195r Sennosides Tablets Usp 25 Mg Sugar Coated Off-white
74a627fc-00e5-eb78-e053-2991aa0abacc
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
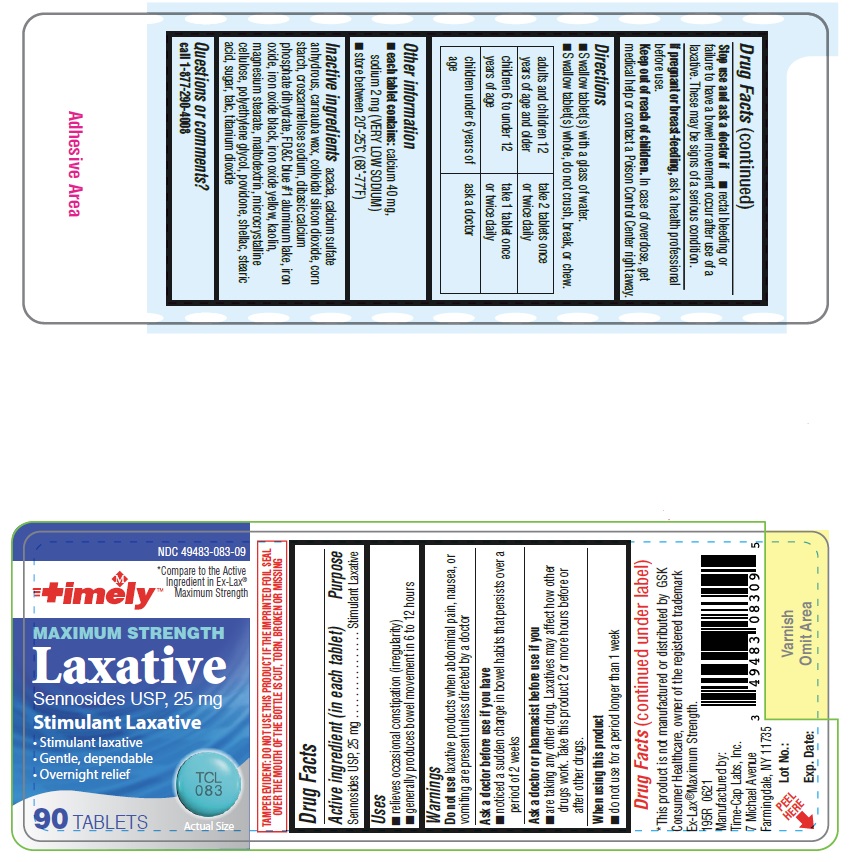
Active Ingredients (in each tablet) Sennosides USP, 25 mg
Medication Information
Warnings and Precautions
Warnings
Indications and Usage
Uses
- Relieves occasional constipation (irregularity)
- generally produces bowel movement in 6 to 12 hours
Dosage and Administration
Directions
- swallow tablet (s) with a glass of water.
- swallow tablet (s) whole, do not crush, break or chew.
adults and children 12 years of age and older
2 tablets once or twice daily
children 6 to under 12 years of age
1 tablet once or twice daily
children under 6 years of age
ask a doctor
Description
Active Ingredients (in each tablet) Sennosides USP, 25 mg
Section 50565-1
Keep out of the reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
- rectal bleeding or failure to have a bowel movement occur after use of a laxative. These may be signs of a serious condition.
Section 50567-7
When using this product
- do not use for a period longer than 1 week
Section 50568-5
Ask a doctor or pharmacist before use if you
are taking any other drug. Laxatives may affect how other drugs work. Take this product 2 or more hours before or after other drugs.
Section 50569-3
Ask a doctor before use if you have
- noticed a sudden change in bowel habits that persists over a period of 2 weeks
Section 50570-1
Do not use laxative products when abdominal pain, nausea, or vomiting are present unless directed by a doctor
Section 51727-6
Inactive ingredients acacia, calcium sulfate anhydrous, carnauba wax, colloidal silicon dioxide, corn starch, croscarmellose sodium, dibasic calcium phosphate dihydrate, FD&C blue #1 aluminum lake, iron oxide, iron oxide black, iron oxide yellow, kaolin, magnesium stearate, maltodextrin, microcrystalline cellulose, polyethylene glycol, povidone, shellac, stearic acid, sugar, talc, titanium dioxide.
Section 53413-1
Questions or comments? Call 1-877-290-4008
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purpose
Stimulant Laxative
Section 55106-9
Active Ingredients (in each tablet)
Sennosides USP, 25 mg
Section 60561-8
Other information
- each tablet contains: calcium 40 mg, sodium 2 mg (VERY LOW SODIUM)
- store between 20°-25°C (68°-77°F)
195r Lax Time (sennosides) 25 Mg
Structured Label Content
Indications and Usage (34067-9)
Uses
- Relieves occasional constipation (irregularity)
- generally produces bowel movement in 6 to 12 hours
Dosage and Administration (34068-7)
Directions
- swallow tablet (s) with a glass of water.
- swallow tablet (s) whole, do not crush, break or chew.
adults and children 12 years of age and older
2 tablets once or twice daily
children 6 to under 12 years of age
1 tablet once or twice daily
children under 6 years of age
ask a doctor
Warnings and Precautions (34071-1)
Warnings
Section 50565-1 (50565-1)
Keep out of the reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- rectal bleeding or failure to have a bowel movement occur after use of a laxative. These may be signs of a serious condition.
Section 50567-7 (50567-7)
When using this product
- do not use for a period longer than 1 week
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you
are taking any other drug. Laxatives may affect how other drugs work. Take this product 2 or more hours before or after other drugs.
Section 50569-3 (50569-3)
Ask a doctor before use if you have
- noticed a sudden change in bowel habits that persists over a period of 2 weeks
Section 50570-1 (50570-1)
Do not use laxative products when abdominal pain, nausea, or vomiting are present unless directed by a doctor
Section 51727-6 (51727-6)
Inactive ingredients acacia, calcium sulfate anhydrous, carnauba wax, colloidal silicon dioxide, corn starch, croscarmellose sodium, dibasic calcium phosphate dihydrate, FD&C blue #1 aluminum lake, iron oxide, iron oxide black, iron oxide yellow, kaolin, magnesium stearate, maltodextrin, microcrystalline cellulose, polyethylene glycol, povidone, shellac, stearic acid, sugar, talc, titanium dioxide.
Section 53413-1 (53413-1)
Questions or comments? Call 1-877-290-4008
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purpose
Stimulant Laxative
Section 55106-9 (55106-9)
Active Ingredients (in each tablet)
Sennosides USP, 25 mg
Section 60561-8 (60561-8)
Other information
- each tablet contains: calcium 40 mg, sodium 2 mg (VERY LOW SODIUM)
- store between 20°-25°C (68°-77°F)
195r Lax Time (sennosides) 25 Mg (195R Lax-Time (Sennosides) 25 mg)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:31.528408 · Updated: 2026-03-14T23:07:26.290118