These Highlights Do Not Include All The Information Needed To Use Tnkase Safely And Effectively. See Full Prescribing Information For Tnkase.
739a3c53-caf6-40d6-a6af-84ce733a948b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
TNKase ® is indicated to reduce the risk of death associated with acute ST elevation myocardial infarction (STEMI).
Indications and Usage
TNKase ® is indicated to reduce the risk of death associated with acute ST elevation myocardial infarction (STEMI).
Dosage and Administration
Initiate treatment as soon as possible after the onset of STEMI symptoms. ( 2.1 ) TNKase is for intravenous administration only, administered as a single bolus over 5 seconds. Individualize dosage based on patient's weight. ( 2.1 )
Warnings and Precautions
Bleeding: Increases the risk of bleeding. Avoid intramuscular injections. Monitor for bleeding. ( 5.1 ) Thromboembolism: The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus. ( 5.2 ) Cholesterol Embolization: Has been reported in patients treated with thrombolytic agents. ( 5.3 ) Arrhythmias: It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered. ( 5.4 ) Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention (PCI) in STEMI: In patients with a large ST segment elevation myocardial infarction, physicians should choose either thrombolysis or PCI as the primary treatment strategy for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate. ( 5.5 ) Hypersensitivity: Monitor patients treated with TNKase during and for several hours after infusion. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids). ( 5.6 )
Contraindications
TNKase is contraindicated in patients with [see Warnings and Precautions (5.1) ] : Active internal bleeding History of cerebrovascular accident Intracranial or intraspinal surgery or trauma within 2 months Intracranial neoplasm, arteriovenous malformation, or aneurysm Known bleeding diathesis Severe uncontrolled hypertension
Adverse Reactions
The following adverse reactions are discussed in other sections of the label: Bleeding [see Contraindications (4) , Warnings and Precautions (5.1) ] Hypersensitivity [see Warnings and Precautions (5.6) ]
Drug Interactions
During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. ( 7.1 )
How Supplied
TNKase (tenecteplase) for injection is supplied as a sterile, white to pale yellow lyophilized powder in a 50 mg single-dose vial under partial vacuum. Each 50 mg single-dose vial of TNKase is packaged with one 10 mL single-dose vial of Sterile Water for Injection, USP for reconstitution, and the B-D ® 10 mL syringe with TwinPak™ Dual Cannula Device: NDC 50242-120-47.
Medication Information
Warnings and Precautions
Bleeding: Increases the risk of bleeding. Avoid intramuscular injections. Monitor for bleeding. ( 5.1 ) Thromboembolism: The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus. ( 5.2 ) Cholesterol Embolization: Has been reported in patients treated with thrombolytic agents. ( 5.3 ) Arrhythmias: It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered. ( 5.4 ) Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention (PCI) in STEMI: In patients with a large ST segment elevation myocardial infarction, physicians should choose either thrombolysis or PCI as the primary treatment strategy for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate. ( 5.5 ) Hypersensitivity: Monitor patients treated with TNKase during and for several hours after infusion. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids). ( 5.6 )
Indications and Usage
TNKase ® is indicated to reduce the risk of death associated with acute ST elevation myocardial infarction (STEMI).
Dosage and Administration
Initiate treatment as soon as possible after the onset of STEMI symptoms. ( 2.1 ) TNKase is for intravenous administration only, administered as a single bolus over 5 seconds. Individualize dosage based on patient's weight. ( 2.1 )
Contraindications
TNKase is contraindicated in patients with [see Warnings and Precautions (5.1) ] : Active internal bleeding History of cerebrovascular accident Intracranial or intraspinal surgery or trauma within 2 months Intracranial neoplasm, arteriovenous malformation, or aneurysm Known bleeding diathesis Severe uncontrolled hypertension
Adverse Reactions
The following adverse reactions are discussed in other sections of the label: Bleeding [see Contraindications (4) , Warnings and Precautions (5.1) ] Hypersensitivity [see Warnings and Precautions (5.6) ]
Drug Interactions
During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. ( 7.1 )
How Supplied
TNKase (tenecteplase) for injection is supplied as a sterile, white to pale yellow lyophilized powder in a 50 mg single-dose vial under partial vacuum. Each 50 mg single-dose vial of TNKase is packaged with one 10 mL single-dose vial of Sterile Water for Injection, USP for reconstitution, and the B-D ® 10 mL syringe with TwinPak™ Dual Cannula Device: NDC 50242-120-47.
Description
TNKase ® is indicated to reduce the risk of death associated with acute ST elevation myocardial infarction (STEMI).
Section 42229-5
Risk Summary
There are risks to the mother and fetus from acute ST elevation myocardial infarction, which is a medical emergency in pregnancy and can be fatal if left untreated (see Clinical Considerations). Published data consisting of a small number of case reports involving the use of related thrombolytic agents in pregnant women have not identified an increased risk of major birth defects. There are no data on the use of tenecteplase during pregnancy to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
TNKase does not elicit maternal and direct embryo toxicity in rabbits following a single IV administration. In developmental toxicity studies conducted in rabbits, the no observable effect level (NOEL) of a single IV administration of TNKase on maternal or developmental toxicity (5 mg/kg) was approximately 7 times human exposure (based on AUC) at the dose for STEMI.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
5.1 Bleeding
TNKase can cause bleeding, including intracranial hemorrhage and fatal bleeding. Concomitant use of other drugs that impair hemostasis increases the risk of bleeding.
Should serious bleeding that is not controlled by local pressure occur, discontinue any concomitant heparin or antiplatelet agents immediately and treat appropriately.
Avoid intramuscular injections and nonessential handling of the patient for the first few hours following treatment with TNKase. Perform arterial and venous punctures carefully and only as required. To minimize bleeding from noncompressible sites, avoid internal jugular and subclavian venous punctures. If an arterial puncture is necessary during TNKase infusion, use an upper extremity vessel that is accessible to manual compression. Apply pressure for at least 30 minutes.
11 Description
Tenecteplase is a tissue plasminogen activator (tPA) produced by recombinant DNA technology using a mammalian cell line (Chinese Hamster Ovary cells). Tenecteplase is a 527-amino acid glycoprotein developed by introducing the following modifications to the complementary DNA (cDNA) for natural human tPA: a substitution of threonine 103 with asparagine, and a substitution of asparagine 117 with glutamine, both within the kringle 1 domain, and a tetra-alanine substitution at amino acids 296–299 in the protease domain. It has a molecular weight of 58,742 daltons. Biological potency is determined by an in vitro clot lysis assay and is expressed in tenecteplase specific units. The specific activity of tenecteplase has been defined as 200 units/mg.
TNKase (tenecteplase) for injection is a sterile, white to pale yellow, lyophilized powder for intravenous bolus administration after reconstitution with Sterile Water for Injection, USP. Each single-dose vial of TNKase nominally contains 50 mg of tenecteplase, arginine (522 mg), phosphoric acid (approximately 160 mg), and polysorbate 20 (4.0 mg). Following reconstitution with the supplied 10 mL single-dose vial of Sterile Water for Injection, USP, the final concentration is 5 mg/mL with a pH of approximately 7.3.
2.2 Preparation
Follow the below steps to prepare TNKase for administration:
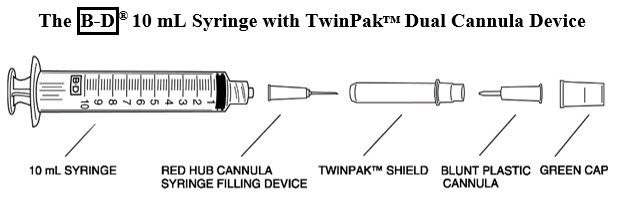
Remove the shield assembly from the supplied B-D® 10 mL syringe with TwinPak™ Dual Cannula Device (see Figure 1) and aseptically withdraw 10 mL of Sterile Water for Injection, USP, from the supplied diluent vial using the red hub cannula syringe filling device. Only use the supplied Sterile Water for Injection, USP for reconstitution.
- Note: Do not discard the shield assembly.
- Aseptically reconstitute the vial with 10 mL Sterile Water for Injection, USP by directing the stream into the lyophilized powder to obtain a final concentration of 5 mg/mL. Slight foaming upon reconstitution is not unusual; any large bubbles will dissipate if the product is allowed to stand undisturbed for several minutes.
- Gently swirl until contents are completely dissolved. DO NOT SHAKE. The reconstituted preparation results in a colorless to pale yellow transparent solution.
- Determine the appropriate dose of TNKase [see Dosage and Administration (2.1)] and withdraw this volume (in milliliters) from the reconstituted vial with the syringe. Discard any unused solution.
- Stand the shield vertically on a flat surface (with green side down) and passively recap the red hub cannula.
- Remove the entire shield assembly, including the red hub cannula, by twisting counterclockwise. Note: The shield assembly also contains the clear-ended blunt plastic cannula; retain for split septum intravenous access.
Figure 1
5.4 Arrhythmias
Coronary thrombolysis may result in arrhythmias associated with reperfusion. These arrhythmias (such as sinus bradycardia, accelerated idioventricular rhythm, ventricular premature depolarizations, ventricular tachycardia) may be managed with standard anti-arrhythmic measures. It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered.
16.1 How Supplied
TNKase (tenecteplase) for injection is supplied as a sterile, white to pale yellow lyophilized powder in a 50 mg single-dose vial under partial vacuum.
Each 50 mg single-dose vial of TNKase is packaged with one 10 mL single-dose vial of Sterile Water for Injection, USP for reconstitution, and the B-D® 10 mL syringe with TwinPak™ Dual Cannula Device: NDC 50242-120-47.
8.4 Pediatric Use
The safety and effectiveness of TNKase in pediatric patients have not been established.
8.5 Geriatric Use
In the ASSENT-2 study, 41% (3500/8458) of patients who were treated with TNKase were aged 65 years or older. In this population, rates of 30-day mortality, stroke, intracranial hemorrhage and major bleeds requiring blood transfusion or leading to hemodynamic complications were higher than in those aged less than 65 years.
2.3 Administration
Follow the below steps for administration of TNKase;
- Inspect the product prior to administration for particulate matter and discoloration. Administer TNKase as reconstituted at 5 mg/mL.
- Precipitation may occur when TNKase is administered in an intravenous line containing dextrose. Flush dextrose-containing lines with a saline-containing solution prior to and following single bolus administration of TNKase.
- Administer reconstituted TNKase as a single intravenous bolus over 5 seconds.
- Because TNKase contains no antibacterial preservatives, reconstitute immediately before use. If the reconstituted TNKase is not used immediately, refrigerate the TNKase vial at 2°C to 8°C (36°F to 46°F) and use within 8 hours.
- Although the supplied syringe is compatible with a conventional needle, this syringe is designed to be used with needleless intravenous systems. From the information below, follow the instructions applicable to the intravenous system in use.
Split septum intravenous system: - Remove the green cap.
- Attach the clear-ended blunt plastic cannula to the syringe.
- Remove the shield and use the blunt plastic cannula to access the split septum injection port.
- Because the blunt plastic cannula has two side ports, air or fluid expelled through the cannula will exit in two sideways directions; direct away from face or mucous membranes.
Luer-Lok® system: Connect syringe directly to intravenous port. Conventional needle
(not supplied in this kit):Attach a large bore needle, e.g., 18 gauge, to the syringe's universal Luer-Lok®. - Dispose of the syringe, cannula and shield per established procedures.
6.1 Immunogenicity
Four of 625 (0.64%) patients tested for antibody formation to TNKase had a positive antibody titer at 30 days in studies with TNKase. The observed incidence of antibody positivity in an assay may be influenced by several factors including sample handling, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to TNKase with the incidence of antibodies to other products may be misleading.
4 Contraindications
TNKase is contraindicated in patients with [see Warnings and Precautions (5.1)]:
- Active internal bleeding
- History of cerebrovascular accident
- Intracranial or intraspinal surgery or trauma within 2 months
- Intracranial neoplasm, arteriovenous malformation, or aneurysm
- Known bleeding diathesis
- Severe uncontrolled hypertension
5.2 Thromboembolism
The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus, such as patients with mitral stenosis or atrial fibrillation.
6 Adverse Reactions
The following adverse reactions are discussed in other sections of the label:
- Bleeding [see Contraindications (4), Warnings and Precautions (5.1)]
- Hypersensitivity [see Warnings and Precautions (5.6)]
7 Drug Interactions
- During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. (7.1)
5.6 Hypersensitivity
Hypersensitivity, including urticarial / anaphylactic reactions, have been reported after administration of TNKase (e.g., anaphylaxis, angioedema, laryngeal edema, rash, and urticaria). Monitor patients treated with TNKase during and for several hours after infusion. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids).
12.2 Pharmacodynamics
Following administration of 30, 40, or 50 mg of TNKase, there are decreases in circulating fibrinogen (4%–15%) and plasminogen (11%–24%).
2.1 Recommended Dosage
Initiate treatment as soon as possible after the onset of STEMI symptoms.
TNKase is for intravenous (IV) administration only, administered as a single bolus over 5 seconds. Individualize dosage based on the patient's weight (see Table 1).
| Patient Weight (kg) |
TNKase (mg) |
Volume TNKase From one vial of TNKase reconstituted with 10 mL Sterile Water for Injection. to be administered (mL) |
|---|---|---|
| < 60 | 30 | 6 |
| ≥ 60 to < 70 | 35 | 7 |
| ≥ 70 to < 80 | 40 | 8 |
| ≥ 80 to < 90 | 45 | 9 |
| ≥ 90 | 50 | 10 |
1 Indications and Usage
TNKase® is indicated to reduce the risk of death associated with acute ST elevation myocardial infarction (STEMI).
12.1 Mechanism of Action
Tenecteplase is a modified form of human tissue plasminogen activator (tPA) that binds to fibrin and converts plasminogen to plasmin. In the presence of fibrin, in vitro studies demonstrate that tenecteplase-mediated conversion of plasminogen to plasmin is increased relative to its conversion in the absence of fibrin. This fibrin specificity decreases systemic activation of plasminogen and the resulting degradation of circulating fibrinogen as compared to a molecule lacking this property. The clinical significance of fibrin-specificity on safety (e.g., bleeding) or efficacy has not been established.
16.2 Stability and Storage
Store lyophilized TNKase at room temperature up to 30°C (86°F) or refrigerated at 2°C to 8°C (36°F to 46°F). Do not use beyond the expiration date stamped on the vial.
5 Warnings and Precautions
- Bleeding: Increases the risk of bleeding. Avoid intramuscular injections. Monitor for bleeding. (5.1)
- Thromboembolism: The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus. (5.2)
- Cholesterol Embolization: Has been reported in patients treated with thrombolytic agents. (5.3)
- Arrhythmias: It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered. (5.4)
- Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention (PCI) in STEMI: In patients with a large ST segment elevation myocardial infarction, physicians should choose either thrombolysis or PCI as the primary treatment strategy for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate. (5.5)
- Hypersensitivity: Monitor patients treated with TNKase during and for several hours after infusion. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids). (5.6)
2 Dosage and Administration
3 Dosage Forms and Strengths
For injection: 50 mg as a white to pale yellow lyophilized powder in a single-dose vial for reconstitution with the co-packaged 10 mL single-dose vial of Sterile Water for Injection, USP (diluent).
5.3 Cholesterol Embolization
Cholesterol embolism has been reported in patients treated with thrombolytic agents. Investigate cause of any new embolic event and treat appropriately.
2.4 Chemical Incompatibilities
TNKase is incompatible with dextrose containing solutions. When used together, precipitation may occur. Flush dextrose containing lines with saline-containing solution before using TNKase.
Principal Display Panel Kit Carton
NDC 50242-120-47
Tenecteplase
TNKase®
50 mg
For use in myocardial infarction
Kit Contents: Each kit contains one 50 mg vial of TNKase, one 10 mL vial of preservative-free
Sterile Water for Injection, USP, one BD® 10 mL syringe with TwinPak™ Dual Cannula Device,
and package insert containing full prescribing information.
Vial Contents: The preservative-free single-use vial of TNKase contains 52.5 mg
Tenecteplase, 0.55 g L-arginine, 0.17 g phosphoric acid, and 4.3 mg polysorbate 20,
under partial vacuum. No U.S. standard of potency.
Rx only
US License No.: 1048
Genentech
10200128
7.1 Drug/laboratory Test Interactions
During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. Tenecteplase is an enzyme that, when present in blood in pharmacologic concentrations, remains active under in vitro conditions. This can lead to degradation of fibrinogen in blood samples removed for analysis.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in animals have not been performed to evaluate the carcinogenic potential, mutagenicity, or the effect on fertility.
5.5 Increased Risk of Heart Failure and Recurrent Ischemia When Used With Planned Percutaneous Coronary Intervention (pci) in Stemi.
In a trial of patients with STEMI, there were trends toward worse outcomes in the individual components of the primary endpoint between TNKase plus PCI versus PCI alone (mortality 6.7% vs. 4.9%, respectively; cardiogenic shock 6.3% vs. 4.8%, respectively; and CHF 12% vs. 9.2%, respectively). In addition, there were trends towards worse outcomes in recurrent MI (6.1% vs. 3.7%, respectively; p = 0.03) and repeat target vessel revascularization (6.6% vs. 3.4%, respectively; p = 0.0045) in patients receiving TNKase plus PCI versus PCI alone [see Clinical Studies (14.1)]. In patients with large ST segment elevation myocardial infarction, physicians should choose either thrombolysis or PCI as the primary treatment strategy for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate; however, the optimal use of adjunctive antithrombotic and antiplatelet therapies in this setting is unknown.
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
There are risks to the mother and fetus from acute ST elevation myocardial infarction, which is a medical emergency in pregnancy and can be fatal if left untreated (see Clinical Considerations). Published data consisting of a small number of case reports involving the use of related thrombolytic agents in pregnant women have not identified an increased risk of major birth defects. There are no data on the use of tenecteplase during pregnancy to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
TNKase does not elicit maternal and direct embryo toxicity in rabbits following a single IV administration. In developmental toxicity studies conducted in rabbits, the no observable effect level (NOEL) of a single IV administration of TNKase on maternal or developmental toxicity (5 mg/kg) was approximately 7 times human exposure (based on AUC) at the dose for STEMI.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
5.1 Bleeding
TNKase can cause bleeding, including intracranial hemorrhage and fatal bleeding. Concomitant use of other drugs that impair hemostasis increases the risk of bleeding.
Should serious bleeding that is not controlled by local pressure occur, discontinue any concomitant heparin or antiplatelet agents immediately and treat appropriately.
Avoid intramuscular injections and nonessential handling of the patient for the first few hours following treatment with TNKase. Perform arterial and venous punctures carefully and only as required. To minimize bleeding from noncompressible sites, avoid internal jugular and subclavian venous punctures. If an arterial puncture is necessary during TNKase infusion, use an upper extremity vessel that is accessible to manual compression. Apply pressure for at least 30 minutes.
11 Description (11 DESCRIPTION)
Tenecteplase is a tissue plasminogen activator (tPA) produced by recombinant DNA technology using a mammalian cell line (Chinese Hamster Ovary cells). Tenecteplase is a 527-amino acid glycoprotein developed by introducing the following modifications to the complementary DNA (cDNA) for natural human tPA: a substitution of threonine 103 with asparagine, and a substitution of asparagine 117 with glutamine, both within the kringle 1 domain, and a tetra-alanine substitution at amino acids 296–299 in the protease domain. It has a molecular weight of 58,742 daltons. Biological potency is determined by an in vitro clot lysis assay and is expressed in tenecteplase specific units. The specific activity of tenecteplase has been defined as 200 units/mg.
TNKase (tenecteplase) for injection is a sterile, white to pale yellow, lyophilized powder for intravenous bolus administration after reconstitution with Sterile Water for Injection, USP. Each single-dose vial of TNKase nominally contains 50 mg of tenecteplase, arginine (522 mg), phosphoric acid (approximately 160 mg), and polysorbate 20 (4.0 mg). Following reconstitution with the supplied 10 mL single-dose vial of Sterile Water for Injection, USP, the final concentration is 5 mg/mL with a pH of approximately 7.3.
2.2 Preparation
Follow the below steps to prepare TNKase for administration:
Remove the shield assembly from the supplied B-D® 10 mL syringe with TwinPak™ Dual Cannula Device (see Figure 1) and aseptically withdraw 10 mL of Sterile Water for Injection, USP, from the supplied diluent vial using the red hub cannula syringe filling device. Only use the supplied Sterile Water for Injection, USP for reconstitution.
- Note: Do not discard the shield assembly.
- Aseptically reconstitute the vial with 10 mL Sterile Water for Injection, USP by directing the stream into the lyophilized powder to obtain a final concentration of 5 mg/mL. Slight foaming upon reconstitution is not unusual; any large bubbles will dissipate if the product is allowed to stand undisturbed for several minutes.
- Gently swirl until contents are completely dissolved. DO NOT SHAKE. The reconstituted preparation results in a colorless to pale yellow transparent solution.
- Determine the appropriate dose of TNKase [see Dosage and Administration (2.1)] and withdraw this volume (in milliliters) from the reconstituted vial with the syringe. Discard any unused solution.
- Stand the shield vertically on a flat surface (with green side down) and passively recap the red hub cannula.
- Remove the entire shield assembly, including the red hub cannula, by twisting counterclockwise. Note: The shield assembly also contains the clear-ended blunt plastic cannula; retain for split septum intravenous access.
Figure 1
5.4 Arrhythmias
Coronary thrombolysis may result in arrhythmias associated with reperfusion. These arrhythmias (such as sinus bradycardia, accelerated idioventricular rhythm, ventricular premature depolarizations, ventricular tachycardia) may be managed with standard anti-arrhythmic measures. It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered.
16.1 How Supplied
TNKase (tenecteplase) for injection is supplied as a sterile, white to pale yellow lyophilized powder in a 50 mg single-dose vial under partial vacuum.
Each 50 mg single-dose vial of TNKase is packaged with one 10 mL single-dose vial of Sterile Water for Injection, USP for reconstitution, and the B-D® 10 mL syringe with TwinPak™ Dual Cannula Device: NDC 50242-120-47.
8.4 Pediatric Use
The safety and effectiveness of TNKase in pediatric patients have not been established.
8.5 Geriatric Use
In the ASSENT-2 study, 41% (3500/8458) of patients who were treated with TNKase were aged 65 years or older. In this population, rates of 30-day mortality, stroke, intracranial hemorrhage and major bleeds requiring blood transfusion or leading to hemodynamic complications were higher than in those aged less than 65 years.
2.3 Administration
Follow the below steps for administration of TNKase;
- Inspect the product prior to administration for particulate matter and discoloration. Administer TNKase as reconstituted at 5 mg/mL.
- Precipitation may occur when TNKase is administered in an intravenous line containing dextrose. Flush dextrose-containing lines with a saline-containing solution prior to and following single bolus administration of TNKase.
- Administer reconstituted TNKase as a single intravenous bolus over 5 seconds.
- Because TNKase contains no antibacterial preservatives, reconstitute immediately before use. If the reconstituted TNKase is not used immediately, refrigerate the TNKase vial at 2°C to 8°C (36°F to 46°F) and use within 8 hours.
- Although the supplied syringe is compatible with a conventional needle, this syringe is designed to be used with needleless intravenous systems. From the information below, follow the instructions applicable to the intravenous system in use.
Split septum intravenous system: - Remove the green cap.
- Attach the clear-ended blunt plastic cannula to the syringe.
- Remove the shield and use the blunt plastic cannula to access the split septum injection port.
- Because the blunt plastic cannula has two side ports, air or fluid expelled through the cannula will exit in two sideways directions; direct away from face or mucous membranes.
Luer-Lok® system: Connect syringe directly to intravenous port. Conventional needle
(not supplied in this kit):Attach a large bore needle, e.g., 18 gauge, to the syringe's universal Luer-Lok®. - Dispose of the syringe, cannula and shield per established procedures.
6.1 Immunogenicity
Four of 625 (0.64%) patients tested for antibody formation to TNKase had a positive antibody titer at 30 days in studies with TNKase. The observed incidence of antibody positivity in an assay may be influenced by several factors including sample handling, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to TNKase with the incidence of antibodies to other products may be misleading.
4 Contraindications (4 CONTRAINDICATIONS)
TNKase is contraindicated in patients with [see Warnings and Precautions (5.1)]:
- Active internal bleeding
- History of cerebrovascular accident
- Intracranial or intraspinal surgery or trauma within 2 months
- Intracranial neoplasm, arteriovenous malformation, or aneurysm
- Known bleeding diathesis
- Severe uncontrolled hypertension
5.2 Thromboembolism
The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus, such as patients with mitral stenosis or atrial fibrillation.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in other sections of the label:
- Bleeding [see Contraindications (4), Warnings and Precautions (5.1)]
- Hypersensitivity [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. (7.1)
5.6 Hypersensitivity
Hypersensitivity, including urticarial / anaphylactic reactions, have been reported after administration of TNKase (e.g., anaphylaxis, angioedema, laryngeal edema, rash, and urticaria). Monitor patients treated with TNKase during and for several hours after infusion. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids).
12.2 Pharmacodynamics
Following administration of 30, 40, or 50 mg of TNKase, there are decreases in circulating fibrinogen (4%–15%) and plasminogen (11%–24%).
2.1 Recommended Dosage
Initiate treatment as soon as possible after the onset of STEMI symptoms.
TNKase is for intravenous (IV) administration only, administered as a single bolus over 5 seconds. Individualize dosage based on the patient's weight (see Table 1).
| Patient Weight (kg) |
TNKase (mg) |
Volume TNKase From one vial of TNKase reconstituted with 10 mL Sterile Water for Injection. to be administered (mL) |
|---|---|---|
| < 60 | 30 | 6 |
| ≥ 60 to < 70 | 35 | 7 |
| ≥ 70 to < 80 | 40 | 8 |
| ≥ 80 to < 90 | 45 | 9 |
| ≥ 90 | 50 | 10 |
1 Indications and Usage (1 INDICATIONS AND USAGE)
TNKase® is indicated to reduce the risk of death associated with acute ST elevation myocardial infarction (STEMI).
12.1 Mechanism of Action
Tenecteplase is a modified form of human tissue plasminogen activator (tPA) that binds to fibrin and converts plasminogen to plasmin. In the presence of fibrin, in vitro studies demonstrate that tenecteplase-mediated conversion of plasminogen to plasmin is increased relative to its conversion in the absence of fibrin. This fibrin specificity decreases systemic activation of plasminogen and the resulting degradation of circulating fibrinogen as compared to a molecule lacking this property. The clinical significance of fibrin-specificity on safety (e.g., bleeding) or efficacy has not been established.
16.2 Stability and Storage
Store lyophilized TNKase at room temperature up to 30°C (86°F) or refrigerated at 2°C to 8°C (36°F to 46°F). Do not use beyond the expiration date stamped on the vial.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Bleeding: Increases the risk of bleeding. Avoid intramuscular injections. Monitor for bleeding. (5.1)
- Thromboembolism: The use of thrombolytics can increase the risk of thrombo-embolic events in patients with high likelihood of left heart thrombus. (5.2)
- Cholesterol Embolization: Has been reported in patients treated with thrombolytic agents. (5.3)
- Arrhythmias: It is recommended that anti-arrhythmic therapy for bradycardia and/or ventricular irritability be available when TNKase is administered. (5.4)
- Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention (PCI) in STEMI: In patients with a large ST segment elevation myocardial infarction, physicians should choose either thrombolysis or PCI as the primary treatment strategy for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate. (5.5)
- Hypersensitivity: Monitor patients treated with TNKase during and for several hours after infusion. If symptoms of hypersensitivity occur, initiate appropriate therapy (e.g., antihistamines, corticosteroids). (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: 50 mg as a white to pale yellow lyophilized powder in a single-dose vial for reconstitution with the co-packaged 10 mL single-dose vial of Sterile Water for Injection, USP (diluent).
5.3 Cholesterol Embolization
Cholesterol embolism has been reported in patients treated with thrombolytic agents. Investigate cause of any new embolic event and treat appropriately.
2.4 Chemical Incompatibilities
TNKase is incompatible with dextrose containing solutions. When used together, precipitation may occur. Flush dextrose containing lines with saline-containing solution before using TNKase.
Principal Display Panel Kit Carton (PRINCIPAL DISPLAY PANEL - Kit Carton)
NDC 50242-120-47
Tenecteplase
TNKase®
50 mg
For use in myocardial infarction
Kit Contents: Each kit contains one 50 mg vial of TNKase, one 10 mL vial of preservative-free
Sterile Water for Injection, USP, one BD® 10 mL syringe with TwinPak™ Dual Cannula Device,
and package insert containing full prescribing information.
Vial Contents: The preservative-free single-use vial of TNKase contains 52.5 mg
Tenecteplase, 0.55 g L-arginine, 0.17 g phosphoric acid, and 4.3 mg polysorbate 20,
under partial vacuum. No U.S. standard of potency.
Rx only
US License No.: 1048
Genentech
10200128
7.1 Drug/laboratory Test Interactions (7.1 Drug/Laboratory Test Interactions)
During TNKase therapy, results of coagulation tests and/or measures of fibrinolytic activity may be unreliable unless specific precautions are taken to prevent in vitro artifacts. Tenecteplase is an enzyme that, when present in blood in pharmacologic concentrations, remains active under in vitro conditions. This can lead to degradation of fibrinogen in blood samples removed for analysis.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in animals have not been performed to evaluate the carcinogenic potential, mutagenicity, or the effect on fertility.
5.5 Increased Risk of Heart Failure and Recurrent Ischemia When Used With Planned Percutaneous Coronary Intervention (pci) in Stemi. (5.5 Increased Risk of Heart Failure and Recurrent Ischemia when used with Planned Percutaneous Coronary Intervention (PCI) in STEMI.)
In a trial of patients with STEMI, there were trends toward worse outcomes in the individual components of the primary endpoint between TNKase plus PCI versus PCI alone (mortality 6.7% vs. 4.9%, respectively; cardiogenic shock 6.3% vs. 4.8%, respectively; and CHF 12% vs. 9.2%, respectively). In addition, there were trends towards worse outcomes in recurrent MI (6.1% vs. 3.7%, respectively; p = 0.03) and repeat target vessel revascularization (6.6% vs. 3.4%, respectively; p = 0.0045) in patients receiving TNKase plus PCI versus PCI alone [see Clinical Studies (14.1)]. In patients with large ST segment elevation myocardial infarction, physicians should choose either thrombolysis or PCI as the primary treatment strategy for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate; however, the optimal use of adjunctive antithrombotic and antiplatelet therapies in this setting is unknown.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:22.079976 · Updated: 2026-03-14T22:40:12.922880