These Highlights Do Not Include All The Information Needed To Use Tracleer Safely And Effectively. See Full Prescribing Information For Tracleer.
749e42fb-2fe0-45dd-9268-b43bb3f4081c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Because of the risk of hepatotoxicity, TRACLEER is available only through a restricted program called the Bosentan Risk Evaluation and Mitigation Strategy (REMS). Under the Bosentan REMS, prescribers, patients, and pharmacies must enroll in the program [see Warnings and Precautions (5.2) ] .
Indications and Usage
TRACLEER is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1): in adults to improve exercise ability and to decrease clinical worsening. Studies establishing effectiveness included predominantly patients with WHO Functional Class II-IV symptoms and etiologies of idiopathic or heritable PAH (60%), PAH associated with connective tissue diseases (21%), and PAH associated with congenital heart disease with left-to-right shunts (18%) [see Clinical Studies (14.1) ] . in pediatric patients aged 3 years and older with idiopathic or congenital PAH to improve pulmonary vascular resistance (PVR), which is expected to result in an improvement in exercise ability.
Dosage and Administration
Patients older than 12 years of age: initiate at 62.5 mg orally twice daily; for patients weighing greater than 40 kg, increase to 125 mg orally twice daily after 4 weeks ( 2.2 ). Patients 12 years of age and younger: dosage is based on weight, see Table 1 ( 2.2 ). Reduce the dose and closely monitor patients developing aminotransferase elevations more than 3×Upper Limit of Normal (ULN) ( 2.4 ).
Warnings and Precautions
Fluid retention: May require intervention ( 5.4 ). Pulmonary veno-occlusive disease (PVOD): If signs of pulmonary edema occur, consider the diagnosis of associated PVOD and consider discontinuing TRACLEER ( 5.5 ). Decreased sperm counts ( 5.6 ). Decreases in hemoglobin and hematocrit: Monitor hemoglobin levels after 1 and 3 months of treatment, then every 3 months thereafter ( 5.7 ).
Contraindications
Pregnancy ( 4.1 ). Use with Cyclosporine A ( 4.2 ). Use with Glyburide ( 4.3 ). Hypersensitivity ( 4.4 ).
Adverse Reactions
The following important adverse reactions are described elsewhere in the labeling: Hepatotoxicity [see Boxed Warning , Warnings and Precautions (5.1) ] Embryo-fetal Toxicity [see Boxed Warning , Warnings and Precautions (5.3) ] Fluid Retention [see Warnings and Precautions (5.4) ]
Drug Interactions
Cytochrome P450: Coadministration of TRACLEER with drugs metabolized by CYP2C9 and CYP3A can increase exposure to TRACLEER and/or the coadministered drug ( 4.2 , 4.3 , 7.1 ). Hormonal contraceptives: TRACLEER use decreases contraceptive exposure and reduces effectiveness ( 7.2 ).
Storage and Handling
62.5 mg film-coated, round, biconvex, orange-white tablets, debossed with identification marking "62,5", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing. NDC 66215-101-06: Bottle containing 60 tablets. NDC 66215-101-03: Carton of 30 tablets in 10 blister strips of 3 tablets. 125 mg film-coated, oval, biconvex, orange-white tablets, debossed with identification marking "125", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing. NDC 66215-102-06: Bottle containing 60 tablets. NDC 66215-102-03: Carton of 30 tablets in 10 blister strips of 3 tablets. 32 mg tablets for oral suspension, pale yellow to off-white, round, bisected on one side and debossed with identification marking "32" on the other side, packaged in child-resistant Aluminum/Aluminum peel-push blisters. NDC 66215-232-56: Carton of 56 tablets for oral suspension in 4 blister-strips of 14 tablets. NDC 66215-232-14: Blister strip of 14 tablets for oral suspension.
How Supplied
62.5 mg film-coated, round, biconvex, orange-white tablets, debossed with identification marking "62,5", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing. NDC 66215-101-06: Bottle containing 60 tablets. NDC 66215-101-03: Carton of 30 tablets in 10 blister strips of 3 tablets. 125 mg film-coated, oval, biconvex, orange-white tablets, debossed with identification marking "125", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing. NDC 66215-102-06: Bottle containing 60 tablets. NDC 66215-102-03: Carton of 30 tablets in 10 blister strips of 3 tablets. 32 mg tablets for oral suspension, pale yellow to off-white, round, bisected on one side and debossed with identification marking "32" on the other side, packaged in child-resistant Aluminum/Aluminum peel-push blisters. NDC 66215-232-56: Carton of 56 tablets for oral suspension in 4 blister-strips of 14 tablets. NDC 66215-232-14: Blister strip of 14 tablets for oral suspension.
Medication Information
Warnings and Precautions
Fluid retention: May require intervention ( 5.4 ). Pulmonary veno-occlusive disease (PVOD): If signs of pulmonary edema occur, consider the diagnosis of associated PVOD and consider discontinuing TRACLEER ( 5.5 ). Decreased sperm counts ( 5.6 ). Decreases in hemoglobin and hematocrit: Monitor hemoglobin levels after 1 and 3 months of treatment, then every 3 months thereafter ( 5.7 ).
Indications and Usage
TRACLEER is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1): in adults to improve exercise ability and to decrease clinical worsening. Studies establishing effectiveness included predominantly patients with WHO Functional Class II-IV symptoms and etiologies of idiopathic or heritable PAH (60%), PAH associated with connective tissue diseases (21%), and PAH associated with congenital heart disease with left-to-right shunts (18%) [see Clinical Studies (14.1) ] . in pediatric patients aged 3 years and older with idiopathic or congenital PAH to improve pulmonary vascular resistance (PVR), which is expected to result in an improvement in exercise ability.
Dosage and Administration
Patients older than 12 years of age: initiate at 62.5 mg orally twice daily; for patients weighing greater than 40 kg, increase to 125 mg orally twice daily after 4 weeks ( 2.2 ). Patients 12 years of age and younger: dosage is based on weight, see Table 1 ( 2.2 ). Reduce the dose and closely monitor patients developing aminotransferase elevations more than 3×Upper Limit of Normal (ULN) ( 2.4 ).
Contraindications
Pregnancy ( 4.1 ). Use with Cyclosporine A ( 4.2 ). Use with Glyburide ( 4.3 ). Hypersensitivity ( 4.4 ).
Adverse Reactions
The following important adverse reactions are described elsewhere in the labeling: Hepatotoxicity [see Boxed Warning , Warnings and Precautions (5.1) ] Embryo-fetal Toxicity [see Boxed Warning , Warnings and Precautions (5.3) ] Fluid Retention [see Warnings and Precautions (5.4) ]
Drug Interactions
Cytochrome P450: Coadministration of TRACLEER with drugs metabolized by CYP2C9 and CYP3A can increase exposure to TRACLEER and/or the coadministered drug ( 4.2 , 4.3 , 7.1 ). Hormonal contraceptives: TRACLEER use decreases contraceptive exposure and reduces effectiveness ( 7.2 ).
Storage and Handling
62.5 mg film-coated, round, biconvex, orange-white tablets, debossed with identification marking "62,5", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing. NDC 66215-101-06: Bottle containing 60 tablets. NDC 66215-101-03: Carton of 30 tablets in 10 blister strips of 3 tablets. 125 mg film-coated, oval, biconvex, orange-white tablets, debossed with identification marking "125", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing. NDC 66215-102-06: Bottle containing 60 tablets. NDC 66215-102-03: Carton of 30 tablets in 10 blister strips of 3 tablets. 32 mg tablets for oral suspension, pale yellow to off-white, round, bisected on one side and debossed with identification marking "32" on the other side, packaged in child-resistant Aluminum/Aluminum peel-push blisters. NDC 66215-232-56: Carton of 56 tablets for oral suspension in 4 blister-strips of 14 tablets. NDC 66215-232-14: Blister strip of 14 tablets for oral suspension.
How Supplied
62.5 mg film-coated, round, biconvex, orange-white tablets, debossed with identification marking "62,5", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing. NDC 66215-101-06: Bottle containing 60 tablets. NDC 66215-101-03: Carton of 30 tablets in 10 blister strips of 3 tablets. 125 mg film-coated, oval, biconvex, orange-white tablets, debossed with identification marking "125", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing. NDC 66215-102-06: Bottle containing 60 tablets. NDC 66215-102-03: Carton of 30 tablets in 10 blister strips of 3 tablets. 32 mg tablets for oral suspension, pale yellow to off-white, round, bisected on one side and debossed with identification marking "32" on the other side, packaged in child-resistant Aluminum/Aluminum peel-push blisters. NDC 66215-232-56: Carton of 56 tablets for oral suspension in 4 blister-strips of 14 tablets. NDC 66215-232-14: Blister strip of 14 tablets for oral suspension.
Description
Because of the risk of hepatotoxicity, TRACLEER is available only through a restricted program called the Bosentan Risk Evaluation and Mitigation Strategy (REMS). Under the Bosentan REMS, prescribers, patients, and pharmacies must enroll in the program [see Warnings and Precautions (5.2) ] .
Section 42229-5
Hepatotoxicity
In clinical studies, TRACLEER caused at least 3-fold upper limit of normal (ULN) elevation of liver aminotransferases (ALT and AST) in about 11% of patients, accompanied by elevated bilirubin in a small number of cases. Because these changes are a marker for potential serious hepatotoxicity, serum aminotransferase levels must be measured prior to initiation of treatment and then monthly [see Dosage and Administration (2.4), Warnings and Precautions (5.1)] . In the postmarketing period, in the setting of close monitoring, rare cases of unexplained hepatic cirrhosis were reported after prolonged (>12 months) therapy with TRACLEER in patients with multiple comorbidities and drug therapies. There have also been reports of liver failure. The contribution of TRACLEER in these cases could not be excluded.
In at least one case, the initial presentation (after >20 months of treatment) included pronounced elevations in aminotransferases and bilirubin levels accompanied by non-specific symptoms, all of which resolved slowly over time after discontinuation of TRACLEER. This case reinforces the importance of strict adherence to the monthly monitoring schedule for the duration of treatment and the treatment algorithm, which includes stopping TRACLEER with a rise of aminotransferases accompanied by signs or symptoms of liver dysfunction [see Dosage and Administration (2.4)] .
Elevations in aminotransferases require close attention [see Dosage and Administration (2.4)] . TRACLEER should generally be avoided in patients with elevated aminotransferases (>3×ULN) at baseline because monitoring for hepatotoxicity may be more difficult. If liver aminotransferase elevations are accompanied by clinical symptoms of hepatotoxicity (such as nausea, vomiting, fever, abdominal pain, jaundice, or unusual lethargy or fatigue) or increases in bilirubin ≥2×ULN, treatment with TRACLEER should be stopped. There is no experience with the reintroduction of TRACLEER in these circumstances.
Section 42231-1
| Medication Guide
TRACLEER ® (TRA-KLEER) (BOSENTAN) TABLETS |
|
|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 11/2025 |
| Read the Medication Guide that comes with TRACLEER before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |
|
What is the most important information I should know about TRACLEER?
|
|
|
TRACLEER 32 mg dispersible tablets contain Aspartame. Phenylketonurics: Contains Phenylalanine 1.87 mg per 32 mg dispersible tablet.
See " What are the possible side effects of TRACLEER? " for more information about side effects. |
|
|
What is TRACLEER?
TRACLEER is a prescription medicine used to treat people with certain types of pulmonary arterial hypertension (PAH), which is high blood pressure in the vessels of the lungs. TRACLEER can improve your ability to exercise and can slow the worsening of your physical condition and symptoms. TRACLEER lowers high blood pressure in your lungs and lets your heart pump blood more efficiently. TRACLEER is only: Prescribed by healthcare providers who are enrolled in the Bosentan REMS. Available to people who understand the risks and requirements and agree to enroll in the Bosentan REMS. |
|
|
Who should not take TRACLEER?
Do not take TRACLEER if you:
|
|
|
What should I tell my healthcare provider before taking TRACLEER?
TRACLEER may not be right for you. Tell your healthcare provider about all your medical conditions, including if you:
|
|
|
|
| There may be more than one brand name medicine. Ask your healthcare provider if you are not sure if your medicine is one that is listed above.
Know the medicines you take. Keep a list of them and show it to your healthcare provider or pharmacist when you get a new medicine. |
|
|
How should I take TRACLEER?
Your healthcare provider will give you detailed information about the Bosentan REMS.
|
|
|
What are the possible side effects of TRACLEER?
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store TRACLEER?
|
|
|
General information about TRACLEER
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use TRACLEER for a condition for which it was not prescribed. Do not give TRACLEER to other people, even if they have the same symptoms that you have. It may harm them. This Medication Guide summarizes the most important information about TRACLEER. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about TRACLEER that is written for health professionals. For more information call 1-800-526-7736, or go to www.TRACLEER.com. |
|
|
What are the ingredients in TRACLEER?
Active ingredient: bosentan Inactive ingredients in 62.5 mg and 125 mg film-coated tablets: corn starch, ethylcellulose, glyceryl behenate, hydroxypropylmethylcellulose, iron oxide red, iron oxide yellow, magnesium stearate, povidone, pregelatinized starch, sodium starch glycolate, talc, titanium dioxide, and triacetin. Inactive ingredients in 32 mg dispersible tablets: acesulfame potassium, aspartame (E951), calcium hydrogen, cellulose microcrystalline, croscarmellose sodium, magnesium stearate, phosphate anhydrous, silica colloidal anhydrous, tartaric acid, and tutti frutti flavor. Manufactured for: Actelion Pharmaceuticals US, Inc. Titusville, NJ 08560, USA For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2001 – 2019 |
Section 43683-2
Section 44425-7
Store at 20 ºC to 25 ºC (68 ºF to 77 ºF). Excursions are permitted between 15 °C and 30 °C (59 °F and 86 °F). [See USP Controlled Room Temperature]. These storage temperatures apply to both film-coated and dispersible tablets. Divided dispersible tablets should be stored under the same conditions and used within 7 days. Tablet pieces may be returned to the opened blister and stored there out of reach of children for up to 7 days.
Keep out of reach of children.
10 Overdosage
Bosentan has been given as a single dose of up to 2400 mg in normal volunteers, or up to 2000 mg/day for 2 months in patients, without any major clinical consequences. The most common side effect was headache of mild to moderate intensity. In the cyclosporine A interaction study, in which doses of 500 and 1000 mg twice daily of bosentan were given concomitantly with cyclosporine A, trough plasma concentrations of bosentan increased 30-fold, resulting in severe headache, nausea, and vomiting, but no serious adverse events. Mild decreases in blood pressure and increases in heart rate were observed.
In the postmarketing period, there was one reported overdose of 10,000 mg of TRACLEER taken by an adolescent male patient. He had symptoms of nausea, vomiting, hypotension, dizziness, sweating, and blurred vision. He recovered within 24 hours with blood pressure support.
Bosentan is unlikely to be effectively removed by dialysis due to the high molecular weight and extensive plasma protein binding.
4.1 Pregnancy
Use of TRACLEER is contraindicated in females who are pregnant [see Boxed Warning, Dosage and Administration (2.1), Warnings and Precautions (5.3), Use in Specific Populations (8.1)].
11 Description
TRACLEER ® is the proprietary name for bosentan, an endothelin receptor antagonist that belongs to a class of highly substituted pyrimidine derivatives, with no chiral centers. It is designated chemically as 4-tert-butyl-N-[6-(2-hydroxy-ethoxy)-5-(2-methoxy-phenoxy)-[2,2´]-bipyrimidin-4-yl]- benzenesulfonamide monohydrate and has the following structural formula:
Bosentan has a molecular weight of 569.64 and a molecular formula of C 27H 29N 5O 6S∙H 2O. Bosentan is a white to yellowish powder. It is poorly soluble in water (1.0 mg/100 mL) and in aqueous solutions at low pH (0.1 mg/100 mL at pH 1.1 and 4.0; 0.2 mg/100 mL at pH 5.0). Solubility increases at higher pH values (43 mg/100 mL at pH 7.5). In the solid state, bosentan is very stable, is not hygroscopic and is not light sensitive.
TRACLEER is available as 62.5 mg and 125 mg film-coated tablets for oral administration, and contains the following excipients: corn starch, ethylcellulose, glyceryl behenate, hydroxypropylmethylcellulose, iron oxide red, iron oxide yellow, magnesium stearate, povidone, pregelatinized starch, sodium starch glycolate, talc, titanium dioxide, and triacetin. Each TRACLEER 62.5 mg tablet contains 64.54 mg of bosentan monohydrate, equivalent to 62.5 mg of anhydrous bosentan. Each TRACLEER 125 mg tablet contains 129.08 mg of bosentan monohydrate, equivalent to 125 mg of anhydrous bosentan.
TRACLEER is also available as a 32 mg tablet for oral suspension and contains the following excipients: acesulfame potassium, aspartame (E951), calcium hydrogen phosphate anhydrous, cellulose microcrystalline, croscarmellose sodium, magnesium stearate, silica colloidal anhydrous, tartaric acid, and tutti frutti flavor. Each dispersible tablet contains 1.87 mg of phenylalanine. Each dispersible tablet contains 33.045 mg of bosentan monohydrate, equivalent to 32 mg anhydrous bosentan.
5.2 Bosentan Rems
Because of the risks of hepatotoxicity , TRACLEER is available only through a restricted program called the Bosentan REMS. As a component of the Bosentan REMS, prescribers, patients, and pharmacies must enroll in the program [see Boxed Warning, Warnings and Precautions (5.1)] .
Required components of the Bosentan REMS are:
- Healthcare professionals who prescribe TRACLEER must review the prescriber educational materials, enroll in the Bosentan REMS and comply with its requirements.
- Healthcare professionals must (1) review serum aminotransferases (ALT/AST) and bilirubin, and agree to order and monitor these tests monthly.
- To receive TRACLEER, all patients must understand the risks and benefits, and complete a patient enrollment form with their prescriber.
- Pharmacies that dispense TRACLEER must enroll in the program and agree to comply with the Bosentan REMS requirements.
Further information about TRACLEER and the Bosentan REMS is available at www.BosentanREMSProgram.com or 1-866-359-2612.
8.4 Pediatric Use
The efficacy of TRACLEER in patients <18 years is supported by data from an uncontrolled trial in which 19 pediatric patients were treated with TRACLEER. In this study, cardiopulmonary hemodynamic improvements were similar to those seen in adults treated with TRACLEER [see Clinical Studies (14.1)] . Safety in pediatric patients is supported by data from 100 pediatric patients treated with TRACLEER for a median of 17 months [see Adverse Reactions (6.1), Clinical Studies (14.1)] .
8.5 Geriatric Use
Clinical studies of TRACLEER did not include sufficient numbers of subjects aged 65 and older to determine whether they respond differently from younger subjects.
2.3 Administration
TRACLEER film-coated tablets and tablets for oral suspension (dispersible tablets) should be administered orally twice daily.
Disperse tablets for oral suspension, or dispersible tablet half, in a minimal amount of water immediately before administration.
Store divided dispersible tablet pieces at 20 ºC to 25 ºC (68 ºF to 77 ºF) in the opened blister for up to 7 days.
5.1 Hepatotoxicity
ALT or AST >3 ×ULN were observed in 11% of TRACLEER-treated patients (n=658) compared to 2% of placebo-treated patients (n=280). Three-fold increases were seen in 12% of 95 pulmonary arterial hypertension (PAH) patients on 125 mg twice daily and 14% of 70 PAH patients on 250 mg twice daily. Eight-fold increases were seen in 2% of PAH patients on 125 mg twice daily and 7% of PAH patients on 250 mg twice daily. Bilirubin increases to ≥3 ×ULN were associated with aminotransferase increases in 2 of 658 (0.3%) of patients treated with TRACLEER. In a pooled analysis of four pediatric studies conducted in PAH (n=100), elevations in liver aminotransferases ≥3×ULN were observed in 2% of patients. The combination of hepatocellular injury (increases in aminotransferases of >3 ×ULN) and increases in total bilirubin (≥2 ×ULN) is a marker for potential serious hepatotoxicity.
Elevations of AST or ALT associated with TRACLEER are dose-dependent, occur both early and late in treatment, usually progress slowly, are typically asymptomatic, and usually have been reversible after treatment interruption or cessation. Aminotransferase elevations also may reverse spontaneously while continuing treatment with TRACLEER.
Liver aminotransferase levels must be measured prior to initiation of treatment and then monthly and therapy adjusted accordingly [see Dosage and Administration (2.1, 2.4)] . Discontinue TRACLEER if liver aminotransferase elevations are accompanied by clinical symptoms of hepatotoxicity (such as nausea, vomiting, fever, abdominal pain, jaundice, or unusual lethargy or fatigue) or increases in bilirubin ≥2 ×ULN.
Avoid initiation of TRACLEER in patients with elevated aminotransferases (>3 ×ULN) prior to drug initiation because monitoring hepatotoxicity in these patients may be more difficult [see Boxed Warning, Dosage and Administration (2.6), Use in Specific Populations (8.6)] .
In WHO Functional Class II patients, consider whether the benefits of TRACLEER are sufficient to offset the risk of hepatotoxicity, which may preclude future use as their disease progresses.
TRACLEER is only available through a restricted program under REMS [see Warnings and Precautions (5.2)].
4 Contraindications
5.4 Fluid Retention
Peripheral edema is a known clinical consequence of PAH and worsening PAH and is also a known effect of TRACLEER and other endothelin receptor antagonists. In PAH clinical trials with TRACLEER, combined adverse events of fluid retention or edema were reported in 1.7% (placebo-corrected) of patients.
In addition, there have been numerous postmarketing reports of fluid retention in patients with pulmonary hypertension occurring within weeks after starting TRACLEER. Patients required intervention with a diuretic, fluid management, or hospitalization for decompensating heart failure.
If clinically significant fluid retention develops, with or without associated weight gain, further evaluation should be undertaken to determine the cause, such as TRACLEER or underlying heart failure, and the possible need for treatment or discontinuation of TRACLEER [see Adverse Reactions (6.1), Clinical Studies (14.2)] .
6 Adverse Reactions
The following important adverse reactions are described elsewhere in the labeling:
- Hepatotoxicity [see Boxed Warning, Warnings and Precautions (5.1)]
- Embryo-fetal Toxicity [see Boxed Warning, Warnings and Precautions (5.3)]
- Fluid Retention [see Warnings and Precautions (5.4)]
7 Drug Interactions
4.4 Hypersensitivity
TRACLEER is contraindicated in patients who are hypersensitive to bosentan or any component of the product. Observed reactions include Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), anaphylaxis, rash, and angioedema [see Adverse Reactions (6.2), Description (11)] .
8.7 Renal Impairment
The effect of renal impairment on the pharmacokinetics of bosentan is small and does not require dosing adjustment [see Clinical Pharmacology (12.3)] .
2.2 Recommended Dosage
Administer TRACLEER orally following the dosing recommendations in Table 1. Doses above 125 mg twice daily did not appear to confer additional benefit sufficient to offset the increased risk of hepatotoxicity.
| Initial 4 weeks | Maintenance (after 4 weeks) | |
|---|---|---|
| Patients >12 years of age and >40 kg | 62.5 mg twice daily | 125 mg twice daily |
| Patients >12 years of age and <40 kg | 62.5 mg twice daily | 62.5 mg twice daily |
| Patients ≤12 years of age | ||
| ≥4–8 kg | 16 mg twice daily | 16 mg twice daily |
| >8–16 kg | 32 mg twice daily | 32 mg twice daily |
| >16–24 kg | 48 mg twice daily | 48 mg twice daily |
| >24–40 kg | 64 mg twice daily | 64 mg twice daily |
4.3 Use With Glyburide
An increased risk of liver enzyme elevations was observed in patients receiving glyburide concomitantly with bosentan. Therefore co-administration of glyburide and TRACLEER is contraindicated [see Drug Interactions (7.1)] .
8.6 Hepatic Impairment
Because there is in vitro and in vivo evidence that the main route of excretion of bosentan is biliary, liver impairment could be expected to increase exposure (C max and AUC) of bosentan. The pharmacokinetics of TRACLEER have not been evaluated in patients with severe liver impairment (Child-Pugh Class C). In patients with moderate hepatic impairment (Child-Pugh Class B), the systemic exposures to bosentan and its active metabolite increased significantly. TRACLEER should generally be avoided in patients with moderate or severe liver impairment. Pharmacokinetics of bosentan were not altered in patients with mild impairment of hepatic function (Child-Pugh Class A) [see Dosage and Administration (2.6), Warnings and Precautions (5.1), Clinical Pharmacology (12.3)] .
1 Indications and Usage
TRACLEER is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1):
- in adults to improve exercise ability and to decrease clinical worsening. Studies establishing effectiveness included predominantly patients with WHO Functional Class II-IV symptoms and etiologies of idiopathic or heritable PAH (60%), PAH associated with connective tissue diseases (21%), and PAH associated with congenital heart disease with left-to-right shunts (18%) [see Clinical Studies (14.1)] .
- in pediatric patients aged 3 years and older with idiopathic or congenital PAH to improve pulmonary vascular resistance (PVR), which is expected to result in an improvement in exercise ability.
2.1 Required Monitoring
Healthcare professionals who prescribe TRACLEER must enroll in the Bosentan REMS and must comply with the required monitoring to minimize the risks associated with TRACLEER [see Warnings and Precautions (5.2)].
Measure liver aminotransferase levels prior to initiation of treatment and then monthly [see Boxed Warning, Warnings and Precautions (5.1, 5.2)].
Exclude pregnancy before initiating treatment with TRACLEER in females of reproductive potential [see Boxed Warning, Contraindications (4.1), Warnings and Precautions (5.3), Use in Specific Populations (8.1, 8.3)] .
12.1 Mechanism of Action
Bosentan is a specific and competitive antagonist at endothelin receptor types ET A and ET B. Bosentan has a slightly higher affinity for ET A receptors than for ET B receptors. The clinical impact of dual endothelin blockage is unknown.
Endothelin-1 (ET-1) is a neurohormone, the effects of which are mediated by binding to ET A and ET B receptors in the endothelium and vascular smooth muscle. ET-1 concentrations are elevated in plasma and lung tissue of patients with PAH, suggesting a pathogenic role for ET-1 in this disease.
5.3 Embryo Fetal Toxicity
Based on data from animal reproduction studies, TRACLEER may cause fetal harm when administered to a pregnant female and is contraindicated in females who are pregnant. The available human data for endothelin receptor antagonists do not establish the presence or absence of major birth defects related to the use of TRACLEER. Advise females of reproductive potential about the potential risk to a fetus. Exclude pregnancy prior to TRACLEER treatment. Advise females of reproductive potential to use effective contraception prior to initiation of treatment with TRACLEER, during treatment, and for at least one month after the last dose. When pregnancy is detected, discontinue TRACLEER as soon as possible [see Dosage and Administration (2.1), Contraindications (4.1), Drug Interactions (7.2), Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions
- Fluid retention: May require intervention ( 5.4).
- Pulmonary veno-occlusive disease (PVOD): If signs of pulmonary edema occur, consider the diagnosis of associated PVOD and consider discontinuing TRACLEER ( 5.5).
- Decreased sperm counts ( 5.6).
- Decreases in hemoglobin and hematocrit: Monitor hemoglobin levels after 1 and 3 months of treatment, then every 3 months thereafter ( 5.7).
5.6 Decreased Sperm Counts
Decreased sperm counts have been observed in patients receiving TRACLEER. Preclinical data also suggest that TRACLEER, similar to other endothelin receptor antagonists, may have an adverse effect on spermatogenesis [see Adverse Reactions (6.1), Nonclinical Toxicology (13.1)] .
2 Dosage and Administration
- Patients older than 12 years of age: initiate at 62.5 mg orally twice daily; for patients weighing greater than 40 kg, increase to 125 mg orally twice daily after 4 weeks ( 2.2).
- Patients 12 years of age and younger: dosage is based on weight, see Table 1( 2.2).
- Reduce the dose and closely monitor patients developing aminotransferase elevations more than 3×Upper Limit of Normal (ULN) ( 2.4).
4.2 Use With Cyclosporine A
Co-administration of cyclosporine A and bosentan resulted in markedly increased plasma concentrations of bosentan. Therefore, concomitant use of TRACLEER and cyclosporine A is contraindicated [see Drug Interactions (7.1)] .
7.2 Hormonal Contraceptives
Hormonal contraceptives, including oral, injectable, transdermal, and implantable forms, may not be reliable when TRACLEER is co-administered. Females should practice additional methods of contraception and not rely on hormonal contraception alone when taking TRACLEER [see Use in Specific Populations (8.3)] .
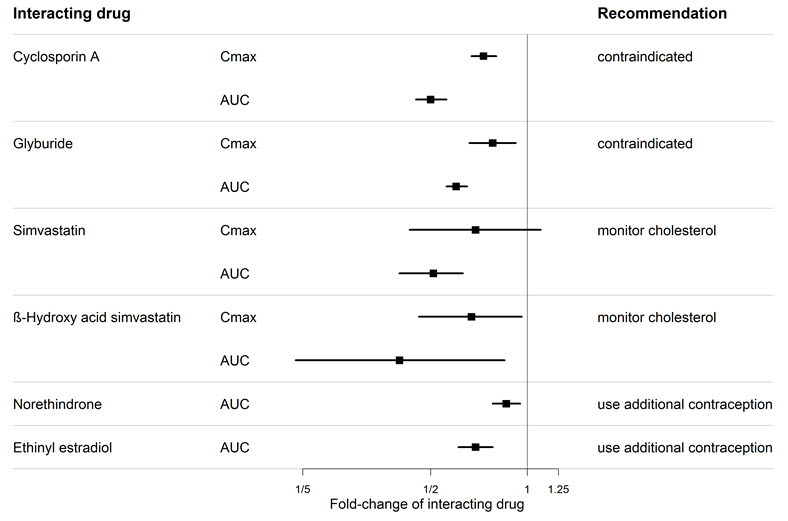
An interaction study demonstrated that co-administration of bosentan and a combination oral hormonal contraceptive produced average decreases of norethindrone and ethinyl estradiol levels of 14% and 31%, respectively. However, decreases in exposure were as much as 56% and 66%, respectively, in individual subjects.
3 Dosage Forms and Strengths
62.5 mg tablets: round, biconvex, orange-white tablets, debossed with identification marking "62,5"
125 mg tablets: oval, biconvex, orange-white tablets, debossed with identification marking "125"
32 mg tablets for oral suspension: round, pale yellow to off-white tablets, bisected on one side and debossed with identification marking “32” on the other side.
6.2 Postmarketing Experience
There have been several postmarketing reports of angioedema associated with the use of TRACLEER. The onset of the reported cases occurred within a range of 8 hours to 21 days after starting therapy. Some patients were treated with an antihistamine and their signs of angioedema resolved without discontinuing TRACLEER.
The following additional adverse reactions have been reported during the post approval use of TRACLEER. Because these adverse reactions are reported from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to TRACLEER exposure:
- Unexplained hepatic cirrhosis [see Boxed Warning]
- Liver failure [see Boxed Warning]
- Hypersensitivity, DRESS, and anaphylaxis [see Contraindications (4.4)]
- Thrombocytopenia
- Rash
- Jaundice
- Anemia requiring transfusion
- Neutropenia and leukopenia
- Nasal congestion
- Autoimmune hepatitis
8 Use in Specific Populations
- Nursing mothers: Choose breastfeeding or TRACLEER ( 8.2).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Safety data on TRACLEER were obtained from 13 clinical studies (9 placebo-controlled and 4 open-label) in 870 adult patients with PAH and other diseases. Doses up to 8 times the currently recommended clinical dose (125 mg twice daily) were administered for a variety of durations. The exposure to TRACLEER in these trials ranged from 1 day to 4.1 years (n=94 for 1 year; n=61 for 1.5 years; and n=39 for more than 2 years). Exposure of PAH patients (n=328) to TRACLEER ranged from 1 day to 1.7 years (n=174 more than 6 months and n=28 more than 12 months).
Treatment discontinuations due to adverse events other than those related to pulmonary hypertension during the clinical trials in adult patients with PAH were more frequent on TRACLEER (6%; 15/258 patients) than on placebo (3%; 5/172 patients). In this database the only cause of discontinuations >1% and occurring more often on TRACLEER was abnormal liver function.
The adverse drug events that occurred in ≥3% of the TRACLEER-treated patients and were more common on TRACLEER in placebo-controlled trials in PAH at doses of 125 or 250 mg twice daily are shown in Table 3:
| Adverse Event | TRACLEER
n=258 |
Placebo
n=172 |
||
|---|---|---|---|---|
| No. | % | No. | % | |
| Respiratory Tract Infection
Respiratory Tract Infection combines the terms "Nasopharyngitis", "Upper Respiratory Tract Infection" and "Respiratory Tract Infection". Combined data from Study 351, BREATHE-1 and EARLY
|
56 | 22% | 30 | 17% |
| Headache | 39 | 15% | 25 | 14% |
| Edema | 28 | 11% | 16 | 9% |
| Chest Pain | 13 | 5% | 8 | 5% |
| Syncope | 12 | 5% | 7 | 4% |
| Flushing | 10 | 4% | 5 | 3% |
| Hypotension | 10 | 4% | 3 | 2% |
| Sinusitis | 9 | 4% | 4 | 2% |
| Arthralgia | 9 | 4% | 3 | 2% |
| Serum Aminotransferases, abnormal | 9 | 4% | 3 | 2% |
| Palpitations | 9 | 4% | 3 | 2% |
| Anemia | 8 | 3% | - | - |
TRACLEER was evaluated for safety in 119 pediatric patients in uncontrolled studies. The safety profile was similar to that observed in adult patients with PAH.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide)
16 How Supplied/storage and Handling
62.5 mgfilm-coated, round, biconvex, orange-white tablets, debossed with identification marking "62,5", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing.
NDC 66215-101-06: Bottle containing 60 tablets.
NDC 66215-101-03: Carton of 30 tablets in 10 blister strips of 3 tablets.
125 mgfilm-coated, oval, biconvex, orange-white tablets, debossed with identification marking "125", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing.
NDC 66215-102-06: Bottle containing 60 tablets.
NDC 66215-102-03: Carton of 30 tablets in 10 blister strips of 3 tablets.
32 mgtablets for oral suspension, pale yellow to off-white, round, bisected on one side and debossed with identification marking "32" on the other side, packaged in child-resistant Aluminum/Aluminum peel-push blisters.
NDC 66215-232-56: Carton of 56 tablets for oral suspension in 4 blister-strips of 14 tablets.
NDC 66215-232-14: Blister strip of 14 tablets for oral suspension.
5.5 Pulmonary Veno Occlusive Disease
If signs of pulmonary edema occur, consider the possibility of associated pulmonary veno-occlusive disease and consider whether TRACLEER should be discontinued.
7.1 Cytochrome P450 Drug Interactions
Bosentan is metabolized by CYP2C9 and CYP3A. Inhibition of these enzymes may increase the plasma concentration of bosentan [see Clinical Pharmacology (12.3)] . Concomitant administration of both a CYP2C9 inhibitor (such as fluconazole or amiodarone) and a strong CYP3A inhibitor (e.g., ketoconazole, itraconazole) or a moderate CYP3A inhibitor (e.g., amprenavir, erythromycin, fluconazole, diltiazem) with TRACLEER will likely lead to large increases in plasma concentrations of bosentan. Co-administration of such combinations of a CYP2C9 inhibitor plus a strong or moderate CYP3A inhibitor with TRACLEER is not recommended.
Bosentan is an inducer of CYP3A and CYP2C9. Consequently plasma concentrations of drugs metabolized by these two isozymes will be decreased when TRACLEER is co-administered. Bosentan had no relevant inhibitory effect on any CYP isozyme in vitro(CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A). Consequently, TRACLEER is not expected to increase the plasma concentrations of drugs metabolized by these enzymes.
Figure 1. CYP3A induction-mediated effect of bosentan on other drugs
Figure 2. Effect of other drugs on bosentan
5.7 Decreases in Hemoglobin and Hematocrit
Treatment with TRACLEER can cause a dose-related decrease in hemoglobin and hematocrit. There have been postmarketing reports of decreases in hemoglobin concentration and hematocrit that have resulted in anemia requiring transfusion. It is recommended that hemoglobin concentrations be checked after 1 and 3 months, and every 3 months thereafter. If a marked decrease in hemoglobin concentration occurs, further evaluation should be undertaken to determine the cause and need for specific treatment [see Adverse Reactions (6.1)] .
8.3 Females and Males of Reproductive Potential
Based on data from animal reproductive toxicity studies, TRACLEER may cause fetal harm, including birth defects and fetal death, when administered to a pregnant patient and is contraindicated during pregnancy [see Contraindications (4.1), Use in Specific Populations (8.1)].
14.2 Lack of Benefit in Congestive Heart Failure
TRACLEER is not effective in the treatment of congestive heart failure with left ventricular dysfunction. In a pair of studies, 1613 subjects with NYHA Class III-IV heart failure, left ventricular ejection fraction <35%, on diuretics, ACE inhibitor, and other therapies, were randomized to placebo or TRACLEER (62.5 mg twice daily titrated as tolerated to 125 mg twice daily) and followed for up to 70 weeks. Use of TRACLEER was associated with no benefit on patient global assessment (the primary end point) or mortality. However, hospitalizations for heart failure were more common during the first 4 to 8 weeks after TRACLEER was initiated. In a placebo-controlled trial of patients with severe chronic heart failure, there was an increased incidence of hospitalization for CHF associated with weight gain and increased leg edema during the first 4–8 weeks of treatment with TRACLEER. Patients required intervention with a diuretic, fluid management, or hospitalization for decompensating heart failure.
Principal Display Panel 125 Mg Tablet Bottle Carton
NDC 66215-102-06
Tracleer
®
(bosentan) tablets
125 mg
Attention: Dispense with
enclosed Medication Guide.
Rx only
60 film-coated tablets
janssen
2.4 Dosage Adjustments for Aminotransferase Elevations
If aminotransferase levels increase, adjust monitoring and treatment plan according to Table 2.
Discontinue TRACLEER if liver aminotransferase elevations are accompanied by clinical symptoms of hepatotoxicity (such as nausea, vomiting, fever, abdominal pain, jaundice, or unusual lethargy or fatigue) or bilirubin ≥2 ×Upper Limit of Normal (ULN). There is no experience with the reintroduction of TRACLEER in these circumstances.
| ALT/AST levels | Treatment and monitoring recommendations |
|---|---|
| >3 and ≤5 ×ULN | Confirm by another aminotransferase test; if confirmed,
|
| >5 and ≤8 ×ULN | Confirm by another aminotransferase test; if confirmed, stop treatment and monitor aminotransferase levels at least every 2 weeks. Once the aminotransferase levels return to pretreatment values,
|
| >8 ×ULN | Stop treatment permanently. There is no experience with reintroduction of TRACLEER in these circumstances. |
Principal Display Panel 62.5 Mg Tablet Bottle Carton
NDC 66215-101-06
Tracleer
®
(bosentan) tablets
62.5 mg
Attention: Dispense with
enclosed Medication Guide.
Rx only
60 film-coated tablets
janssen
2.6 Use in Patients With Pre Existing Hepatic Impairment
Avoid initiation of TRACLEER in patients with aminotransferases >3 ×ULN. No dose adjustment is required in patients with mildly impaired liver function [see Warnings and Precautions (5.1), Use in Specific Populations (8.6), Clinical Pharmacology (12.3)] .
Warning: Risks of Hepatotoxicity and Embryo Fetal Toxicity
Because of the risk of hepatotoxicity, TRACLEER is available only through a restricted program called the Bosentan Risk Evaluation and Mitigation Strategy (REMS). Under the Bosentan REMS, prescribers, patients, and pharmacies must enroll in the program [see Warnings and Precautions (5.2)] .
Principal Display Panel 32 Mg Tablet Blister Pack Carton 232
NDC 66215-232-56
Tracleer
®
(bosentan) Tablets for Oral Suspension
32 mg
Phenylketonurics:
Contains Phenylalanine
1.87 mg per tablet for oral suspension
Rx only
Contains: 56 tablets for oral suspension per carton.
Each carton contains 4 blister-strips of 14 tablets each.
Attention: Dispense with
enclosed Medication Guide.
This package is child-resistant.
Store this medicine out of
the reach of children.
janssen
Structured Label Content
Section 42229-5 (42229-5)
Hepatotoxicity
In clinical studies, TRACLEER caused at least 3-fold upper limit of normal (ULN) elevation of liver aminotransferases (ALT and AST) in about 11% of patients, accompanied by elevated bilirubin in a small number of cases. Because these changes are a marker for potential serious hepatotoxicity, serum aminotransferase levels must be measured prior to initiation of treatment and then monthly [see Dosage and Administration (2.4), Warnings and Precautions (5.1)] . In the postmarketing period, in the setting of close monitoring, rare cases of unexplained hepatic cirrhosis were reported after prolonged (>12 months) therapy with TRACLEER in patients with multiple comorbidities and drug therapies. There have also been reports of liver failure. The contribution of TRACLEER in these cases could not be excluded.
In at least one case, the initial presentation (after >20 months of treatment) included pronounced elevations in aminotransferases and bilirubin levels accompanied by non-specific symptoms, all of which resolved slowly over time after discontinuation of TRACLEER. This case reinforces the importance of strict adherence to the monthly monitoring schedule for the duration of treatment and the treatment algorithm, which includes stopping TRACLEER with a rise of aminotransferases accompanied by signs or symptoms of liver dysfunction [see Dosage and Administration (2.4)] .
Elevations in aminotransferases require close attention [see Dosage and Administration (2.4)] . TRACLEER should generally be avoided in patients with elevated aminotransferases (>3×ULN) at baseline because monitoring for hepatotoxicity may be more difficult. If liver aminotransferase elevations are accompanied by clinical symptoms of hepatotoxicity (such as nausea, vomiting, fever, abdominal pain, jaundice, or unusual lethargy or fatigue) or increases in bilirubin ≥2×ULN, treatment with TRACLEER should be stopped. There is no experience with the reintroduction of TRACLEER in these circumstances.
Section 42231-1 (42231-1)
| Medication Guide
TRACLEER ® (TRA-KLEER) (BOSENTAN) TABLETS |
|
|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 11/2025 |
| Read the Medication Guide that comes with TRACLEER before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |
|
What is the most important information I should know about TRACLEER?
|
|
|
TRACLEER 32 mg dispersible tablets contain Aspartame. Phenylketonurics: Contains Phenylalanine 1.87 mg per 32 mg dispersible tablet.
See " What are the possible side effects of TRACLEER? " for more information about side effects. |
|
|
What is TRACLEER?
TRACLEER is a prescription medicine used to treat people with certain types of pulmonary arterial hypertension (PAH), which is high blood pressure in the vessels of the lungs. TRACLEER can improve your ability to exercise and can slow the worsening of your physical condition and symptoms. TRACLEER lowers high blood pressure in your lungs and lets your heart pump blood more efficiently. TRACLEER is only: Prescribed by healthcare providers who are enrolled in the Bosentan REMS. Available to people who understand the risks and requirements and agree to enroll in the Bosentan REMS. |
|
|
Who should not take TRACLEER?
Do not take TRACLEER if you:
|
|
|
What should I tell my healthcare provider before taking TRACLEER?
TRACLEER may not be right for you. Tell your healthcare provider about all your medical conditions, including if you:
|
|
|
|
| There may be more than one brand name medicine. Ask your healthcare provider if you are not sure if your medicine is one that is listed above.
Know the medicines you take. Keep a list of them and show it to your healthcare provider or pharmacist when you get a new medicine. |
|
|
How should I take TRACLEER?
Your healthcare provider will give you detailed information about the Bosentan REMS.
|
|
|
What are the possible side effects of TRACLEER?
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store TRACLEER?
|
|
|
General information about TRACLEER
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use TRACLEER for a condition for which it was not prescribed. Do not give TRACLEER to other people, even if they have the same symptoms that you have. It may harm them. This Medication Guide summarizes the most important information about TRACLEER. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about TRACLEER that is written for health professionals. For more information call 1-800-526-7736, or go to www.TRACLEER.com. |
|
|
What are the ingredients in TRACLEER?
Active ingredient: bosentan Inactive ingredients in 62.5 mg and 125 mg film-coated tablets: corn starch, ethylcellulose, glyceryl behenate, hydroxypropylmethylcellulose, iron oxide red, iron oxide yellow, magnesium stearate, povidone, pregelatinized starch, sodium starch glycolate, talc, titanium dioxide, and triacetin. Inactive ingredients in 32 mg dispersible tablets: acesulfame potassium, aspartame (E951), calcium hydrogen, cellulose microcrystalline, croscarmellose sodium, magnesium stearate, phosphate anhydrous, silica colloidal anhydrous, tartaric acid, and tutti frutti flavor. Manufactured for: Actelion Pharmaceuticals US, Inc. Titusville, NJ 08560, USA For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2001 – 2019 |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store at 20 ºC to 25 ºC (68 ºF to 77 ºF). Excursions are permitted between 15 °C and 30 °C (59 °F and 86 °F). [See USP Controlled Room Temperature]. These storage temperatures apply to both film-coated and dispersible tablets. Divided dispersible tablets should be stored under the same conditions and used within 7 days. Tablet pieces may be returned to the opened blister and stored there out of reach of children for up to 7 days.
Keep out of reach of children.
10 Overdosage (10 OVERDOSAGE)
Bosentan has been given as a single dose of up to 2400 mg in normal volunteers, or up to 2000 mg/day for 2 months in patients, without any major clinical consequences. The most common side effect was headache of mild to moderate intensity. In the cyclosporine A interaction study, in which doses of 500 and 1000 mg twice daily of bosentan were given concomitantly with cyclosporine A, trough plasma concentrations of bosentan increased 30-fold, resulting in severe headache, nausea, and vomiting, but no serious adverse events. Mild decreases in blood pressure and increases in heart rate were observed.
In the postmarketing period, there was one reported overdose of 10,000 mg of TRACLEER taken by an adolescent male patient. He had symptoms of nausea, vomiting, hypotension, dizziness, sweating, and blurred vision. He recovered within 24 hours with blood pressure support.
Bosentan is unlikely to be effectively removed by dialysis due to the high molecular weight and extensive plasma protein binding.
4.1 Pregnancy
Use of TRACLEER is contraindicated in females who are pregnant [see Boxed Warning, Dosage and Administration (2.1), Warnings and Precautions (5.3), Use in Specific Populations (8.1)].
11 Description (11 DESCRIPTION)
TRACLEER ® is the proprietary name for bosentan, an endothelin receptor antagonist that belongs to a class of highly substituted pyrimidine derivatives, with no chiral centers. It is designated chemically as 4-tert-butyl-N-[6-(2-hydroxy-ethoxy)-5-(2-methoxy-phenoxy)-[2,2´]-bipyrimidin-4-yl]- benzenesulfonamide monohydrate and has the following structural formula:
Bosentan has a molecular weight of 569.64 and a molecular formula of C 27H 29N 5O 6S∙H 2O. Bosentan is a white to yellowish powder. It is poorly soluble in water (1.0 mg/100 mL) and in aqueous solutions at low pH (0.1 mg/100 mL at pH 1.1 and 4.0; 0.2 mg/100 mL at pH 5.0). Solubility increases at higher pH values (43 mg/100 mL at pH 7.5). In the solid state, bosentan is very stable, is not hygroscopic and is not light sensitive.
TRACLEER is available as 62.5 mg and 125 mg film-coated tablets for oral administration, and contains the following excipients: corn starch, ethylcellulose, glyceryl behenate, hydroxypropylmethylcellulose, iron oxide red, iron oxide yellow, magnesium stearate, povidone, pregelatinized starch, sodium starch glycolate, talc, titanium dioxide, and triacetin. Each TRACLEER 62.5 mg tablet contains 64.54 mg of bosentan monohydrate, equivalent to 62.5 mg of anhydrous bosentan. Each TRACLEER 125 mg tablet contains 129.08 mg of bosentan monohydrate, equivalent to 125 mg of anhydrous bosentan.
TRACLEER is also available as a 32 mg tablet for oral suspension and contains the following excipients: acesulfame potassium, aspartame (E951), calcium hydrogen phosphate anhydrous, cellulose microcrystalline, croscarmellose sodium, magnesium stearate, silica colloidal anhydrous, tartaric acid, and tutti frutti flavor. Each dispersible tablet contains 1.87 mg of phenylalanine. Each dispersible tablet contains 33.045 mg of bosentan monohydrate, equivalent to 32 mg anhydrous bosentan.
5.2 Bosentan Rems (5.2 Bosentan REMS)
Because of the risks of hepatotoxicity , TRACLEER is available only through a restricted program called the Bosentan REMS. As a component of the Bosentan REMS, prescribers, patients, and pharmacies must enroll in the program [see Boxed Warning, Warnings and Precautions (5.1)] .
Required components of the Bosentan REMS are:
- Healthcare professionals who prescribe TRACLEER must review the prescriber educational materials, enroll in the Bosentan REMS and comply with its requirements.
- Healthcare professionals must (1) review serum aminotransferases (ALT/AST) and bilirubin, and agree to order and monitor these tests monthly.
- To receive TRACLEER, all patients must understand the risks and benefits, and complete a patient enrollment form with their prescriber.
- Pharmacies that dispense TRACLEER must enroll in the program and agree to comply with the Bosentan REMS requirements.
Further information about TRACLEER and the Bosentan REMS is available at www.BosentanREMSProgram.com or 1-866-359-2612.
8.4 Pediatric Use
The efficacy of TRACLEER in patients <18 years is supported by data from an uncontrolled trial in which 19 pediatric patients were treated with TRACLEER. In this study, cardiopulmonary hemodynamic improvements were similar to those seen in adults treated with TRACLEER [see Clinical Studies (14.1)] . Safety in pediatric patients is supported by data from 100 pediatric patients treated with TRACLEER for a median of 17 months [see Adverse Reactions (6.1), Clinical Studies (14.1)] .
8.5 Geriatric Use
Clinical studies of TRACLEER did not include sufficient numbers of subjects aged 65 and older to determine whether they respond differently from younger subjects.
2.3 Administration
TRACLEER film-coated tablets and tablets for oral suspension (dispersible tablets) should be administered orally twice daily.
Disperse tablets for oral suspension, or dispersible tablet half, in a minimal amount of water immediately before administration.
Store divided dispersible tablet pieces at 20 ºC to 25 ºC (68 ºF to 77 ºF) in the opened blister for up to 7 days.
5.1 Hepatotoxicity
ALT or AST >3 ×ULN were observed in 11% of TRACLEER-treated patients (n=658) compared to 2% of placebo-treated patients (n=280). Three-fold increases were seen in 12% of 95 pulmonary arterial hypertension (PAH) patients on 125 mg twice daily and 14% of 70 PAH patients on 250 mg twice daily. Eight-fold increases were seen in 2% of PAH patients on 125 mg twice daily and 7% of PAH patients on 250 mg twice daily. Bilirubin increases to ≥3 ×ULN were associated with aminotransferase increases in 2 of 658 (0.3%) of patients treated with TRACLEER. In a pooled analysis of four pediatric studies conducted in PAH (n=100), elevations in liver aminotransferases ≥3×ULN were observed in 2% of patients. The combination of hepatocellular injury (increases in aminotransferases of >3 ×ULN) and increases in total bilirubin (≥2 ×ULN) is a marker for potential serious hepatotoxicity.
Elevations of AST or ALT associated with TRACLEER are dose-dependent, occur both early and late in treatment, usually progress slowly, are typically asymptomatic, and usually have been reversible after treatment interruption or cessation. Aminotransferase elevations also may reverse spontaneously while continuing treatment with TRACLEER.
Liver aminotransferase levels must be measured prior to initiation of treatment and then monthly and therapy adjusted accordingly [see Dosage and Administration (2.1, 2.4)] . Discontinue TRACLEER if liver aminotransferase elevations are accompanied by clinical symptoms of hepatotoxicity (such as nausea, vomiting, fever, abdominal pain, jaundice, or unusual lethargy or fatigue) or increases in bilirubin ≥2 ×ULN.
Avoid initiation of TRACLEER in patients with elevated aminotransferases (>3 ×ULN) prior to drug initiation because monitoring hepatotoxicity in these patients may be more difficult [see Boxed Warning, Dosage and Administration (2.6), Use in Specific Populations (8.6)] .
In WHO Functional Class II patients, consider whether the benefits of TRACLEER are sufficient to offset the risk of hepatotoxicity, which may preclude future use as their disease progresses.
TRACLEER is only available through a restricted program under REMS [see Warnings and Precautions (5.2)].
4 Contraindications (4 CONTRAINDICATIONS)
5.4 Fluid Retention
Peripheral edema is a known clinical consequence of PAH and worsening PAH and is also a known effect of TRACLEER and other endothelin receptor antagonists. In PAH clinical trials with TRACLEER, combined adverse events of fluid retention or edema were reported in 1.7% (placebo-corrected) of patients.
In addition, there have been numerous postmarketing reports of fluid retention in patients with pulmonary hypertension occurring within weeks after starting TRACLEER. Patients required intervention with a diuretic, fluid management, or hospitalization for decompensating heart failure.
If clinically significant fluid retention develops, with or without associated weight gain, further evaluation should be undertaken to determine the cause, such as TRACLEER or underlying heart failure, and the possible need for treatment or discontinuation of TRACLEER [see Adverse Reactions (6.1), Clinical Studies (14.2)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are described elsewhere in the labeling:
- Hepatotoxicity [see Boxed Warning, Warnings and Precautions (5.1)]
- Embryo-fetal Toxicity [see Boxed Warning, Warnings and Precautions (5.3)]
- Fluid Retention [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
4.4 Hypersensitivity
TRACLEER is contraindicated in patients who are hypersensitive to bosentan or any component of the product. Observed reactions include Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), anaphylaxis, rash, and angioedema [see Adverse Reactions (6.2), Description (11)] .
8.7 Renal Impairment
The effect of renal impairment on the pharmacokinetics of bosentan is small and does not require dosing adjustment [see Clinical Pharmacology (12.3)] .
2.2 Recommended Dosage
Administer TRACLEER orally following the dosing recommendations in Table 1. Doses above 125 mg twice daily did not appear to confer additional benefit sufficient to offset the increased risk of hepatotoxicity.
| Initial 4 weeks | Maintenance (after 4 weeks) | |
|---|---|---|
| Patients >12 years of age and >40 kg | 62.5 mg twice daily | 125 mg twice daily |
| Patients >12 years of age and <40 kg | 62.5 mg twice daily | 62.5 mg twice daily |
| Patients ≤12 years of age | ||
| ≥4–8 kg | 16 mg twice daily | 16 mg twice daily |
| >8–16 kg | 32 mg twice daily | 32 mg twice daily |
| >16–24 kg | 48 mg twice daily | 48 mg twice daily |
| >24–40 kg | 64 mg twice daily | 64 mg twice daily |
4.3 Use With Glyburide (4.3 Use with Glyburide)
An increased risk of liver enzyme elevations was observed in patients receiving glyburide concomitantly with bosentan. Therefore co-administration of glyburide and TRACLEER is contraindicated [see Drug Interactions (7.1)] .
8.6 Hepatic Impairment
Because there is in vitro and in vivo evidence that the main route of excretion of bosentan is biliary, liver impairment could be expected to increase exposure (C max and AUC) of bosentan. The pharmacokinetics of TRACLEER have not been evaluated in patients with severe liver impairment (Child-Pugh Class C). In patients with moderate hepatic impairment (Child-Pugh Class B), the systemic exposures to bosentan and its active metabolite increased significantly. TRACLEER should generally be avoided in patients with moderate or severe liver impairment. Pharmacokinetics of bosentan were not altered in patients with mild impairment of hepatic function (Child-Pugh Class A) [see Dosage and Administration (2.6), Warnings and Precautions (5.1), Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
TRACLEER is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1):
- in adults to improve exercise ability and to decrease clinical worsening. Studies establishing effectiveness included predominantly patients with WHO Functional Class II-IV symptoms and etiologies of idiopathic or heritable PAH (60%), PAH associated with connective tissue diseases (21%), and PAH associated with congenital heart disease with left-to-right shunts (18%) [see Clinical Studies (14.1)] .
- in pediatric patients aged 3 years and older with idiopathic or congenital PAH to improve pulmonary vascular resistance (PVR), which is expected to result in an improvement in exercise ability.
2.1 Required Monitoring
Healthcare professionals who prescribe TRACLEER must enroll in the Bosentan REMS and must comply with the required monitoring to minimize the risks associated with TRACLEER [see Warnings and Precautions (5.2)].
Measure liver aminotransferase levels prior to initiation of treatment and then monthly [see Boxed Warning, Warnings and Precautions (5.1, 5.2)].
Exclude pregnancy before initiating treatment with TRACLEER in females of reproductive potential [see Boxed Warning, Contraindications (4.1), Warnings and Precautions (5.3), Use in Specific Populations (8.1, 8.3)] .
12.1 Mechanism of Action
Bosentan is a specific and competitive antagonist at endothelin receptor types ET A and ET B. Bosentan has a slightly higher affinity for ET A receptors than for ET B receptors. The clinical impact of dual endothelin blockage is unknown.
Endothelin-1 (ET-1) is a neurohormone, the effects of which are mediated by binding to ET A and ET B receptors in the endothelium and vascular smooth muscle. ET-1 concentrations are elevated in plasma and lung tissue of patients with PAH, suggesting a pathogenic role for ET-1 in this disease.
5.3 Embryo Fetal Toxicity (5.3 Embryo-Fetal Toxicity)
Based on data from animal reproduction studies, TRACLEER may cause fetal harm when administered to a pregnant female and is contraindicated in females who are pregnant. The available human data for endothelin receptor antagonists do not establish the presence or absence of major birth defects related to the use of TRACLEER. Advise females of reproductive potential about the potential risk to a fetus. Exclude pregnancy prior to TRACLEER treatment. Advise females of reproductive potential to use effective contraception prior to initiation of treatment with TRACLEER, during treatment, and for at least one month after the last dose. When pregnancy is detected, discontinue TRACLEER as soon as possible [see Dosage and Administration (2.1), Contraindications (4.1), Drug Interactions (7.2), Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Fluid retention: May require intervention ( 5.4).
- Pulmonary veno-occlusive disease (PVOD): If signs of pulmonary edema occur, consider the diagnosis of associated PVOD and consider discontinuing TRACLEER ( 5.5).
- Decreased sperm counts ( 5.6).
- Decreases in hemoglobin and hematocrit: Monitor hemoglobin levels after 1 and 3 months of treatment, then every 3 months thereafter ( 5.7).
5.6 Decreased Sperm Counts
Decreased sperm counts have been observed in patients receiving TRACLEER. Preclinical data also suggest that TRACLEER, similar to other endothelin receptor antagonists, may have an adverse effect on spermatogenesis [see Adverse Reactions (6.1), Nonclinical Toxicology (13.1)] .
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Patients older than 12 years of age: initiate at 62.5 mg orally twice daily; for patients weighing greater than 40 kg, increase to 125 mg orally twice daily after 4 weeks ( 2.2).
- Patients 12 years of age and younger: dosage is based on weight, see Table 1( 2.2).
- Reduce the dose and closely monitor patients developing aminotransferase elevations more than 3×Upper Limit of Normal (ULN) ( 2.4).
4.2 Use With Cyclosporine A (4.2 Use with Cyclosporine A)
Co-administration of cyclosporine A and bosentan resulted in markedly increased plasma concentrations of bosentan. Therefore, concomitant use of TRACLEER and cyclosporine A is contraindicated [see Drug Interactions (7.1)] .
7.2 Hormonal Contraceptives
Hormonal contraceptives, including oral, injectable, transdermal, and implantable forms, may not be reliable when TRACLEER is co-administered. Females should practice additional methods of contraception and not rely on hormonal contraception alone when taking TRACLEER [see Use in Specific Populations (8.3)] .
An interaction study demonstrated that co-administration of bosentan and a combination oral hormonal contraceptive produced average decreases of norethindrone and ethinyl estradiol levels of 14% and 31%, respectively. However, decreases in exposure were as much as 56% and 66%, respectively, in individual subjects.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
62.5 mg tablets: round, biconvex, orange-white tablets, debossed with identification marking "62,5"
125 mg tablets: oval, biconvex, orange-white tablets, debossed with identification marking "125"
32 mg tablets for oral suspension: round, pale yellow to off-white tablets, bisected on one side and debossed with identification marking “32” on the other side.
6.2 Postmarketing Experience
There have been several postmarketing reports of angioedema associated with the use of TRACLEER. The onset of the reported cases occurred within a range of 8 hours to 21 days after starting therapy. Some patients were treated with an antihistamine and their signs of angioedema resolved without discontinuing TRACLEER.
The following additional adverse reactions have been reported during the post approval use of TRACLEER. Because these adverse reactions are reported from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to TRACLEER exposure:
- Unexplained hepatic cirrhosis [see Boxed Warning]
- Liver failure [see Boxed Warning]
- Hypersensitivity, DRESS, and anaphylaxis [see Contraindications (4.4)]
- Thrombocytopenia
- Rash
- Jaundice
- Anemia requiring transfusion
- Neutropenia and leukopenia
- Nasal congestion
- Autoimmune hepatitis
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Nursing mothers: Choose breastfeeding or TRACLEER ( 8.2).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Safety data on TRACLEER were obtained from 13 clinical studies (9 placebo-controlled and 4 open-label) in 870 adult patients with PAH and other diseases. Doses up to 8 times the currently recommended clinical dose (125 mg twice daily) were administered for a variety of durations. The exposure to TRACLEER in these trials ranged from 1 day to 4.1 years (n=94 for 1 year; n=61 for 1.5 years; and n=39 for more than 2 years). Exposure of PAH patients (n=328) to TRACLEER ranged from 1 day to 1.7 years (n=174 more than 6 months and n=28 more than 12 months).
Treatment discontinuations due to adverse events other than those related to pulmonary hypertension during the clinical trials in adult patients with PAH were more frequent on TRACLEER (6%; 15/258 patients) than on placebo (3%; 5/172 patients). In this database the only cause of discontinuations >1% and occurring more often on TRACLEER was abnormal liver function.
The adverse drug events that occurred in ≥3% of the TRACLEER-treated patients and were more common on TRACLEER in placebo-controlled trials in PAH at doses of 125 or 250 mg twice daily are shown in Table 3:
| Adverse Event | TRACLEER
n=258 |
Placebo
n=172 |
||
|---|---|---|---|---|
| No. | % | No. | % | |
| Respiratory Tract Infection
Respiratory Tract Infection combines the terms "Nasopharyngitis", "Upper Respiratory Tract Infection" and "Respiratory Tract Infection". Combined data from Study 351, BREATHE-1 and EARLY
|
56 | 22% | 30 | 17% |
| Headache | 39 | 15% | 25 | 14% |
| Edema | 28 | 11% | 16 | 9% |
| Chest Pain | 13 | 5% | 8 | 5% |
| Syncope | 12 | 5% | 7 | 4% |
| Flushing | 10 | 4% | 5 | 3% |
| Hypotension | 10 | 4% | 3 | 2% |
| Sinusitis | 9 | 4% | 4 | 2% |
| Arthralgia | 9 | 4% | 3 | 2% |
| Serum Aminotransferases, abnormal | 9 | 4% | 3 | 2% |
| Palpitations | 9 | 4% | 3 | 2% |
| Anemia | 8 | 3% | - | - |
TRACLEER was evaluated for safety in 119 pediatric patients in uncontrolled studies. The safety profile was similar to that observed in adult patients with PAH.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide)
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
62.5 mgfilm-coated, round, biconvex, orange-white tablets, debossed with identification marking "62,5", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing.
NDC 66215-101-06: Bottle containing 60 tablets.
NDC 66215-101-03: Carton of 30 tablets in 10 blister strips of 3 tablets.
125 mgfilm-coated, oval, biconvex, orange-white tablets, debossed with identification marking "125", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap or in foil blister-strips for hospital unit-dosing.
NDC 66215-102-06: Bottle containing 60 tablets.
NDC 66215-102-03: Carton of 30 tablets in 10 blister strips of 3 tablets.
32 mgtablets for oral suspension, pale yellow to off-white, round, bisected on one side and debossed with identification marking "32" on the other side, packaged in child-resistant Aluminum/Aluminum peel-push blisters.
NDC 66215-232-56: Carton of 56 tablets for oral suspension in 4 blister-strips of 14 tablets.
NDC 66215-232-14: Blister strip of 14 tablets for oral suspension.
5.5 Pulmonary Veno Occlusive Disease (5.5 Pulmonary Veno-Occlusive Disease)
If signs of pulmonary edema occur, consider the possibility of associated pulmonary veno-occlusive disease and consider whether TRACLEER should be discontinued.
7.1 Cytochrome P450 Drug Interactions
Bosentan is metabolized by CYP2C9 and CYP3A. Inhibition of these enzymes may increase the plasma concentration of bosentan [see Clinical Pharmacology (12.3)] . Concomitant administration of both a CYP2C9 inhibitor (such as fluconazole or amiodarone) and a strong CYP3A inhibitor (e.g., ketoconazole, itraconazole) or a moderate CYP3A inhibitor (e.g., amprenavir, erythromycin, fluconazole, diltiazem) with TRACLEER will likely lead to large increases in plasma concentrations of bosentan. Co-administration of such combinations of a CYP2C9 inhibitor plus a strong or moderate CYP3A inhibitor with TRACLEER is not recommended.
Bosentan is an inducer of CYP3A and CYP2C9. Consequently plasma concentrations of drugs metabolized by these two isozymes will be decreased when TRACLEER is co-administered. Bosentan had no relevant inhibitory effect on any CYP isozyme in vitro(CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A). Consequently, TRACLEER is not expected to increase the plasma concentrations of drugs metabolized by these enzymes.
Figure 1. CYP3A induction-mediated effect of bosentan on other drugs
Figure 2. Effect of other drugs on bosentan
5.7 Decreases in Hemoglobin and Hematocrit
Treatment with TRACLEER can cause a dose-related decrease in hemoglobin and hematocrit. There have been postmarketing reports of decreases in hemoglobin concentration and hematocrit that have resulted in anemia requiring transfusion. It is recommended that hemoglobin concentrations be checked after 1 and 3 months, and every 3 months thereafter. If a marked decrease in hemoglobin concentration occurs, further evaluation should be undertaken to determine the cause and need for specific treatment [see Adverse Reactions (6.1)] .
8.3 Females and Males of Reproductive Potential
Based on data from animal reproductive toxicity studies, TRACLEER may cause fetal harm, including birth defects and fetal death, when administered to a pregnant patient and is contraindicated during pregnancy [see Contraindications (4.1), Use in Specific Populations (8.1)].
14.2 Lack of Benefit in Congestive Heart Failure
TRACLEER is not effective in the treatment of congestive heart failure with left ventricular dysfunction. In a pair of studies, 1613 subjects with NYHA Class III-IV heart failure, left ventricular ejection fraction <35%, on diuretics, ACE inhibitor, and other therapies, were randomized to placebo or TRACLEER (62.5 mg twice daily titrated as tolerated to 125 mg twice daily) and followed for up to 70 weeks. Use of TRACLEER was associated with no benefit on patient global assessment (the primary end point) or mortality. However, hospitalizations for heart failure were more common during the first 4 to 8 weeks after TRACLEER was initiated. In a placebo-controlled trial of patients with severe chronic heart failure, there was an increased incidence of hospitalization for CHF associated with weight gain and increased leg edema during the first 4–8 weeks of treatment with TRACLEER. Patients required intervention with a diuretic, fluid management, or hospitalization for decompensating heart failure.
Principal Display Panel 125 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 125 mg Tablet Bottle Carton)
NDC 66215-102-06
Tracleer
®
(bosentan) tablets
125 mg
Attention: Dispense with
enclosed Medication Guide.
Rx only
60 film-coated tablets
janssen
2.4 Dosage Adjustments for Aminotransferase Elevations
If aminotransferase levels increase, adjust monitoring and treatment plan according to Table 2.
Discontinue TRACLEER if liver aminotransferase elevations are accompanied by clinical symptoms of hepatotoxicity (such as nausea, vomiting, fever, abdominal pain, jaundice, or unusual lethargy or fatigue) or bilirubin ≥2 ×Upper Limit of Normal (ULN). There is no experience with the reintroduction of TRACLEER in these circumstances.
| ALT/AST levels | Treatment and monitoring recommendations |
|---|---|
| >3 and ≤5 ×ULN | Confirm by another aminotransferase test; if confirmed,
|
| >5 and ≤8 ×ULN | Confirm by another aminotransferase test; if confirmed, stop treatment and monitor aminotransferase levels at least every 2 weeks. Once the aminotransferase levels return to pretreatment values,
|
| >8 ×ULN | Stop treatment permanently. There is no experience with reintroduction of TRACLEER in these circumstances. |
Principal Display Panel 62.5 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 62.5 mg Tablet Bottle Carton)
NDC 66215-101-06
Tracleer
®
(bosentan) tablets
62.5 mg
Attention: Dispense with
enclosed Medication Guide.
Rx only
60 film-coated tablets
janssen
2.6 Use in Patients With Pre Existing Hepatic Impairment (2.6 Use in Patients with Pre-existing Hepatic Impairment)
Avoid initiation of TRACLEER in patients with aminotransferases >3 ×ULN. No dose adjustment is required in patients with mildly impaired liver function [see Warnings and Precautions (5.1), Use in Specific Populations (8.6), Clinical Pharmacology (12.3)] .
Warning: Risks of Hepatotoxicity and Embryo Fetal Toxicity (WARNING: RISKS OF HEPATOTOXICITY and EMBRYO-FETAL TOXICITY)
Because of the risk of hepatotoxicity, TRACLEER is available only through a restricted program called the Bosentan Risk Evaluation and Mitigation Strategy (REMS). Under the Bosentan REMS, prescribers, patients, and pharmacies must enroll in the program [see Warnings and Precautions (5.2)] .
Principal Display Panel 32 Mg Tablet Blister Pack Carton 232 (PRINCIPAL DISPLAY PANEL - 32 mg Tablet Blister Pack Carton - 232)
NDC 66215-232-56
Tracleer
®
(bosentan) Tablets for Oral Suspension
32 mg
Phenylketonurics:
Contains Phenylalanine
1.87 mg per tablet for oral suspension
Rx only
Contains: 56 tablets for oral suspension per carton.
Each carton contains 4 blister-strips of 14 tablets each.
Attention: Dispense with
enclosed Medication Guide.
This package is child-resistant.
Store this medicine out of
the reach of children.
janssen
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:34.999536 · Updated: 2026-03-14T22:44:06.575433