Conjugated Estrogens Tablets
7277bab4-d8c2-4e4b-a0d9-5dfe1885d547
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Conjugated estrogens tablets is a mixture of estrogens indicated for: •Treatment of Moderate to Severe Vasomotor Symptoms due to Menopause. ( 1.1 ) •Treatment of Moderate to Severe Vulvar and Vaginal Atrophy due to Menopause ( 1.2 ) •Treatment of Hypoestrogenism due to Hypogonadism, Castration or Primary Ovarian Failure ( 1.3 ) •Treatment of Breast Cancer (for Palliation Only) in Appropriately Selected Women and Men with Metastatic Disease ( 1.4 ) •Treatment of Advanced Androgen-Dependent Carcinoma of the Prostate (for Palliation Only) ( 1.5 ) •Prevention of Postmenopausal Osteoporosis ( 1.6 )
Dosage and Administration
Generally, when estrogen therapy is prescribed for a postmenopausal woman with a uterus, a progestin should be considered to reduce the risk of endometrial cancer [see Boxed Warning ]. A woman without a uterus does not need progestin. In some cases, however, hysterectomized women with a history of endometriosis may need a progestin [see Warnings and Precautions (5.2, 5.16) ] . Use of estrogen-alone, or in combination with a progestin, should be with the lowest effective dose and for the shortest duration consistent with treatment goals and risks for the individual woman. Postmenopausal women should be re-evaluated periodically as clinically appropriate to determine if treatment is still necessary. Conjugated estrogens tablets may be taken without regard to meals.
Contraindications
Conjugated estrogens tablets therapy is contraindicated in individuals with any of the following conditions: • Undiagnosed abnormal genital bleeding [see Warnings and Precautions ( 5.2 )] • Breast cancer or a history of breast cancer except in appropriately selected patients being treated for metastatic disease [see Warnings and Precautions ( 5.2 )] • Estrogen-dependent neoplasia [see Warnings and Precautions ( 5.2 )] • Active DVT, PE, or a history of these conditions [see Warnings and Precautions ( 5.1 )] • Active arterial thromboembolic disease (for example stroke and MI), or a history of these conditions [see Warnings and Precautions ( 5.1 )] • Known anaphylactic reaction or angioedema with conjugated estrogens tablets [see Warnings and Precautions ( 5.7 , 5.15 )] • Hepatic impairment or disease [see Warnings and Precautions ( 5.11 )] • Protein C, protein S or antithrombin deficiency, or other known thrombophilic disorders.
Warnings and Precautions
•Estrogens increase the risk of gallbladder disease ( 5.4 ) •Discontinue estrogen if severe hypercalcemia, loss of vision, severe hypertriglyceridemia or cholestatic jaundice occurs ( 5.5 , 5.6 , 5.10, 5.11 ) •Monitor thyroid function in patients on thyroid replacement therapy ( 5.12 , 5.19 )
Adverse Reactions
Inform postmenopausal women of possible less serious but common adverse reactions of estrogen therapy such as headache, breast pain and tenderness, nausea and vomiting.
Drug Interactions
Data from a single-dose drug-drug interaction study involving CE and MPA indicate that the pharmacokinetic disposition of both drugs is not altered when the drugs are coadministered. No other clinical drug-drug interaction studies have been conducted with CE.
How Supplied
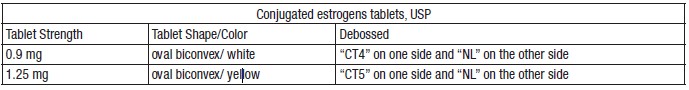
Conjugated estrogens tablets, USP Each white oval biconvex coated tablet contains 0.9 mg, in bottles of 100 (NDC 50742-390-01) and 1,000 (NDC 50742-390-10). Each yellow oval biconvex coated tablet contains 1.25 mg, in bottles of 100 (NDC 50742-391-01) and 1,000 (NDC 50742-391-10).
Storage and Handling
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Dispense in a well-closed container, as defined in the USP.
Patient Information
Conjugated estrogens tablets, USP ("kon′ju gay″ted es′troe jenz") Read this PATIENT INFORMATION before you start taking conjugated estrogens tablets and read what you get each time you refill your conjugated estrogens tablets prescription. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Description
WARNING: ENDOMETRIAL CANCER, CARDIOVASCULAR DISORDERS, BREAST CANCER and PROBABLE DEMENTIA See full prescribing information for complete boxed warning. Estrogen-Alone Therapy •There is an increased risk of endometrial cancer in a woman with a uterus who uses unopposed estrogens ( 5.2 ) • Estrogen-alone therapy should not be used for the prevention of cardiovascular disease or dementia ( 5.1 , 5.3 ) • Women's Health Initiative (WHI) estrogen-alone substudy reported increased risks of stroke and deep vein thrombosis (DVT) ( 5.1 ) • The WHI Memory Study (WHIMS) estrogen-alone ancillary study of WHI reported an increased risk of probable dementia in postmenopausal women 65 years of age and older ( 5.3 ) Estrogen Plus Progestin Therapy • Estrogen plus progestin therapy should not be used for the prevention of cardiovascular disease or dementia ( 5.1 , 5.3 ) • The WHI estrogen plus progestin substudy reported increased risks of stroke, DVT, pulmonary embolism (PE), and myocardial infarction (MI) ( 5.1 ) • The WHI estrogen plus progestin substudy reported increased risks of invasive breast cancer ( 5.2 ) • The WHIMS estrogen plus progestin ancillary study of WHI reported an increased risk of probable dementia in postmenopausal women 65 years of age and older ( 5.3 )
Medication Information
Recent Major Changes
WHAT IS THE MOST IMPORTANT INFORMATION I SHOULD KNOW ABOUT CONJUGATED ESTROGENS TABLETS (AN ESTROGEN MIXTURE)?
•Using estrogen-alone may increase your chance of getting cancer of the uterus (womb)
Report any unusual vaginal bleeding right away while you are using conjugated estrogens tablets. Vaginal bleeding after menopause may be a warning sign of cancer of the uterus (womb). Your healthcare provider should check any unusual vaginal bleeding to find the cause.
•Do not use estrogen-alone to prevent heart disease, heart attacks, or dementia (decline of brain function)
•Using estrogen-alone may increase your chances of getting strokes or blood clots
•Using estrogen-alone may increase your chance of getting dementia, based on a study of women 65 years of age or older
•Do not use estrogens with progestins to prevent heart disease, heart attacks, strokes, or dementia
•Using estrogens with progestins may increase your chances of getting heart attacks, strokes, breast cancer, or blood clots
•Using estrogens with progestins may increase your chance of getting dementia, based on a study of women 65 years of age or older
•You and your healthcare provider should talk regularly about whether you still need treatment with conjugated estrogens tablets
Warnings and Precautions
•Estrogens increase the risk of gallbladder disease ( 5.4 ) •Discontinue estrogen if severe hypercalcemia, loss of vision, severe hypertriglyceridemia or cholestatic jaundice occurs ( 5.5 , 5.6 , 5.10, 5.11 ) •Monitor thyroid function in patients on thyroid replacement therapy ( 5.12 , 5.19 )
Indications and Usage
Conjugated estrogens tablets is a mixture of estrogens indicated for: •Treatment of Moderate to Severe Vasomotor Symptoms due to Menopause. ( 1.1 ) •Treatment of Moderate to Severe Vulvar and Vaginal Atrophy due to Menopause ( 1.2 ) •Treatment of Hypoestrogenism due to Hypogonadism, Castration or Primary Ovarian Failure ( 1.3 ) •Treatment of Breast Cancer (for Palliation Only) in Appropriately Selected Women and Men with Metastatic Disease ( 1.4 ) •Treatment of Advanced Androgen-Dependent Carcinoma of the Prostate (for Palliation Only) ( 1.5 ) •Prevention of Postmenopausal Osteoporosis ( 1.6 )
Dosage and Administration
Generally, when estrogen therapy is prescribed for a postmenopausal woman with a uterus, a progestin should be considered to reduce the risk of endometrial cancer [see Boxed Warning ]. A woman without a uterus does not need progestin. In some cases, however, hysterectomized women with a history of endometriosis may need a progestin [see Warnings and Precautions (5.2, 5.16) ] . Use of estrogen-alone, or in combination with a progestin, should be with the lowest effective dose and for the shortest duration consistent with treatment goals and risks for the individual woman. Postmenopausal women should be re-evaluated periodically as clinically appropriate to determine if treatment is still necessary. Conjugated estrogens tablets may be taken without regard to meals.
Contraindications
Conjugated estrogens tablets therapy is contraindicated in individuals with any of the following conditions: • Undiagnosed abnormal genital bleeding [see Warnings and Precautions ( 5.2 )] • Breast cancer or a history of breast cancer except in appropriately selected patients being treated for metastatic disease [see Warnings and Precautions ( 5.2 )] • Estrogen-dependent neoplasia [see Warnings and Precautions ( 5.2 )] • Active DVT, PE, or a history of these conditions [see Warnings and Precautions ( 5.1 )] • Active arterial thromboembolic disease (for example stroke and MI), or a history of these conditions [see Warnings and Precautions ( 5.1 )] • Known anaphylactic reaction or angioedema with conjugated estrogens tablets [see Warnings and Precautions ( 5.7 , 5.15 )] • Hepatic impairment or disease [see Warnings and Precautions ( 5.11 )] • Protein C, protein S or antithrombin deficiency, or other known thrombophilic disorders.
Adverse Reactions
Inform postmenopausal women of possible less serious but common adverse reactions of estrogen therapy such as headache, breast pain and tenderness, nausea and vomiting.
Drug Interactions
Data from a single-dose drug-drug interaction study involving CE and MPA indicate that the pharmacokinetic disposition of both drugs is not altered when the drugs are coadministered. No other clinical drug-drug interaction studies have been conducted with CE.
Storage and Handling
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Dispense in a well-closed container, as defined in the USP.
How Supplied
Conjugated estrogens tablets, USP Each white oval biconvex coated tablet contains 0.9 mg, in bottles of 100 (NDC 50742-390-01) and 1,000 (NDC 50742-390-10). Each yellow oval biconvex coated tablet contains 1.25 mg, in bottles of 100 (NDC 50742-391-01) and 1,000 (NDC 50742-391-10).
Patient Information
Conjugated estrogens tablets, USP
("kon′ju gay″ted es′troe jenz")
Read this PATIENT INFORMATION before you start taking conjugated estrogens tablets and read what you get each time you refill your conjugated estrogens tablets prescription. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Description
WARNING: ENDOMETRIAL CANCER, CARDIOVASCULAR DISORDERS, BREAST CANCER and PROBABLE DEMENTIA See full prescribing information for complete boxed warning. Estrogen-Alone Therapy •There is an increased risk of endometrial cancer in a woman with a uterus who uses unopposed estrogens ( 5.2 ) • Estrogen-alone therapy should not be used for the prevention of cardiovascular disease or dementia ( 5.1 , 5.3 ) • Women's Health Initiative (WHI) estrogen-alone substudy reported increased risks of stroke and deep vein thrombosis (DVT) ( 5.1 ) • The WHI Memory Study (WHIMS) estrogen-alone ancillary study of WHI reported an increased risk of probable dementia in postmenopausal women 65 years of age and older ( 5.3 ) Estrogen Plus Progestin Therapy • Estrogen plus progestin therapy should not be used for the prevention of cardiovascular disease or dementia ( 5.1 , 5.3 ) • The WHI estrogen plus progestin substudy reported increased risks of stroke, DVT, pulmonary embolism (PE), and myocardial infarction (MI) ( 5.1 ) • The WHI estrogen plus progestin substudy reported increased risks of invasive breast cancer ( 5.2 ) • The WHIMS estrogen plus progestin ancillary study of WHI reported an increased risk of probable dementia in postmenopausal women 65 years of age and older ( 5.3 )
Section 42229-5
Estrogen-Alone Therapy
Section 51945-4
10 Overdosage
Overdosage of estrogen may cause nausea, vomiting, breast tenderness, abdominal pain, drowsiness and fatigue, and withdrawal bleeding may occur in women. Treatment of overdose consists of discontinuation of conjugated estrogens tablets therapy with institution of appropriate symptomatic care.
15 References
1. Rossouw JE, et al. Postmenopausal Hormone Therapy and Risk of Cardiovascular Disease by Age and Years Since Menopause. JAMA. 2007;297:1465–1477.
2. Hsia J, et al. Conjugated Equine Estrogens and Coronary Heart Disease. Arch Int Med. 2006;166:357–365.
3. Curb JD, et al. Venous Thrombosis and Conjugated Equine Estrogen in Women Without a Uterus. Arch Int Med. 2006;166:772–780.
4. Cushman M, et al. Estrogen Plus Progestin and Risk of Venous Thrombosis. JAMA. 2004;292:1573–1580.
5. Stefanick ML, et al. Effects of Conjugated Equine Estrogens on Breast Cancer and Mammography Screening in Postmenopausal Women With Hysterectomy. JAMA. 2006;295:1647–1657.
6. Chlebowski RT, et al. Influence of Estrogen Plus Progestin on Breast Cancer and Mammography in Healthy Postmenopausal Women. JAMA. 2003;289:3234–3253.
7. Anderson GL, et al. Effects of Estrogen Plus Progestin on Gynecologic Cancers and Associated Diagnostic Procedures. JAMA. 2003;290:1739–1748.
8. Shumaker SA, et al. Conjugated Equine Estrogens and Incidence of Probable Dementia and Mild Cognitive Impairment in Postmenopausal Women. JAMA. 2004;291:2947–2958.
9. Jackson RD, et al. Effects of Conjugated Equine Estrogen on Risk of Fractures and BMD in Postmenopausal Women With Hysterectomy: Results From the Women's Health Initiative Randomized Trial. J Bone Miner Res. 2006;21:817–828.
10. Hendrix SL, et al. Effects of Conjugated Equine Estrogen on Stroke in the Women's Health Initiative. Circulation. 2006;113:2425–2434.
8.1 Pregnancy
Risk Summary
Conjugated estrogens tablets is not indicated for use during pregnancy.
There are no data with the use of conjugated estrogens tablet in pregnant women; however, epidemiologic studies and meta-analyses have not found an increased risk of genital or nongenital birth defects (including cardiac anomalies and limb-reduction defects) following exposure to combined hormonal contraceptives (estrogen and progestins) before conception or during early pregnancy.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
Estrogens and progestins and metabolites are present in human milk. These hormones can reduce milk production in breast-feeding women. This reduction can occur at any time but is less likely to occur once breast-feeding is well established. The developmental and health benefits of breast-feeding should be considered along with the mother’s clinical need for conjugated estrogens tablets and any potential adverse effects on the breast-fed child from conjugated estrogens tablets or from the underlying maternal condition.
11 Description
Conjugated estrogens tablets, USP for oral administration contains a mixture of CE purified from pregnant mares' urine and consists of the sodium salts of water-soluble estrogen sulfates blended to represent the average composition of material derived from pregnant mares' urine. It is a mixture of sodium estrone sulfate and sodium equilin sulfate. It contains concomitant components as sodium sulfate conjugates, 17α-dihydroequilin, 17α-estradiol, and 17β-dihydroequilin. Tablets for oral administration are available in 0.9 mg, and 1.25 mg strengths of CE.
Conjugated estrogens tablets USP, 0.9 mg, and 1.25 mg tablets also contain the following inactive ingredients: hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, sodium acetate anhydrous, and titanium dioxide. Each tablet strength contains the following colors:
FDA approved dissolution test specifications differ from USP.
16.1 How Supplied
Conjugated estrogens tablets, USP
- Each white oval biconvex coated tablet contains 0.9 mg, in bottles of 100 (NDC 50742-390-01) and 1,000 (NDC 50742-390-10).
- Each yellow oval biconvex coated tablet contains 1.25 mg, in bottles of 100 (NDC 50742-391-01) and 1,000 (NDC 50742-391-10).
5.14 Hypocalcemia
Estrogen therapy should be used with caution in all patients with hypoparathyroidism as estrogen-induced hypocalcemia may occur.
5.5 Hypercalcemia
Estrogen administration may lead to severe hypercalcemia in patients with breast cancer and bone metastases. If hypercalcemia occurs, use of the drug should be stopped and appropriate measures taken to reduce the serum calcium level.
8.4 Pediatric Use
Estrogen therapy has been used for the induction of puberty in adolescents with some forms of pubertal delay. Safety and effectiveness in pediatric patients have not otherwise been established.
Large and repeated doses of estrogen over an extended time period have been shown to accelerate epiphyseal closure, which could result in short stature if treatment is initiated before the completion of physiologic puberty in normally developing children. If estrogen is administered to patients whose bone growth is not complete, periodic monitoring of bone maturation and effects on epiphyseal centers is recommended during estrogen administration.
Estrogen treatment of prepubertal girls also induces premature breast development and vaginal cornification, and may induce vaginal bleeding. In boys, estrogen treatment may modify the normal pubertal process and induce gynecomastia.
8.5 Geriatric Use
There have not been sufficient numbers of geriatric patients involved in studies utilizing conjugated estrogens tablets to determine whether those over 65 years of age differ from younger subjects in their response to conjugated estrogens tablets.
4 Contraindications
Conjugated estrogens tablets therapy is contraindicated in individuals with any of the following conditions:
• Undiagnosed abnormal genital bleeding [see Warnings and Precautions (5.2)]
• Breast cancer or a history of breast cancer except in appropriately selected patients being treated for metastatic disease [see Warnings and Precautions (5.2)]
• Estrogen-dependent neoplasia [see Warnings and Precautions (5.2)]
• Active DVT, PE, or a history of these conditions [see Warnings and Precautions (5.1)]
• Active arterial thromboembolic disease (for example stroke and MI), or a history of these conditions [see Warnings and Precautions (5.1)]
• Known anaphylactic reaction or angioedema with conjugated estrogens tablets [see Warnings and Precautions (5.7,5.15)]
• Hepatic impairment or disease [see Warnings and Precautions (5.11)]
• Protein C, protein S or antithrombin deficiency, or other known thrombophilic disorders.
6 Adverse Reactions
The following serious adverse reactions are discussed elsewhere in labeling:
•Cardiovascular Disorders [see Boxed Warning, Warnings and Precautions (5.1)]
•Malignant Neoplasms [see Boxed Warning, Warnings and Precautions (5.2)]
7 Drug Interactions
Data from a single-dose drug-drug interaction study involving CE and MPA indicate that the pharmacokinetic disposition of both drugs is not altered when the drugs are coadministered. No other clinical drug-drug interaction studies have been conducted with CE.
5.13 Fluid Retention
Estrogens may cause some degree of fluid retention. Women with conditions that might be influenced by this factor, such as cardiac or renal dysfunction, warrant careful observation when estrogens are prescribed.
8.6 Renal Impairment
The effect of renal impairment on the pharmacokinetics of conjugated estrogens tablets has not been studied.
12.2 Pharmacodynamics
There are no pharmacodynamic data for conjugated estrogens tablets.
17.1 Vaginal Bleeding
Inform postmenopausal women of the importance of reporting vaginal bleeding to their healthcare provider as soon as possible [see Warnings and Precautions (5.2)].
5.18 Laboratory Tests
Serum follicle stimulating hormone (FSH) and estradiol levels have not been shown to be useful in the management of moderate to severe vasomotor symptoms and moderate to severe symptoms of vulvar and vaginal atrophy.
Laboratory parameters may be useful in guiding dosage for the treatment of hypoestrogenism due to hypogonadism, castration and primary ovarian failure.
5.3 Probable Dementia
In the WHIMS estrogen-alone ancillary study of WHI, a population of 2,947 hysterectomized women 65 to 79 years of age was randomized to daily CE (0.625 mg)-alone or placebo.
After an average follow-up of 5.2 years, 28 women in the estrogen-alone group and 19 women in the placebo group were diagnosed with probable dementia. The relative risk of probable dementia for CE-alone versus placebo was 1.49 (95% CI 0.83–2.66). The absolute risk of probable dementia for CE-alone versus placebo was 37 versus 25 cases per 10,000 women-years8 [see Use in Specific Populations (8.5), and Clinical Studies (14.6)].
In the WHIMS estrogen plus progestin ancillary study of WHI, a population of 4,532 postmenopausal women 65 to 79 years of age was randomized to daily CE (0.625 mg) plus MPA (2.5 mg) or placebo. After an average follow-up of 4 years, 40 women in the CE plus MPA group and 21 women in the placebo group were diagnosed with probable dementia. The relative risk of probable dementia for CE plus MPA versus placebo was 2.05 (95% CI 1.21–3.48). The absolute risk of probable dementia for CE plus MPA versus placebo was 45 versus 22 cases per 10,000 women-years8 [see Use in Specific Populations (8.5), and Clinical Studies (14.6)].
When data from the two populations in the WHIMS estrogen-alone and estrogen plus progestin ancillary studies were pooled as planned in the WHIMS protocol, the reported overall relative risk for probable dementia was 1.76 (95% CI 1.19–2.60). Since both substudies were conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women8 [see Use in Specific Populations (8.5), and Clinical Studies (14.6)].
8.7 Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of conjugated estrogens tablets has not been studied.
1 Indications and Usage
Conjugated estrogens tablets is a mixture of estrogens indicated for:
•Treatment of Moderate to Severe Vasomotor Symptoms due to Menopause. (1.1)
•Treatment of Moderate to Severe Vulvar and Vaginal Atrophy due to Menopause (1.2)
•Treatment of Hypoestrogenism due to Hypogonadism, Castration or Primary Ovarian Failure (1.3)
•Treatment of Breast Cancer (for Palliation Only) in Appropriately Selected Women and Men with Metastatic Disease (1.4)
•Treatment of Advanced Androgen-Dependent Carcinoma of the Prostate (for Palliation Only) (1.5)
•Prevention of Postmenopausal Osteoporosis (1.6)
5.4 Gallbladder Disease
A 2- to 4-fold increase in the risk of gallbladder disease requiring surgery in postmenopausal women receiving estrogens has been reported.
12.1 Mechanism of Action
Endogenous estrogens are largely responsible for the development and maintenance of the female reproductive system and secondary sexual characteristics. Although circulating estrogens exist in a dynamic equilibrium of metabolic interconversions, estradiol is the principal intracellular human estrogen and is substantially more potent than its metabolites, estrone and estriol, at the receptor level.
The primary source of estrogen in normally cycling adult women is the ovarian follicle, which secretes 70 to 500 mcg of estradiol daily, depending on the phase of the menstrual cycle. After menopause, most endogenous estrogen is produced by conversion of androstenedione, secreted by the adrenal cortex, to estrone in the peripheral tissues. Thus, estrone and the sulfate-conjugated form, estrone sulfate, are the most abundant circulating estrogens in postmenopausal women.
Estrogens act through binding to nuclear receptors in estrogen-responsive tissues. To date, two estrogen receptors have been identified. These vary in proportion from tissue to tissue.
Circulating estrogens modulate the pituitary secretion of the gonadotropins, luteinizing hormone (LH) and FSH, through a negative feedback mechanism. Estrogens act to reduce the elevated levels of these gonadotropins seen in postmenopausal women.
5.6 Visual Abnormalities
Retinal vascular thrombosis has been reported in patients receiving estrogens. Discontinue medication pending examination if there is sudden partial or complete loss of vision, or a sudden onset of proptosis, diplopia, or migraine. If examination reveals papilledema or retinal vascular lesions, estrogens should be permanently discontinued.
16.2 Storage and Handling
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Dispense in a well-closed container, as defined in the USP.
5 Warnings and Precautions
5.15 Hereditary Angioedema
Exogenous estrogens may exacerbate symptoms of angioedema in women with hereditary angioedema.
7.1 Metabolic Interactions
In vitro and in vivo studies have shown that estrogens are metabolized partially by cytochrome P450 3A4 (CYP3A4). Therefore, inducers or inhibitors of CYP3A4 may affect estrogen drug metabolism. Inducers of CYP3A4, such as St. John's Wort (Hypericum perforatum) preparations, phenobarbital, carbamazepine, and rifampin, may reduce plasma concentrations of estrogens, possibly resulting in a decrease in therapeutic effects and/or changes in the uterine bleeding profile. Inhibitors of CYP3A4, such as erythromycin, clarithromycin, ketoconazole, itraconazole, ritonavir and grapefruit juice, may increase plasma concentrations of estrogens and may result in side effects.
2 Dosage and Administration
Generally, when estrogen therapy is prescribed for a postmenopausal woman with a uterus, a progestin should be considered to reduce the risk of endometrial cancer [see Boxed Warning].
A woman without a uterus does not need progestin. In some cases, however, hysterectomized women with a history of endometriosis may need a progestin [see Warnings and Precautions (5.2, 5.16)].
Use of estrogen-alone, or in combination with a progestin, should be with the lowest effective dose and for the shortest duration consistent with treatment goals and risks for the individual woman. Postmenopausal women should be re-evaluated periodically as clinically appropriate to determine if treatment is still necessary.
Conjugated estrogens tablets may be taken without regard to meals.
5.9 Elevated Blood Pressure
In a small number of case reports, substantial increases in blood pressure have been attributed to idiosyncratic reactions to estrogens. In a large, randomized, placebo-controlled clinical trial, a generalized effect of estrogen therapy on blood pressure was not seen.
3 Dosage Forms and Strengths
5.1 Cardiovascular Disorders
An increased risk of stroke and DVT has been reported with estrogen-alone therapy. An increased risk of PE, DVT, stroke and MI has been reported with estrogen plus progestin therapy. Should any of these events occur or be suspected, estrogen with or without progestin therapy should be discontinued immediately.
Risk factors for arterial vascular disease (for example, hypertension, diabetes mellitus, tobacco use, hypercholesterolemia, and obesity) and/or venous thromboembolism (VTE) (for example, personal or family history of VTE, obesity, and systemic lupus erythematosus) should be managed appropriately.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post-approval use of conjugated estrogens tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible always to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Study Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During the first year of a 2-year clinical trial with 2,333 postmenopausal women with a uterus between 40 and 65 years of age (88% Caucasian), 1,012 women were treated with CE , and 332 were treated with placebo.
Table 1 summarizes treatment-related adverse reactions that occurred at a rate of ≥ 1% in any treatment group.
| Conjugated estrogens tablets 0.625 mg (n=348) |
Conjugated estrogens tablets 0.45 mg (n=338) |
Conjugated estrogens tablets 0.3 mg (n=326) |
Placebo
(n=332) |
|
|---|---|---|---|---|
|
Body as a whole |
||||
|
Abdominal pain |
38 (11) |
28 (8) |
30 (9) |
21 (6) |
|
Asthenia |
16 (5) |
8 (2) |
14 (4) |
3 (1) |
|
Back pain |
18 (5) |
11 (3) |
13 (4) |
4 (1) |
|
Chest pain |
2 (1) |
3 (1) |
4 (1) |
2 (1) |
|
Generalized edema |
7 (2) |
6 (2) |
4 (1) |
8 (2) |
|
Headache |
45 (13) |
47 (14) |
44 (13) |
46 (14) |
|
Moniliasis |
5 (1) |
4 (1) |
4 (1) |
1 (0) |
|
Pain |
17 (5) |
10 (3) |
12 (4) |
14 (4) |
|
Pelvic pain |
10 (3) |
9 (3) |
8 (2) |
4 (1) |
|
Cardiovascular system |
||||
|
Hypertension |
4 (1) |
4 (1) |
7 (2) |
5 (2) |
|
Migraine |
7 (2) |
1 (0) |
0 |
3 (1) |
|
Palpitation |
3 (1) |
3 (1) |
3 (1) |
4 (1) |
|
Vasodilatation |
2 (1) |
2 (1) |
3 (1) |
5 (2) |
|
Digestive system |
||||
|
Constipation |
7 (2) |
6 (2) |
4 (1) |
3 (1) |
|
Diarrhea |
4 (1) |
5 (1) |
5 (2) |
8 (2) |
|
Dyspepsia |
7 (2) |
5 (1) |
6 (2) |
14 (4) |
|
Eructation |
1 (0) |
1 (0) |
4 (1) |
1 (0) |
|
Flatulence |
22 (6) |
18 (5) |
13 (4) |
8 (2) |
|
Increased appetite |
4 (1) |
1 (0) |
1 (0) |
2 (1) |
|
Nausea |
16 (5) |
10 (3) |
15 (5) |
16 (5) |
|
Metabolic and nutritional |
||||
|
Hyperlipidemia |
2 (1) |
4 (1) |
3 (1) |
2 (1) |
|
Peripheral edema |
5 (1) |
2 (1) |
4 (1) |
3 (1) |
|
Weight gain |
11 (3) |
10 (3) |
8 (2) |
14 (4) |
|
Musculoskeletal system |
||||
|
Arthralgia |
6 (2) |
3 (1) |
2 (1) |
5 (2) |
|
Leg cramps |
10 (3) |
5 (1) |
9 (3) |
4 (1) |
|
Myalgia |
2 (1) |
1 (0) |
4 (1) |
1 (0) |
|
Nervous system |
||||
|
Anxiety |
6 (2) |
4 (1) |
2 (1) |
4 (1) |
|
Depression |
17 (5) |
15 (4) |
10 (3) |
17 (5) |
|
Dizziness |
9 (3) |
7 (2) |
4 (1) |
5 (2) |
|
Emotional lability |
3 (1) |
4 (1) |
5 (2) |
8 (2) |
|
Hypertonia |
1 (0) |
1 (0) |
5 (2) |
3 (1) |
|
Insomnia |
16 (5) |
10 (3) |
13 (4) |
14 (4) |
|
Nervousness |
9 (3) |
12 (4) |
2 (1) |
6 (2) |
|
Skin and appendages |
||||
|
Acne |
3 (1) |
1 (0) |
8 (2) |
3 (1) |
|
Alopecia |
6 (2) |
6 (2) |
5 (2) |
2 (1) |
|
Hirsutism |
4 (1) |
2 (1) |
1 (0) |
0 |
|
Pruritus |
11 (3) |
11 (3) |
10 (3) |
3 (1) |
|
Rash |
6 (2) |
3 (1) |
1 (0) |
2 (1) |
|
Skin discoloration |
4 (1) |
2 (1) |
0 |
1 (0) |
|
Sweating |
4 (1) |
1 (0) |
3 (1) |
4 (1) |
|
Urogenital system |
||||
|
Breast disorder |
6 (2) |
3 (1) |
3 (1) |
6 (2) |
|
Breast enlargement |
3 (1) |
4 (1) |
7 (2) |
3 (1) |
|
Breast neoplasm |
4 (1) |
4 (1) |
7 (2) |
7 (2) |
|
Breast pain |
37 (11) |
39 (12) |
24 (7) |
26 (8) |
|
Cervix disorder |
8 (2) |
4 (1) |
5 (2) |
0 |
|
Dysmenorrhea |
12 (3) |
10 (3) |
4 (1) |
2 (1) |
|
Endometrial disorder |
4 (1) |
2 (1) |
2 (1) |
0 |
|
Endometrial hyperplasia |
16 (5) |
8 (2) |
1 (0) |
0 |
|
Leukorrhea |
17 (5) |
17 (5) |
12 (4) |
6 (2) |
|
Metrorrhagia |
11 (3) |
4 (1) |
3 (1) |
1 (0) |
|
Urinary tract infection |
1 (0) |
2 (1) |
1 (0) |
4 (1) |
|
Uterine fibroids enlarged |
6 (2) |
1 (0) |
2 (1) |
2 (1) |
|
Uterine spasm |
11 (3) |
5 (1) |
3 (1) |
2 (1) |
|
Vaginal dryness |
1 (0) |
2 (1) |
1 (0) |
6 (2) |
|
Vaginal hemorrhage |
46 (13) |
13 (4) |
6 (2) |
0 |
|
Vaginal moniliasis |
14 (4) |
10 (3) |
12 (4) |
5 (2) |
|
Vaginitis |
18 (5) |
7 (2) |
9 (3) |
1 (0) |
8 Use in Specific Populations
•Lactation: Estrogen administration to lactating women has been shown to decrease the quantity and quality of breast milk(8.2)
•Geriatric Use: An increased risk of probable dementia in women over 65 years of age was reported in the Women's Health Initiative Memory ancillary studies of the Women's Health Initiative (5.3, 8.5)
17 Patient Counseling Information
Advise the patients to read the FDA-approved patient labeling (Patient Information).
14.1 Effects On Vasomotor Symptoms
In the first year of the Health and Osteoporosis, Progestin and Estrogen (HOPE) Study, a total of 2,805 postmenopausal women (average age 53.3 ± 4.9 years) were randomly assigned to one of eight treatment groups of either placebo or CE, with or without MPA. Efficacy for vasomotor symptoms was assessed during the first 12 weeks of treatment in a subset of symptomatic women (n = 241) who had at least seven moderate to severe hot flushes daily, or at least 50 moderate to severe hot flushes during the week before randomization. Conjugated estrogens tablets (0.3 mg, 0.45 mg, and 0.625 mg tablets) was shown to be statistically better than placebo at weeks 4 and 12 for relief of both frequency and severity of moderate to severe vasomotor symptoms. Table 3 shows the adjusted mean number of hot flushes in the conjugated estrogens tablets 0.3 mg, 0.45 mg, and 0.625 mg and placebo groups during the initial 12-week period.
5.16 Exacerbation of Endometriosis
A few cases of malignant transformation of residual endometrial implants have been reported in women treated post-hysterectomy with estrogen-alone therapy. For women known to have residual endometriosis post-hysterectomy, the addition of progestin should be considered.
14.4 Effects On Female Hypogonadism
In clinical studies of delayed puberty due to female hypogonadism, breast development was induced by doses as low as 0.15 mg. The dosage may be gradually titrated upward at 6-to 12 month intervals as needed to achieve appropriate bone age advancement and eventual epiphyseal closure. Clinical studies suggest that doses of 0.15 mg, 0.3 mg, and 0.6 mg are associated with mean ratios of bone age advancement to chronological age progression (ΔBA/ΔCA) of 1.1, 1.5, and 2.1, respectively. (Conjugated estrogens tablets in the dose strength of 0.15 mg is not available commercially). Available data suggest that chronic dosing with 0.625 mg is sufficient to induce artificial cyclic menses with sequential progestin treatment and to maintain bone mineral density after skeletal maturity is achieved.
5.12 exacerbation of Hypothyroidism
Estrogen administration leads to increased thyroid-binding globulin (TBG) levels. Women with normal thyroid function can compensate for the increased TBG by making more thyroid hormone, thus maintaining free T4 and T3 serum concentrations in the normal range. Women dependent on thyroid hormone replacement therapy who are also receiving estrogens may require increased doses of their thyroid replacement therapy. These women should have their thyroid function monitored in order to maintain their free thyroid hormone levels in an acceptable range.
5.17 Exacerbation of Other Conditions
Estrogen therapy may cause an exacerbation of asthma, diabetes mellitus, epilepsy, migraine, porphyria, systemic lupus erythematosus, and hepatic hemangiomas and should be used with caution in women with these conditions.
14.5 Women's Health Initiative Studies
The WHI enrolled approximately 27,000 predominantly healthy postmenopausal women in two substudies to assess the risks and benefits of daily oral CE (0.625 mg)-alone or in combination with MPA (2.5 mg) compared to placebo in the prevention of certain chronic diseases. The primary endpoint was the incidence of CHD (defined as nonfatal MI, silent MI and CHD death), with invasive breast cancer as the primary adverse outcome. A "global index" included the earliest occurrence of CHD, invasive breast cancer, stroke, PE, endometrial cancer (only in the CE plus MPA substudy), colorectal cancer, hip fracture, or death due to other causes. These substudies did not evaluate the effects of CE-alone or CE plus MPA on menopausal symptoms.
5.19 Drug Laboratory Test Interactions
• Accelerated prothrombin time, partial thromboplastin time, and platelet aggregation time; increased platelet count; increased factors II, VII antigen, VIII antigen, VIII coagulant activity, IX, X, XII, VII-X complex, II-VII-X complex, and beta-thromboglobulin; decreased levels of anti-factor Xa and antithrombin III, decreased antithrombin III activity; increased levels of fibrinogen and fibrinogen activity; increased plasminogen antigen and activity.
• Increased thyroid-binding globulin (TBG) levels leading to increased circulating total thyroid hormone levels as measured by protein-bound iodine (PBI), T4 levels (by column or by radioimmunoassay) or T3 levels by radioimmunoassay. T3 resin uptake is decreased, reflecting the elevated TBG. Free T4 and free T3 concentrations are unaltered. Patients on thyroid replacement therapy may require higher doses of thyroid hormone.
• Other binding proteins may be elevated in serum, for example, corticosteroid binding globulin (CBG), sex hormone-binding globulin (SHBG), leading to increased total circulating corticosteroids and sex steroids, respectively. Free hormone concentrations, such as testosterone and estradiol, may be decreased. Other plasma proteins may be increased (angiotensinogen/renin substrate, alpha-1-antitrypsin, ceruloplasmin).
• Increased plasma high-density lipoprotein (HDL) and HDL2 cholesterol subfraction, reduced low-density lipoprotein (LDL) cholesterol, increased triglyceride levels.
• Impaired glucose tolerance.
5.7 Anaphylactic Reaction and Angioedema
Cases of anaphylaxis, which developed within minutes to hours after taking conjugated estrogens tablets and require emergency medical management, have been reported in the postmarketing setting. Skin (hives, pruritis, swollen lips-tongue-face) and either respiratory tract (respiratory compromise) or gastrointestinal tract (abdominal pain, vomiting) involvement has been noted.
Angioedema involving the tongue, larynx, face, hands, and feet requiring medical intervention has occurred postmarketing in patients taking conjugated estrogens tablets. If angioedema involves the tongue, glottis, or larynx, airway obstruction may occur. Patients who develop an anaphylactic reaction with or without angioedema after treatment with conjugated estrogens tablets should not receive conjugated estrogens tablets again.
14.2 Effects On Vulvar and Vaginal Atrophy
Results of vaginal maturation indexes at cycles 6 and 13 showed that the differences from placebo were statistically significant (p < 0.001) for all treatment groups. (CE alone and CE/MPA treatment groups).
5.10 exacerbation of Hypertriglyceridemia
In women with pre-existing hypertriglyceridemia, estrogen therapy may be associated with elevations of plasma triglycerides leading to pancreatitis. Consider discontinuation of treatment if pancreatitis occurs.
14.6 Women's Health Initiative Memory Study
The WHIMS estrogen-alone ancillary study of WHI enrolled 2,947 predominantly healthy hysterectomized postmenopausal women 65 to 79 years of age (45 % were 65 to 69 years of age; 36 % were 70 to 74 years of age; 19 % were 75 years of age and older) to evaluate the effects of daily CE (0.625 mg)- alone on the incidence of probable dementia (primary outcome) compared to placebo.
After an average follow-up of 5.2 years, the relative risk of probable dementia for CE-alone versus placebo was 1.49 (95 % CI 0.83–2.66). The absolute risk of probable dementia for CE-alone versus placebo was 37 versus 25 cases per 10,000 women-years. Probable dementia as defined in this study included Alzheimer's disease (AD), vascular dementia (VaD) and mixed types (having features of both AD and VaD). The most common classification of probable dementia in the treatment group and the placebo groups was AD. Since the ancillary study was conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women [see Warnings and Precautions (5.3) , and Use in Specific Populations (8.5)].
The WHIMS estrogen plus progestin ancillary study enrolled 4,532 predominantly healthy postmenopausal women 65 years of age and older (47 % were 65 to 69 years of age; 35 % were 70 to 74 years; 18 % were 75 years of age and older) to evaluate the effects of daily CE (0.625 mg) plus MPA (2.5 mg) on the incidence of probable dementia (primary outcome) compared to placebo.
After an average follow-up of 4 years, the relative risk of probable dementia for CE plus MPA was 2.05 (95 % CI, 1.21–3.48). The absolute risk of probable dementia for CE (0.625 mg) plus MPA (2.5 mg) versus placebo was 45 versus 22 per 10,000 women-years. Probable dementia as defined in this study included AD, VaD and mixed types (having features of both AD and VaD). The most common classification of probable dementia in both the treatment and placebo groups was AD. Since the ancillary study was conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women [see Warnings and Precautions (5.3), and Use in Specific Populations (8.5)].
When data from the two populations were pooled as planned in the WHIMS protocol, the reported overall relative risk for probable dementia was 1.76 (95 percent CI 1.19–2.60). Differences between groups became apparent in the first year of treatment. It is unknown whether these findings apply to younger postmenopausal women [see Warnings and Precautions (5.3), and Use in Specific Populations (8.5)].
2.6 Prevention of Postmenopausal Osteoporosis
Conjugated estrogens tablets therapy may be given continuously, with no interruption in therapy, or in cyclical regimens (regimens such as 25 days on drug followed by 5 days off drug), as is medically appropriate on an individual basis.
Patients should be treated with the lowest effective dose. Generally, women should be started at 0.3 mg conjugated estrogens tablets daily. Subsequent dosage adjustment may be made based upon the individual clinical and bone mineral density responses. This dose should be periodically reassessed by the healthcare provider.
17.2 Possible Serious Adverse Reactions With Estrogens
Inform postmenopausal women of possible serious adverse reactions of estrogen therapy including Cardiovascular Disorders, Malignant Neoplasms, and Probable Dementia [see Warnings and Precautions (5.1, 5.2, 5.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term continuous administration of natural and synthetic estrogens in certain animal species increases the frequency of carcinomas of the breast, uterus, cervix, vagina, testis, and liver. [see Warnings and Precautions (5.2)].
5.11 Hepatic Impairment And/or Past History of Cholestatic Jaundice
Estrogens may be poorly metabolized in patients with impaired liver function. For women with a history of cholestatic jaundice associated with past estrogen use or with pregnancy, caution should be exercised, and in the case of recurrence, medication should be discontinued.
5.8 Addition of A Progestin When A Woman Has Not Had A Hysterectomy
Studies of the addition of a progestin for 10 or more days of a cycle of estrogen administration or daily with estrogen in a continuous regimen, have reported a lowered incidence of endometrial hyperplasia than would be induced by estrogen treatment alone. Endometrial hyperplasia may be a precursor to endometrial cancer.
There are, however, possible risks that may be associated with the use of progestins with estrogens compared to estrogen-alone regimens. These include an increased risk of breast cancer.
17.3 Possible Less Serious But Common Adverse Reactions With Estrogens
Inform postmenopausal women of possible less serious but common adverse reactions of estrogen therapy such as headache, breast pain and tenderness, nausea and vomiting.
2.1 Treatment of Moderate to Severe Vasomotor Symptoms Due to Menopause
Patients should be treated with the lowest effective dose. Generally, women should be started at 0.3 mg conjugated estrogens tablets daily. Subsequent dosage adjustment may be made based upon the individual patient response. This dose should be periodically reassessed by the healthcare provider.
Conjugated estrogens tablets therapy may be given continuously, with no interruption in therapy, or in cyclical regimens (regimens such as 25 days on drug followed by 5 days off drug), as is medically appropriate on an individual basis.
Warning: Endometrial Cancer, Cardiovascular Disorders, Breast Cancer and Probable Dementia
WARNING: ENDOMETRIAL CANCER, CARDIOVASCULAR DISORDERS, BREAST CANCER and PROBABLE DEMENTIA
See full prescribing information for complete boxed warning.
Estrogen-Alone Therapy
•There is an increased risk of endometrial cancer in a woman with a uterus who uses unopposed estrogens ( 5.2 )
•Estrogen-alone therapy should not be used for the prevention of cardiovascular disease or dementia ( 5.1, 5.3)
•Women's Health Initiative (WHI) estrogen-alone substudy reported increased risks of stroke and deep vein thrombosis (DVT) ( 5.1 )
•The WHI Memory Study (WHIMS) estrogen-alone ancillary study of WHI reported an increased risk of probable dementia in postmenopausal women 65 years of age and older ( 5.3 )
Estrogen Plus Progestin Therapy
•Estrogen plus progestin therapy should not be used for the prevention of cardiovascular disease or dementia ( 5.1 , 5.3 )
•The WHI estrogen plus progestin substudy reported increased risks of stroke, DVT, pulmonary embolism (PE), and myocardial infarction (MI) ( 5.1 )
•The WHI estrogen plus progestin substudy reported increased risks of invasive breast cancer ( 5.2 )
•The WHIMS estrogen plus progestin ancillary study of WHI reported an increased risk of probable dementia in postmenopausal women 65 years of age and older ( 5.3 )
1.2 Treatment of Moderate to Severe Symptoms of Vulvar and Vaginal Atrophy Due to Menopause
Limitations of Use
When prescribing solely for the treatment of moderate to severe symptoms of vulvar and vaginal atrophy due to menopause, topical vaginal products should be considered.
2.2 Treatment of Moderate to Severe Symptoms of Vulvar and Vaginal Atrophy Due to Menopause
Patients should be treated with the lowest effective dose. Generally, women should be started at 0.3 mg conjugated estrogens tablets daily. Subsequent dosage adjustment may be made based upon the individual patient response. This dose should be periodically reassessed by the healthcare provider.
Conjugated estrogens tablets therapy may be given continuously, with no interruption in therapy, or in cyclical regimens (regimens such as 25 days on drug followed by 5 days off drug), as is medically appropriate on an individual basis.
2.3 Treatment of Hypoestrogenism Due to Hypogonadism, Castration, Or Primary Ovarian Failure
Conjugated estrogens tablets therapy should be initiated and maintained with the lowest effective dose to achieve clinical goals. Female hypogonadism: 0.3 mg or 0.625 mg daily, administered cyclically (e.g., three weeks on and one week off). Doses are adjusted depending on the severity of symptoms and responsiveness of the endometrium [see Clinical Studies (14.4) ].
Female castration or primary ovarian failure: 1.25 mg daily, cyclically. Adjust dosage, upward or downward, according to severity of symptoms and response of the patient. For maintenance, adjust dosage to lowest level that will provide effective control.
2.5 Treatment of Advanced Androgen Dependent Carcinoma of the Prostate (for Palliation Only)
1.25 mg to 2 × 1.25 mg three times daily. The effectiveness of therapy can be judged by phosphatase determinations as well as by symptomatic improvement of the patient.
2.4 Treatment of Breast Cancer (for Palliation Only) in Appropriately Selected Women and Men With Metastatic Disease
Suggested dosage is 10 mg three times daily, for a period of at least three months.
Structured Label Content
Recent Major Changes (34066-1)
WHAT IS THE MOST IMPORTANT INFORMATION I SHOULD KNOW ABOUT CONJUGATED ESTROGENS TABLETS (AN ESTROGEN MIXTURE)?
•Using estrogen-alone may increase your chance of getting cancer of the uterus (womb)
Report any unusual vaginal bleeding right away while you are using conjugated estrogens tablets. Vaginal bleeding after menopause may be a warning sign of cancer of the uterus (womb). Your healthcare provider should check any unusual vaginal bleeding to find the cause.
•Do not use estrogen-alone to prevent heart disease, heart attacks, or dementia (decline of brain function)
•Using estrogen-alone may increase your chances of getting strokes or blood clots
•Using estrogen-alone may increase your chance of getting dementia, based on a study of women 65 years of age or older
•Do not use estrogens with progestins to prevent heart disease, heart attacks, strokes, or dementia
•Using estrogens with progestins may increase your chances of getting heart attacks, strokes, breast cancer, or blood clots
•Using estrogens with progestins may increase your chance of getting dementia, based on a study of women 65 years of age or older
•You and your healthcare provider should talk regularly about whether you still need treatment with conjugated estrogens tablets
Patient Information (PATIENT INFORMATION)
Conjugated estrogens tablets, USP
("kon′ju gay″ted es′troe jenz")
Read this PATIENT INFORMATION before you start taking conjugated estrogens tablets and read what you get each time you refill your conjugated estrogens tablets prescription. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Section 42229-5 (42229-5)
Estrogen-Alone Therapy
Section 51945-4 (51945-4)
10 Overdosage (10 OVERDOSAGE)
Overdosage of estrogen may cause nausea, vomiting, breast tenderness, abdominal pain, drowsiness and fatigue, and withdrawal bleeding may occur in women. Treatment of overdose consists of discontinuation of conjugated estrogens tablets therapy with institution of appropriate symptomatic care.
15 References (15 REFERENCES)
1. Rossouw JE, et al. Postmenopausal Hormone Therapy and Risk of Cardiovascular Disease by Age and Years Since Menopause. JAMA. 2007;297:1465–1477.
2. Hsia J, et al. Conjugated Equine Estrogens and Coronary Heart Disease. Arch Int Med. 2006;166:357–365.
3. Curb JD, et al. Venous Thrombosis and Conjugated Equine Estrogen in Women Without a Uterus. Arch Int Med. 2006;166:772–780.
4. Cushman M, et al. Estrogen Plus Progestin and Risk of Venous Thrombosis. JAMA. 2004;292:1573–1580.
5. Stefanick ML, et al. Effects of Conjugated Equine Estrogens on Breast Cancer and Mammography Screening in Postmenopausal Women With Hysterectomy. JAMA. 2006;295:1647–1657.
6. Chlebowski RT, et al. Influence of Estrogen Plus Progestin on Breast Cancer and Mammography in Healthy Postmenopausal Women. JAMA. 2003;289:3234–3253.
7. Anderson GL, et al. Effects of Estrogen Plus Progestin on Gynecologic Cancers and Associated Diagnostic Procedures. JAMA. 2003;290:1739–1748.
8. Shumaker SA, et al. Conjugated Equine Estrogens and Incidence of Probable Dementia and Mild Cognitive Impairment in Postmenopausal Women. JAMA. 2004;291:2947–2958.
9. Jackson RD, et al. Effects of Conjugated Equine Estrogen on Risk of Fractures and BMD in Postmenopausal Women With Hysterectomy: Results From the Women's Health Initiative Randomized Trial. J Bone Miner Res. 2006;21:817–828.
10. Hendrix SL, et al. Effects of Conjugated Equine Estrogen on Stroke in the Women's Health Initiative. Circulation. 2006;113:2425–2434.
8.1 Pregnancy
Risk Summary
Conjugated estrogens tablets is not indicated for use during pregnancy.
There are no data with the use of conjugated estrogens tablet in pregnant women; however, epidemiologic studies and meta-analyses have not found an increased risk of genital or nongenital birth defects (including cardiac anomalies and limb-reduction defects) following exposure to combined hormonal contraceptives (estrogen and progestins) before conception or during early pregnancy.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
Estrogens and progestins and metabolites are present in human milk. These hormones can reduce milk production in breast-feeding women. This reduction can occur at any time but is less likely to occur once breast-feeding is well established. The developmental and health benefits of breast-feeding should be considered along with the mother’s clinical need for conjugated estrogens tablets and any potential adverse effects on the breast-fed child from conjugated estrogens tablets or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
Conjugated estrogens tablets, USP for oral administration contains a mixture of CE purified from pregnant mares' urine and consists of the sodium salts of water-soluble estrogen sulfates blended to represent the average composition of material derived from pregnant mares' urine. It is a mixture of sodium estrone sulfate and sodium equilin sulfate. It contains concomitant components as sodium sulfate conjugates, 17α-dihydroequilin, 17α-estradiol, and 17β-dihydroequilin. Tablets for oral administration are available in 0.9 mg, and 1.25 mg strengths of CE.
Conjugated estrogens tablets USP, 0.9 mg, and 1.25 mg tablets also contain the following inactive ingredients: hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, sodium acetate anhydrous, and titanium dioxide. Each tablet strength contains the following colors:
FDA approved dissolution test specifications differ from USP.
16.1 How Supplied
Conjugated estrogens tablets, USP
- Each white oval biconvex coated tablet contains 0.9 mg, in bottles of 100 (NDC 50742-390-01) and 1,000 (NDC 50742-390-10).
- Each yellow oval biconvex coated tablet contains 1.25 mg, in bottles of 100 (NDC 50742-391-01) and 1,000 (NDC 50742-391-10).
5.14 Hypocalcemia
Estrogen therapy should be used with caution in all patients with hypoparathyroidism as estrogen-induced hypocalcemia may occur.
5.5 Hypercalcemia
Estrogen administration may lead to severe hypercalcemia in patients with breast cancer and bone metastases. If hypercalcemia occurs, use of the drug should be stopped and appropriate measures taken to reduce the serum calcium level.
8.4 Pediatric Use
Estrogen therapy has been used for the induction of puberty in adolescents with some forms of pubertal delay. Safety and effectiveness in pediatric patients have not otherwise been established.
Large and repeated doses of estrogen over an extended time period have been shown to accelerate epiphyseal closure, which could result in short stature if treatment is initiated before the completion of physiologic puberty in normally developing children. If estrogen is administered to patients whose bone growth is not complete, periodic monitoring of bone maturation and effects on epiphyseal centers is recommended during estrogen administration.
Estrogen treatment of prepubertal girls also induces premature breast development and vaginal cornification, and may induce vaginal bleeding. In boys, estrogen treatment may modify the normal pubertal process and induce gynecomastia.
8.5 Geriatric Use
There have not been sufficient numbers of geriatric patients involved in studies utilizing conjugated estrogens tablets to determine whether those over 65 years of age differ from younger subjects in their response to conjugated estrogens tablets.
4 Contraindications (4 CONTRAINDICATIONS)
Conjugated estrogens tablets therapy is contraindicated in individuals with any of the following conditions:
• Undiagnosed abnormal genital bleeding [see Warnings and Precautions (5.2)]
• Breast cancer or a history of breast cancer except in appropriately selected patients being treated for metastatic disease [see Warnings and Precautions (5.2)]
• Estrogen-dependent neoplasia [see Warnings and Precautions (5.2)]
• Active DVT, PE, or a history of these conditions [see Warnings and Precautions (5.1)]
• Active arterial thromboembolic disease (for example stroke and MI), or a history of these conditions [see Warnings and Precautions (5.1)]
• Known anaphylactic reaction or angioedema with conjugated estrogens tablets [see Warnings and Precautions (5.7,5.15)]
• Hepatic impairment or disease [see Warnings and Precautions (5.11)]
• Protein C, protein S or antithrombin deficiency, or other known thrombophilic disorders.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed elsewhere in labeling:
•Cardiovascular Disorders [see Boxed Warning, Warnings and Precautions (5.1)]
•Malignant Neoplasms [see Boxed Warning, Warnings and Precautions (5.2)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Data from a single-dose drug-drug interaction study involving CE and MPA indicate that the pharmacokinetic disposition of both drugs is not altered when the drugs are coadministered. No other clinical drug-drug interaction studies have been conducted with CE.
5.13 Fluid Retention
Estrogens may cause some degree of fluid retention. Women with conditions that might be influenced by this factor, such as cardiac or renal dysfunction, warrant careful observation when estrogens are prescribed.
8.6 Renal Impairment
The effect of renal impairment on the pharmacokinetics of conjugated estrogens tablets has not been studied.
12.2 Pharmacodynamics
There are no pharmacodynamic data for conjugated estrogens tablets.
17.1 Vaginal Bleeding
Inform postmenopausal women of the importance of reporting vaginal bleeding to their healthcare provider as soon as possible [see Warnings and Precautions (5.2)].
5.18 Laboratory Tests
Serum follicle stimulating hormone (FSH) and estradiol levels have not been shown to be useful in the management of moderate to severe vasomotor symptoms and moderate to severe symptoms of vulvar and vaginal atrophy.
Laboratory parameters may be useful in guiding dosage for the treatment of hypoestrogenism due to hypogonadism, castration and primary ovarian failure.
5.3 Probable Dementia
In the WHIMS estrogen-alone ancillary study of WHI, a population of 2,947 hysterectomized women 65 to 79 years of age was randomized to daily CE (0.625 mg)-alone or placebo.
After an average follow-up of 5.2 years, 28 women in the estrogen-alone group and 19 women in the placebo group were diagnosed with probable dementia. The relative risk of probable dementia for CE-alone versus placebo was 1.49 (95% CI 0.83–2.66). The absolute risk of probable dementia for CE-alone versus placebo was 37 versus 25 cases per 10,000 women-years8 [see Use in Specific Populations (8.5), and Clinical Studies (14.6)].
In the WHIMS estrogen plus progestin ancillary study of WHI, a population of 4,532 postmenopausal women 65 to 79 years of age was randomized to daily CE (0.625 mg) plus MPA (2.5 mg) or placebo. After an average follow-up of 4 years, 40 women in the CE plus MPA group and 21 women in the placebo group were diagnosed with probable dementia. The relative risk of probable dementia for CE plus MPA versus placebo was 2.05 (95% CI 1.21–3.48). The absolute risk of probable dementia for CE plus MPA versus placebo was 45 versus 22 cases per 10,000 women-years8 [see Use in Specific Populations (8.5), and Clinical Studies (14.6)].
When data from the two populations in the WHIMS estrogen-alone and estrogen plus progestin ancillary studies were pooled as planned in the WHIMS protocol, the reported overall relative risk for probable dementia was 1.76 (95% CI 1.19–2.60). Since both substudies were conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women8 [see Use in Specific Populations (8.5), and Clinical Studies (14.6)].
8.7 Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of conjugated estrogens tablets has not been studied.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Conjugated estrogens tablets is a mixture of estrogens indicated for:
•Treatment of Moderate to Severe Vasomotor Symptoms due to Menopause. (1.1)
•Treatment of Moderate to Severe Vulvar and Vaginal Atrophy due to Menopause (1.2)
•Treatment of Hypoestrogenism due to Hypogonadism, Castration or Primary Ovarian Failure (1.3)
•Treatment of Breast Cancer (for Palliation Only) in Appropriately Selected Women and Men with Metastatic Disease (1.4)
•Treatment of Advanced Androgen-Dependent Carcinoma of the Prostate (for Palliation Only) (1.5)
•Prevention of Postmenopausal Osteoporosis (1.6)
5.4 Gallbladder Disease
A 2- to 4-fold increase in the risk of gallbladder disease requiring surgery in postmenopausal women receiving estrogens has been reported.
12.1 Mechanism of Action
Endogenous estrogens are largely responsible for the development and maintenance of the female reproductive system and secondary sexual characteristics. Although circulating estrogens exist in a dynamic equilibrium of metabolic interconversions, estradiol is the principal intracellular human estrogen and is substantially more potent than its metabolites, estrone and estriol, at the receptor level.
The primary source of estrogen in normally cycling adult women is the ovarian follicle, which secretes 70 to 500 mcg of estradiol daily, depending on the phase of the menstrual cycle. After menopause, most endogenous estrogen is produced by conversion of androstenedione, secreted by the adrenal cortex, to estrone in the peripheral tissues. Thus, estrone and the sulfate-conjugated form, estrone sulfate, are the most abundant circulating estrogens in postmenopausal women.
Estrogens act through binding to nuclear receptors in estrogen-responsive tissues. To date, two estrogen receptors have been identified. These vary in proportion from tissue to tissue.
Circulating estrogens modulate the pituitary secretion of the gonadotropins, luteinizing hormone (LH) and FSH, through a negative feedback mechanism. Estrogens act to reduce the elevated levels of these gonadotropins seen in postmenopausal women.
5.6 Visual Abnormalities
Retinal vascular thrombosis has been reported in patients receiving estrogens. Discontinue medication pending examination if there is sudden partial or complete loss of vision, or a sudden onset of proptosis, diplopia, or migraine. If examination reveals papilledema or retinal vascular lesions, estrogens should be permanently discontinued.
16.2 Storage and Handling
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Dispense in a well-closed container, as defined in the USP.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
5.15 Hereditary Angioedema
Exogenous estrogens may exacerbate symptoms of angioedema in women with hereditary angioedema.
7.1 Metabolic Interactions
In vitro and in vivo studies have shown that estrogens are metabolized partially by cytochrome P450 3A4 (CYP3A4). Therefore, inducers or inhibitors of CYP3A4 may affect estrogen drug metabolism. Inducers of CYP3A4, such as St. John's Wort (Hypericum perforatum) preparations, phenobarbital, carbamazepine, and rifampin, may reduce plasma concentrations of estrogens, possibly resulting in a decrease in therapeutic effects and/or changes in the uterine bleeding profile. Inhibitors of CYP3A4, such as erythromycin, clarithromycin, ketoconazole, itraconazole, ritonavir and grapefruit juice, may increase plasma concentrations of estrogens and may result in side effects.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Generally, when estrogen therapy is prescribed for a postmenopausal woman with a uterus, a progestin should be considered to reduce the risk of endometrial cancer [see Boxed Warning].
A woman without a uterus does not need progestin. In some cases, however, hysterectomized women with a history of endometriosis may need a progestin [see Warnings and Precautions (5.2, 5.16)].
Use of estrogen-alone, or in combination with a progestin, should be with the lowest effective dose and for the shortest duration consistent with treatment goals and risks for the individual woman. Postmenopausal women should be re-evaluated periodically as clinically appropriate to determine if treatment is still necessary.
Conjugated estrogens tablets may be taken without regard to meals.
5.9 Elevated Blood Pressure
In a small number of case reports, substantial increases in blood pressure have been attributed to idiosyncratic reactions to estrogens. In a large, randomized, placebo-controlled clinical trial, a generalized effect of estrogen therapy on blood pressure was not seen.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
5.1 Cardiovascular Disorders
An increased risk of stroke and DVT has been reported with estrogen-alone therapy. An increased risk of PE, DVT, stroke and MI has been reported with estrogen plus progestin therapy. Should any of these events occur or be suspected, estrogen with or without progestin therapy should be discontinued immediately.
Risk factors for arterial vascular disease (for example, hypertension, diabetes mellitus, tobacco use, hypercholesterolemia, and obesity) and/or venous thromboembolism (VTE) (for example, personal or family history of VTE, obesity, and systemic lupus erythematosus) should be managed appropriately.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post-approval use of conjugated estrogens tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible always to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Study Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During the first year of a 2-year clinical trial with 2,333 postmenopausal women with a uterus between 40 and 65 years of age (88% Caucasian), 1,012 women were treated with CE , and 332 were treated with placebo.
Table 1 summarizes treatment-related adverse reactions that occurred at a rate of ≥ 1% in any treatment group.
| Conjugated estrogens tablets 0.625 mg (n=348) |
Conjugated estrogens tablets 0.45 mg (n=338) |
Conjugated estrogens tablets 0.3 mg (n=326) |
Placebo
(n=332) |
|
|---|---|---|---|---|
|
Body as a whole |
||||
|
Abdominal pain |
38 (11) |
28 (8) |
30 (9) |
21 (6) |
|
Asthenia |
16 (5) |
8 (2) |
14 (4) |
3 (1) |
|
Back pain |
18 (5) |
11 (3) |
13 (4) |
4 (1) |
|
Chest pain |
2 (1) |
3 (1) |
4 (1) |
2 (1) |
|
Generalized edema |
7 (2) |
6 (2) |
4 (1) |
8 (2) |
|
Headache |
45 (13) |
47 (14) |
44 (13) |
46 (14) |
|
Moniliasis |
5 (1) |
4 (1) |
4 (1) |
1 (0) |
|
Pain |
17 (5) |
10 (3) |
12 (4) |
14 (4) |
|
Pelvic pain |
10 (3) |
9 (3) |
8 (2) |
4 (1) |
|
Cardiovascular system |
||||
|
Hypertension |
4 (1) |
4 (1) |
7 (2) |
5 (2) |
|
Migraine |
7 (2) |
1 (0) |
0 |
3 (1) |
|
Palpitation |
3 (1) |
3 (1) |
3 (1) |
4 (1) |
|
Vasodilatation |
2 (1) |
2 (1) |
3 (1) |
5 (2) |
|
Digestive system |
||||
|
Constipation |
7 (2) |
6 (2) |
4 (1) |
3 (1) |
|
Diarrhea |
4 (1) |
5 (1) |
5 (2) |
8 (2) |
|
Dyspepsia |
7 (2) |
5 (1) |
6 (2) |
14 (4) |
|
Eructation |
1 (0) |
1 (0) |
4 (1) |
1 (0) |
|
Flatulence |
22 (6) |
18 (5) |
13 (4) |
8 (2) |
|
Increased appetite |
4 (1) |
1 (0) |
1 (0) |
2 (1) |
|
Nausea |
16 (5) |
10 (3) |
15 (5) |
16 (5) |
|
Metabolic and nutritional |
||||
|
Hyperlipidemia |
2 (1) |
4 (1) |
3 (1) |
2 (1) |
|
Peripheral edema |
5 (1) |
2 (1) |
4 (1) |
3 (1) |
|
Weight gain |
11 (3) |
10 (3) |
8 (2) |
14 (4) |
|
Musculoskeletal system |
||||
|
Arthralgia |
6 (2) |
3 (1) |
2 (1) |
5 (2) |
|
Leg cramps |
10 (3) |
5 (1) |
9 (3) |
4 (1) |
|
Myalgia |
2 (1) |
1 (0) |
4 (1) |
1 (0) |
|
Nervous system |
||||
|
Anxiety |
6 (2) |
4 (1) |
2 (1) |
4 (1) |
|
Depression |
17 (5) |
15 (4) |
10 (3) |
17 (5) |
|
Dizziness |
9 (3) |
7 (2) |
4 (1) |
5 (2) |
|
Emotional lability |
3 (1) |
4 (1) |
5 (2) |
8 (2) |
|
Hypertonia |
1 (0) |
1 (0) |
5 (2) |
3 (1) |
|
Insomnia |
16 (5) |
10 (3) |
13 (4) |
14 (4) |
|
Nervousness |
9 (3) |
12 (4) |
2 (1) |
6 (2) |
|
Skin and appendages |
||||
|
Acne |
3 (1) |
1 (0) |
8 (2) |
3 (1) |
|
Alopecia |
6 (2) |
6 (2) |
5 (2) |
2 (1) |
|
Hirsutism |
4 (1) |
2 (1) |
1 (0) |
0 |
|
Pruritus |
11 (3) |
11 (3) |
10 (3) |
3 (1) |
|
Rash |
6 (2) |
3 (1) |
1 (0) |
2 (1) |
|
Skin discoloration |
4 (1) |
2 (1) |
0 |
1 (0) |
|
Sweating |
4 (1) |
1 (0) |
3 (1) |
4 (1) |
|
Urogenital system |
||||
|
Breast disorder |
6 (2) |
3 (1) |
3 (1) |
6 (2) |
|
Breast enlargement |
3 (1) |
4 (1) |
7 (2) |
3 (1) |
|
Breast neoplasm |
4 (1) |
4 (1) |
7 (2) |
7 (2) |
|
Breast pain |
37 (11) |
39 (12) |
24 (7) |
26 (8) |
|
Cervix disorder |
8 (2) |
4 (1) |
5 (2) |
0 |
|
Dysmenorrhea |
12 (3) |
10 (3) |
4 (1) |
2 (1) |
|
Endometrial disorder |
4 (1) |
2 (1) |
2 (1) |
0 |
|
Endometrial hyperplasia |
16 (5) |
8 (2) |
1 (0) |
0 |
|
Leukorrhea |
17 (5) |
17 (5) |
12 (4) |
6 (2) |
|
Metrorrhagia |
11 (3) |
4 (1) |
3 (1) |
1 (0) |
|
Urinary tract infection |
1 (0) |
2 (1) |
1 (0) |
4 (1) |
|
Uterine fibroids enlarged |
6 (2) |
1 (0) |
2 (1) |
2 (1) |
|
Uterine spasm |
11 (3) |
5 (1) |
3 (1) |
2 (1) |
|
Vaginal dryness |
1 (0) |
2 (1) |
1 (0) |
6 (2) |
|
Vaginal hemorrhage |
46 (13) |
13 (4) |
6 (2) |
0 |
|
Vaginal moniliasis |
14 (4) |
10 (3) |
12 (4) |
5 (2) |
|
Vaginitis |
18 (5) |
7 (2) |
9 (3) |
1 (0) |
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
•Lactation: Estrogen administration to lactating women has been shown to decrease the quantity and quality of breast milk(8.2)
•Geriatric Use: An increased risk of probable dementia in women over 65 years of age was reported in the Women's Health Initiative Memory ancillary studies of the Women's Health Initiative (5.3, 8.5)
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patients to read the FDA-approved patient labeling (Patient Information).
14.1 Effects On Vasomotor Symptoms (14.1 Effects on Vasomotor Symptoms)
In the first year of the Health and Osteoporosis, Progestin and Estrogen (HOPE) Study, a total of 2,805 postmenopausal women (average age 53.3 ± 4.9 years) were randomly assigned to one of eight treatment groups of either placebo or CE, with or without MPA. Efficacy for vasomotor symptoms was assessed during the first 12 weeks of treatment in a subset of symptomatic women (n = 241) who had at least seven moderate to severe hot flushes daily, or at least 50 moderate to severe hot flushes during the week before randomization. Conjugated estrogens tablets (0.3 mg, 0.45 mg, and 0.625 mg tablets) was shown to be statistically better than placebo at weeks 4 and 12 for relief of both frequency and severity of moderate to severe vasomotor symptoms. Table 3 shows the adjusted mean number of hot flushes in the conjugated estrogens tablets 0.3 mg, 0.45 mg, and 0.625 mg and placebo groups during the initial 12-week period.
5.16 Exacerbation of Endometriosis
A few cases of malignant transformation of residual endometrial implants have been reported in women treated post-hysterectomy with estrogen-alone therapy. For women known to have residual endometriosis post-hysterectomy, the addition of progestin should be considered.
14.4 Effects On Female Hypogonadism (14.4 Effects on Female Hypogonadism)
In clinical studies of delayed puberty due to female hypogonadism, breast development was induced by doses as low as 0.15 mg. The dosage may be gradually titrated upward at 6-to 12 month intervals as needed to achieve appropriate bone age advancement and eventual epiphyseal closure. Clinical studies suggest that doses of 0.15 mg, 0.3 mg, and 0.6 mg are associated with mean ratios of bone age advancement to chronological age progression (ΔBA/ΔCA) of 1.1, 1.5, and 2.1, respectively. (Conjugated estrogens tablets in the dose strength of 0.15 mg is not available commercially). Available data suggest that chronic dosing with 0.625 mg is sufficient to induce artificial cyclic menses with sequential progestin treatment and to maintain bone mineral density after skeletal maturity is achieved.
5.12 exacerbation of Hypothyroidism (5.12 Exacerbation of Hypothyroidism)
Estrogen administration leads to increased thyroid-binding globulin (TBG) levels. Women with normal thyroid function can compensate for the increased TBG by making more thyroid hormone, thus maintaining free T4 and T3 serum concentrations in the normal range. Women dependent on thyroid hormone replacement therapy who are also receiving estrogens may require increased doses of their thyroid replacement therapy. These women should have their thyroid function monitored in order to maintain their free thyroid hormone levels in an acceptable range.
5.17 Exacerbation of Other Conditions
Estrogen therapy may cause an exacerbation of asthma, diabetes mellitus, epilepsy, migraine, porphyria, systemic lupus erythematosus, and hepatic hemangiomas and should be used with caution in women with these conditions.
14.5 Women's Health Initiative Studies
The WHI enrolled approximately 27,000 predominantly healthy postmenopausal women in two substudies to assess the risks and benefits of daily oral CE (0.625 mg)-alone or in combination with MPA (2.5 mg) compared to placebo in the prevention of certain chronic diseases. The primary endpoint was the incidence of CHD (defined as nonfatal MI, silent MI and CHD death), with invasive breast cancer as the primary adverse outcome. A "global index" included the earliest occurrence of CHD, invasive breast cancer, stroke, PE, endometrial cancer (only in the CE plus MPA substudy), colorectal cancer, hip fracture, or death due to other causes. These substudies did not evaluate the effects of CE-alone or CE plus MPA on menopausal symptoms.
5.19 Drug Laboratory Test Interactions (5.19 Drug-Laboratory Test Interactions)
• Accelerated prothrombin time, partial thromboplastin time, and platelet aggregation time; increased platelet count; increased factors II, VII antigen, VIII antigen, VIII coagulant activity, IX, X, XII, VII-X complex, II-VII-X complex, and beta-thromboglobulin; decreased levels of anti-factor Xa and antithrombin III, decreased antithrombin III activity; increased levels of fibrinogen and fibrinogen activity; increased plasminogen antigen and activity.
• Increased thyroid-binding globulin (TBG) levels leading to increased circulating total thyroid hormone levels as measured by protein-bound iodine (PBI), T4 levels (by column or by radioimmunoassay) or T3 levels by radioimmunoassay. T3 resin uptake is decreased, reflecting the elevated TBG. Free T4 and free T3 concentrations are unaltered. Patients on thyroid replacement therapy may require higher doses of thyroid hormone.
• Other binding proteins may be elevated in serum, for example, corticosteroid binding globulin (CBG), sex hormone-binding globulin (SHBG), leading to increased total circulating corticosteroids and sex steroids, respectively. Free hormone concentrations, such as testosterone and estradiol, may be decreased. Other plasma proteins may be increased (angiotensinogen/renin substrate, alpha-1-antitrypsin, ceruloplasmin).
• Increased plasma high-density lipoprotein (HDL) and HDL2 cholesterol subfraction, reduced low-density lipoprotein (LDL) cholesterol, increased triglyceride levels.
• Impaired glucose tolerance.
5.7 Anaphylactic Reaction and Angioedema
Cases of anaphylaxis, which developed within minutes to hours after taking conjugated estrogens tablets and require emergency medical management, have been reported in the postmarketing setting. Skin (hives, pruritis, swollen lips-tongue-face) and either respiratory tract (respiratory compromise) or gastrointestinal tract (abdominal pain, vomiting) involvement has been noted.
Angioedema involving the tongue, larynx, face, hands, and feet requiring medical intervention has occurred postmarketing in patients taking conjugated estrogens tablets. If angioedema involves the tongue, glottis, or larynx, airway obstruction may occur. Patients who develop an anaphylactic reaction with or without angioedema after treatment with conjugated estrogens tablets should not receive conjugated estrogens tablets again.
14.2 Effects On Vulvar and Vaginal Atrophy (14.2 Effects on Vulvar and Vaginal Atrophy)
Results of vaginal maturation indexes at cycles 6 and 13 showed that the differences from placebo were statistically significant (p < 0.001) for all treatment groups. (CE alone and CE/MPA treatment groups).
5.10 exacerbation of Hypertriglyceridemia (5.10 Exacerbation of Hypertriglyceridemia)
In women with pre-existing hypertriglyceridemia, estrogen therapy may be associated with elevations of plasma triglycerides leading to pancreatitis. Consider discontinuation of treatment if pancreatitis occurs.
14.6 Women's Health Initiative Memory Study
The WHIMS estrogen-alone ancillary study of WHI enrolled 2,947 predominantly healthy hysterectomized postmenopausal women 65 to 79 years of age (45 % were 65 to 69 years of age; 36 % were 70 to 74 years of age; 19 % were 75 years of age and older) to evaluate the effects of daily CE (0.625 mg)- alone on the incidence of probable dementia (primary outcome) compared to placebo.
After an average follow-up of 5.2 years, the relative risk of probable dementia for CE-alone versus placebo was 1.49 (95 % CI 0.83–2.66). The absolute risk of probable dementia for CE-alone versus placebo was 37 versus 25 cases per 10,000 women-years. Probable dementia as defined in this study included Alzheimer's disease (AD), vascular dementia (VaD) and mixed types (having features of both AD and VaD). The most common classification of probable dementia in the treatment group and the placebo groups was AD. Since the ancillary study was conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women [see Warnings and Precautions (5.3) , and Use in Specific Populations (8.5)].
The WHIMS estrogen plus progestin ancillary study enrolled 4,532 predominantly healthy postmenopausal women 65 years of age and older (47 % were 65 to 69 years of age; 35 % were 70 to 74 years; 18 % were 75 years of age and older) to evaluate the effects of daily CE (0.625 mg) plus MPA (2.5 mg) on the incidence of probable dementia (primary outcome) compared to placebo.
After an average follow-up of 4 years, the relative risk of probable dementia for CE plus MPA was 2.05 (95 % CI, 1.21–3.48). The absolute risk of probable dementia for CE (0.625 mg) plus MPA (2.5 mg) versus placebo was 45 versus 22 per 10,000 women-years. Probable dementia as defined in this study included AD, VaD and mixed types (having features of both AD and VaD). The most common classification of probable dementia in both the treatment and placebo groups was AD. Since the ancillary study was conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women [see Warnings and Precautions (5.3), and Use in Specific Populations (8.5)].
When data from the two populations were pooled as planned in the WHIMS protocol, the reported overall relative risk for probable dementia was 1.76 (95 percent CI 1.19–2.60). Differences between groups became apparent in the first year of treatment. It is unknown whether these findings apply to younger postmenopausal women [see Warnings and Precautions (5.3), and Use in Specific Populations (8.5)].
2.6 Prevention of Postmenopausal Osteoporosis
Conjugated estrogens tablets therapy may be given continuously, with no interruption in therapy, or in cyclical regimens (regimens such as 25 days on drug followed by 5 days off drug), as is medically appropriate on an individual basis.
Patients should be treated with the lowest effective dose. Generally, women should be started at 0.3 mg conjugated estrogens tablets daily. Subsequent dosage adjustment may be made based upon the individual clinical and bone mineral density responses. This dose should be periodically reassessed by the healthcare provider.
17.2 Possible Serious Adverse Reactions With Estrogens (17.2 Possible Serious Adverse Reactions with Estrogens)
Inform postmenopausal women of possible serious adverse reactions of estrogen therapy including Cardiovascular Disorders, Malignant Neoplasms, and Probable Dementia [see Warnings and Precautions (5.1, 5.2, 5.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term continuous administration of natural and synthetic estrogens in certain animal species increases the frequency of carcinomas of the breast, uterus, cervix, vagina, testis, and liver. [see Warnings and Precautions (5.2)].
5.11 Hepatic Impairment And/or Past History of Cholestatic Jaundice (5.11 Hepatic Impairment and/or Past History of Cholestatic Jaundice)
Estrogens may be poorly metabolized in patients with impaired liver function. For women with a history of cholestatic jaundice associated with past estrogen use or with pregnancy, caution should be exercised, and in the case of recurrence, medication should be discontinued.
5.8 Addition of A Progestin When A Woman Has Not Had A Hysterectomy (5.8 Addition of a Progestin When a Woman Has Not Had a Hysterectomy)
Studies of the addition of a progestin for 10 or more days of a cycle of estrogen administration or daily with estrogen in a continuous regimen, have reported a lowered incidence of endometrial hyperplasia than would be induced by estrogen treatment alone. Endometrial hyperplasia may be a precursor to endometrial cancer.
There are, however, possible risks that may be associated with the use of progestins with estrogens compared to estrogen-alone regimens. These include an increased risk of breast cancer.
17.3 Possible Less Serious But Common Adverse Reactions With Estrogens (17.3 Possible Less Serious but Common Adverse Reactions with Estrogens)
Inform postmenopausal women of possible less serious but common adverse reactions of estrogen therapy such as headache, breast pain and tenderness, nausea and vomiting.
2.1 Treatment of Moderate to Severe Vasomotor Symptoms Due to Menopause (2.1 Treatment of Moderate to Severe Vasomotor Symptoms due to Menopause)
Patients should be treated with the lowest effective dose. Generally, women should be started at 0.3 mg conjugated estrogens tablets daily. Subsequent dosage adjustment may be made based upon the individual patient response. This dose should be periodically reassessed by the healthcare provider.
Conjugated estrogens tablets therapy may be given continuously, with no interruption in therapy, or in cyclical regimens (regimens such as 25 days on drug followed by 5 days off drug), as is medically appropriate on an individual basis.
Warning: Endometrial Cancer, Cardiovascular Disorders, Breast Cancer and Probable Dementia (WARNING: ENDOMETRIAL CANCER, CARDIOVASCULAR DISORDERS, BREAST CANCER and PROBABLE DEMENTIA)
WARNING: ENDOMETRIAL CANCER, CARDIOVASCULAR DISORDERS, BREAST CANCER and PROBABLE DEMENTIA
See full prescribing information for complete boxed warning.
Estrogen-Alone Therapy
•There is an increased risk of endometrial cancer in a woman with a uterus who uses unopposed estrogens ( 5.2 )
•Estrogen-alone therapy should not be used for the prevention of cardiovascular disease or dementia ( 5.1, 5.3)
•Women's Health Initiative (WHI) estrogen-alone substudy reported increased risks of stroke and deep vein thrombosis (DVT) ( 5.1 )
•The WHI Memory Study (WHIMS) estrogen-alone ancillary study of WHI reported an increased risk of probable dementia in postmenopausal women 65 years of age and older ( 5.3 )
Estrogen Plus Progestin Therapy
•Estrogen plus progestin therapy should not be used for the prevention of cardiovascular disease or dementia ( 5.1 , 5.3 )
•The WHI estrogen plus progestin substudy reported increased risks of stroke, DVT, pulmonary embolism (PE), and myocardial infarction (MI) ( 5.1 )
•The WHI estrogen plus progestin substudy reported increased risks of invasive breast cancer ( 5.2 )
•The WHIMS estrogen plus progestin ancillary study of WHI reported an increased risk of probable dementia in postmenopausal women 65 years of age and older ( 5.3 )
1.2 Treatment of Moderate to Severe Symptoms of Vulvar and Vaginal Atrophy Due to Menopause (1.2 Treatment of Moderate to Severe Symptoms of Vulvar and Vaginal Atrophy due to Menopause)
Limitations of Use
When prescribing solely for the treatment of moderate to severe symptoms of vulvar and vaginal atrophy due to menopause, topical vaginal products should be considered.
2.2 Treatment of Moderate to Severe Symptoms of Vulvar and Vaginal Atrophy Due to Menopause (2.2 Treatment of Moderate to Severe Symptoms of Vulvar and Vaginal Atrophy due to Menopause)
Patients should be treated with the lowest effective dose. Generally, women should be started at 0.3 mg conjugated estrogens tablets daily. Subsequent dosage adjustment may be made based upon the individual patient response. This dose should be periodically reassessed by the healthcare provider.
Conjugated estrogens tablets therapy may be given continuously, with no interruption in therapy, or in cyclical regimens (regimens such as 25 days on drug followed by 5 days off drug), as is medically appropriate on an individual basis.
2.3 Treatment of Hypoestrogenism Due to Hypogonadism, Castration, Or Primary Ovarian Failure (2.3 Treatment of Hypoestrogenism due to Hypogonadism, Castration, or Primary Ovarian Failure)
Conjugated estrogens tablets therapy should be initiated and maintained with the lowest effective dose to achieve clinical goals. Female hypogonadism: 0.3 mg or 0.625 mg daily, administered cyclically (e.g., three weeks on and one week off). Doses are adjusted depending on the severity of symptoms and responsiveness of the endometrium [see Clinical Studies (14.4) ].
Female castration or primary ovarian failure: 1.25 mg daily, cyclically. Adjust dosage, upward or downward, according to severity of symptoms and response of the patient. For maintenance, adjust dosage to lowest level that will provide effective control.
2.5 Treatment of Advanced Androgen Dependent Carcinoma of the Prostate (for Palliation Only) (2.5 Treatment of Advanced Androgen-Dependent Carcinoma of the Prostate (for Palliation Only))
1.25 mg to 2 × 1.25 mg three times daily. The effectiveness of therapy can be judged by phosphatase determinations as well as by symptomatic improvement of the patient.
2.4 Treatment of Breast Cancer (for Palliation Only) in Appropriately Selected Women and Men With Metastatic Disease (2.4 Treatment of Breast Cancer (for Palliation Only) in Appropriately Selected Women and Men with Metastatic Disease)
Suggested dosage is 10 mg three times daily, for a period of at least three months.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:25.613837 · Updated: 2026-03-14T22:37:33.826