Plevent™
721f4405-241b-4770-9bd7-ff6c69a1877f
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
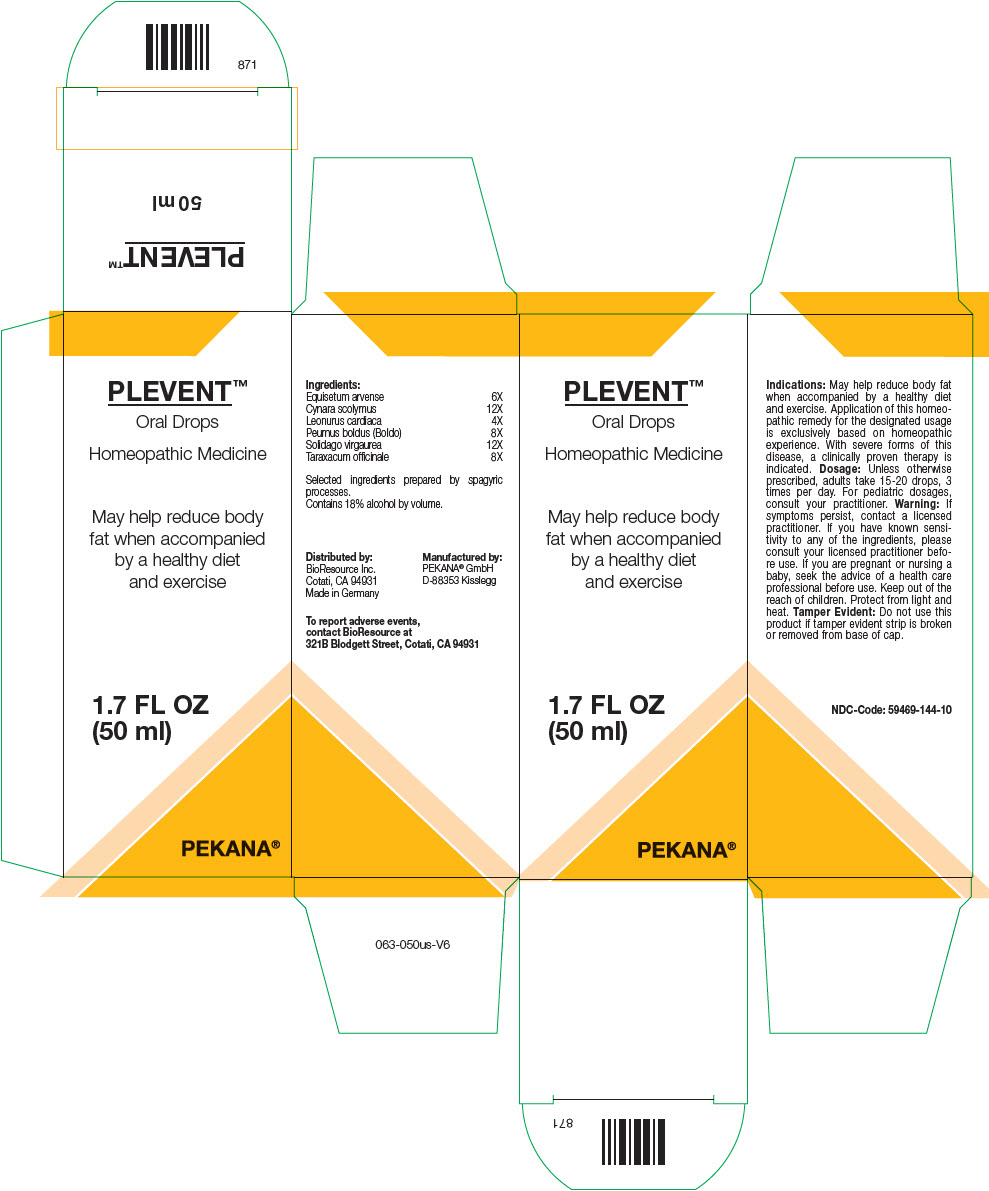
Ingredients: Equisetum arvense 6X Cynara scolymus 12X Leonurus cardiaca 4X Peumus boldus (Boldo) 8X Solidago virgaurea 12X Taraxacum officinale 8X Selected ingredients prepared by spagyric processes.
Medication Information
Description
Ingredients: Equisetum arvense 6X Cynara scolymus 12X Leonurus cardiaca 4X Peumus boldus (Boldo) 8X Solidago virgaurea 12X Taraxacum officinale 8X Selected ingredients prepared by spagyric processes.
Dosage
Unless otherwise prescribed, adults take 15-20 drops, 3 times per day. For pediatric dosages, consult your practitioner.
Section 42229-5
Distributed by:
BioResource Inc.
Cotati, CA 94931
Manufactured by:
PEKANA®
GmbH
D-88353 Kisslegg
Section 44425-7
Protect from light and heat.
Section 50565-1
Keep out of the reach of children.
Section 51727-6
Contains 18% alcohol by volume.
Section 53413-1
To report adverse events, contact BioResource at 321B Blodgett Street, Cotati, CA 94931
Section 55105-1
May help reduce body fat when accompanied by a healthy diet and exercise. Application of this homeopathic remedy for the designated usage is exclusively based on homeopathic experience. With severe forms of this disease, a clinically proven therapy is indicated.
Section 55106-9
| Ingredients: | |
|---|---|
| Equisetum arvense | 6X |
| Cynara scolymus | 12X |
| Leonurus cardiaca | 4X |
| Peumus boldus (Boldo) | 8X |
| Solidago virgaurea | 12X |
| Taraxacum officinale | 8X |
Selected ingredients prepared by spagyric processes.
Warning
If symptoms persist, contact a licensed practitioner. If you have known sensitivity to any of the ingredients, please consult your licensed practitioner before use. If you are pregnant or nursing a baby, seek the advice of a health care professional before use.
Tamper Evident
Do not use this product if tamper evident strip is broken or removed from base of cap.
Principal Display Panel 50 Ml Bottle Box
PLEVENT™
Oral Drops
Homeopathic Medicine
May help reduce body
fat when accompanied
by a healthy diet
and exercise
1.7 FL OZ
(50 ml)
PEKANA®
Structured Label Content
Dosage
Unless otherwise prescribed, adults take 15-20 drops, 3 times per day. For pediatric dosages, consult your practitioner.
Section 42229-5 (42229-5)
Distributed by:
BioResource Inc.
Cotati, CA 94931
Manufactured by:
PEKANA®
GmbH
D-88353 Kisslegg
Section 44425-7 (44425-7)
Protect from light and heat.
Section 50565-1 (50565-1)
Keep out of the reach of children.
Section 51727-6 (51727-6)
Contains 18% alcohol by volume.
Section 53413-1 (53413-1)
To report adverse events, contact BioResource at 321B Blodgett Street, Cotati, CA 94931
Section 55105-1 (55105-1)
May help reduce body fat when accompanied by a healthy diet and exercise. Application of this homeopathic remedy for the designated usage is exclusively based on homeopathic experience. With severe forms of this disease, a clinically proven therapy is indicated.
Section 55106-9 (55106-9)
| Ingredients: | |
|---|---|
| Equisetum arvense | 6X |
| Cynara scolymus | 12X |
| Leonurus cardiaca | 4X |
| Peumus boldus (Boldo) | 8X |
| Solidago virgaurea | 12X |
| Taraxacum officinale | 8X |
Selected ingredients prepared by spagyric processes.
Warning
If symptoms persist, contact a licensed practitioner. If you have known sensitivity to any of the ingredients, please consult your licensed practitioner before use. If you are pregnant or nursing a baby, seek the advice of a health care professional before use.
Tamper Evident
Do not use this product if tamper evident strip is broken or removed from base of cap.
Principal Display Panel 50 Ml Bottle Box (PRINCIPAL DISPLAY PANEL - 50 ml Bottle Box)
PLEVENT™
Oral Drops
Homeopathic Medicine
May help reduce body
fat when accompanied
by a healthy diet
and exercise
1.7 FL OZ
(50 ml)
PEKANA®
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:10:53.621980 · Updated: 2026-03-14T23:15:51.329649