These Highlights Do Not Include All The Information Needed To Use Fexinidazole Tablets Safely And Effectively. See Full Prescribing Information For Fexinidazole Tablets.
74a611bc-9977-46bb-b626-0370b3031628
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Contraindications ( 4 ) 9/2024
Indications and Usage
Fexinidazole Tablets are indicated for the treatment of both the first-stage (hemolymphatic) and second-stage (meningoencephalitic) human African trypanosomiasis (HAT) due to Trypanosoma brucei gambiense in patients 6 years of age and older and weighing at least 20 kg.
Dosage and Administration
Administer Fexinidazole Tablets once daily with food each day at about the same time of the day. Do not break or crush tablets. ( 2.1 , 2.2 ) Recommended Dosage of Fexinidazole Tablets in Patients 6 years of age and older and weighing at least 20 kg ( 2.2 ) Body Weight Type of Dose Daily Dose Number of Tablets Duration of Treatment Greater than or equal to 35 kg Loading dose 1,800 mg 3 4 days Maintenance dose 1,200 mg 2 6 days Greater than or equal to 20 kg to less than 35 kg Loading dose 1,200 mg 2 4 days Maintenance dose 600 mg 1 6 days
Warnings and Precautions
Decreased Efficacy in Severe Human African Trypanosomiasis caused by Trypanosoma brucei gambiense. ( 1 , 5.1 ) QT Interval Prolongation: Prolongation of the QT interval due to Fexinidazole Tablets occurs in a concentration-dependent manner. Avoid use in patients with known prolongation, proarrhythmic conditions, and concomitant use with drugs that prolong the QT interval, those that block cardiac potassium channels, and/or those that induce bradycardia, or are inducers of hepatic CYP450. ( 5.2 , 7.1 , 7.2 , 12.2 ) Neuropsychiatric Adverse Reactions: Adverse reactions such as agitation, anxiety, abnormal behavior, depression, suicidal ideation, nightmares, hallucination, and personality change have been observed during therapy. Inform patients and their caregivers of the risk. Consider alternative therapy or increased monitoring of the patient, including hospitalization in patients with psychiatric disorders, or if these adverse reactions occur. ( 5.3 ) Neutropenia: Avoid concomitant use of drugs which may cause neutropenia and monitor leukocyte count periodically. Monitor patients with neutropenia for symptoms or signs of infection. ( 5.4 , 6.1 ) Potential for Hepatotoxicity: Evaluate liver-related laboratory tests at the start and during treatment. ( 4 , 5.5 ) Risk of Disulfiram-like Reactions Due to Concomitant Use with Alcohol: Nitroimidazole-class drugs may cause a disulfiram-like reaction in patients who concurrently consume alcohol. Advise patients to avoid consumption of alcohol during treatment with and for at least 48 hours after completing therapy. ( 2.1 , 5.6 ) Risk of Psychotic Reactions Due to Concomitant Use with Disulfiram: Psychotic reactions have been reported in patients concurrently taking disulfiram and nitroimidazole-class drugs. Avoid use in patients who have taken disulfiram within the last two weeks. ( 5.7 ).
Contraindications
Fexinidazole Tablets are contraindicated in: Patients with known hypersensitivity to Fexinidazole Tablets and/or any nitroimidazole-class drugs (e.g., metronidazole, tinidazole). Patients with severe hepatic impairment [see Warnings and Precautions (5.5) , Adverse Reactions (6.1) , and Use in Specific Populations (8.7) ] . Patients with Cockayne syndrome. Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole, another nitroimidazole drug, structurally related to fexinidazole, in patients with Cockayne syndrome [see Adverse Reactions (6.2) ] .
Adverse Reactions
Adult patients treated with Fexinidazole Tablets reported a higher percentage of Central Nervous System (CNS) and psychiatric-related adverse reactions than those treated with nifurtimox eflornithine combination therapy (NECT) in a clinical trial [see Adverse Reactions (6.1) ] . Increased incidence in insomnia, headache, and tremor was noted in the patients treated with Fexinidazole Tablets compared to NECT. In the same trial, adverse reactions representing mood changes and psychiatric disorders (such as agitation, anxiety, abnormal behavior, depression, nightmares, hallucination, and personality change) were more common in the patients treated with Fexinidazole Tablets compared to the NECT arm. Suicidal ideation has also been observed with Fexinidazole Tablets [see Adverse Reactions (6.1) ] . Healthcare providers should inform patients and their caregivers of the risk for neuropsychiatric adverse reactions during treatment with Fexinidazole Tablets. In patients with current or a history of psychiatric disorders, or should such adverse reactions occur, healthcare providers should consider alternative therapy or increased monitoring of the patient, including hospitalization.
Drug Interactions
Table 3: Effect of Fexinidazole on other Drugs Drugs Metabolized by Cytochrome P450 (CYP) 3A4/5 Examples (not fully inclusive): Lovastatin, simvastatin, nisoldipine, saquinavir, midazolam, certain hormonal contraceptives (e.g., birth control pills, skin patches, implant) Clinical Impact Fexinidazole is considered a moderate CYP3A4/5 inducer [see Clinical Pharmacology (12.3) ] . Co-administration of Fexinidazole Tablets and CYP3A4/5 substrates may reduce the systemic exposures of CYP3A4/5 substrate drug(s), which may lead to reduced efficacy [see Clinical Pharmacology (12.3) ] . Prevention or Management Avoid concomitant use with Fexinidazole Tablets . If coadministration cannot be avoided, monitor for lack of efficacy of sensitive substrate drugs. If concomitant use of oral contraceptives cannot be avoided, an alternative contraceptive that is not affected by enzyme inducers (e.g., intrauterine system) or additional nonhormonal contraception (e.g., condoms) is recommended during treatment with Fexinidazole Tablets and for at least 5 days after the last dose. Drugs Metabolized by CYP1A2 or CYP2C19 Examples (not fully inclusive): CYP1A2: duloxetine, tacrine, tizanidine, theophylline CYP2C19: lansoprazole, mephenytoin, diazepam Clinical Impact Increased risk for adverse reactions associated with increased concentrations of the drug due to inhibition of either CYP1A2 or CYP2C19 by fexinidazole [see Clinical Pharmacology (12.3) ] . Prevention or Management Monitor for adverse reactions associated with these drugs when used concomitantly with Fexinidazole Tablets. Drugs Metabolized by CYP2B6 Examples (not fully inclusive): Bupropion, efavirenz Clinical Impact Increased risk for the lack of efficacy associated with decreased plasma concentrations of the drug due to induction of CYP2B6 by fexinidazole and Metabolite M1 [see Clinical Pharmacology (12.3) ] . Prevention or Management Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for lack of efficacy of these drugs. Drugs Metabolized by uridine 5'-diphospho--glucuronosyl transferase (UGT) Examples (not fully inclusive): Zidovudine, bictegravir, cabotegravir, dolutegravir, bexagliflozin, canagliflozin, sotagliflozin, deferasirox, lamotrigine, selexipag Clinical Impact Fexinidazole metabolites have the potential to be UGT inducers [see Clinical Pharmacology (12.3) ] . Co-administration of Fexinidazole Tablets and UGT substrates may reduce the systemic exposures of UGT substrate drug(s), which may lead to reduced efficacy. Prevention or Management When fexinidazole is concomitantly administered with UGT substrates, monitor for lack of efficacy of UGT substrate drugs. Drugs Substrates of OCT2, OAT1, OAT3, MATE1, and MATE2-K Transporters Examples (not fully inclusive): Metformin, dofetilide, adefovir, cefaclor, furosemide Clinical Impact Increased risk for adverse reactions associated with increased concentrations of the drug due to inhibition of these transporters by fexinidazole [see Clinical Pharmacology (12.3) ] . Prevention or Management Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for adverse reactions associated with these drugs. Table 4: Effect of other Drugs on Fexinidazole CYP450 Inducers Examples (not fully inclusive): Rifampin, phenytoin, St. John's wort, carbamazepine Clinical Impact Increased risk for adverse reactions associated with increased systemic exposure to the M1 and M2 metabolites of fexinidazole. M2 plasma concentrations have been associated with the increased risk of QT interval prolongation. Prevention or Management Avoid concomitant use with Fexinidazole Tablets [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3) ]. CYP450 Inhibitors Examples (not fully inclusive): Clarithromycin, itraconazole, voriconazole, erythromycin, fluconazole Clinical Impact Multiple CYP450 enzymes are involved in the metabolism of fexinidazole to its pharmacologically active M1 and M2 metabolites. Although no clinical drug interaction studies were performed with CYP450 inhibitors, the formation of the M1 and M2 metabolites may be decreased. Prevention or Management Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for lack of efficacy of fexinidazole due to potential for decreased plasma concentrations of the M1 and M2 metabolites [see Clinical Pharmacology (12.3) ] .
Storage and Handling
Fexinidazole 600 mg tablets are pale-yellow, round, biconvex tablets debossed with "4512" on one side. Fexinidazole 600 mg tablets are supplied as: NDC 0024-4512-24, child-resistant blister pack of 24 tablets (10-day dose pack) for adults and pediatric patients weighing 35 kg or more. NDC 0024-4512-14, child-resistant blister pack of 14 tablets (10-day dose pack) for pediatric patients older than 6 years weighing 20 kg to less than 35 kg.
How Supplied
Fexinidazole 600 mg tablets are pale-yellow, round, biconvex tablets debossed with "4512" on one side. Fexinidazole 600 mg tablets are supplied as: NDC 0024-4512-24, child-resistant blister pack of 24 tablets (10-day dose pack) for adults and pediatric patients weighing 35 kg or more. NDC 0024-4512-14, child-resistant blister pack of 14 tablets (10-day dose pack) for pediatric patients older than 6 years weighing 20 kg to less than 35 kg.
Medication Information
Warnings and Precautions
Decreased Efficacy in Severe Human African Trypanosomiasis caused by Trypanosoma brucei gambiense. ( 1 , 5.1 ) QT Interval Prolongation: Prolongation of the QT interval due to Fexinidazole Tablets occurs in a concentration-dependent manner. Avoid use in patients with known prolongation, proarrhythmic conditions, and concomitant use with drugs that prolong the QT interval, those that block cardiac potassium channels, and/or those that induce bradycardia, or are inducers of hepatic CYP450. ( 5.2 , 7.1 , 7.2 , 12.2 ) Neuropsychiatric Adverse Reactions: Adverse reactions such as agitation, anxiety, abnormal behavior, depression, suicidal ideation, nightmares, hallucination, and personality change have been observed during therapy. Inform patients and their caregivers of the risk. Consider alternative therapy or increased monitoring of the patient, including hospitalization in patients with psychiatric disorders, or if these adverse reactions occur. ( 5.3 ) Neutropenia: Avoid concomitant use of drugs which may cause neutropenia and monitor leukocyte count periodically. Monitor patients with neutropenia for symptoms or signs of infection. ( 5.4 , 6.1 ) Potential for Hepatotoxicity: Evaluate liver-related laboratory tests at the start and during treatment. ( 4 , 5.5 ) Risk of Disulfiram-like Reactions Due to Concomitant Use with Alcohol: Nitroimidazole-class drugs may cause a disulfiram-like reaction in patients who concurrently consume alcohol. Advise patients to avoid consumption of alcohol during treatment with and for at least 48 hours after completing therapy. ( 2.1 , 5.6 ) Risk of Psychotic Reactions Due to Concomitant Use with Disulfiram: Psychotic reactions have been reported in patients concurrently taking disulfiram and nitroimidazole-class drugs. Avoid use in patients who have taken disulfiram within the last two weeks. ( 5.7 ).
Indications and Usage
Fexinidazole Tablets are indicated for the treatment of both the first-stage (hemolymphatic) and second-stage (meningoencephalitic) human African trypanosomiasis (HAT) due to Trypanosoma brucei gambiense in patients 6 years of age and older and weighing at least 20 kg.
Dosage and Administration
Administer Fexinidazole Tablets once daily with food each day at about the same time of the day. Do not break or crush tablets. ( 2.1 , 2.2 ) Recommended Dosage of Fexinidazole Tablets in Patients 6 years of age and older and weighing at least 20 kg ( 2.2 ) Body Weight Type of Dose Daily Dose Number of Tablets Duration of Treatment Greater than or equal to 35 kg Loading dose 1,800 mg 3 4 days Maintenance dose 1,200 mg 2 6 days Greater than or equal to 20 kg to less than 35 kg Loading dose 1,200 mg 2 4 days Maintenance dose 600 mg 1 6 days
Contraindications
Fexinidazole Tablets are contraindicated in: Patients with known hypersensitivity to Fexinidazole Tablets and/or any nitroimidazole-class drugs (e.g., metronidazole, tinidazole). Patients with severe hepatic impairment [see Warnings and Precautions (5.5) , Adverse Reactions (6.1) , and Use in Specific Populations (8.7) ] . Patients with Cockayne syndrome. Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole, another nitroimidazole drug, structurally related to fexinidazole, in patients with Cockayne syndrome [see Adverse Reactions (6.2) ] .
Adverse Reactions
Adult patients treated with Fexinidazole Tablets reported a higher percentage of Central Nervous System (CNS) and psychiatric-related adverse reactions than those treated with nifurtimox eflornithine combination therapy (NECT) in a clinical trial [see Adverse Reactions (6.1) ] . Increased incidence in insomnia, headache, and tremor was noted in the patients treated with Fexinidazole Tablets compared to NECT. In the same trial, adverse reactions representing mood changes and psychiatric disorders (such as agitation, anxiety, abnormal behavior, depression, nightmares, hallucination, and personality change) were more common in the patients treated with Fexinidazole Tablets compared to the NECT arm. Suicidal ideation has also been observed with Fexinidazole Tablets [see Adverse Reactions (6.1) ] . Healthcare providers should inform patients and their caregivers of the risk for neuropsychiatric adverse reactions during treatment with Fexinidazole Tablets. In patients with current or a history of psychiatric disorders, or should such adverse reactions occur, healthcare providers should consider alternative therapy or increased monitoring of the patient, including hospitalization.
Drug Interactions
Table 3: Effect of Fexinidazole on other Drugs Drugs Metabolized by Cytochrome P450 (CYP) 3A4/5 Examples (not fully inclusive): Lovastatin, simvastatin, nisoldipine, saquinavir, midazolam, certain hormonal contraceptives (e.g., birth control pills, skin patches, implant) Clinical Impact Fexinidazole is considered a moderate CYP3A4/5 inducer [see Clinical Pharmacology (12.3) ] . Co-administration of Fexinidazole Tablets and CYP3A4/5 substrates may reduce the systemic exposures of CYP3A4/5 substrate drug(s), which may lead to reduced efficacy [see Clinical Pharmacology (12.3) ] . Prevention or Management Avoid concomitant use with Fexinidazole Tablets . If coadministration cannot be avoided, monitor for lack of efficacy of sensitive substrate drugs. If concomitant use of oral contraceptives cannot be avoided, an alternative contraceptive that is not affected by enzyme inducers (e.g., intrauterine system) or additional nonhormonal contraception (e.g., condoms) is recommended during treatment with Fexinidazole Tablets and for at least 5 days after the last dose. Drugs Metabolized by CYP1A2 or CYP2C19 Examples (not fully inclusive): CYP1A2: duloxetine, tacrine, tizanidine, theophylline CYP2C19: lansoprazole, mephenytoin, diazepam Clinical Impact Increased risk for adverse reactions associated with increased concentrations of the drug due to inhibition of either CYP1A2 or CYP2C19 by fexinidazole [see Clinical Pharmacology (12.3) ] . Prevention or Management Monitor for adverse reactions associated with these drugs when used concomitantly with Fexinidazole Tablets. Drugs Metabolized by CYP2B6 Examples (not fully inclusive): Bupropion, efavirenz Clinical Impact Increased risk for the lack of efficacy associated with decreased plasma concentrations of the drug due to induction of CYP2B6 by fexinidazole and Metabolite M1 [see Clinical Pharmacology (12.3) ] . Prevention or Management Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for lack of efficacy of these drugs. Drugs Metabolized by uridine 5'-diphospho--glucuronosyl transferase (UGT) Examples (not fully inclusive): Zidovudine, bictegravir, cabotegravir, dolutegravir, bexagliflozin, canagliflozin, sotagliflozin, deferasirox, lamotrigine, selexipag Clinical Impact Fexinidazole metabolites have the potential to be UGT inducers [see Clinical Pharmacology (12.3) ] . Co-administration of Fexinidazole Tablets and UGT substrates may reduce the systemic exposures of UGT substrate drug(s), which may lead to reduced efficacy. Prevention or Management When fexinidazole is concomitantly administered with UGT substrates, monitor for lack of efficacy of UGT substrate drugs. Drugs Substrates of OCT2, OAT1, OAT3, MATE1, and MATE2-K Transporters Examples (not fully inclusive): Metformin, dofetilide, adefovir, cefaclor, furosemide Clinical Impact Increased risk for adverse reactions associated with increased concentrations of the drug due to inhibition of these transporters by fexinidazole [see Clinical Pharmacology (12.3) ] . Prevention or Management Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for adverse reactions associated with these drugs. Table 4: Effect of other Drugs on Fexinidazole CYP450 Inducers Examples (not fully inclusive): Rifampin, phenytoin, St. John's wort, carbamazepine Clinical Impact Increased risk for adverse reactions associated with increased systemic exposure to the M1 and M2 metabolites of fexinidazole. M2 plasma concentrations have been associated with the increased risk of QT interval prolongation. Prevention or Management Avoid concomitant use with Fexinidazole Tablets [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3) ]. CYP450 Inhibitors Examples (not fully inclusive): Clarithromycin, itraconazole, voriconazole, erythromycin, fluconazole Clinical Impact Multiple CYP450 enzymes are involved in the metabolism of fexinidazole to its pharmacologically active M1 and M2 metabolites. Although no clinical drug interaction studies were performed with CYP450 inhibitors, the formation of the M1 and M2 metabolites may be decreased. Prevention or Management Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for lack of efficacy of fexinidazole due to potential for decreased plasma concentrations of the M1 and M2 metabolites [see Clinical Pharmacology (12.3) ] .
Storage and Handling
Fexinidazole 600 mg tablets are pale-yellow, round, biconvex tablets debossed with "4512" on one side. Fexinidazole 600 mg tablets are supplied as: NDC 0024-4512-24, child-resistant blister pack of 24 tablets (10-day dose pack) for adults and pediatric patients weighing 35 kg or more. NDC 0024-4512-14, child-resistant blister pack of 14 tablets (10-day dose pack) for pediatric patients older than 6 years weighing 20 kg to less than 35 kg.
How Supplied
Fexinidazole 600 mg tablets are pale-yellow, round, biconvex tablets debossed with "4512" on one side. Fexinidazole 600 mg tablets are supplied as: NDC 0024-4512-24, child-resistant blister pack of 24 tablets (10-day dose pack) for adults and pediatric patients weighing 35 kg or more. NDC 0024-4512-14, child-resistant blister pack of 14 tablets (10-day dose pack) for pediatric patients older than 6 years weighing 20 kg to less than 35 kg.
Description
Contraindications ( 4 ) 9/2024
Section 42229-5
Limitations of Use
Due to the decreased efficacy observed in patients with severe second stage HAT (cerebrospinal fluid white blood cell count (CSF-WBC) >100 cells/µL) due to T. brucei gambiense disease, Fexinidazole Tablets should only be used in these patients if there are no other available treatment options [see Warnings and Precautions (5.1)].
Section 43683-2
| Contraindications (4) | 9/2024 |
Section 44425-7
Store below 30°C (86°F). Store in the original package in order to protect from light and moisture.
10 Overdosage
Randomized, controlled clinical studies were conducted in normal adult male subjects who were administered single or multiple oral doses of fexinidazole of up to 3,600 mg daily for 14 days (not an approved dose). The subjects experienced adverse reactions of increased transaminases, vomiting and panic attack [see Adverse Reactions (6.1)].
Reported symptoms of overdosage in a pediatric HAT patient following ingestion of a higher than recommended dosing regimen in Trial 3 included vomiting over the first 5 days of treatment and increased potassium and decreased calcium levels from Day 11 to Week 9.
There is no specific antidote for Fexinidazole Tablets. Treatment should be supportive with appropriate monitoring.
11 Description
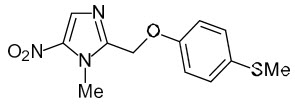
Fexinidazole Tablets contain fexinidazole, a nitroimidazole antimicrobial drug for oral use.
The chemical name of fexinidazole is 1-methyl-2-{[4-(methylthio)phenoxy]methyl}-5-nitro-1H-imidazole.
Its molecular formula is C12H13N3O3S and the molecular weight is 279.3 g/mol. The structural formula is:
Fexinidazole is a yellow powder. It is practically insoluble in water, sparingly soluble in acetone and acetonitrile, very slightly soluble in ethanol and slightly soluble in methanol.
Fexinidazole 600 mg Tablets contain the active ingredient fexinidazole and the following inactive ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, povidone, and sodium lauryl sulfate.
5.4 Neutropenia
Neutropenia (absolute neutrophil count less than 1,000 cells/mm3) has been reported in patients receiving Fexinidazole Tablets [see Adverse Reactions (6.1)]. In Trial 1, the adverse reaction occurred in patients with a baseline absolute neutrophil count of less than 5,000 cells/mm3. Avoid concomitant use of drugs which may cause neutropenia and monitor leukocyte count periodically. Carefully monitor patients with neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur.
8.4 Pediatric Use
The safety and effectiveness of Fexinidazole Tablets for the treatment of both the first-stage (hemolymphatic) and second-stage (meningoencephalitic) HAT due to Trypanosoma brucei gambiense have been established in pediatric patients aged 6 years and older and weighing at least 20 kg. Use of Fexinidazole Tablets for this indication is supported by evidence from an adequate and well-controlled trial in adults with additional efficacy, pharmacokinetic and safety data in pediatric patients aged 6 years and older [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Pediatric patients may be more sensitive to vomiting. The safety profile for Fexinidazole Tablets in pediatric patients was generally similar to that of adult patients with the exception of more frequent vomiting within 2 hours of administration of Fexinidazole Tablets. Vomiting did not result in permanent treatment discontinuation [see Adverse Reactions (6.1)].
The safety and efficacy of Fexinidazole Tablets have not been established in pediatric patients younger than 6 years old and/or less than 20 kg in body weight.
8.5 Geriatric Use
Of the 619 subjects in the three clinical trials treated with Fexinidazole Tablets for HAT, there were 11 subjects who were 65 years of age or older, and no subjects greater than 75 years of age. There were an insufficient number of elderly subjects to detect differences in safety and/or effectiveness between elderly and younger adult patients.
4 Contraindications
Fexinidazole Tablets are contraindicated in:
- Patients with known hypersensitivity to Fexinidazole Tablets and/or any nitroimidazole-class drugs (e.g., metronidazole, tinidazole).
- Patients with severe hepatic impairment [see Warnings and Precautions (5.5), Adverse Reactions (6.1), and Use in Specific Populations (8.7)].
- Patients with Cockayne syndrome. Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole, another nitroimidazole drug, structurally related to fexinidazole, in patients with Cockayne syndrome [see Adverse Reactions (6.2)].
6 Adverse Reactions
The following serious and otherwise important adverse reactions are discussed in greater detail in other sections of labeling:
- Decreased Efficacy in Severe Human African Trypanosomiasis Caused by Trypanosoma brucei gambiense [see Warnings and Precautions (5.1)]
- QT Interval Prolongation [see Warnings and Precautions (5.2)]
- Neuropsychiatric Adverse Reactions [see Warnings and Precautions (5.3)]
- Neutropenia [see Warnings and Precautions (5.4)]
- Potential for Hepatotoxicity [see Warnings and Precautions (5.5)]
7 Drug Interactions
8.6 Renal Impairment
No dosage adjustment is needed for patients with mild to moderate renal impairment with estimated glomerular filtration rates (eGFR) from 30 mL/min/1.73 m2 to less than or equal to 89 mL/min/1.73 m2 [see Clinical Pharmacology (12.3)]. The pharmacokinetics of fexinidazole in patients with severe renal impairment (eGFR less than 30 mL/min/1.73 m2) is unknown. Avoid the use of Fexinidazole Tablets in patients with severe renal impairment.
12.3 Pharmacokinetics
The pharmacokinetics (PK) of fexinidazole and its two pharmacologically active M1 (sulfoxide) and M2 (sulfone) metabolites following administration of the recommended adult dosage regimen of Fexinidazole Tablets in 12 healthy adult male subjects under fed conditions are presented in Table 5.
| Fexinidazole | M1 | M2 | ||
|---|---|---|---|---|
| Cmax = maximum plasma concentration; Tmax = time to maximum concentration; CSF = cerebrospinal fluid; CYP = cytochrome P450 enzymes; AUC0–24 hours = area under the plasma concentration-time curve from time zero to 24 hours, AUC0–t = area under the plasma concentration-time curve from time zero to the last timepoint with measurable analyte concentrations; DBS = dried blood spot | ||||
| NA: Not available or not applicable | ||||
| Mean (±SD) Cmax mcg/mL | Day 1 | 1.6 (±0.4) | 8.1 (±2.2) | 7.5 (±3.3) |
| Day 4 | 0.8 (±0.3) | 8.0 (±2.3) | 19.6 (±5.4) | |
| Day 10 | 0.5 (±0.2) | 5.9 (±2.1) | 12.5 (±3.5) | |
| Mean (±SD) AUC(0–24 hours) mcg∙h/mL | Day 1 | 14.3 (±2.6) | 102.3 (±28.5) | 110.1 (±41.1) |
| Day 4 | 11.6 (±2.2) | 127.9 (±49.2) | 391.5 (±126.7) | |
| Day 10 | 7.0 (±2.5) | 84.2 (±36.3) | 252.4 (±73.6) | |
| Absorption | ||||
| Median Tmax (Range) on Day 4, hours | 4 (0–9) | 4 (0–6) | 6 (0–24) | |
|
Effect of Food
The effect of food following administration of a single 1,200 mg dose with a meal containing approximately 963 Kcal with 62% of total calories from fat, 17% from protein, and 21% from carbohydrate (n=12).
|
The AUC of fexinidazole, M1, and M2 were approximately 4 to 5-fold higher following administration with food compared to the fasted state. | |||
| Distribution | ||||
| Apparent Volume of Distribution on Day 4, L | 3222 (±1199) | NA | NA | |
| Plasma Protein Binding | 98% | 41% | 57% | |
| Mean (Range) CSF concentrations at 24 hours after the last fexinidazole dose on Day 10, mcg/mL From study in patient with HAT.
|
NA | 1.39 (0–4.5) | 6.45 (0.3–14.9) | |
| Mean (Range) CSF to DBS Ratios | NA | 0.53 (0.1–2.2) | 0.36 (0.1–0.8) | |
| Elimination | ||||
| Mean (±SD) Day 10 Half-life, hours | 15 (±6) | 16 (±6) | 23 (±4) | |
| Mean (±SD) Apparent Clearance on Day 4, L/hour | 161 (±37) | NA | NA | |
| Metabolism | ||||
| Fexinidazole |
|
|||
| Active Metabolites |
|
|||
| Excretion | ||||
| Urine | Less than 3.2% of a given dose of Fexinidazole Tablets, primarily as M1 and M2 metabolites |
2.2 Recommended Dosage
Administer Fexinidazole Tablets, orally, once daily for a total of 10 days (loading dose plus maintenance dose) with food each day at about the same time of the day. Do not break or crush Fexinidazole Tablets.
The recommended dosage of Fexinidazole Tablets for patients 6 years of age and older is according to body weight as described in Table 1 below.
| Body weight | Type of Dose | Recommended Administer Fexinidazole Tablets once daily with food each day at about the same time of the day Daily Dose |
Number of 600 mg Fexinidazole Tablets Daily | Duration of Treatment |
|---|---|---|---|---|
| Greater than or equal to 35 kg | Loading dose | 1,800 mg | 3 | 4 days |
| Maintenance dose | 1,200 mg | 2 | 6 days | |
| Greater than or equal to 20 kg to less than 35 kg | Loading dose | 1,200 mg | 2 | 4 days |
| Maintenance dose | 600 mg | 1 | 6 days |
8.7 Hepatic Impairment
No dosage adjustment of Fexinidazole Tablets is recommended for patients with mild hepatic impairment (Child-Pugh Class A). Moderate hepatic impairment (Child-Pugh Class B) increases fexinidazole and its active metabolite M1 exposures, which may increase the risk of adverse reactions [see Clinical Pharmacology (12.3)]. Use of Fexinidazole Tablets is contraindicated in patients with severe hepatic impairment [see Contraindications (4) and Warnings and Precautions (5.5)].
1 Indications and Usage
Fexinidazole Tablets are indicated for the treatment of both the first-stage (hemolymphatic) and second-stage (meningoencephalitic) human African trypanosomiasis (HAT) due to Trypanosoma brucei gambiense in patients 6 years of age and older and weighing at least 20 kg.
12.1 Mechanism of Action
Fexinidazole is an antiprotozoal drug [see Microbiology (12.4)].
5 Warnings and Precautions
- Decreased Efficacy in Severe Human African Trypanosomiasis caused by Trypanosoma brucei gambiense. (1, 5.1)
- QT Interval Prolongation: Prolongation of the QT interval due to Fexinidazole Tablets occurs in a concentration-dependent manner. Avoid use in patients with known prolongation, proarrhythmic conditions, and concomitant use with drugs that prolong the QT interval, those that block cardiac potassium channels, and/or those that induce bradycardia, or are inducers of hepatic CYP450. (5.2, 7.1, 7.2, 12.2)
- Neuropsychiatric Adverse Reactions: Adverse reactions such as agitation, anxiety, abnormal behavior, depression, suicidal ideation, nightmares, hallucination, and personality change have been observed during therapy. Inform patients and their caregivers of the risk. Consider alternative therapy or increased monitoring of the patient, including hospitalization in patients with psychiatric disorders, or if these adverse reactions occur. (5.3)
- Neutropenia: Avoid concomitant use of drugs which may cause neutropenia and monitor leukocyte count periodically. Monitor patients with neutropenia for symptoms or signs of infection. (5.4, 6.1)
- Potential for Hepatotoxicity: Evaluate liver-related laboratory tests at the start and during treatment. (4, 5.5)
- Risk of Disulfiram-like Reactions Due to Concomitant Use with Alcohol: Nitroimidazole-class drugs may cause a disulfiram-like reaction in patients who concurrently consume alcohol. Advise patients to avoid consumption of alcohol during treatment with and for at least 48 hours after completing therapy. (2.1, 5.6)
- Risk of Psychotic Reactions Due to Concomitant Use with Disulfiram: Psychotic reactions have been reported in patients concurrently taking disulfiram and nitroimidazole-class drugs. Avoid use in patients who have taken disulfiram within the last two weeks. (5.7).
2 Dosage and Administration
Administer Fexinidazole Tablets once daily with food each day at about the same time of the day. Do not break or crush tablets. (2.1, 2.2)
| Body Weight | Type of Dose | Daily Dose | Number of Tablets | Duration of Treatment |
|---|---|---|---|---|
| Greater than or equal to 35 kg | Loading dose | 1,800 mg | 3 | 4 days |
| Maintenance dose | 1,200 mg | 2 | 6 days | |
| Greater than or equal to 20 kg to less than 35 kg | Loading dose | 1,200 mg | 2 | 4 days |
| Maintenance dose | 600 mg | 1 | 6 days |
3 Dosage Forms and Strengths
Tablets: 600 mg of fexinidazole per tablet as pale-yellow, round, biconvex tablets debossed with "4512" on one side.
5.2 Qt Interval Prolongation
Fexinidazole Tablets have been shown to prolong the QT interval in a concentration-dependent manner [see Clinical Pharmacology (12.2)]. Treatment with Fexinidazole Tablets caused an average increase of 19 msec in the QTcF interval. In clinical trials in HAT patients, three (<1%) patients in the fexinidazole group had a QTcF value of >500 ms versus none in the nifurtimox-eflornithine combination therapy (NECT) group.
Avoid use of Fexinidazole Tablets in patients who have:
- QTcF interval greater than 470 msec
- A history of torsade de pointes, congenital long QT syndrome, cardiac arrhythmias, uncompensated heart failure, or family history of sudden death
- Uncorrected hypokalemia
Avoid concomitant administration of Fexinidazole Tablets with other drugs that are known to prolong the QT interval, those that block cardiac potassium channels, and/or those that induce bradycardia [see Drug Interactions (7.1)].
Avoid concomitant administration of Fexinidazole Tablets with drugs that are inducers of hepatic CYP450 as these drugs may significantly increase plasma concentrations of fexinidazole's active metabolites: fexinidazole sulfoxide (M1) and fexinidazole sulfone (M2). M2 plasma concentrations have been associated with increased QT prolongation risks [see Drug Interactions (7.2)].
If patients are, or need to be, treated with drugs known to prolong QTcF interval or to induce bradycardia either do not initiate therapy with Fexinidazole Tablets until such drugs are eliminated from the body (allow a washout period of 5 half-lives for such other drugs), or do not start such drugs until fexinidazole is eliminated from the body (allow a washout period of 7 days for Fexinidazole Tablets).
6.2 Postmarketing Experience
The following adverse reaction has been identified and reported during post-approval use of other nitroimidazole agents. Because the reports of this reaction are voluntary and the population is of uncertain size, it is not always possible to reliably estimate the frequency of the reaction or establish a causal relationship to drug exposure.
Metronidazole, Another Nitroimidazole Product, Structurally Related to Fexinidazole: Cases of severe irreversible hepatotoxicity/acute liver failure, including cases with fatal outcomes with very rapid onset after initiation of systemic use of metronidazole, another nitroimidazole agent structurally related to fexinidazole, have been reported in patients with Cockayne syndrome (latency from drug start to signs of liver failure as short as 2 days) [see Contraindications (4)].
8 Use in Specific Populations
- Avoid use in patients with severe renal impairment. (8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Fexinidazole Tablets were evaluated for the treatment of HAT due to T. brucei gambiense in three clinical trials, of which one was comparative and two were noncomparative. Trial 1 compared the safety of Fexinidazole Tablets to nifurtimox-eflornithine combination therapy (NECT) in second stage, meningoencephalitic HAT (N=394). Trial 2 enrolled patients with stage 1 hemolymphatic and early stage 2 HAT (N=230), and Trial 3 assessed the safety of fexinidazole in pediatric patients aged 6 years or older with any stage HAT (N=125).
The three trials were primarily conducted in the Democratic Republic of Congo (DRC) and a total of 749 patients received at least one dose of study medication. The patients ranged from 6 to 73 years of age, and 11 were more than 65 years old. Trials 1 and 3 enrolled more males than females (61% and 54% were males, respectively), while the gender distribution was equally balanced in Trial 2. The mean BMI ranged from 16.1 to 19.3 kg/m2 across trials which was consistent with the nutritional status of the study population. Patients with AST/ALT >2 times the upper limit of normal or total bilirubin >1.5 the upper limit of normal were excluded from the trials.
Trial 1 included 264 patients in the fexinidazole treatment arm and 130 patients in the NECT treatment arm. The patients were followed for up to 24 months from the completion of treatment.
5.5 Potential for Hepatotoxicity
Elevations in liver transaminases occurred in less than two percent of patients receiving Fexinidazole Tablets for the treatment of HAT [see Adverse reactions (6.1) and Overdosage (10)]. Evaluate liver-related laboratory tests at the start [see Contraindications (4)] and during treatment with Fexinidazole Tablets. Monitor patients who develop abnormal liver-related laboratory tests during treatment with Fexinidazole Tablets.
16 How Supplied/storage and Handling
Fexinidazole 600 mg tablets are pale-yellow, round, biconvex tablets debossed with "4512" on one side.
Fexinidazole 600 mg tablets are supplied as:
- NDC 0024-4512-24, child-resistant blister pack of 24 tablets (10-day dose pack) for adults and pediatric patients weighing 35 kg or more.
- NDC 0024-4512-14, child-resistant blister pack of 14 tablets (10-day dose pack) for pediatric patients older than 6 years weighing 20 kg to less than 35 kg.
7.2 Pharmacokinetic Drug Interactions
| Drugs Metabolized by Cytochrome P450 (CYP) 3A4/5 | |
| Examples (not fully inclusive): | Lovastatin, simvastatin, nisoldipine, saquinavir, midazolam, certain hormonal contraceptives (e.g., birth control pills, skin patches, implant) |
| Clinical Impact | Fexinidazole is considered a moderate CYP3A4/5 inducer [see Clinical Pharmacology (12.3)]. Co-administration of Fexinidazole Tablets and CYP3A4/5 substrates may reduce the systemic exposures of CYP3A4/5 substrate drug(s), which may lead to reduced efficacy [see Clinical Pharmacology (12.3)]. |
| Prevention or Management | Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for lack of efficacy of sensitive substrate drugs. If concomitant use of oral contraceptives cannot be avoided, an alternative contraceptive that is not affected by enzyme inducers (e.g., intrauterine system) or additional nonhormonal contraception (e.g., condoms) is recommended during treatment with Fexinidazole Tablets and for at least 5 days after the last dose. |
| Drugs Metabolized by CYP1A2 or CYP2C19 | |
| Examples (not fully inclusive): | CYP1A2: duloxetine, tacrine, tizanidine, theophylline CYP2C19: lansoprazole, mephenytoin, diazepam |
| Clinical Impact | Increased risk for adverse reactions associated with increased concentrations of the drug due to inhibition of either CYP1A2 or CYP2C19 by fexinidazole [see Clinical Pharmacology (12.3)]. |
| Prevention or Management | Monitor for adverse reactions associated with these drugs when used concomitantly with Fexinidazole Tablets. |
| Drugs Metabolized by CYP2B6 | |
| Examples (not fully inclusive): | Bupropion, efavirenz |
| Clinical Impact | Increased risk for the lack of efficacy associated with decreased plasma concentrations of the drug due to induction of CYP2B6 by fexinidazole and Metabolite M1 [see Clinical Pharmacology (12.3)]. |
| Prevention or Management | Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for lack of efficacy of these drugs. |
| Drugs Metabolized by uridine 5'-diphospho--glucuronosyl transferase (UGT) | |
| Examples (not fully inclusive): | Zidovudine, bictegravir, cabotegravir, dolutegravir, bexagliflozin, canagliflozin, sotagliflozin, deferasirox, lamotrigine, selexipag |
| Clinical Impact | Fexinidazole metabolites have the potential to be UGT inducers [see Clinical Pharmacology (12.3)]. Co-administration of Fexinidazole Tablets and UGT substrates may reduce the systemic exposures of UGT substrate drug(s), which may lead to reduced efficacy. |
| Prevention or Management | When fexinidazole is concomitantly administered with UGT substrates, monitor for lack of efficacy of UGT substrate drugs. |
| Drugs Substrates of OCT2, OAT1, OAT3, MATE1, and MATE2-K Transporters | |
| Examples (not fully inclusive): | Metformin, dofetilide, adefovir, cefaclor, furosemide |
| Clinical Impact | Increased risk for adverse reactions associated with increased concentrations of the drug due to inhibition of these transporters by fexinidazole [see Clinical Pharmacology (12.3)]. |
| Prevention or Management | Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for adverse reactions associated with these drugs. |
| CYP450 Inducers | |
| Examples (not fully inclusive): | Rifampin, phenytoin, St. John's wort, carbamazepine |
| Clinical Impact | Increased risk for adverse reactions associated with increased systemic exposure to the M1 and M2 metabolites of fexinidazole. M2 plasma concentrations have been associated with the increased risk of QT interval prolongation. |
| Prevention or Management | Avoid concomitant use with Fexinidazole Tablets [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. |
| CYP450 Inhibitors | |
| Examples (not fully inclusive): | Clarithromycin, itraconazole, voriconazole, erythromycin, fluconazole |
| Clinical Impact | Multiple CYP450 enzymes are involved in the metabolism of fexinidazole to its pharmacologically active M1 and M2 metabolites. Although no clinical drug interaction studies were performed with CYP450 inhibitors, the formation of the M1 and M2 metabolites may be decreased. |
| Prevention or Management | Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for lack of efficacy of fexinidazole due to potential for decreased plasma concentrations of the M1 and M2 metabolites [see Clinical Pharmacology (12.3)]. |
5.3 Neuropsychiatric Adverse Reactions
Adult patients treated with Fexinidazole Tablets reported a higher percentage of Central Nervous System (CNS) and psychiatric-related adverse reactions than those treated with nifurtimox eflornithine combination therapy (NECT) in a clinical trial [see Adverse Reactions (6.1)]. Increased incidence in insomnia, headache, and tremor was noted in the patients treated with Fexinidazole Tablets compared to NECT. In the same trial, adverse reactions representing mood changes and psychiatric disorders (such as agitation, anxiety, abnormal behavior, depression, nightmares, hallucination, and personality change) were more common in the patients treated with Fexinidazole Tablets compared to the NECT arm. Suicidal ideation has also been observed with Fexinidazole Tablets [see Adverse Reactions (6.1)]. Healthcare providers should inform patients and their caregivers of the risk for neuropsychiatric adverse reactions during treatment with Fexinidazole Tablets. In patients with current or a history of psychiatric disorders, or should such adverse reactions occur, healthcare providers should consider alternative therapy or increased monitoring of the patient, including hospitalization.
2.1 Important Administration Instructions
- Patients should be closely followed by their healthcare provider during treatment with Fexinidazole Tablets.
- Fexinidazole Tablets must be administered with food [see Dosage and Administration (2.2)].
- Avoid consumption of alcoholic beverages during treatment with Fexinidazole Tablets and for at least 48 hours after completing therapy [see Warnings and Precautions (5.6)].
- If a first event of vomiting occurs after receiving Fexinidazole Tablets, do not re-dose. Administer the next dose the following day using the recommended treatment schedule [see Adverse Reactions (6.1)].
- If a scheduled dose is missed (not taken on the assigned day), normal dosing should resume the following day until the full course (10 days) of treatment has been completed. The clinical consequences of multiple missed doses of Fexinidazole Tablets are not known.
Principal Display Panel 600 Mg Tablet Dose Pack
Opening Instructions:
Press and hold down
button while pulling out
medication card.
NDC 0024-4512-24
Rx only
Fexinidazole
Tablets
600 mg per tablet
10-Day Dose Pack
for adults and children
weighing 35 kg or more
DISPENSE ONLY IN ORIGINAL PACKAGE
24 tablets
SANOFI
5.7 Risk of Psychotic Reactions Due to Concomitant Use With Disulfiram
Psychotic reactions have been reported in patients who were concurrently taking disulfiram and nitroimidazole drugs. Avoid use of Fexinidazole Tablets in patients who have taken disulfiram within the last two weeks.
5.6 Risk of Disulfiram Like Reaction Due to Concomitant Use With Alcohol
Nitroimidazole-class drugs may cause a disulfiram-like reaction characterized by flushing, rash, weakness, abdominal cramps, nausea, vomiting and headache in patients who concurrently consume alcohol. Advise patients to avoid consumption of alcohol during treatment with Fexinidazole Tablets and for at least 48 hours after completing therapy [see Dosage and Administration (2.1)].
5.1 Decreased Efficacy in Severe Human African Trypanosomiasis Caused By Trypanosoma Brucei Gambiense
Decreased efficacy was observed in patients treated with Fexinidazole Tablets as compared to nifurtimox-eflornithine combination therapy (NECT)-treated patients in a randomized, comparative open-label study in the subgroup of patients with severe second stage disease, as defined by cerebrospinal fluid white blood cell count (CSF-WBC) >100 cells/µL at baseline [see Clinical Studies (14)]. The 18-month success rate in this subgroup of patients with severe second stage disease was 86.9% with Fexinidazole Tablets compared to 98.7% with NECT with a difference of -11.8%, 95% confidence interval (CI) (-18.3%, -2.1%). All-cause mortality was higher in patients with severe disease treated with Fexinidazole Tablets than in patients treated with NECT through 24 months (7/160 [4.4%] vs 0/78 [0%], treatment difference 4.4%, 95% CI [-0.9%, 8.9%]).
Patients with severe second stage HAT (CSF-WBC >100 cells/µL) due to T. brucei gambiense disease should only be treated with Fexinidazole Tablets if there are no other available treatment options.
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
Due to the decreased efficacy observed in patients with severe second stage HAT (cerebrospinal fluid white blood cell count (CSF-WBC) >100 cells/µL) due to T. brucei gambiense disease, Fexinidazole Tablets should only be used in these patients if there are no other available treatment options [see Warnings and Precautions (5.1)].
Section 43683-2 (43683-2)
| Contraindications (4) | 9/2024 |
Section 44425-7 (44425-7)
Store below 30°C (86°F). Store in the original package in order to protect from light and moisture.
10 Overdosage (10 OVERDOSAGE)
Randomized, controlled clinical studies were conducted in normal adult male subjects who were administered single or multiple oral doses of fexinidazole of up to 3,600 mg daily for 14 days (not an approved dose). The subjects experienced adverse reactions of increased transaminases, vomiting and panic attack [see Adverse Reactions (6.1)].
Reported symptoms of overdosage in a pediatric HAT patient following ingestion of a higher than recommended dosing regimen in Trial 3 included vomiting over the first 5 days of treatment and increased potassium and decreased calcium levels from Day 11 to Week 9.
There is no specific antidote for Fexinidazole Tablets. Treatment should be supportive with appropriate monitoring.
11 Description (11 DESCRIPTION)
Fexinidazole Tablets contain fexinidazole, a nitroimidazole antimicrobial drug for oral use.
The chemical name of fexinidazole is 1-methyl-2-{[4-(methylthio)phenoxy]methyl}-5-nitro-1H-imidazole.
Its molecular formula is C12H13N3O3S and the molecular weight is 279.3 g/mol. The structural formula is:
Fexinidazole is a yellow powder. It is practically insoluble in water, sparingly soluble in acetone and acetonitrile, very slightly soluble in ethanol and slightly soluble in methanol.
Fexinidazole 600 mg Tablets contain the active ingredient fexinidazole and the following inactive ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, povidone, and sodium lauryl sulfate.
5.4 Neutropenia
Neutropenia (absolute neutrophil count less than 1,000 cells/mm3) has been reported in patients receiving Fexinidazole Tablets [see Adverse Reactions (6.1)]. In Trial 1, the adverse reaction occurred in patients with a baseline absolute neutrophil count of less than 5,000 cells/mm3. Avoid concomitant use of drugs which may cause neutropenia and monitor leukocyte count periodically. Carefully monitor patients with neutropenia for fever or other symptoms or signs of infection and treat promptly if such symptoms or signs occur.
8.4 Pediatric Use
The safety and effectiveness of Fexinidazole Tablets for the treatment of both the first-stage (hemolymphatic) and second-stage (meningoencephalitic) HAT due to Trypanosoma brucei gambiense have been established in pediatric patients aged 6 years and older and weighing at least 20 kg. Use of Fexinidazole Tablets for this indication is supported by evidence from an adequate and well-controlled trial in adults with additional efficacy, pharmacokinetic and safety data in pediatric patients aged 6 years and older [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Pediatric patients may be more sensitive to vomiting. The safety profile for Fexinidazole Tablets in pediatric patients was generally similar to that of adult patients with the exception of more frequent vomiting within 2 hours of administration of Fexinidazole Tablets. Vomiting did not result in permanent treatment discontinuation [see Adverse Reactions (6.1)].
The safety and efficacy of Fexinidazole Tablets have not been established in pediatric patients younger than 6 years old and/or less than 20 kg in body weight.
8.5 Geriatric Use
Of the 619 subjects in the three clinical trials treated with Fexinidazole Tablets for HAT, there were 11 subjects who were 65 years of age or older, and no subjects greater than 75 years of age. There were an insufficient number of elderly subjects to detect differences in safety and/or effectiveness between elderly and younger adult patients.
4 Contraindications (4 CONTRAINDICATIONS)
Fexinidazole Tablets are contraindicated in:
- Patients with known hypersensitivity to Fexinidazole Tablets and/or any nitroimidazole-class drugs (e.g., metronidazole, tinidazole).
- Patients with severe hepatic impairment [see Warnings and Precautions (5.5), Adverse Reactions (6.1), and Use in Specific Populations (8.7)].
- Patients with Cockayne syndrome. Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole, another nitroimidazole drug, structurally related to fexinidazole, in patients with Cockayne syndrome [see Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious and otherwise important adverse reactions are discussed in greater detail in other sections of labeling:
- Decreased Efficacy in Severe Human African Trypanosomiasis Caused by Trypanosoma brucei gambiense [see Warnings and Precautions (5.1)]
- QT Interval Prolongation [see Warnings and Precautions (5.2)]
- Neuropsychiatric Adverse Reactions [see Warnings and Precautions (5.3)]
- Neutropenia [see Warnings and Precautions (5.4)]
- Potential for Hepatotoxicity [see Warnings and Precautions (5.5)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.6 Renal Impairment
No dosage adjustment is needed for patients with mild to moderate renal impairment with estimated glomerular filtration rates (eGFR) from 30 mL/min/1.73 m2 to less than or equal to 89 mL/min/1.73 m2 [see Clinical Pharmacology (12.3)]. The pharmacokinetics of fexinidazole in patients with severe renal impairment (eGFR less than 30 mL/min/1.73 m2) is unknown. Avoid the use of Fexinidazole Tablets in patients with severe renal impairment.
12.3 Pharmacokinetics
The pharmacokinetics (PK) of fexinidazole and its two pharmacologically active M1 (sulfoxide) and M2 (sulfone) metabolites following administration of the recommended adult dosage regimen of Fexinidazole Tablets in 12 healthy adult male subjects under fed conditions are presented in Table 5.
| Fexinidazole | M1 | M2 | ||
|---|---|---|---|---|
| Cmax = maximum plasma concentration; Tmax = time to maximum concentration; CSF = cerebrospinal fluid; CYP = cytochrome P450 enzymes; AUC0–24 hours = area under the plasma concentration-time curve from time zero to 24 hours, AUC0–t = area under the plasma concentration-time curve from time zero to the last timepoint with measurable analyte concentrations; DBS = dried blood spot | ||||
| NA: Not available or not applicable | ||||
| Mean (±SD) Cmax mcg/mL | Day 1 | 1.6 (±0.4) | 8.1 (±2.2) | 7.5 (±3.3) |
| Day 4 | 0.8 (±0.3) | 8.0 (±2.3) | 19.6 (±5.4) | |
| Day 10 | 0.5 (±0.2) | 5.9 (±2.1) | 12.5 (±3.5) | |
| Mean (±SD) AUC(0–24 hours) mcg∙h/mL | Day 1 | 14.3 (±2.6) | 102.3 (±28.5) | 110.1 (±41.1) |
| Day 4 | 11.6 (±2.2) | 127.9 (±49.2) | 391.5 (±126.7) | |
| Day 10 | 7.0 (±2.5) | 84.2 (±36.3) | 252.4 (±73.6) | |
| Absorption | ||||
| Median Tmax (Range) on Day 4, hours | 4 (0–9) | 4 (0–6) | 6 (0–24) | |
|
Effect of Food
The effect of food following administration of a single 1,200 mg dose with a meal containing approximately 963 Kcal with 62% of total calories from fat, 17% from protein, and 21% from carbohydrate (n=12).
|
The AUC of fexinidazole, M1, and M2 were approximately 4 to 5-fold higher following administration with food compared to the fasted state. | |||
| Distribution | ||||
| Apparent Volume of Distribution on Day 4, L | 3222 (±1199) | NA | NA | |
| Plasma Protein Binding | 98% | 41% | 57% | |
| Mean (Range) CSF concentrations at 24 hours after the last fexinidazole dose on Day 10, mcg/mL From study in patient with HAT.
|
NA | 1.39 (0–4.5) | 6.45 (0.3–14.9) | |
| Mean (Range) CSF to DBS Ratios | NA | 0.53 (0.1–2.2) | 0.36 (0.1–0.8) | |
| Elimination | ||||
| Mean (±SD) Day 10 Half-life, hours | 15 (±6) | 16 (±6) | 23 (±4) | |
| Mean (±SD) Apparent Clearance on Day 4, L/hour | 161 (±37) | NA | NA | |
| Metabolism | ||||
| Fexinidazole |
|
|||
| Active Metabolites |
|
|||
| Excretion | ||||
| Urine | Less than 3.2% of a given dose of Fexinidazole Tablets, primarily as M1 and M2 metabolites |
2.2 Recommended Dosage
Administer Fexinidazole Tablets, orally, once daily for a total of 10 days (loading dose plus maintenance dose) with food each day at about the same time of the day. Do not break or crush Fexinidazole Tablets.
The recommended dosage of Fexinidazole Tablets for patients 6 years of age and older is according to body weight as described in Table 1 below.
| Body weight | Type of Dose | Recommended Administer Fexinidazole Tablets once daily with food each day at about the same time of the day Daily Dose |
Number of 600 mg Fexinidazole Tablets Daily | Duration of Treatment |
|---|---|---|---|---|
| Greater than or equal to 35 kg | Loading dose | 1,800 mg | 3 | 4 days |
| Maintenance dose | 1,200 mg | 2 | 6 days | |
| Greater than or equal to 20 kg to less than 35 kg | Loading dose | 1,200 mg | 2 | 4 days |
| Maintenance dose | 600 mg | 1 | 6 days |
8.7 Hepatic Impairment
No dosage adjustment of Fexinidazole Tablets is recommended for patients with mild hepatic impairment (Child-Pugh Class A). Moderate hepatic impairment (Child-Pugh Class B) increases fexinidazole and its active metabolite M1 exposures, which may increase the risk of adverse reactions [see Clinical Pharmacology (12.3)]. Use of Fexinidazole Tablets is contraindicated in patients with severe hepatic impairment [see Contraindications (4) and Warnings and Precautions (5.5)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Fexinidazole Tablets are indicated for the treatment of both the first-stage (hemolymphatic) and second-stage (meningoencephalitic) human African trypanosomiasis (HAT) due to Trypanosoma brucei gambiense in patients 6 years of age and older and weighing at least 20 kg.
12.1 Mechanism of Action
Fexinidazole is an antiprotozoal drug [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Decreased Efficacy in Severe Human African Trypanosomiasis caused by Trypanosoma brucei gambiense. (1, 5.1)
- QT Interval Prolongation: Prolongation of the QT interval due to Fexinidazole Tablets occurs in a concentration-dependent manner. Avoid use in patients with known prolongation, proarrhythmic conditions, and concomitant use with drugs that prolong the QT interval, those that block cardiac potassium channels, and/or those that induce bradycardia, or are inducers of hepatic CYP450. (5.2, 7.1, 7.2, 12.2)
- Neuropsychiatric Adverse Reactions: Adverse reactions such as agitation, anxiety, abnormal behavior, depression, suicidal ideation, nightmares, hallucination, and personality change have been observed during therapy. Inform patients and their caregivers of the risk. Consider alternative therapy or increased monitoring of the patient, including hospitalization in patients with psychiatric disorders, or if these adverse reactions occur. (5.3)
- Neutropenia: Avoid concomitant use of drugs which may cause neutropenia and monitor leukocyte count periodically. Monitor patients with neutropenia for symptoms or signs of infection. (5.4, 6.1)
- Potential for Hepatotoxicity: Evaluate liver-related laboratory tests at the start and during treatment. (4, 5.5)
- Risk of Disulfiram-like Reactions Due to Concomitant Use with Alcohol: Nitroimidazole-class drugs may cause a disulfiram-like reaction in patients who concurrently consume alcohol. Advise patients to avoid consumption of alcohol during treatment with and for at least 48 hours after completing therapy. (2.1, 5.6)
- Risk of Psychotic Reactions Due to Concomitant Use with Disulfiram: Psychotic reactions have been reported in patients concurrently taking disulfiram and nitroimidazole-class drugs. Avoid use in patients who have taken disulfiram within the last two weeks. (5.7).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Administer Fexinidazole Tablets once daily with food each day at about the same time of the day. Do not break or crush tablets. (2.1, 2.2)
| Body Weight | Type of Dose | Daily Dose | Number of Tablets | Duration of Treatment |
|---|---|---|---|---|
| Greater than or equal to 35 kg | Loading dose | 1,800 mg | 3 | 4 days |
| Maintenance dose | 1,200 mg | 2 | 6 days | |
| Greater than or equal to 20 kg to less than 35 kg | Loading dose | 1,200 mg | 2 | 4 days |
| Maintenance dose | 600 mg | 1 | 6 days |
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 600 mg of fexinidazole per tablet as pale-yellow, round, biconvex tablets debossed with "4512" on one side.
5.2 Qt Interval Prolongation (5.2 QT Interval Prolongation)
Fexinidazole Tablets have been shown to prolong the QT interval in a concentration-dependent manner [see Clinical Pharmacology (12.2)]. Treatment with Fexinidazole Tablets caused an average increase of 19 msec in the QTcF interval. In clinical trials in HAT patients, three (<1%) patients in the fexinidazole group had a QTcF value of >500 ms versus none in the nifurtimox-eflornithine combination therapy (NECT) group.
Avoid use of Fexinidazole Tablets in patients who have:
- QTcF interval greater than 470 msec
- A history of torsade de pointes, congenital long QT syndrome, cardiac arrhythmias, uncompensated heart failure, or family history of sudden death
- Uncorrected hypokalemia
Avoid concomitant administration of Fexinidazole Tablets with other drugs that are known to prolong the QT interval, those that block cardiac potassium channels, and/or those that induce bradycardia [see Drug Interactions (7.1)].
Avoid concomitant administration of Fexinidazole Tablets with drugs that are inducers of hepatic CYP450 as these drugs may significantly increase plasma concentrations of fexinidazole's active metabolites: fexinidazole sulfoxide (M1) and fexinidazole sulfone (M2). M2 plasma concentrations have been associated with increased QT prolongation risks [see Drug Interactions (7.2)].
If patients are, or need to be, treated with drugs known to prolong QTcF interval or to induce bradycardia either do not initiate therapy with Fexinidazole Tablets until such drugs are eliminated from the body (allow a washout period of 5 half-lives for such other drugs), or do not start such drugs until fexinidazole is eliminated from the body (allow a washout period of 7 days for Fexinidazole Tablets).
6.2 Postmarketing Experience
The following adverse reaction has been identified and reported during post-approval use of other nitroimidazole agents. Because the reports of this reaction are voluntary and the population is of uncertain size, it is not always possible to reliably estimate the frequency of the reaction or establish a causal relationship to drug exposure.
Metronidazole, Another Nitroimidazole Product, Structurally Related to Fexinidazole: Cases of severe irreversible hepatotoxicity/acute liver failure, including cases with fatal outcomes with very rapid onset after initiation of systemic use of metronidazole, another nitroimidazole agent structurally related to fexinidazole, have been reported in patients with Cockayne syndrome (latency from drug start to signs of liver failure as short as 2 days) [see Contraindications (4)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Avoid use in patients with severe renal impairment. (8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Fexinidazole Tablets were evaluated for the treatment of HAT due to T. brucei gambiense in three clinical trials, of which one was comparative and two were noncomparative. Trial 1 compared the safety of Fexinidazole Tablets to nifurtimox-eflornithine combination therapy (NECT) in second stage, meningoencephalitic HAT (N=394). Trial 2 enrolled patients with stage 1 hemolymphatic and early stage 2 HAT (N=230), and Trial 3 assessed the safety of fexinidazole in pediatric patients aged 6 years or older with any stage HAT (N=125).
The three trials were primarily conducted in the Democratic Republic of Congo (DRC) and a total of 749 patients received at least one dose of study medication. The patients ranged from 6 to 73 years of age, and 11 were more than 65 years old. Trials 1 and 3 enrolled more males than females (61% and 54% were males, respectively), while the gender distribution was equally balanced in Trial 2. The mean BMI ranged from 16.1 to 19.3 kg/m2 across trials which was consistent with the nutritional status of the study population. Patients with AST/ALT >2 times the upper limit of normal or total bilirubin >1.5 the upper limit of normal were excluded from the trials.
Trial 1 included 264 patients in the fexinidazole treatment arm and 130 patients in the NECT treatment arm. The patients were followed for up to 24 months from the completion of treatment.
5.5 Potential for Hepatotoxicity
Elevations in liver transaminases occurred in less than two percent of patients receiving Fexinidazole Tablets for the treatment of HAT [see Adverse reactions (6.1) and Overdosage (10)]. Evaluate liver-related laboratory tests at the start [see Contraindications (4)] and during treatment with Fexinidazole Tablets. Monitor patients who develop abnormal liver-related laboratory tests during treatment with Fexinidazole Tablets.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Fexinidazole 600 mg tablets are pale-yellow, round, biconvex tablets debossed with "4512" on one side.
Fexinidazole 600 mg tablets are supplied as:
- NDC 0024-4512-24, child-resistant blister pack of 24 tablets (10-day dose pack) for adults and pediatric patients weighing 35 kg or more.
- NDC 0024-4512-14, child-resistant blister pack of 14 tablets (10-day dose pack) for pediatric patients older than 6 years weighing 20 kg to less than 35 kg.
7.2 Pharmacokinetic Drug Interactions
| Drugs Metabolized by Cytochrome P450 (CYP) 3A4/5 | |
| Examples (not fully inclusive): | Lovastatin, simvastatin, nisoldipine, saquinavir, midazolam, certain hormonal contraceptives (e.g., birth control pills, skin patches, implant) |
| Clinical Impact | Fexinidazole is considered a moderate CYP3A4/5 inducer [see Clinical Pharmacology (12.3)]. Co-administration of Fexinidazole Tablets and CYP3A4/5 substrates may reduce the systemic exposures of CYP3A4/5 substrate drug(s), which may lead to reduced efficacy [see Clinical Pharmacology (12.3)]. |
| Prevention or Management | Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for lack of efficacy of sensitive substrate drugs. If concomitant use of oral contraceptives cannot be avoided, an alternative contraceptive that is not affected by enzyme inducers (e.g., intrauterine system) or additional nonhormonal contraception (e.g., condoms) is recommended during treatment with Fexinidazole Tablets and for at least 5 days after the last dose. |
| Drugs Metabolized by CYP1A2 or CYP2C19 | |
| Examples (not fully inclusive): | CYP1A2: duloxetine, tacrine, tizanidine, theophylline CYP2C19: lansoprazole, mephenytoin, diazepam |
| Clinical Impact | Increased risk for adverse reactions associated with increased concentrations of the drug due to inhibition of either CYP1A2 or CYP2C19 by fexinidazole [see Clinical Pharmacology (12.3)]. |
| Prevention or Management | Monitor for adverse reactions associated with these drugs when used concomitantly with Fexinidazole Tablets. |
| Drugs Metabolized by CYP2B6 | |
| Examples (not fully inclusive): | Bupropion, efavirenz |
| Clinical Impact | Increased risk for the lack of efficacy associated with decreased plasma concentrations of the drug due to induction of CYP2B6 by fexinidazole and Metabolite M1 [see Clinical Pharmacology (12.3)]. |
| Prevention or Management | Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for lack of efficacy of these drugs. |
| Drugs Metabolized by uridine 5'-diphospho--glucuronosyl transferase (UGT) | |
| Examples (not fully inclusive): | Zidovudine, bictegravir, cabotegravir, dolutegravir, bexagliflozin, canagliflozin, sotagliflozin, deferasirox, lamotrigine, selexipag |
| Clinical Impact | Fexinidazole metabolites have the potential to be UGT inducers [see Clinical Pharmacology (12.3)]. Co-administration of Fexinidazole Tablets and UGT substrates may reduce the systemic exposures of UGT substrate drug(s), which may lead to reduced efficacy. |
| Prevention or Management | When fexinidazole is concomitantly administered with UGT substrates, monitor for lack of efficacy of UGT substrate drugs. |
| Drugs Substrates of OCT2, OAT1, OAT3, MATE1, and MATE2-K Transporters | |
| Examples (not fully inclusive): | Metformin, dofetilide, adefovir, cefaclor, furosemide |
| Clinical Impact | Increased risk for adverse reactions associated with increased concentrations of the drug due to inhibition of these transporters by fexinidazole [see Clinical Pharmacology (12.3)]. |
| Prevention or Management | Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for adverse reactions associated with these drugs. |
| CYP450 Inducers | |
| Examples (not fully inclusive): | Rifampin, phenytoin, St. John's wort, carbamazepine |
| Clinical Impact | Increased risk for adverse reactions associated with increased systemic exposure to the M1 and M2 metabolites of fexinidazole. M2 plasma concentrations have been associated with the increased risk of QT interval prolongation. |
| Prevention or Management | Avoid concomitant use with Fexinidazole Tablets [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. |
| CYP450 Inhibitors | |
| Examples (not fully inclusive): | Clarithromycin, itraconazole, voriconazole, erythromycin, fluconazole |
| Clinical Impact | Multiple CYP450 enzymes are involved in the metabolism of fexinidazole to its pharmacologically active M1 and M2 metabolites. Although no clinical drug interaction studies were performed with CYP450 inhibitors, the formation of the M1 and M2 metabolites may be decreased. |
| Prevention or Management | Avoid concomitant use with Fexinidazole Tablets. If coadministration cannot be avoided, monitor for lack of efficacy of fexinidazole due to potential for decreased plasma concentrations of the M1 and M2 metabolites [see Clinical Pharmacology (12.3)]. |
5.3 Neuropsychiatric Adverse Reactions
Adult patients treated with Fexinidazole Tablets reported a higher percentage of Central Nervous System (CNS) and psychiatric-related adverse reactions than those treated with nifurtimox eflornithine combination therapy (NECT) in a clinical trial [see Adverse Reactions (6.1)]. Increased incidence in insomnia, headache, and tremor was noted in the patients treated with Fexinidazole Tablets compared to NECT. In the same trial, adverse reactions representing mood changes and psychiatric disorders (such as agitation, anxiety, abnormal behavior, depression, nightmares, hallucination, and personality change) were more common in the patients treated with Fexinidazole Tablets compared to the NECT arm. Suicidal ideation has also been observed with Fexinidazole Tablets [see Adverse Reactions (6.1)]. Healthcare providers should inform patients and their caregivers of the risk for neuropsychiatric adverse reactions during treatment with Fexinidazole Tablets. In patients with current or a history of psychiatric disorders, or should such adverse reactions occur, healthcare providers should consider alternative therapy or increased monitoring of the patient, including hospitalization.
2.1 Important Administration Instructions
- Patients should be closely followed by their healthcare provider during treatment with Fexinidazole Tablets.
- Fexinidazole Tablets must be administered with food [see Dosage and Administration (2.2)].
- Avoid consumption of alcoholic beverages during treatment with Fexinidazole Tablets and for at least 48 hours after completing therapy [see Warnings and Precautions (5.6)].
- If a first event of vomiting occurs after receiving Fexinidazole Tablets, do not re-dose. Administer the next dose the following day using the recommended treatment schedule [see Adverse Reactions (6.1)].
- If a scheduled dose is missed (not taken on the assigned day), normal dosing should resume the following day until the full course (10 days) of treatment has been completed. The clinical consequences of multiple missed doses of Fexinidazole Tablets are not known.
Principal Display Panel 600 Mg Tablet Dose Pack (PRINCIPAL DISPLAY PANEL - 600 mg Tablet Dose Pack)
Opening Instructions:
Press and hold down
button while pulling out
medication card.
NDC 0024-4512-24
Rx only
Fexinidazole
Tablets
600 mg per tablet
10-Day Dose Pack
for adults and children
weighing 35 kg or more
DISPENSE ONLY IN ORIGINAL PACKAGE
24 tablets
SANOFI
5.7 Risk of Psychotic Reactions Due to Concomitant Use With Disulfiram (5.7 Risk of Psychotic Reactions Due to Concomitant Use with Disulfiram)
Psychotic reactions have been reported in patients who were concurrently taking disulfiram and nitroimidazole drugs. Avoid use of Fexinidazole Tablets in patients who have taken disulfiram within the last two weeks.
5.6 Risk of Disulfiram Like Reaction Due to Concomitant Use With Alcohol (5.6 Risk of Disulfiram-like Reaction Due to Concomitant Use with Alcohol)
Nitroimidazole-class drugs may cause a disulfiram-like reaction characterized by flushing, rash, weakness, abdominal cramps, nausea, vomiting and headache in patients who concurrently consume alcohol. Advise patients to avoid consumption of alcohol during treatment with Fexinidazole Tablets and for at least 48 hours after completing therapy [see Dosage and Administration (2.1)].
5.1 Decreased Efficacy in Severe Human African Trypanosomiasis Caused By Trypanosoma Brucei Gambiense (5.1 Decreased Efficacy in Severe Human African Trypanosomiasis Caused by Trypanosoma brucei gambiense)
Decreased efficacy was observed in patients treated with Fexinidazole Tablets as compared to nifurtimox-eflornithine combination therapy (NECT)-treated patients in a randomized, comparative open-label study in the subgroup of patients with severe second stage disease, as defined by cerebrospinal fluid white blood cell count (CSF-WBC) >100 cells/µL at baseline [see Clinical Studies (14)]. The 18-month success rate in this subgroup of patients with severe second stage disease was 86.9% with Fexinidazole Tablets compared to 98.7% with NECT with a difference of -11.8%, 95% confidence interval (CI) (-18.3%, -2.1%). All-cause mortality was higher in patients with severe disease treated with Fexinidazole Tablets than in patients treated with NECT through 24 months (7/160 [4.4%] vs 0/78 [0%], treatment difference 4.4%, 95% CI [-0.9%, 8.9%]).
Patients with severe second stage HAT (CSF-WBC >100 cells/µL) due to T. brucei gambiense disease should only be treated with Fexinidazole Tablets if there are no other available treatment options.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:00.970868 · Updated: 2026-03-14T22:26:10.810615