Medicaine Sting®
753e6cbe-31e0-4017-93d0-8b4bf5972f37
34390-5
HUMAN OTC DRUG LABEL
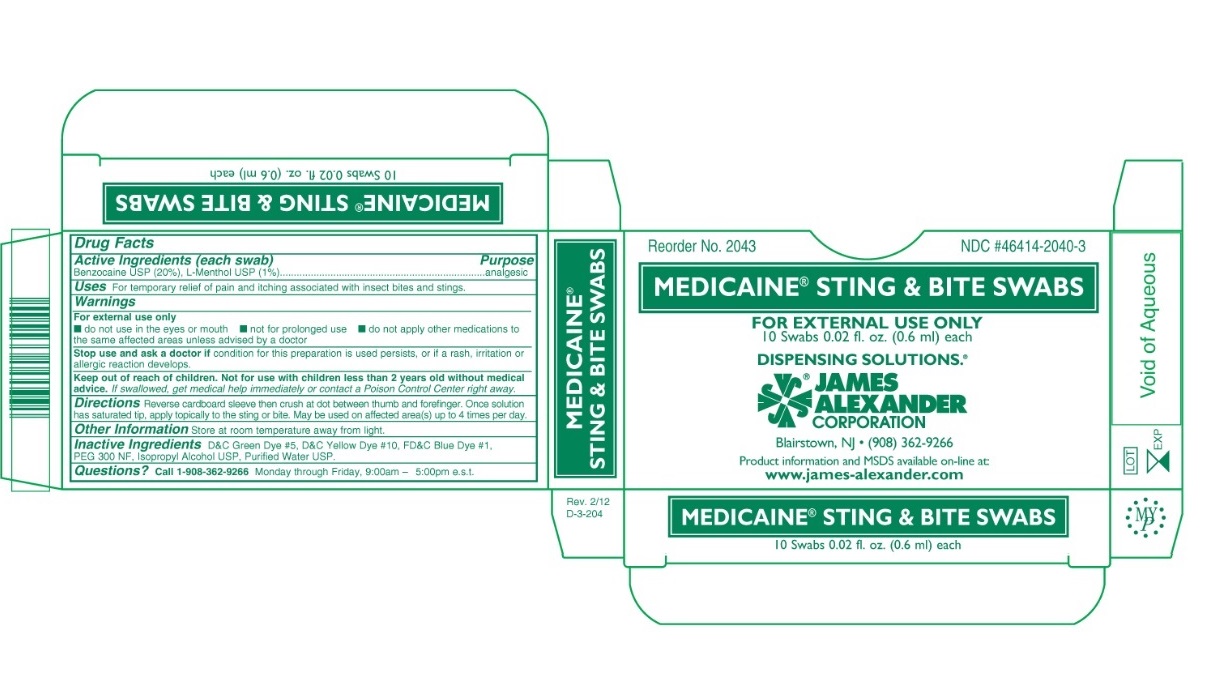
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Benzocaine USP (20%), L-Menthol USP (1%)
Purpose
analgesic
Medication Information
Purpose
analgesic
Description
Benzocaine USP (20%), L-Menthol USP (1%)
Uses
For temporary relief of pain and itching associated with insect bites and stings.
Section 42229-5
DISPENSING SOLUTIONS®
JAMES ALEXANDER CORPORATION
Blairstown, NJ. (908) 362-9266
Product information and MSDS available on-line at:
www.james-alexander.com
Void of Aqueous
Section 50565-1
Keep out of reach of children. Not for use with children less than 2 years old without medical advice. If swallowed, get medical help immediately or contact a Poison Control Center right away.
Warnings
For external use only
- do not use in the eyes or mouth
- not for prolonged use
- do not apply other medications to the same affected areas unless advised by a doctor
Stop use and ask a doctor if condition for this preparation is used persists, or if a rash or irritation or allergic reaction develops.
Packaging
Directions
Reverse cardboard sleeve then crush at dot between thumb and forefinger. Once solution has saturated tip, apply topically to the sting or bite. May be used on affected area(s) up to 4 times per day.
Drug Facts
Questions?
Call 1-908-362-9266 Monday through Friday, 9:00am - 5:00pm e.s.t
Other Information
Store at room temperature away from light.
Inactive Ingredients
D&C Green Dye #5, D&C Yellow Dye #10, FD&C Blue Dye #1, PEG 300 NF, Isopropyl Alcohol USP, Purified Water USP.
Active Ingredients (each Swab)
Benzocaine USP (20%), L-Menthol USP (1%)
Structured Label Content
Uses
For temporary relief of pain and itching associated with insect bites and stings.
Section 42229-5 (42229-5)
DISPENSING SOLUTIONS®
JAMES ALEXANDER CORPORATION
Blairstown, NJ. (908) 362-9266
Product information and MSDS available on-line at:
www.james-alexander.com
Void of Aqueous
Section 50565-1 (50565-1)
Keep out of reach of children. Not for use with children less than 2 years old without medical advice. If swallowed, get medical help immediately or contact a Poison Control Center right away.
Purpose
analgesic
Warnings
For external use only
- do not use in the eyes or mouth
- not for prolonged use
- do not apply other medications to the same affected areas unless advised by a doctor
Stop use and ask a doctor if condition for this preparation is used persists, or if a rash or irritation or allergic reaction develops.
Packaging
Directions
Reverse cardboard sleeve then crush at dot between thumb and forefinger. Once solution has saturated tip, apply topically to the sting or bite. May be used on affected area(s) up to 4 times per day.
Drug Facts
Questions?
Call 1-908-362-9266 Monday through Friday, 9:00am - 5:00pm e.s.t
Other Information
Store at room temperature away from light.
Inactive Ingredients
D&C Green Dye #5, D&C Yellow Dye #10, FD&C Blue Dye #1, PEG 300 NF, Isopropyl Alcohol USP, Purified Water USP.
Active Ingredients (each Swab) (Active Ingredients (each swab))
Benzocaine USP (20%), L-Menthol USP (1%)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:44.413929 · Updated: 2026-03-14T23:05:38.431426