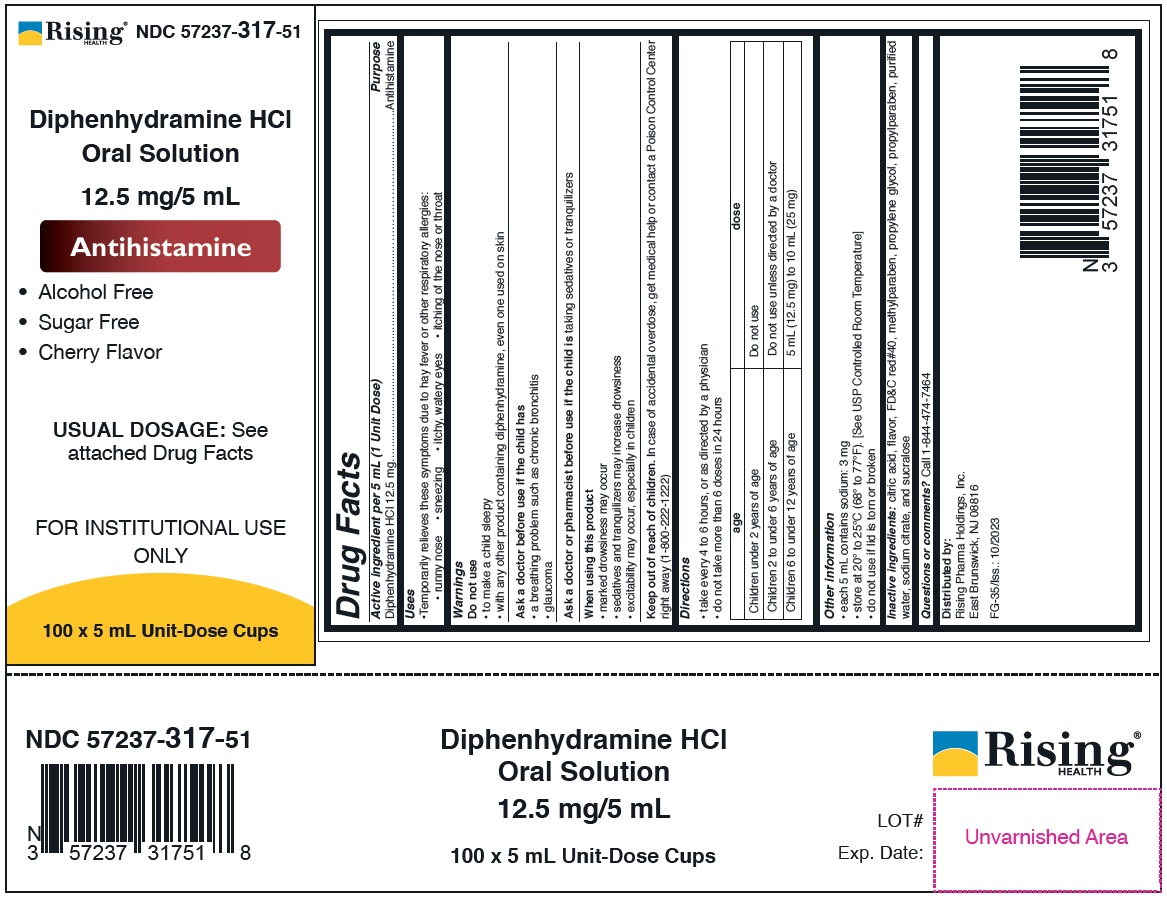
diphenhydramine hcl oral solution 12.5 mg/5 ml
7406e4e4-3093-4cd1-80ce-93f7b6db2a87
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antihistamine
Description
Drug Facts
Medication Information
Active Ingredient
Diphenhydramine HCl 12.5 mg
Uses
• Temporarily relieves these symptoms due to hay fever or other respiratory allergies:
• runny nose • sneezing • itchy, watery eyes • itching of the nose or throat
Purpose
Antihistamine
Directions
• take every 4 to 6 hours, or as directed by a physician
• do not take more than 6 doses in 24 hours
| age | dose |
|---|---|
| Children under 2 years of age | Do not use |
| Children 2 to under 6 years of age | Do not use unless directed by a doctor |
| Children 6 to under 12 years of age | 5 mL (12.5 mg) to 10 mL (25 mg) |
Other Information
• each 5 mL contains sodium: 3 mg
• store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature]
• do not use if lid is torn or broken
Description
Drug Facts
Principal Display Panel
12.5 mg/5 mL
Case Label NDC 57237-317-51
Lid Label NDC 57237-317-05
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222)
Section 50568-5
Ask a doctor or pharmacist before use if the child is taking sedatives or tranquilizers
Section 51727-6
Inactive ingredients: citric acid, flavor, FD&C red#40, methylparaben, propylene glycol, propylparaben, purified water, sodium citrate, and sucralose
Section 53413-1
Questions or comments? Call 1-844-474-7464
Do Not Use
• to make a child sleepy
• with any other product containing diphenhydramine, even one used on skin
Ndc Information
12.5 mg/5 mL
Each 5 mL of Diphenhydramine HCl Oral Solution contains Diphenhydramine HCl 12.5 mg and is supplied in the following oral dosage forms:
NDC 57237-317-05: 5 mL unit dose cup
NDC 57237-317-51: 100 x 5 mL Unit-Dose Cups
When Using This Product
• marked drowsiness may occur
• sedatives and tranquilizers may increase drowsiness
• excitability may occur, especially in children
Ask A Doctor Before Use If the Child Has
• a breathing problem such as chronic bronchitis
• glaucoma
Structured Label Content
Active Ingredient (55106-9)
Diphenhydramine HCl 12.5 mg
Uses
• Temporarily relieves these symptoms due to hay fever or other respiratory allergies:
• runny nose • sneezing • itchy, watery eyes • itching of the nose or throat
Purpose
Antihistamine
Directions
• take every 4 to 6 hours, or as directed by a physician
• do not take more than 6 doses in 24 hours
| age | dose |
|---|---|
| Children under 2 years of age | Do not use |
| Children 2 to under 6 years of age | Do not use unless directed by a doctor |
| Children 6 to under 12 years of age | 5 mL (12.5 mg) to 10 mL (25 mg) |
Other Information (Other information)
• each 5 mL contains sodium: 3 mg
• store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature]
• do not use if lid is torn or broken
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
12.5 mg/5 mL
Case Label NDC 57237-317-51
Lid Label NDC 57237-317-05
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222)
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if the child is taking sedatives or tranquilizers
Section 51727-6 (51727-6)
Inactive ingredients: citric acid, flavor, FD&C red#40, methylparaben, propylene glycol, propylparaben, purified water, sodium citrate, and sucralose
Section 53413-1 (53413-1)
Questions or comments? Call 1-844-474-7464
Do Not Use (Do not use)
• to make a child sleepy
• with any other product containing diphenhydramine, even one used on skin
Ndc Information (NDC Information -)
12.5 mg/5 mL
Each 5 mL of Diphenhydramine HCl Oral Solution contains Diphenhydramine HCl 12.5 mg and is supplied in the following oral dosage forms:
NDC 57237-317-05: 5 mL unit dose cup
NDC 57237-317-51: 100 x 5 mL Unit-Dose Cups
When Using This Product (When using this product)
• marked drowsiness may occur
• sedatives and tranquilizers may increase drowsiness
• excitability may occur, especially in children
Ask A Doctor Before Use If the Child Has (Ask a doctor before use if the child has)
• a breathing problem such as chronic bronchitis
• glaucoma
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:29.056117 · Updated: 2026-03-14T23:09:16.713853