive Ingredients
lanolin, mineral oil, purified water, white petrolatum
Description
Sodium chloride 50 mg (5%)
Questions
Call 1-800-645-2158
Active Ingredient
Sodium chloride 50 mg (5%)
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel Carton
Rugby ®
NDC 0536-1253-91
Sodium Chloride Hypertonicity
Ophthalmic Ointment, 5%
- STERILE NET WT. 1/8 OZ. (3.5 G)
Structured Label Content
Warnings
For external use only
Do not useexcept under the advice and supervision of a doctor
When using this product
- it may cause temporary burning and irritation
- replace cap after use
- to avoid contamination do not touch tip of container to any surface
Stop use and ask a doctor if
- condition worsens or persists for more than 72 hours
- you experience eye pain, changes in vision, continued redness or irritation of the eye
Uses
temporary relief of corneal edema
Purpose
Hypertonicity agent
Directions
- pull down the lower lid of the affected eye
- apply a small amount (1/4 inch) of ointment to the inside of eyelid
- apply every 3 or 4 hours or as directed by a doctor
Other Information (Other information)
- store at 15° - 25°C (59° - 77°F)
- keep tightly closed
- DO NOT FREEZE
- see crimp of tube or carton for Lot Number and Expiration Date
- do not use if difficult to dispense or visible particles are seen in the product
- serious side effects associated with use of the product may be reported to the phone number below
Inactive Ingredients (Inactive ingredients)
lanolin, mineral oil, purified water, white petrolatum
Questions
Call 1-800-645-2158
Active Ingredient (Active ingredient)
Sodium chloride 50 mg (5%)
Keep Out of Reach of Children
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel Carton (Package/Label Principal Display Panel Carton)
Rugby ®
NDC 0536-1253-91
Sodium Chloride Hypertonicity
Ophthalmic Ointment, 5%
- STERILE NET WT. 1/8 OZ. (3.5 G)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:36.581927 · Updated: 2026-03-14T23:03:47.044278