Irbesartan And Hydrochlorothiazide Tablets
72fa8cd4-bc2a-4a7d-8feb-2d60e077d801
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
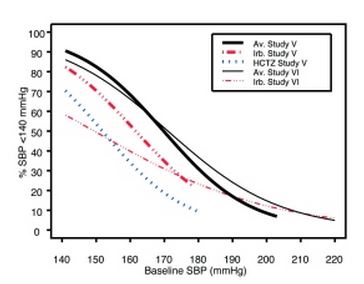
Irbesartan and hydrochlorothiazide tablets are indicated for the treatment of hypertension. Irbesartan and hydrochlorothiazide tablets may be used in patients whose blood pressure is not adequately controlled on monotherapy. Irbesartan and hydrochlorothiazide tablets may also be used as initial therapy in patients who are likely to need multiple drugs to achieve their blood pressure goals. The choice of irbesartan and hydrochlorothiazide tablets as initial therapy for hypertension should be based on an assessment of potential benefits and risks. Patients with stage 2 (moderate or severe) hypertension are at relatively high risk for cardiovascular events (such as strokes, heart attacks, and heart failure), kidney failure, and vision problems, so prompt treatment is clinically relevant. The decision to use a combination as initial therapy should be individualized and may be shaped by considerations such as the baseline blood pressure, the target goal, and the incremental likelihood of achieving goal with a combination compared with monotherapy. Data from Studies V and VI [see Clinical Studies (14.2) ] provide estimates of the probability of reaching a blood pressure goal with irbesartan and hydrochlorothiazide tablets compared to irbesartan or hydrochlorothiazide (HCTZ) monotherapy. The relationship between baseline blood pressure and achievement of a SeSBP <140 or <130 mmHg or SeDBP <90 or <80 mmHg in patients treated with irbesartan and hydrochlorothiazide tablets compared to patients treated with irbesartan or HCTZ monotherapy are shown in Figures 1a through 2b. Figure 1a: Probability of Achieving SBP <140 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)* Figure 1b: Probability of Achieving SBP <130 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)* Figure 2a: Probability of Achieving DBP <90 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)* Figure 2b: Probability of Achieving DBP <80 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)* The above graphs provide a rough approximation of the likelihood of reaching a targeted blood pressure goal (e.g., Week 8 sitting systolic blood pressure ≤140 mmHg) for the treatment groups. The curve of each treatment group in each study was estimated by logistic regression modeling from all available data of that treatment group. The estimated likelihood at the right tail of each curve is less reliable due to small numbers of subjects with high baseline blood pressures. For example, a patient with a blood pressure of 180/105 mmHg has about a 25% likelihood of achieving a goal of <140 mmHg (systolic) and 50% likelihood of achieving <90 mmHg (diastolic) on irbesartan alone (and lower still likelihoods on HCTZ alone). The likelihood of achieving these goals on irbesartan and hydrochlorothiazide tablets rises to about 40% (systolic) or 70% (diastolic).
Dosage and Administration
General Considerations • Maximum effects within 2 to 4 weeks after dose change. (2.1) • Renal impairment: Not recommended for patients with severe renal impairment (creatinine clearance <30 mL/min). (2.1 , 5.8) Hypertension • Initiate with 150 mg/12.5 mg. Titrate to 300 mg/12.5 mg then 300 mg/25 mg if needed. (2.2) • Replacement therapy: May be substituted for titrated components. (2.3)
Contraindications
• Irbesartan and hydrochlorothiazide tablets are contraindicated in patients who are hypersensitive to any component of this product. • Because of the hydrochlorothiazide component, this product is contraindicated in patients with anuria or hypersensitivity to other sulfonamide-derived drugs. • Do not coadminister aliskiren with irbesartan and hydrochlorothiazide tablets in patients with diabetes [see Drug Interactions (7) ] .
Warnings and Precautions
• Hypotension: Correct volume depletion prior to administration. (5.2) • Impaired renal function. ( 5.7 ) • Thiazide diuretics may cause an exacerbation or activation of systemic lupus erythematosus. (5.4) • Acute angle-closure glaucoma, acute myopia, and choroidal effusion. ( 5.8 )
Adverse Reactions
• Most common adverse events (≥5% on irbesartan and hydrochlorothiazide tablets and more often than on placebo) are dizziness, fatigue, and musculoskeletal pain. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Solco Healthcare US, LLC at 1-866-257-2597 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
No significant drug-drug pharmacokinetic (or pharmacodynamic) interactions have been found in interaction studies with hydrochlorothiazide, digoxin, warfarin, and nifedipine. In vitro studies show significant inhibition of the formation of oxidized irbesartan metabolites with the known cytochrome CYP2C9 substrates/inhibitors sulphenazole, tolbutamide, and nifedipine. However, in clinical studies the consequences of concomitant irbesartan on the pharmacodynamics of warfarin were negligible. Concomitant nifedipine or hydrochlorothiazide had no effect on irbesartan pharmacokinetics. Based on in vitro data, no interaction would be expected with drugs whose metabolism is dependent upon cytochrome P450 isoenzymes 1A1, 1A2, 2A6, 2B6, 2D6, 2E1, or 3A4. In separate studies of patients receiving maintenance doses of warfarin, hydrochlorothiazide, or digoxin, irbesartan administration for 7 days had no effect on the pharmacodynamics of warfarin (prothrombin time) or the pharmacokinetics of digoxin. The pharmacokinetics of irbesartan were not affected by coadministration of nifedipine or hydrochlorothiazide.
How Supplied
Irbesartan and Hydrochlorothiazide film-coated tablets have markings on both sides and are available in the strengths and packages listed in the following table: Tablet Strength (irbesartan and hydrochlorothiazide) Film-Coated Tablet Color/Shape Tablet Markings Package Size NDC Code 150 mg and 12.5 mg Pink, oval-shaped Debossed ‘325’ on one side and ‘HH’ on the other side Bottles of 30 43547-330-03 Bottles of 90 43547-330-09 Bottles of 500 43547-330-50 300 mg and 12.5 mg Pink, capsule-shaped Debossed ‘327’ on one side and ‘HH’ on the other side Bottles of 30 43547-331-03 Bottles of 90 43547-331-09 Bottles of 500 43547-331-50
Description
• When pregnancy is detected, discontinue irbesartan and hydrochlorothiazide tablets as soon as possible [see Warnings and Precautions (5.1) and Use in Specific Populations ( 8.1 )]. • Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus [see Warnings and Precautions (5.1) and Use in Specific Populations ( 8.1 )].
Medication Information
Warnings and Precautions
• Hypotension: Correct volume depletion prior to administration. (5.2) • Impaired renal function. ( 5.7 ) • Thiazide diuretics may cause an exacerbation or activation of systemic lupus erythematosus. (5.4) • Acute angle-closure glaucoma, acute myopia, and choroidal effusion. ( 5.8 )
Indications and Usage
Irbesartan and hydrochlorothiazide tablets are indicated for the treatment of hypertension. Irbesartan and hydrochlorothiazide tablets may be used in patients whose blood pressure is not adequately controlled on monotherapy. Irbesartan and hydrochlorothiazide tablets may also be used as initial therapy in patients who are likely to need multiple drugs to achieve their blood pressure goals. The choice of irbesartan and hydrochlorothiazide tablets as initial therapy for hypertension should be based on an assessment of potential benefits and risks. Patients with stage 2 (moderate or severe) hypertension are at relatively high risk for cardiovascular events (such as strokes, heart attacks, and heart failure), kidney failure, and vision problems, so prompt treatment is clinically relevant. The decision to use a combination as initial therapy should be individualized and may be shaped by considerations such as the baseline blood pressure, the target goal, and the incremental likelihood of achieving goal with a combination compared with monotherapy. Data from Studies V and VI [see Clinical Studies (14.2) ] provide estimates of the probability of reaching a blood pressure goal with irbesartan and hydrochlorothiazide tablets compared to irbesartan or hydrochlorothiazide (HCTZ) monotherapy. The relationship between baseline blood pressure and achievement of a SeSBP <140 or <130 mmHg or SeDBP <90 or <80 mmHg in patients treated with irbesartan and hydrochlorothiazide tablets compared to patients treated with irbesartan or HCTZ monotherapy are shown in Figures 1a through 2b. Figure 1a: Probability of Achieving SBP <140 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)* Figure 1b: Probability of Achieving SBP <130 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)* Figure 2a: Probability of Achieving DBP <90 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)* Figure 2b: Probability of Achieving DBP <80 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)* The above graphs provide a rough approximation of the likelihood of reaching a targeted blood pressure goal (e.g., Week 8 sitting systolic blood pressure ≤140 mmHg) for the treatment groups. The curve of each treatment group in each study was estimated by logistic regression modeling from all available data of that treatment group. The estimated likelihood at the right tail of each curve is less reliable due to small numbers of subjects with high baseline blood pressures. For example, a patient with a blood pressure of 180/105 mmHg has about a 25% likelihood of achieving a goal of <140 mmHg (systolic) and 50% likelihood of achieving <90 mmHg (diastolic) on irbesartan alone (and lower still likelihoods on HCTZ alone). The likelihood of achieving these goals on irbesartan and hydrochlorothiazide tablets rises to about 40% (systolic) or 70% (diastolic).
Dosage and Administration
General Considerations • Maximum effects within 2 to 4 weeks after dose change. (2.1) • Renal impairment: Not recommended for patients with severe renal impairment (creatinine clearance <30 mL/min). (2.1 , 5.8) Hypertension • Initiate with 150 mg/12.5 mg. Titrate to 300 mg/12.5 mg then 300 mg/25 mg if needed. (2.2) • Replacement therapy: May be substituted for titrated components. (2.3)
Contraindications
• Irbesartan and hydrochlorothiazide tablets are contraindicated in patients who are hypersensitive to any component of this product. • Because of the hydrochlorothiazide component, this product is contraindicated in patients with anuria or hypersensitivity to other sulfonamide-derived drugs. • Do not coadminister aliskiren with irbesartan and hydrochlorothiazide tablets in patients with diabetes [see Drug Interactions (7) ] .
Adverse Reactions
• Most common adverse events (≥5% on irbesartan and hydrochlorothiazide tablets and more often than on placebo) are dizziness, fatigue, and musculoskeletal pain. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Solco Healthcare US, LLC at 1-866-257-2597 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
No significant drug-drug pharmacokinetic (or pharmacodynamic) interactions have been found in interaction studies with hydrochlorothiazide, digoxin, warfarin, and nifedipine. In vitro studies show significant inhibition of the formation of oxidized irbesartan metabolites with the known cytochrome CYP2C9 substrates/inhibitors sulphenazole, tolbutamide, and nifedipine. However, in clinical studies the consequences of concomitant irbesartan on the pharmacodynamics of warfarin were negligible. Concomitant nifedipine or hydrochlorothiazide had no effect on irbesartan pharmacokinetics. Based on in vitro data, no interaction would be expected with drugs whose metabolism is dependent upon cytochrome P450 isoenzymes 1A1, 1A2, 2A6, 2B6, 2D6, 2E1, or 3A4. In separate studies of patients receiving maintenance doses of warfarin, hydrochlorothiazide, or digoxin, irbesartan administration for 7 days had no effect on the pharmacodynamics of warfarin (prothrombin time) or the pharmacokinetics of digoxin. The pharmacokinetics of irbesartan were not affected by coadministration of nifedipine or hydrochlorothiazide.
How Supplied
Irbesartan and Hydrochlorothiazide film-coated tablets have markings on both sides and are available in the strengths and packages listed in the following table: Tablet Strength (irbesartan and hydrochlorothiazide) Film-Coated Tablet Color/Shape Tablet Markings Package Size NDC Code 150 mg and 12.5 mg Pink, oval-shaped Debossed ‘325’ on one side and ‘HH’ on the other side Bottles of 30 43547-330-03 Bottles of 90 43547-330-09 Bottles of 500 43547-330-50 300 mg and 12.5 mg Pink, capsule-shaped Debossed ‘327’ on one side and ‘HH’ on the other side Bottles of 30 43547-331-03 Bottles of 90 43547-331-09 Bottles of 500 43547-331-50
Description
• When pregnancy is detected, discontinue irbesartan and hydrochlorothiazide tablets as soon as possible [see Warnings and Precautions (5.1) and Use in Specific Populations ( 8.1 )]. • Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus [see Warnings and Precautions (5.1) and Use in Specific Populations ( 8.1 )].
Section 42229-5
Irbesartan and Hydrochlorothiazide
In double-blind clinical trials of various doses of irbesartan and hydrochlorothiazide, the incidence of hypertensive patients who developed hypokalemia (serum potassium <3.5 mEq/L) was 7.5% versus 6.0% for placebo; the incidence of hyperkalemia (serum potassium >5.7 mEq/L) was <1.0% versus 1.7% for placebo. No patient discontinued due to increases or decreases in serum potassium. On average, the combination of irbesartan and hydrochlorothiazide had no effect on serum potassium. Higher doses of irbesartan ameliorated the hypokalemic response to hydrochlorothiazide.
Coadministration of irbesartan and hydrochlorothiazide tablets with potassium-sparing diuretics, potassium supplements, potassium-containing salt substitutes or other drugs that raise serum potassium levels may result in hyperkalemia, sometimes severe. Monitor serum potassium in such patients.
7.6 Lithium
Increases in serum lithium concentrations and lithium toxicity have been reported with concomitant use of irbesartan or thiazide diuretics. Monitor lithium levels in patients receiving irbesartan and hydrochlorothiazide tablets and lithium.
16.2 Storage
Store at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30°C (59°F-86°F) [see USP Controlled Room Temperature].
Risk Summary
Irbesartan and hydrochlorothiazide tablets can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death [see Clinical Considerations]. Most epidemiologic studies examining fetal abnormalities after exposure to antihypertensive use in the first trimester have not distinguished drugs affecting the renin-angiotensin system from other antihypertensive agents. When pregnancy is detected, discontinue irbesartan and hydrochlorothiazide tablets as soon as possible.
All pregnancies have a background risk of birth defect, loss, or other adverse outcomes regardless of drug exposure. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo-fetal risk
Hypertension in pregnancy increases the maternal risk for preeclampsia, gestational diabetes, premature delivery, and delivery complications (e.g., need for cesarean section and postpartum hemorrhage). Hypertension increases the fetal risk for intrauterine growth restriction and intrauterine death. Pregnant women with hypertension should be carefully monitored and managed accordingly.
Fetal/neonatal adverse reactions
Oligohydramnios in pregnant women who use drugs affecting the renin-angiotensin system in the second and third trimesters of pregnancy can result in the following: reduced fetal renal function leading to anuria and renal failure, fetal lung hypoplasia, skeletal deformations, including skull hypoplasia, hypotension, and death. Perform serial ultrasound examinations to assess the intra-amniotic environment. Fetal testing may be appropriate, based on the week of pregnancy. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury. Closely observe infants with histories of in utero exposure to irbesartan and hydrochlorothiazide tablets for hypotension, oliguria, and hyperkalemia and other symptoms of renal impairment. In neonates with a history of in utero exposure to irbesartan and hydrochlorothiazide tablets, if oliguria or hypotension occurs, direct attention toward support of blood pressure and renal perfusion. Exchange transfusion or dialysis may be required as means of reversing hypotension and/or substituting for disordered renal function. Thiazides cross the placenta, and use of thiazides during pregnancy is associated with a risk of fetal or neonatal jaundice, thrombocytopenia, and possibly other adverse reactions that have occurred in adults [see Warnings and Precautions (5.1)].
Data
Animal data
Irbesartan crosses the placenta in rats and rabbits. In female rats given irbesartan prior to mating through gestation and lactation at oral doses of 50, 180, or 650 mg/kg/day (1.6 to 21.1 times the maximum recommended human dose (MRHD) based on body surface area), fetuses examined on Gestation Day 20 showed increased incidences of hydroureter and renal pelvic cavitation and/or absence of renal papilla in all irbesartan-treated groups. Subcutaneous edema also occurred in fetuses at maternal doses ≥180 mg/kg/day (5.8 times the MRHD). These anomalies occurred when female rats received irbesartan from prior to mating through Day 20 of gestation but were not observed in pups postnatally in the same study, or when irbesartan was given to pregnant rats only during organogenesis (Gestation Day 6 through Gestation Day 15) at oral doses from 50 to 450 mg/kg/day (up to 14.6 times the MRHD). In addition, no adverse effects on kidney development were observed in pups from dams given irbesartan from Gestation Day 15 through Lactation Day 24 at doses of 50, 180, or 650 mg/kg/day (up to 21.1 times the MRHD). The observed effects are believed to be late gestational effects of the drug. Pregnant rabbits given oral doses of irbesartan of 30 mg/kg/day (1.9 times the MRHD based on body surface area) experienced a high rate of maternal mortality and abortion. Surviving females had a slight increase in early resorptions and a corresponding decrease in live fetuses.
Radioactivity was present in the rat and rabbit fetuses during late gestation following oral doses of radiolabeled irbesartan.
When pregnant mice and rats were given hydrochlorothiazide at doses up to 3,000 and 1,000 mg/kg/day, respectively (about 600 and 400 times the MRHD) during their respective periods of major organogenesis, there was no evidence of fetal harm.
A development toxicity study was performed in rats with doses of 50/50 mg/kg/day and 150/150 mg/kg/day irbesartan and hydrochlorothiazide. Although the high dose combination appeared to be more toxic to the dams than either drug alone, there did not appear to be an increase in toxicity to the developing embryos.
8.2 Lactation
There are no available data on the presence of irbesartan in human milk, effects on milk production, or the breastfed infant. Irbesartan or some metabolite of irbesartan is secreted in the milk of lactating rats. Thiazides appear in human milk [see Clinical Pharmacology (12.3)]. Because of the potential for adverse effects on the nursing infant, the use of irbesartan and hydrochlorothiazide tablets in breastfeeding women is not recommended.
11 Description
Irbesartan and hydrochlorothiazide tablets are a combination of an angiotensin II receptor antagonist (AT1 subtype), irbesartan, and a thiazide diuretic, hydrochlorothiazide (HCTZ).
Irbesartan is a non-peptide compound, chemically described as a 2-butyl-3-[p-(o-1H-tetrazol-5-ylphenyl)benzyl]-1,3-diazaspiro[4.4]non-1-en-4-one. Its empirical formula is C25H28N6O, and its structural formula is:
Irbesartan, USP is a white to off-white crystalline powder with a molecular weight of 428.5. It is a nonpolar compound with a partition coefficient (octanol/water) of 10.1 at pH of 7.4. Irbesartan is slightly soluble in alcohol and methylene chloride and practically insoluble in water.
Hydrochlorothiazide is 6-chloro-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide. Its empirical formula is C7H8ClN3O4S2 and its structural formula is:
Hydrochlorothiazide, USP is a white, or practically white, crystalline powder with a molecular weight of 297.7. Hydrochlorothiazide is slightly soluble in water and freely soluble in sodium hydroxide solution.
Irbesartan and hydrochlorothiazide tablets, USP are available for oral administration in film-coated tablets containing either 150 mg or 300 mg of irbesartan combined with 12.5 mg of hydrochlorothiazide. All dosage strengths contain the following inactive ingredients: croscarmellose sodium, ferric oxide black, ferric oxide red, ferric oxide yellow, hypromellose, lactose monohydrate, lecithin, macrogol/PEG 3350, magnesium stearate, microcrystalline cellulose, polyvinyl alcohol-part.hydrolyzed, talc, and titanium dioxide.
Initial Therapy
In the moderate hypertension Study V (mean SeDBP between 90 and 110 mmHg), the types and incidences of adverse events reported for patients treated with irbesartan and hydrochlorothiazide tablets were similar to the adverse event profile in patients on initial irbesartan or HCTZ monotherapy. There were no reported events of syncope in the irbesartan and hydrochlorothiazide tablets treatment group and there was one reported event in the HCTZ treatment group. The incidences of prespecified adverse events on irbesartan and hydrochlorothiazide tablets, irbesartan, and HCTZ, respectively, were: 0.9%, 0%, and 0% for hypotension; 3.0%, 3.8%, and 1.0% for dizziness; 5.5%, 3.8%, and 4.8% for headache; 1.2%, 0%, and 1.0% for hyperkalemia; and 0.9%, 0%, and 0% for hypokalemia. The rates of discontinuation due to adverse events on irbesartan and hydrochlorothiazide tablets, irbesartan alone, and HCTZ alone were 6.7%, 3.8%, and 4.8%.
In the severe hypertension (SeDBP ≥110 mmHg) Study VI, the overall pattern of adverse events reported through 7 weeks of follow-up was similar in patients treated with irbesartan and hydrochlorothiazide tablets as initial therapy and in patients treated with irbesartan as initial therapy. The incidences of the prespecified adverse events on irbesartan and hydrochlorothiazide tablets and irbesartan, respectively, were: 0% and 0% for syncope; 0.6% and 0% for hypotension; 3.6% and 4.0% for dizziness; 4.3% and 6.6% for headache; 0.2% and 0% for hyperkalemia; and 0.6% and 0.4% for hypokalemia. The rates of discontinuation due to adverse events were 2.1% and 2.2%. [See Clinical Studies (14.2).]
16.1 How Supplied
Irbesartan and Hydrochlorothiazide film-coated tablets have markings on both sides and are available in the strengths and packages listed in the following table:
|
Tablet Strength
|
Film-Coated Tablet
|
Tablet Markings |
Package Size |
NDC Code |
|
150 mg and 12.5 mg |
Pink, |
|
Bottles of 30 |
43547-330-03 |
|
Bottles of 90 |
43547-330-09 |
|||
|
Bottles of 500 |
43547-330-50 |
|||
|
300 mg and 12.5 mg |
Pink, |
Debossed ‘327’ on one |
Bottles of 30 |
43547-331-03 |
|
Bottles of 90 |
43547-331-09 |
|||
|
Bottles of 500 |
43547-331-50 |
7.7 Carbamazepine
Concomitant use of carbamazepine and hydrochlorothiazide has been associated with the risk of symptomatic hyponatremia. Monitor electrolytes during concomitant use.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Of 1,694 patients receiving irbesartan and hydrochlorothiazide tablets in controlled clinical studies of hypertension, 264 (15.6%) were 65 years and over, while 45 (2.7%) were 75 years and over. No overall differences in safety or effectiveness were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out. [See Clinical Pharmacology (12.3) and Clinical Studies (14) .]
2.2 Add On Therapy
In patients not controlled on monotherapy with irbesartan or hydrochlorothiazide, the recommended doses of irbesartan and hydrochlorothiazide tablets, in order of increasing mean effect, are (irbesartan and hydrochlorothiazide) 150/12.5 mg, 300/12.5 mg, and 300/25 mg. The largest incremental effect will likely be in the transition from monotherapy to 150/12.5 mg. [See Clinical Studies (14.2).]
5.1 Fetal Toxicity
Irbesartan and hydrochlorothiazide tablets can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue irbesartan and hydrochlorothiazide tablets as soon as possible [see Use in Specific Populations (8.1)].
Thiazides cross the placenta and use of thiazides during pregnancy is associated with a risk of fetal or neonatal jaundice, thrombocytopenia, and possibly other adverse reactions that have occurred in adults.
2.4 Initial Therapy
The usual starting dose is irbesartan and hydrochlorothiazide tablets 150/12.5 mg once daily. The dosage can be increased after 1 to 2 weeks of therapy to a maximum of 300/25 mg once daily as needed to control blood pressure [see Clinical Studies (14.2)]. Irbesartan and hydrochlorothiazide tablets are not recommended as initial therapy in patients with intravascular volume depletion [see Warnings and Precautions (5.2)].
4 Contraindications
-
•Irbesartan and hydrochlorothiazide tablets are contraindicated in patients who are hypersensitive to any component of this product.
-
•Because of the hydrochlorothiazide component, this product is contraindicated in patients with anuria or hypersensitivity to other sulfonamide-derived drugs.
-
•Do not coadminister aliskiren with irbesartan and hydrochlorothiazide tablets in patients with diabetes [see Drug Interactions (7)].
6 Adverse Reactions
-
•Most common adverse events (≥5% on irbesartan and hydrochlorothiazide tablets and more often than on placebo) are dizziness, fatigue, and musculoskeletal pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Solco Healthcare US, LLC at 1-866-257-2597 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions
-
•NSAIDs and selective COX-2 inhibitors: Can reduce diuretic, natriuretic of diuretic, may lead to increased risk of renal impairment and reduced antihypertensive effect. Monitor renal function periodically. (7)
-
•Dual blockade of the renin-angiotensin system: Increased risk of renal impairment, hypotension, and hyperkalemia. (7)
-
•Antidiabetic drugs: Dosage adjustment of antidiabetic may be required. (7)
-
•Cholestyramine and colestipol: Reduced absorption of thiazides. (7)
-
•Lithium: Increases in serum lithium concentrations and lithium toxicity. (7)
-
•Carbamazepine: Increased risk of hyponatremia. (7)
12.3 Pharmacokinetics
Irbesartan
Irbesartan is an orally active agent that does not require biotransformation into an active form. The oral absorption of irbesartan is rapid and complete with an average absolute bioavailability of 60% to 80%. Following oral administration of irbesartan, peak plasma concentrations of irbesartan are attained at 1.5 to 2 hours after dosing. Food does not affect the bioavailability of irbesartan.
Irbesartan exhibits linear pharmacokinetics over the therapeutic dose range.
The terminal elimination half-life of irbesartan averaged 11 to 15 hours. Steady-state concentrations are achieved within 3 days. Limited accumulation of irbesartan (<20%) is observed in plasma upon repeated once-daily dosing.
5.6 Hepatic Impairment
Hydrochlorothiazide
Thiazides should be used with caution in patients with impaired hepatic function or progressive liver disease since minor alterations of fluid and electrolyte balance may precipitate hepatic coma.
Drug Drug Interactions
No significant drug-drug pharmacokinetic (or pharmacodynamic) interactions have been found in interaction studies with hydrochlorothiazide, digoxin, warfarin, and nifedipine.
In vitro studies show significant inhibition of the formation of oxidized irbesartan metabolites with the known cytochrome CYP2C9 substrates/inhibitors sulphenazole, tolbutamide, and nifedipine. However, in clinical studies the consequences of concomitant irbesartan on the pharmacodynamics of warfarin were negligible. Concomitant nifedipine or hydrochlorothiazide had no effect on irbesartan pharmacokinetics. Based on in vitro data, no interaction would be expected with drugs whose metabolism is dependent upon cytochrome P450 isoenzymes 1A1, 1A2, 2A6, 2B6, 2D6, 2E1, or 3A4.
In separate studies of patients receiving maintenance doses of warfarin, hydrochlorothiazide, or digoxin, irbesartan administration for 7 days had no effect on the pharmacodynamics of warfarin (prothrombin time) or the pharmacokinetics of digoxin. The pharmacokinetics of irbesartan were not affected by coadministration of nifedipine or hydrochlorothiazide.
1 Indications and Usage
Irbesartan and hydrochlorothiazide tablets are indicated for the treatment of hypertension.
Irbesartan and hydrochlorothiazide tablets may be used in patients whose blood pressure is not adequately controlled on monotherapy.
Irbesartan and hydrochlorothiazide tablets may also be used as initial therapy in patients who are likely to need multiple drugs to achieve their blood pressure goals.
-
The choice of irbesartan and hydrochlorothiazide tablets as initial therapy for hypertension should be based on an assessment of potential benefits and risks.
Patients with stage 2 (moderate or severe) hypertension are at relatively high risk for cardiovascular events (such as strokes, heart attacks, and heart failure), kidney failure, and vision problems, so prompt treatment is clinically relevant. The decision to use a combination as initial therapy should be individualized and may be shaped by considerations such as the baseline blood pressure, the target goal, and the incremental likelihood of achieving goal with a combination compared with monotherapy.
Data from Studies V and VI [see Clinical Studies (14.2)] provide estimates of the probability of reaching a blood pressure goal with irbesartan and hydrochlorothiazide tablets compared to irbesartan or hydrochlorothiazide (HCTZ) monotherapy. The relationship between baseline blood pressure and achievement of a SeSBP <140 or <130 mmHg or SeDBP <90 or <80 mmHg in patients treated with irbesartan and hydrochlorothiazide tablets compared to patients treated with irbesartan or HCTZ monotherapy are shown in Figures 1a through 2b.
|
Figure 1a: Probability of Achieving SBP <140 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)*
|
Figure 1b: Probability of Achieving SBP <130 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)*
|
|
Figure 2a: Probability of Achieving DBP <90 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)*
|
Figure 2b: Probability of Achieving DBP <80 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)*
|
The above graphs provide a rough approximation of the likelihood of reaching a targeted blood pressure goal (e.g., Week 8 sitting systolic blood pressure ≤140 mmHg) for the treatment groups. The curve of each treatment group in each study was estimated by logistic regression modeling from all available data of that treatment group. The estimated likelihood at the right tail of each curve is less reliable due to small numbers of subjects with high baseline blood pressures.
For example, a patient with a blood pressure of 180/105 mmHg has about a 25% likelihood of achieving a goal of <140 mmHg (systolic) and 50% likelihood of achieving <90 mmHg (diastolic) on irbesartan alone (and lower still likelihoods on HCTZ alone).
The likelihood of achieving these goals on irbesartan and hydrochlorothiazide tablets rises to about 40% (systolic) or 70% (diastolic).
2.3 Replacement Therapy
Irbesartan and hydrochlorothiazide tablets may be substituted for the titrated components.
Warning: Fetal Toxicity
-
•When pregnancy is detected, discontinue irbesartan and hydrochlorothiazide tablets as soon as possible [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
-
•Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
2.1 General Considerations
The side effects of irbesartan are generally rare and apparently independent of dose; those of hydrochlorothiazide are a mixture of dose-dependent (primarily hypokalemia) and dose-independent phenomena (e.g., pancreatitis), the former much more common than the latter. [See Adverse Reactions (6).]
Maximum antihypertensive effects are attained within 2 to 4 weeks after a change in dose.
Irbesartan and hydrochlorothiazide tablets may be administered with or without food.
Irbesartan and hydrochlorothiazide tablets may be administered with other antihypertensive agents.
Renal Impairment
The usual regimens of therapy with irbesartan and hydrochlorothiazide tablets may be followed as long as the patient’s creatinine clearance is >30 mL/min. In patients with more severe renal impairment, loop diuretics are preferred to thiazides, so irbesartan and hydrochlorothiazide tablets are not recommended.
Hepatic Impairment
No dosage adjustment is necessary in patients with hepatic impairment.
5 Warnings and Precautions
14.1 Irbesartan Monotherapy
The antihypertensive effects of irbesartan were examined in 7 major placebo-controlled, 8 to 12-week trials in patients with baseline diastolic blood pressures of 95 to 110 mmHg. Doses of 1 to 900 mg were included in these trials in order to fully explore the dose-range of irbesartan. These studies allowed a comparison of once or twice-daily regimens at 150 mg/day, comparisons of peak and trough effects, and comparisons of response by gender, age, and race. Two of the 7 placebo-controlled trials identified above and 2 additional placebo-controlled studies examined the antihypertensive effects of irbesartan and hydrochlorothiazide in combination.
The 7 studies of irbesartan monotherapy included a total of 1915 patients randomized to irbesartan (1 to 900 mg) and 611 patients randomized to placebo. Once-daily doses of 150 to 300 mg provided statistically and clinically significant decreases in systolic and diastolic blood pressure with trough (24-hour post dose) effects after 6 to 12 weeks of treatment compared to placebo, of about 8 to 10/5 to 6 mmHg and 8 to 12/5 to 8 mmHg, respectively. No further increase in effect was seen at dosages greater than 300 mg. The dose-response relationships for effects on systolic and diastolic pressure are shown in Figures 3 and 4.
Once-daily administration of therapeutic doses of irbesartan gave peak effects at around 3 to 6 hours and, in one continuous ambulatory blood pressure monitoring study, again around 14 hours. This was seen with both once-daily and twice-daily dosing. Trough-to-peak ratios for systolic and diastolic response were generally between 60% and 70%. In a continuous ambulatory blood pressure monitoring study, once-daily dosing with 150 mg gave trough and mean 24-hour responses similar to those observed in patients receiving twice-daily dosing at the same total daily dose.
Analysis of age, gender, and race subgroups of patients showed that men and women, and patients over and under 65 years of age, had generally similar responses. Irbesartan was effective in reducing blood pressure regardless of race, although the effect was somewhat less in blacks (usually a low-renin population). Black patients typically show an improved response with the addition of a low dose diuretic (e.g., 12.5 mg hydrochlorothiazide).
The effect of irbesartan is apparent after the first dose and is close to the full observed effect at 2 weeks. At the end of the 8-week exposure, about 2/3 of the antihypertensive effect was still present 1 week after the last dose. Rebound hypertension was not observed. There was essentially no change in average heart rate in irbesartan-treated patients in controlled trials.
2 Dosage and Administration
5.7 Impaired Renal Function
As a consequence of inhibiting the renin-angiotensin-aldosterone system, changes in renal function may be anticipated in susceptible individuals [see Drug Interactions (7)]. In patients whose renal function may depend on the activity of the renin-angiotensin-aldosterone system (e.g., patients with severe congestive heart failure), treatment with ACE inhibitors has been associated with oliguria and/or progressive azotemia and (rarely) with acute renal failure and/or death. Irbesartan would be expected to behave similarly. In studies of ACE inhibitors in patients with unilateral or bilateral renal artery stenosis, increases in serum creatinine or BUN have been reported. There has been no known use of irbesartan in patients with unilateral or bilateral renal artery stenosis, but a similar effect should be anticipated.
Thiazides should be used with caution in severe renal disease. In patients with renal disease, thiazides may precipitate azotemia. Cumulative effects of the drug may develop in patients with impaired renal function.
3 Dosage Forms and Strengths
Irbesartan and hydrochlorothiazide tablets USP, 150 mg/12.5 mg are pink, oval-shaped, biconvex-coated tablet debossed ‘325’ on one side and ‘HH’ on the other side.
Irbesartan and hydrochlorothiazide tablets USP, 300 mg/12.5 mg are pink, capsule-shaped, biconvex-coated tablet debossed ‘327’ on one side and ‘HH’ on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of irbesartan and hydrochlorothiazide tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Decisions to include these reactions in labeling are typically based on one or more of the following factors: (1) seriousness of the reaction, (2) frequency of reporting, or (3) strength of causal connection to irbesartan and hydrochlorothiazide tablets.
Irbesartan and hydrochlorothiazide
Blood and lymphatic system: Thrombocytopenia
Hepatobiliary: Hepatitis, Jaundice
Renal and urinary: Impaired renal function including renal failure
Skin and subcutaneous tissue: Urticaria
Irbesartan
Blood and lymphatic system: Anemia Ear and labyrinth: Tinnitus
Gastrointestinal: Intestinal angioedema
Skin and subcutaneous tissue: Angioedema (involving swelling of the face, lips, pharynx, and/or tongue)
Immune system: Anaphylactic reaction including anaphylactic shock Investigations: Increased CPK (Creatine Phosphokinase)
Metabolism and nutrition: Hyperkalemia, Hypoglycemia in diabetic patients
Hydrochlorothiazide
Eye: acute angle-closure glaucoma, acute myopia, and choroidal effusion
Non-melanoma Skin Cancer
Hydrochlorothiazide is associated with an increased risk of non-melanoma skin cancer. In a study conducted in the Sentinel System, increased risk was predominantly for squamous cell carcinoma (SCC) and in white patients taking large cumulative doses. The increased risk for SCC in the overall population was approximately 1 additional case per 16,000 patients per year, and for white patients taking a cumulative dose of ≥50,000 mg the risk increase was approximately 1 additional SCC case for every 6,700 patients per year.
6.3 Laboratory Abnormalities
In controlled clinical trials, clinically important changes in standard laboratory parameters were rarely associated with administration of irbesartan and hydrochlorothiazide tablets.
Creatinine, Blood Urea Nitrogen: Minor increases in blood urea nitrogen (BUN) or serum creatinine were observed in 2.3% and 1.1%, respectively, of patients with essential hypertension treated with irbesartan and hydrochlorothiazide tablets alone. No patient discontinued taking irbesartan and hydrochlorothiazide tablets due to increased BUN. One patient discontinued taking irbesartan and hydrochlorothiazide tablets due to a minor increase in serum creatinine.
Liver Function Tests: Occasional elevations of liver enzymes and/or serum bilirubin have occurred. In patients with essential hypertension treated with irbesartan and hydrochlorothiazide tablets alone, one patient was discontinued due to elevated liver enzymes.
Serum Electrolytes: [See Warnings and Precautions (5.2, 5.6).]
5.3 Hypersensitivity Reaction
Hydrochlorothiazide
Hypersensitivity reactions to hydrochlorothiazide may occur in patients with or without a history of allergy or bronchial asthma, but are more likely in patients with such a history.
8 Use in Specific Populations
-
•Lactation: Potential for adverse effects in infant. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The adverse reaction information from clinical trials does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating rates.
5.4 Systemic Lupus Erythematosus
Hydrochlorothiazide
Thiazide diuretics have been reported to cause exacerbation or activation of systemic lupus erythematosus.
Click Or Tap Here to Enter Text.
[Click here to enter Recent Major Changes]
17 Patient Counseling Information
Pregnancy
Tell female patients of childbearing age about the consequences of exposure to irbesartan and hydrochlorothiazide tablets during pregnancy. Discuss treatment options with women planning to become pregnant. Ask patients to report pregnancies to their physician as soon as possible.
Symptomatic Hypotension
Tell patients using irbesartan and hydrochlorothiazide tablets that they may feel lightheaded, especially during the first days of use. Tell patients to inform their physician if they feel lightheaded or faint. Tell the patient, if fainting occurs, stop using irbesartan and hydrochlorothiazide tablets and contact the prescribing doctor.
Tell patients using irbesartan and hydrochlorothiazide tablets that getting dehydrated can lower their blood pressure too much and lead to lightheadedness and possible fainting. Dehydration may occur with excessive sweating, diarrhea, or vomiting and with not drinking enough liquids.
Potassium Supplements
Advise patients not to use potassium supplements or salt substitutes containing potassium without consulting their healthcare provider [see Drug Interactions (7.3)].
Acute Angle-Closure Glaucoma, Acute Myopia, and Choroidal Effusion
Advise patients to discontinue irbesartan and hydrochlorothiazide tablets and seek immediate medical attention if they experience symptoms of acute angle-closure glaucoma, acute myopia, and choroidal effusion [see Warnings and Precautions (5.8)].
Non-melanoma Skin Cancer
Instruct patients taking hydrochlorothiazide to protect skin from the sun and undergo regular skin cancer screening.
7.3 Agents Increasing Serum Potassium
Coadministration of irbesartan and hydrochlorothiazide tablets with other drugs that raise serum potassium levels may result in hyperkalemia, sometimes severe. Monitor serum potassium in such patients.
14.2 Irbesartan and Hydrochlorothiazide
The antihypertensive effects of irbesartan and hydrochlorothiazide tablets were examined in 4 placebo-controlled studies in patients with mild-moderate hypertension (mean seated diastolic blood pressure [SeDBP] between 90 and 110 mmHg), one study in patients with moderate hypertension (mean seated systolic blood pressure [SeSBP] 160 to 179 mmHg or SeDBP 100 to 109 mmHg), and one study in patients with severe hypertension (mean SeDBP ≥110 mmHg) of 8 to 12 weeks. These trials included 3149 patients randomized to fixed doses of irbesartan (37.5 to 300 mg) and concomitant hydrochlorothiazide (6.25 to 25 mg).
Study I was a factorial study that compared all combinations of irbesartan (37.5 mg, 100 mg, and 300 mg or placebo) and hydrochlorothiazide (6.25 mg, 12.5 mg, and 25 mg or placebo).
-
Study II compared the irbesartan and hydrochlorothiazide combinations of 75/12.5 mg and 150/12.5 mg to their individual components and placebo.
Study III investigated the ambulatory blood pressure responses to irbesartan and hydrochlorothiazide (75/12.5 mg and 150/12.5 mg) and placebo after 8 weeks of dosing.
Study IV investigated the effects of the addition of irbesartan (75 or 150 mg) in patients not controlled (SeDBP 93-120 mmHg) on hydrochlorothiazide (25 mg) alone. In Studies I–III, the addition of irbesartan 150 to 300 mg to hydrochlorothiazide doses of 6.25, 12.5, or 25 mg produced further dose-related reductions in blood pressure at trough of 8 to 10 mmHg/3 to 6 mmHg, similar to those achieved with the same monotherapy dose of irbesartan. The addition of hydrochlorothiazide to irbesartan produced further dose-related reductions in blood pressure at trough (24 hours post dose) of 5 to 6/2 to 3 mmHg (12.5 mg) and 7 to 11/4 to 5 mmHg (25 mg), also similar to effects achieved with hydrochlorothiazide alone. Once-daily dosing with 150 mg irbesartan and 12.5 mg hydrochlorothiazide, 300 mg irbesartan and 12.5 mg hydrochlorothiazide, or 300 mg irbesartan and 25 mg hydrochlorothiazide produced mean placebo-adjusted blood pressure reductions at trough (24 hours post dosing) of about 13 to 15/7 to 9 mmHg, 14/9 to 12 mmHg, and 19 to 21/11 to 12 mmHg, respectively. Peak effects occurred at 3 to 6 hours, with the trough-to-peak ratios >65%.
-
In Study IV, the addition of irbesartan (75-150 mg) gave an additive effect (systolic/diastolic) at trough (24 hours post dosing) of 11/7 mmHg.
7.5 Cholestyramine and Colestipol Resins
Absorption of hydrochlorothiazide is impaired in the presence of anionic exchange resins. Stagger the dosage of hydrochlorothiazide and the resin such that irbesartan and hydrochlorothiazide tablets is administered at least 4 hours before or 4 to 6 hours after the administration of the resin.
7.4 Antidiabetic Drugs (oral Agents and Insulin)
Dosage adjustment of the antidiabetic drug may be required when coadministered with hydrochlorothiazide.
5.2 Hypotension in Volume Or Salt Depleted Patients
Excessive reduction of blood pressure was rarely seen in patients with uncomplicated hypertension treated with irbesartan alone (<0.1%) or with irbesartan and hydrochlorothiazide (approximately 1%). Initiation of antihypertensive therapy may cause symptomatic hypotension in patients with intravascular volume or sodium depletion, e.g., in patients treated vigorously with diuretics or in patients on dialysis. Such volume depletion should be corrected prior to administration of antihypertensive therapy.
If hypotension occurs, the patient should be placed in the supine position and, if necessary, given an intravenous infusion of normal saline. A transient hypotensive response is not a contraindication to further treatment, which usually can be continued without difficulty once the blood pressure has stabilized.
7.2 Dual Blockade of the Renin Angiotensin System (ras)
Dual blockade of the RAS with angiotensin-receptor blockers, ACE inhibitors, or aliskiren is associated with increased risks of hypotension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Closely monitor blood pressure, renal function, and electrolytes in patients on irbesartan and hydrochlorothiazide tablets and other agents that affect the RAS.
In most patients no benefit has been associated with using two RAS inhibitors concomitantly. In general, avoid combined use of RAS inhibitors.
Do not coadminister aliskiren with irbesartan and hydrochlorothiazide tablets in patients with diabetes. Avoid use of aliskiren with irbesartan and hydrochlorothiazide tablets in patients with renal impairment (GFR <60 mL/min).
Package Label.principal Display Panel 300/12.5 Mg Label
NDC 43547-331-03 Rx only
Irbesartan and
Hydrochlorothiazide Tablets USP
300 mg/12.5 mg
30 Tablets
Each film-coated tablet contains: 300 mg of Irbesartan, USP and 12.5 mg of Hydrochlorothiazide, USP.
See package insert for dosing information.
Store at 20°C - 25°C (68°F - 77°F); excursion permitted to 15°C - 30°C (59°F - 86°F). [See USP Controlled Room
Temperature]
Manufactured by:
Zhejiang Huahai Pharmaceutical Co., Ltd.
Xunqiao, Linhai, Zhejiang 317024, China
Distributed by:
Solco Healthcare US, LLC
Somerset, NJ 08873, USA
Rev.: 06/2025
200238-03
Package Label.principal Display Panel – 150 Mg/12.5 Mg Label
NDC 43547-330-03 Rx only
Irbesartan and
Hydrochlorothiazide Tablets USP
150 mg/12.5 mg
30 Tablets
Each film-coated tablet contains: 150 mg of Irbesartan, USP and 12.5 mg of Hydrochlorothiazide, USP.
See package insert for dosing information.
Store at 20°C - 25°C (68°F - 77°F); excursion permitted to 15°C - 30°C (59°F - 86°F). [See USP Controlled Room
Temperature]
Manufactured by:
Zhejiang Huahai Pharmaceutical Co., Ltd.
Xunqiao, Linhai, Zhejiang 317024, China
Distributed by:
Solco Healthcare US, LLC
Somerset, NJ 08873, USA
Rev.: 06/2025
200236-03
5.8 Acute Angle Closure Glaucoma, Acute Myopia, and Choroidal Effusion
Hydrochlorothiazide
Hydrochlorothiazide, a sulfonamide, can cause an idiosyncratic reaction resulting in acute angle-closure glaucoma and elevated intraocular pressure with or without a noticeable acute myopic shift and/or choroidal effusions. Cases of acute angle-closure glaucoma have been reported with hydrochlorothiazide. Symptoms include acute onset of decreased visual acuity or ocular pain and typically occur within hours to weeks of drug initiation. Untreated acute angle-closure glaucoma may result in permanent vision loss. The primary treatment is to discontinue drug intake as rapidly as possible. Prompt medical or surgical treatments may need to be considered if the intraocular pressure remains uncontrolled. Risk factors for developing acute angle-closure glaucoma may include a history of sulfonamide or penicillin allergy.
7.1 Nonsteroidal Anti Inflammatory Agents Including Selective Cyclooxygenase 2 Inhibitors (cox 2 Inhibitors)
Irbesartan
In patients who are elderly, volume depleted (including those on diuretic therapy), or with compromised renal function, coadministration of NSAIDs, including selective COX-2 inhibitors, with angiotensin II receptor antagonists, including irbesartan, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Therefore, monitor renal function and blood pressure periodically in patients receiving irbesartan and NSAID therapy.
Hydrochlorothiazide
Administration of a nonsteroidal anti-inflammatory agent, including a selective COX-2 inhibitor can reduce the diuretic, natriuretic, and antihypertensive effects of loop, potassium-sparing, and thiazide diuretics. Therefore, when irbesartan and hydrochlorothiazide tablets and nonsteroidal anti-inflammatory agents are used concomitantly, the patient should be observed closely to determine if the desired effect of the diuretic is obtained.
Structured Label Content
Section 42229-5 (42229-5)
Irbesartan and Hydrochlorothiazide
In double-blind clinical trials of various doses of irbesartan and hydrochlorothiazide, the incidence of hypertensive patients who developed hypokalemia (serum potassium <3.5 mEq/L) was 7.5% versus 6.0% for placebo; the incidence of hyperkalemia (serum potassium >5.7 mEq/L) was <1.0% versus 1.7% for placebo. No patient discontinued due to increases or decreases in serum potassium. On average, the combination of irbesartan and hydrochlorothiazide had no effect on serum potassium. Higher doses of irbesartan ameliorated the hypokalemic response to hydrochlorothiazide.
Coadministration of irbesartan and hydrochlorothiazide tablets with potassium-sparing diuretics, potassium supplements, potassium-containing salt substitutes or other drugs that raise serum potassium levels may result in hyperkalemia, sometimes severe. Monitor serum potassium in such patients.
7.6 Lithium
Increases in serum lithium concentrations and lithium toxicity have been reported with concomitant use of irbesartan or thiazide diuretics. Monitor lithium levels in patients receiving irbesartan and hydrochlorothiazide tablets and lithium.
16.2 Storage
Store at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30°C (59°F-86°F) [see USP Controlled Room Temperature].
Risk Summary
Irbesartan and hydrochlorothiazide tablets can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death [see Clinical Considerations]. Most epidemiologic studies examining fetal abnormalities after exposure to antihypertensive use in the first trimester have not distinguished drugs affecting the renin-angiotensin system from other antihypertensive agents. When pregnancy is detected, discontinue irbesartan and hydrochlorothiazide tablets as soon as possible.
All pregnancies have a background risk of birth defect, loss, or other adverse outcomes regardless of drug exposure. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo-fetal risk
Hypertension in pregnancy increases the maternal risk for preeclampsia, gestational diabetes, premature delivery, and delivery complications (e.g., need for cesarean section and postpartum hemorrhage). Hypertension increases the fetal risk for intrauterine growth restriction and intrauterine death. Pregnant women with hypertension should be carefully monitored and managed accordingly.
Fetal/neonatal adverse reactions
Oligohydramnios in pregnant women who use drugs affecting the renin-angiotensin system in the second and third trimesters of pregnancy can result in the following: reduced fetal renal function leading to anuria and renal failure, fetal lung hypoplasia, skeletal deformations, including skull hypoplasia, hypotension, and death. Perform serial ultrasound examinations to assess the intra-amniotic environment. Fetal testing may be appropriate, based on the week of pregnancy. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury. Closely observe infants with histories of in utero exposure to irbesartan and hydrochlorothiazide tablets for hypotension, oliguria, and hyperkalemia and other symptoms of renal impairment. In neonates with a history of in utero exposure to irbesartan and hydrochlorothiazide tablets, if oliguria or hypotension occurs, direct attention toward support of blood pressure and renal perfusion. Exchange transfusion or dialysis may be required as means of reversing hypotension and/or substituting for disordered renal function. Thiazides cross the placenta, and use of thiazides during pregnancy is associated with a risk of fetal or neonatal jaundice, thrombocytopenia, and possibly other adverse reactions that have occurred in adults [see Warnings and Precautions (5.1)].
Data
Animal data
Irbesartan crosses the placenta in rats and rabbits. In female rats given irbesartan prior to mating through gestation and lactation at oral doses of 50, 180, or 650 mg/kg/day (1.6 to 21.1 times the maximum recommended human dose (MRHD) based on body surface area), fetuses examined on Gestation Day 20 showed increased incidences of hydroureter and renal pelvic cavitation and/or absence of renal papilla in all irbesartan-treated groups. Subcutaneous edema also occurred in fetuses at maternal doses ≥180 mg/kg/day (5.8 times the MRHD). These anomalies occurred when female rats received irbesartan from prior to mating through Day 20 of gestation but were not observed in pups postnatally in the same study, or when irbesartan was given to pregnant rats only during organogenesis (Gestation Day 6 through Gestation Day 15) at oral doses from 50 to 450 mg/kg/day (up to 14.6 times the MRHD). In addition, no adverse effects on kidney development were observed in pups from dams given irbesartan from Gestation Day 15 through Lactation Day 24 at doses of 50, 180, or 650 mg/kg/day (up to 21.1 times the MRHD). The observed effects are believed to be late gestational effects of the drug. Pregnant rabbits given oral doses of irbesartan of 30 mg/kg/day (1.9 times the MRHD based on body surface area) experienced a high rate of maternal mortality and abortion. Surviving females had a slight increase in early resorptions and a corresponding decrease in live fetuses.
Radioactivity was present in the rat and rabbit fetuses during late gestation following oral doses of radiolabeled irbesartan.
When pregnant mice and rats were given hydrochlorothiazide at doses up to 3,000 and 1,000 mg/kg/day, respectively (about 600 and 400 times the MRHD) during their respective periods of major organogenesis, there was no evidence of fetal harm.
A development toxicity study was performed in rats with doses of 50/50 mg/kg/day and 150/150 mg/kg/day irbesartan and hydrochlorothiazide. Although the high dose combination appeared to be more toxic to the dams than either drug alone, there did not appear to be an increase in toxicity to the developing embryos.
8.2 Lactation
There are no available data on the presence of irbesartan in human milk, effects on milk production, or the breastfed infant. Irbesartan or some metabolite of irbesartan is secreted in the milk of lactating rats. Thiazides appear in human milk [see Clinical Pharmacology (12.3)]. Because of the potential for adverse effects on the nursing infant, the use of irbesartan and hydrochlorothiazide tablets in breastfeeding women is not recommended.
11 Description (11 DESCRIPTION)
Irbesartan and hydrochlorothiazide tablets are a combination of an angiotensin II receptor antagonist (AT1 subtype), irbesartan, and a thiazide diuretic, hydrochlorothiazide (HCTZ).
Irbesartan is a non-peptide compound, chemically described as a 2-butyl-3-[p-(o-1H-tetrazol-5-ylphenyl)benzyl]-1,3-diazaspiro[4.4]non-1-en-4-one. Its empirical formula is C25H28N6O, and its structural formula is:
Irbesartan, USP is a white to off-white crystalline powder with a molecular weight of 428.5. It is a nonpolar compound with a partition coefficient (octanol/water) of 10.1 at pH of 7.4. Irbesartan is slightly soluble in alcohol and methylene chloride and practically insoluble in water.
Hydrochlorothiazide is 6-chloro-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide. Its empirical formula is C7H8ClN3O4S2 and its structural formula is:
Hydrochlorothiazide, USP is a white, or practically white, crystalline powder with a molecular weight of 297.7. Hydrochlorothiazide is slightly soluble in water and freely soluble in sodium hydroxide solution.
Irbesartan and hydrochlorothiazide tablets, USP are available for oral administration in film-coated tablets containing either 150 mg or 300 mg of irbesartan combined with 12.5 mg of hydrochlorothiazide. All dosage strengths contain the following inactive ingredients: croscarmellose sodium, ferric oxide black, ferric oxide red, ferric oxide yellow, hypromellose, lactose monohydrate, lecithin, macrogol/PEG 3350, magnesium stearate, microcrystalline cellulose, polyvinyl alcohol-part.hydrolyzed, talc, and titanium dioxide.
Initial Therapy
In the moderate hypertension Study V (mean SeDBP between 90 and 110 mmHg), the types and incidences of adverse events reported for patients treated with irbesartan and hydrochlorothiazide tablets were similar to the adverse event profile in patients on initial irbesartan or HCTZ monotherapy. There were no reported events of syncope in the irbesartan and hydrochlorothiazide tablets treatment group and there was one reported event in the HCTZ treatment group. The incidences of prespecified adverse events on irbesartan and hydrochlorothiazide tablets, irbesartan, and HCTZ, respectively, were: 0.9%, 0%, and 0% for hypotension; 3.0%, 3.8%, and 1.0% for dizziness; 5.5%, 3.8%, and 4.8% for headache; 1.2%, 0%, and 1.0% for hyperkalemia; and 0.9%, 0%, and 0% for hypokalemia. The rates of discontinuation due to adverse events on irbesartan and hydrochlorothiazide tablets, irbesartan alone, and HCTZ alone were 6.7%, 3.8%, and 4.8%.
In the severe hypertension (SeDBP ≥110 mmHg) Study VI, the overall pattern of adverse events reported through 7 weeks of follow-up was similar in patients treated with irbesartan and hydrochlorothiazide tablets as initial therapy and in patients treated with irbesartan as initial therapy. The incidences of the prespecified adverse events on irbesartan and hydrochlorothiazide tablets and irbesartan, respectively, were: 0% and 0% for syncope; 0.6% and 0% for hypotension; 3.6% and 4.0% for dizziness; 4.3% and 6.6% for headache; 0.2% and 0% for hyperkalemia; and 0.6% and 0.4% for hypokalemia. The rates of discontinuation due to adverse events were 2.1% and 2.2%. [See Clinical Studies (14.2).]
16.1 How Supplied
Irbesartan and Hydrochlorothiazide film-coated tablets have markings on both sides and are available in the strengths and packages listed in the following table:
|
Tablet Strength
|
Film-Coated Tablet
|
Tablet Markings |
Package Size |
NDC Code |
|
150 mg and 12.5 mg |
Pink, |
|
Bottles of 30 |
43547-330-03 |
|
Bottles of 90 |
43547-330-09 |
|||
|
Bottles of 500 |
43547-330-50 |
|||
|
300 mg and 12.5 mg |
Pink, |
Debossed ‘327’ on one |
Bottles of 30 |
43547-331-03 |
|
Bottles of 90 |
43547-331-09 |
|||
|
Bottles of 500 |
43547-331-50 |
7.7 Carbamazepine
Concomitant use of carbamazepine and hydrochlorothiazide has been associated with the risk of symptomatic hyponatremia. Monitor electrolytes during concomitant use.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Of 1,694 patients receiving irbesartan and hydrochlorothiazide tablets in controlled clinical studies of hypertension, 264 (15.6%) were 65 years and over, while 45 (2.7%) were 75 years and over. No overall differences in safety or effectiveness were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out. [See Clinical Pharmacology (12.3) and Clinical Studies (14) .]
2.2 Add On Therapy (2.2 Add-On Therapy)
In patients not controlled on monotherapy with irbesartan or hydrochlorothiazide, the recommended doses of irbesartan and hydrochlorothiazide tablets, in order of increasing mean effect, are (irbesartan and hydrochlorothiazide) 150/12.5 mg, 300/12.5 mg, and 300/25 mg. The largest incremental effect will likely be in the transition from monotherapy to 150/12.5 mg. [See Clinical Studies (14.2).]
5.1 Fetal Toxicity
Irbesartan and hydrochlorothiazide tablets can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue irbesartan and hydrochlorothiazide tablets as soon as possible [see Use in Specific Populations (8.1)].
Thiazides cross the placenta and use of thiazides during pregnancy is associated with a risk of fetal or neonatal jaundice, thrombocytopenia, and possibly other adverse reactions that have occurred in adults.
2.4 Initial Therapy
The usual starting dose is irbesartan and hydrochlorothiazide tablets 150/12.5 mg once daily. The dosage can be increased after 1 to 2 weeks of therapy to a maximum of 300/25 mg once daily as needed to control blood pressure [see Clinical Studies (14.2)]. Irbesartan and hydrochlorothiazide tablets are not recommended as initial therapy in patients with intravascular volume depletion [see Warnings and Precautions (5.2)].
4 Contraindications (4 CONTRAINDICATIONS)
-
•Irbesartan and hydrochlorothiazide tablets are contraindicated in patients who are hypersensitive to any component of this product.
-
•Because of the hydrochlorothiazide component, this product is contraindicated in patients with anuria or hypersensitivity to other sulfonamide-derived drugs.
-
•Do not coadminister aliskiren with irbesartan and hydrochlorothiazide tablets in patients with diabetes [see Drug Interactions (7)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
-
•Most common adverse events (≥5% on irbesartan and hydrochlorothiazide tablets and more often than on placebo) are dizziness, fatigue, and musculoskeletal pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Solco Healthcare US, LLC at 1-866-257-2597 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions (7 DRUG INTERACTIONS)
-
•NSAIDs and selective COX-2 inhibitors: Can reduce diuretic, natriuretic of diuretic, may lead to increased risk of renal impairment and reduced antihypertensive effect. Monitor renal function periodically. (7)
-
•Dual blockade of the renin-angiotensin system: Increased risk of renal impairment, hypotension, and hyperkalemia. (7)
-
•Antidiabetic drugs: Dosage adjustment of antidiabetic may be required. (7)
-
•Cholestyramine and colestipol: Reduced absorption of thiazides. (7)
-
•Lithium: Increases in serum lithium concentrations and lithium toxicity. (7)
-
•Carbamazepine: Increased risk of hyponatremia. (7)
12.3 Pharmacokinetics
Irbesartan
Irbesartan is an orally active agent that does not require biotransformation into an active form. The oral absorption of irbesartan is rapid and complete with an average absolute bioavailability of 60% to 80%. Following oral administration of irbesartan, peak plasma concentrations of irbesartan are attained at 1.5 to 2 hours after dosing. Food does not affect the bioavailability of irbesartan.
Irbesartan exhibits linear pharmacokinetics over the therapeutic dose range.
The terminal elimination half-life of irbesartan averaged 11 to 15 hours. Steady-state concentrations are achieved within 3 days. Limited accumulation of irbesartan (<20%) is observed in plasma upon repeated once-daily dosing.
5.6 Hepatic Impairment
Hydrochlorothiazide
Thiazides should be used with caution in patients with impaired hepatic function or progressive liver disease since minor alterations of fluid and electrolyte balance may precipitate hepatic coma.
Drug Drug Interactions (Drug-Drug Interactions)
No significant drug-drug pharmacokinetic (or pharmacodynamic) interactions have been found in interaction studies with hydrochlorothiazide, digoxin, warfarin, and nifedipine.
In vitro studies show significant inhibition of the formation of oxidized irbesartan metabolites with the known cytochrome CYP2C9 substrates/inhibitors sulphenazole, tolbutamide, and nifedipine. However, in clinical studies the consequences of concomitant irbesartan on the pharmacodynamics of warfarin were negligible. Concomitant nifedipine or hydrochlorothiazide had no effect on irbesartan pharmacokinetics. Based on in vitro data, no interaction would be expected with drugs whose metabolism is dependent upon cytochrome P450 isoenzymes 1A1, 1A2, 2A6, 2B6, 2D6, 2E1, or 3A4.
In separate studies of patients receiving maintenance doses of warfarin, hydrochlorothiazide, or digoxin, irbesartan administration for 7 days had no effect on the pharmacodynamics of warfarin (prothrombin time) or the pharmacokinetics of digoxin. The pharmacokinetics of irbesartan were not affected by coadministration of nifedipine or hydrochlorothiazide.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Irbesartan and hydrochlorothiazide tablets are indicated for the treatment of hypertension.
Irbesartan and hydrochlorothiazide tablets may be used in patients whose blood pressure is not adequately controlled on monotherapy.
Irbesartan and hydrochlorothiazide tablets may also be used as initial therapy in patients who are likely to need multiple drugs to achieve their blood pressure goals.
-
The choice of irbesartan and hydrochlorothiazide tablets as initial therapy for hypertension should be based on an assessment of potential benefits and risks.
Patients with stage 2 (moderate or severe) hypertension are at relatively high risk for cardiovascular events (such as strokes, heart attacks, and heart failure), kidney failure, and vision problems, so prompt treatment is clinically relevant. The decision to use a combination as initial therapy should be individualized and may be shaped by considerations such as the baseline blood pressure, the target goal, and the incremental likelihood of achieving goal with a combination compared with monotherapy.
Data from Studies V and VI [see Clinical Studies (14.2)] provide estimates of the probability of reaching a blood pressure goal with irbesartan and hydrochlorothiazide tablets compared to irbesartan or hydrochlorothiazide (HCTZ) monotherapy. The relationship between baseline blood pressure and achievement of a SeSBP <140 or <130 mmHg or SeDBP <90 or <80 mmHg in patients treated with irbesartan and hydrochlorothiazide tablets compared to patients treated with irbesartan or HCTZ monotherapy are shown in Figures 1a through 2b.
|
Figure 1a: Probability of Achieving SBP <140 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)*
|
Figure 1b: Probability of Achieving SBP <130 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)*
|
|
Figure 2a: Probability of Achieving DBP <90 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)*
|
Figure 2b: Probability of Achieving DBP <80 mmHg in Patients from Initial Therapy Studies V (Week 8) and VI (Week 7)*
|
The above graphs provide a rough approximation of the likelihood of reaching a targeted blood pressure goal (e.g., Week 8 sitting systolic blood pressure ≤140 mmHg) for the treatment groups. The curve of each treatment group in each study was estimated by logistic regression modeling from all available data of that treatment group. The estimated likelihood at the right tail of each curve is less reliable due to small numbers of subjects with high baseline blood pressures.
For example, a patient with a blood pressure of 180/105 mmHg has about a 25% likelihood of achieving a goal of <140 mmHg (systolic) and 50% likelihood of achieving <90 mmHg (diastolic) on irbesartan alone (and lower still likelihoods on HCTZ alone).
The likelihood of achieving these goals on irbesartan and hydrochlorothiazide tablets rises to about 40% (systolic) or 70% (diastolic).
2.3 Replacement Therapy
Irbesartan and hydrochlorothiazide tablets may be substituted for the titrated components.
Warning: Fetal Toxicity (WARNING: FETAL TOXICITY)
-
•When pregnancy is detected, discontinue irbesartan and hydrochlorothiazide tablets as soon as possible [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
-
•Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
2.1 General Considerations
The side effects of irbesartan are generally rare and apparently independent of dose; those of hydrochlorothiazide are a mixture of dose-dependent (primarily hypokalemia) and dose-independent phenomena (e.g., pancreatitis), the former much more common than the latter. [See Adverse Reactions (6).]
Maximum antihypertensive effects are attained within 2 to 4 weeks after a change in dose.
Irbesartan and hydrochlorothiazide tablets may be administered with or without food.
Irbesartan and hydrochlorothiazide tablets may be administered with other antihypertensive agents.
Renal Impairment
The usual regimens of therapy with irbesartan and hydrochlorothiazide tablets may be followed as long as the patient’s creatinine clearance is >30 mL/min. In patients with more severe renal impairment, loop diuretics are preferred to thiazides, so irbesartan and hydrochlorothiazide tablets are not recommended.
Hepatic Impairment
No dosage adjustment is necessary in patients with hepatic impairment.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
14.1 Irbesartan Monotherapy
The antihypertensive effects of irbesartan were examined in 7 major placebo-controlled, 8 to 12-week trials in patients with baseline diastolic blood pressures of 95 to 110 mmHg. Doses of 1 to 900 mg were included in these trials in order to fully explore the dose-range of irbesartan. These studies allowed a comparison of once or twice-daily regimens at 150 mg/day, comparisons of peak and trough effects, and comparisons of response by gender, age, and race. Two of the 7 placebo-controlled trials identified above and 2 additional placebo-controlled studies examined the antihypertensive effects of irbesartan and hydrochlorothiazide in combination.
The 7 studies of irbesartan monotherapy included a total of 1915 patients randomized to irbesartan (1 to 900 mg) and 611 patients randomized to placebo. Once-daily doses of 150 to 300 mg provided statistically and clinically significant decreases in systolic and diastolic blood pressure with trough (24-hour post dose) effects after 6 to 12 weeks of treatment compared to placebo, of about 8 to 10/5 to 6 mmHg and 8 to 12/5 to 8 mmHg, respectively. No further increase in effect was seen at dosages greater than 300 mg. The dose-response relationships for effects on systolic and diastolic pressure are shown in Figures 3 and 4.
Once-daily administration of therapeutic doses of irbesartan gave peak effects at around 3 to 6 hours and, in one continuous ambulatory blood pressure monitoring study, again around 14 hours. This was seen with both once-daily and twice-daily dosing. Trough-to-peak ratios for systolic and diastolic response were generally between 60% and 70%. In a continuous ambulatory blood pressure monitoring study, once-daily dosing with 150 mg gave trough and mean 24-hour responses similar to those observed in patients receiving twice-daily dosing at the same total daily dose.
Analysis of age, gender, and race subgroups of patients showed that men and women, and patients over and under 65 years of age, had generally similar responses. Irbesartan was effective in reducing blood pressure regardless of race, although the effect was somewhat less in blacks (usually a low-renin population). Black patients typically show an improved response with the addition of a low dose diuretic (e.g., 12.5 mg hydrochlorothiazide).
The effect of irbesartan is apparent after the first dose and is close to the full observed effect at 2 weeks. At the end of the 8-week exposure, about 2/3 of the antihypertensive effect was still present 1 week after the last dose. Rebound hypertension was not observed. There was essentially no change in average heart rate in irbesartan-treated patients in controlled trials.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
5.7 Impaired Renal Function
As a consequence of inhibiting the renin-angiotensin-aldosterone system, changes in renal function may be anticipated in susceptible individuals [see Drug Interactions (7)]. In patients whose renal function may depend on the activity of the renin-angiotensin-aldosterone system (e.g., patients with severe congestive heart failure), treatment with ACE inhibitors has been associated with oliguria and/or progressive azotemia and (rarely) with acute renal failure and/or death. Irbesartan would be expected to behave similarly. In studies of ACE inhibitors in patients with unilateral or bilateral renal artery stenosis, increases in serum creatinine or BUN have been reported. There has been no known use of irbesartan in patients with unilateral or bilateral renal artery stenosis, but a similar effect should be anticipated.
Thiazides should be used with caution in severe renal disease. In patients with renal disease, thiazides may precipitate azotemia. Cumulative effects of the drug may develop in patients with impaired renal function.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Irbesartan and hydrochlorothiazide tablets USP, 150 mg/12.5 mg are pink, oval-shaped, biconvex-coated tablet debossed ‘325’ on one side and ‘HH’ on the other side.
Irbesartan and hydrochlorothiazide tablets USP, 300 mg/12.5 mg are pink, capsule-shaped, biconvex-coated tablet debossed ‘327’ on one side and ‘HH’ on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of irbesartan and hydrochlorothiazide tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Decisions to include these reactions in labeling are typically based on one or more of the following factors: (1) seriousness of the reaction, (2) frequency of reporting, or (3) strength of causal connection to irbesartan and hydrochlorothiazide tablets.
Irbesartan and hydrochlorothiazide
Blood and lymphatic system: Thrombocytopenia
Hepatobiliary: Hepatitis, Jaundice
Renal and urinary: Impaired renal function including renal failure
Skin and subcutaneous tissue: Urticaria
Irbesartan
Blood and lymphatic system: Anemia Ear and labyrinth: Tinnitus
Gastrointestinal: Intestinal angioedema
Skin and subcutaneous tissue: Angioedema (involving swelling of the face, lips, pharynx, and/or tongue)
Immune system: Anaphylactic reaction including anaphylactic shock Investigations: Increased CPK (Creatine Phosphokinase)
Metabolism and nutrition: Hyperkalemia, Hypoglycemia in diabetic patients
Hydrochlorothiazide
Eye: acute angle-closure glaucoma, acute myopia, and choroidal effusion
Non-melanoma Skin Cancer
Hydrochlorothiazide is associated with an increased risk of non-melanoma skin cancer. In a study conducted in the Sentinel System, increased risk was predominantly for squamous cell carcinoma (SCC) and in white patients taking large cumulative doses. The increased risk for SCC in the overall population was approximately 1 additional case per 16,000 patients per year, and for white patients taking a cumulative dose of ≥50,000 mg the risk increase was approximately 1 additional SCC case for every 6,700 patients per year.
6.3 Laboratory Abnormalities
In controlled clinical trials, clinically important changes in standard laboratory parameters were rarely associated with administration of irbesartan and hydrochlorothiazide tablets.
Creatinine, Blood Urea Nitrogen: Minor increases in blood urea nitrogen (BUN) or serum creatinine were observed in 2.3% and 1.1%, respectively, of patients with essential hypertension treated with irbesartan and hydrochlorothiazide tablets alone. No patient discontinued taking irbesartan and hydrochlorothiazide tablets due to increased BUN. One patient discontinued taking irbesartan and hydrochlorothiazide tablets due to a minor increase in serum creatinine.
Liver Function Tests: Occasional elevations of liver enzymes and/or serum bilirubin have occurred. In patients with essential hypertension treated with irbesartan and hydrochlorothiazide tablets alone, one patient was discontinued due to elevated liver enzymes.
Serum Electrolytes: [See Warnings and Precautions (5.2, 5.6).]
5.3 Hypersensitivity Reaction
Hydrochlorothiazide
Hypersensitivity reactions to hydrochlorothiazide may occur in patients with or without a history of allergy or bronchial asthma, but are more likely in patients with such a history.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
-
•Lactation: Potential for adverse effects in infant. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The adverse reaction information from clinical trials does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating rates.
5.4 Systemic Lupus Erythematosus
Hydrochlorothiazide
Thiazide diuretics have been reported to cause exacerbation or activation of systemic lupus erythematosus.
Click Or Tap Here to Enter Text. (Click or tap here to enter text.)
[Click here to enter Recent Major Changes]
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Pregnancy
Tell female patients of childbearing age about the consequences of exposure to irbesartan and hydrochlorothiazide tablets during pregnancy. Discuss treatment options with women planning to become pregnant. Ask patients to report pregnancies to their physician as soon as possible.
Symptomatic Hypotension
Tell patients using irbesartan and hydrochlorothiazide tablets that they may feel lightheaded, especially during the first days of use. Tell patients to inform their physician if they feel lightheaded or faint. Tell the patient, if fainting occurs, stop using irbesartan and hydrochlorothiazide tablets and contact the prescribing doctor.
Tell patients using irbesartan and hydrochlorothiazide tablets that getting dehydrated can lower their blood pressure too much and lead to lightheadedness and possible fainting. Dehydration may occur with excessive sweating, diarrhea, or vomiting and with not drinking enough liquids.
Potassium Supplements
Advise patients not to use potassium supplements or salt substitutes containing potassium without consulting their healthcare provider [see Drug Interactions (7.3)].
Acute Angle-Closure Glaucoma, Acute Myopia, and Choroidal Effusion
Advise patients to discontinue irbesartan and hydrochlorothiazide tablets and seek immediate medical attention if they experience symptoms of acute angle-closure glaucoma, acute myopia, and choroidal effusion [see Warnings and Precautions (5.8)].
Non-melanoma Skin Cancer
Instruct patients taking hydrochlorothiazide to protect skin from the sun and undergo regular skin cancer screening.
7.3 Agents Increasing Serum Potassium
Coadministration of irbesartan and hydrochlorothiazide tablets with other drugs that raise serum potassium levels may result in hyperkalemia, sometimes severe. Monitor serum potassium in such patients.
14.2 Irbesartan and Hydrochlorothiazide
The antihypertensive effects of irbesartan and hydrochlorothiazide tablets were examined in 4 placebo-controlled studies in patients with mild-moderate hypertension (mean seated diastolic blood pressure [SeDBP] between 90 and 110 mmHg), one study in patients with moderate hypertension (mean seated systolic blood pressure [SeSBP] 160 to 179 mmHg or SeDBP 100 to 109 mmHg), and one study in patients with severe hypertension (mean SeDBP ≥110 mmHg) of 8 to 12 weeks. These trials included 3149 patients randomized to fixed doses of irbesartan (37.5 to 300 mg) and concomitant hydrochlorothiazide (6.25 to 25 mg).
Study I was a factorial study that compared all combinations of irbesartan (37.5 mg, 100 mg, and 300 mg or placebo) and hydrochlorothiazide (6.25 mg, 12.5 mg, and 25 mg or placebo).
-
Study II compared the irbesartan and hydrochlorothiazide combinations of 75/12.5 mg and 150/12.5 mg to their individual components and placebo.
Study III investigated the ambulatory blood pressure responses to irbesartan and hydrochlorothiazide (75/12.5 mg and 150/12.5 mg) and placebo after 8 weeks of dosing.
Study IV investigated the effects of the addition of irbesartan (75 or 150 mg) in patients not controlled (SeDBP 93-120 mmHg) on hydrochlorothiazide (25 mg) alone. In Studies I–III, the addition of irbesartan 150 to 300 mg to hydrochlorothiazide doses of 6.25, 12.5, or 25 mg produced further dose-related reductions in blood pressure at trough of 8 to 10 mmHg/3 to 6 mmHg, similar to those achieved with the same monotherapy dose of irbesartan. The addition of hydrochlorothiazide to irbesartan produced further dose-related reductions in blood pressure at trough (24 hours post dose) of 5 to 6/2 to 3 mmHg (12.5 mg) and 7 to 11/4 to 5 mmHg (25 mg), also similar to effects achieved with hydrochlorothiazide alone. Once-daily dosing with 150 mg irbesartan and 12.5 mg hydrochlorothiazide, 300 mg irbesartan and 12.5 mg hydrochlorothiazide, or 300 mg irbesartan and 25 mg hydrochlorothiazide produced mean placebo-adjusted blood pressure reductions at trough (24 hours post dosing) of about 13 to 15/7 to 9 mmHg, 14/9 to 12 mmHg, and 19 to 21/11 to 12 mmHg, respectively. Peak effects occurred at 3 to 6 hours, with the trough-to-peak ratios >65%.
-
In Study IV, the addition of irbesartan (75-150 mg) gave an additive effect (systolic/diastolic) at trough (24 hours post dosing) of 11/7 mmHg.
7.5 Cholestyramine and Colestipol Resins
Absorption of hydrochlorothiazide is impaired in the presence of anionic exchange resins. Stagger the dosage of hydrochlorothiazide and the resin such that irbesartan and hydrochlorothiazide tablets is administered at least 4 hours before or 4 to 6 hours after the administration of the resin.
7.4 Antidiabetic Drugs (oral Agents and Insulin) (7.4 Antidiabetic Drugs (oral agents and insulin))
Dosage adjustment of the antidiabetic drug may be required when coadministered with hydrochlorothiazide.
5.2 Hypotension in Volume Or Salt Depleted Patients (5.2 Hypotension in Volume or Salt-Depleted Patients)
Excessive reduction of blood pressure was rarely seen in patients with uncomplicated hypertension treated with irbesartan alone (<0.1%) or with irbesartan and hydrochlorothiazide (approximately 1%). Initiation of antihypertensive therapy may cause symptomatic hypotension in patients with intravascular volume or sodium depletion, e.g., in patients treated vigorously with diuretics or in patients on dialysis. Such volume depletion should be corrected prior to administration of antihypertensive therapy.
If hypotension occurs, the patient should be placed in the supine position and, if necessary, given an intravenous infusion of normal saline. A transient hypotensive response is not a contraindication to further treatment, which usually can be continued without difficulty once the blood pressure has stabilized.
7.2 Dual Blockade of the Renin Angiotensin System (ras) (7.2 Dual Blockade of the Renin-Angiotensin System (RAS))
Dual blockade of the RAS with angiotensin-receptor blockers, ACE inhibitors, or aliskiren is associated with increased risks of hypotension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Closely monitor blood pressure, renal function, and electrolytes in patients on irbesartan and hydrochlorothiazide tablets and other agents that affect the RAS.
In most patients no benefit has been associated with using two RAS inhibitors concomitantly. In general, avoid combined use of RAS inhibitors.
Do not coadminister aliskiren with irbesartan and hydrochlorothiazide tablets in patients with diabetes. Avoid use of aliskiren with irbesartan and hydrochlorothiazide tablets in patients with renal impairment (GFR <60 mL/min).
Package Label.principal Display Panel 300/12.5 Mg Label (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 300/12.5 mg label)
NDC 43547-331-03 Rx only
Irbesartan and
Hydrochlorothiazide Tablets USP
300 mg/12.5 mg
30 Tablets
Each film-coated tablet contains: 300 mg of Irbesartan, USP and 12.5 mg of Hydrochlorothiazide, USP.
See package insert for dosing information.
Store at 20°C - 25°C (68°F - 77°F); excursion permitted to 15°C - 30°C (59°F - 86°F). [See USP Controlled Room
Temperature]
Manufactured by:
Zhejiang Huahai Pharmaceutical Co., Ltd.
Xunqiao, Linhai, Zhejiang 317024, China
Distributed by:
Solco Healthcare US, LLC
Somerset, NJ 08873, USA
Rev.: 06/2025
200238-03
Package Label.principal Display Panel – 150 Mg/12.5 Mg Label (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL – 150 mg/12.5 mg label)
NDC 43547-330-03 Rx only
Irbesartan and
Hydrochlorothiazide Tablets USP
150 mg/12.5 mg
30 Tablets
Each film-coated tablet contains: 150 mg of Irbesartan, USP and 12.5 mg of Hydrochlorothiazide, USP.
See package insert for dosing information.
Store at 20°C - 25°C (68°F - 77°F); excursion permitted to 15°C - 30°C (59°F - 86°F). [See USP Controlled Room
Temperature]
Manufactured by:
Zhejiang Huahai Pharmaceutical Co., Ltd.
Xunqiao, Linhai, Zhejiang 317024, China
Distributed by:
Solco Healthcare US, LLC
Somerset, NJ 08873, USA
Rev.: 06/2025
200236-03
5.8 Acute Angle Closure Glaucoma, Acute Myopia, and Choroidal Effusion (5.8 Acute Angle-Closure Glaucoma, Acute Myopia, and Choroidal Effusion)
Hydrochlorothiazide
Hydrochlorothiazide, a sulfonamide, can cause an idiosyncratic reaction resulting in acute angle-closure glaucoma and elevated intraocular pressure with or without a noticeable acute myopic shift and/or choroidal effusions. Cases of acute angle-closure glaucoma have been reported with hydrochlorothiazide. Symptoms include acute onset of decreased visual acuity or ocular pain and typically occur within hours to weeks of drug initiation. Untreated acute angle-closure glaucoma may result in permanent vision loss. The primary treatment is to discontinue drug intake as rapidly as possible. Prompt medical or surgical treatments may need to be considered if the intraocular pressure remains uncontrolled. Risk factors for developing acute angle-closure glaucoma may include a history of sulfonamide or penicillin allergy.
7.1 Nonsteroidal Anti Inflammatory Agents Including Selective Cyclooxygenase 2 Inhibitors (cox 2 Inhibitors) (7.1 Nonsteroidal Anti-inflammatory Agents Including Selective Cyclooxygenase-2 Inhibitors (COX-2 Inhibitors))
Irbesartan
In patients who are elderly, volume depleted (including those on diuretic therapy), or with compromised renal function, coadministration of NSAIDs, including selective COX-2 inhibitors, with angiotensin II receptor antagonists, including irbesartan, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Therefore, monitor renal function and blood pressure periodically in patients receiving irbesartan and NSAID therapy.
Hydrochlorothiazide
Administration of a nonsteroidal anti-inflammatory agent, including a selective COX-2 inhibitor can reduce the diuretic, natriuretic, and antihypertensive effects of loop, potassium-sparing, and thiazide diuretics. Therefore, when irbesartan and hydrochlorothiazide tablets and nonsteroidal anti-inflammatory agents are used concomitantly, the patient should be observed closely to determine if the desired effect of the diuretic is obtained.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:34.984308 · Updated: 2026-03-14T22:44:06.575433