These Highlights Do Not Include All The Information Needed To Use Dysport Safely And Effectively. See Full Prescribing Information For Dysport.
71313a04-1349-4c26-b840-a39e4a3ddaed
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Postmarketing reports indicate that the effects of DYSPORT and all botulinum toxin products may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These may include asthenia, generalized muscle weakness, diplopia, blurred vision, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence and breathing difficulties. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life threatening and there have been reports of death. The risk of symptoms is probably greatest in children treated for spasticity but symptoms can also occur in adults treated for spasticity and other conditions, particularly in those patients who have underlying conditions that would predispose them to these symptoms. In unapproved uses and in approved indications, cases of spread of effect have been reported at doses comparable to or lower than the maximum recommended total dose [ see Warnings and Precautions (5.1) ].
Indications and Usage
DYSPORT is an acetylcholine release inhibitor and neuromuscular blocking agent indicated for: the treatment of cervical dystonia in adults ( 1.1 ) the temporary improvement in the appearance of moderate to severe glabellar lines associated with procerus and corrugator muscle activity in adults < 65 years of age ( 1.2 ) The treatment of spasticity in patients 2 years of age and older ( 1.3 )
Dosage and Administration
Preparation of DYSPORT Solution for Administration ( 2.2 ) Once reconstituted, store in original container in a refrigerator 2°C to 8°C (36°F to 46°F) and use within 24 hours Do not freeze after reconstitution Reconstitution instructions are specific for the 300 Unit and 500 Unit vials Reconstituted DYSPORT is intended for intramuscular injection only. After reconstitution, DYSPORT should be used for only one injection session and for only one patient. Cervical Dystonia ( 2.3 ) • Initial dose is 500 Units given intramuscularly as a divided dose among the affected muscles • Re-treatment every 12 to 16 weeks or longer, as necessary, based on return of clinical symptoms with doses administered between 250 and 1000 Units to optimize clinical benefit • Re-treatment should not occur in intervals of less than 12 weeks • Titrate in 250 Unit steps according to patient's response Glabellar Lines ( 2.4 ) Administer a total dose of 50 Units, divided in five equal aliquots of 10 Units each, intramuscularly to affected muscles to achieve clinical effect Re-treatment should be administered no more frequently than every 3 months Spasticity in Adults ( 2.5 ) Select dose based on muscles affected, severity of spasticity, and treatment and adverse reaction history botulinum toxins Dosing for upper limb spasticity: between 500 Units and 1000 Units Dosing for lower limb spasticity: up to 1500 Units The maximum recommended total dose per treatment session (upper and lower limb combined) in adults is 1500 Units Re-treatment, based on return of clinical symptoms, should not occur in intervals of less than 12 weeks Spasticity in Pediatric Patients ( 2.6 ) Select dose based on the affected muscle, severity of spasticity, and treatment and adverse reaction history with all botulinum toxins. Recommended dosing for upper limb spasticity: 8 Units/kg to 16 Units/kg per limb. The maximum recommended total dose administered per treatment session must not exceed 16 Units/kg or 640 Units, whichever is lower. Recommended dosing for lower limb spasticity: 10 Units/kg to 15 Units/kg per limb. Total dose per treatment session must not exceed 15 Units/kg for unilateral lower limb injections, 30 Units/kg for bilateral injections, or 1000 Units, whichever is lower. The maximum recommended total dose per treatment session is 30 Units/kg or 1000 Units, whichever is lower. Re-treatment, based on return of clinical symptoms, should not occur in intervals of less than 3 months.
Warnings and Precautions
The potency units of DYSPORT are not interchangeable with other preparations of botulinum toxin products ( 5.2 ) Immediate medical attention may be required in cases of respiratory, speech or swallowing difficulties ( 5.4 ) Recommended dose and frequency of administration should not be exceeded ( 5.5 ) Dry eye may occur with glabellar line treatment; if symptoms persist, consider referring patient to an ophthalmologist ( 5.6 ) Concomitant neuromuscular disorder may exacerbate clinical effects of treatment ( 5.7 )
Contraindications
DYSPORT is contraindicated in patients with: Known hypersensitivity to any botulinum toxin products, cow's milk protein, or to any of the components in the formulation [ see Warnings and Precautions (5.3) ] . This product may contain trace amounts of cow's milk protein [ see Description (11) ] . Infection at the proposed injection site(s).
Adverse Reactions
The following serious adverse reactions are discussed below and elsewhere in labeling: Spread of Toxin Effect [ see Warning s and Precautions (5.1)] Lack of Interchangeability between Botulinum Toxin Products [ see Warnings and Precautions (5.2) ] Hypersensitivity Reactions [ see Warnings and Precautions (5.3) ] Dysphagia and Breathing Difficulties [ see Warnings and Precautions (5.4) ] Facial Anatomy in the Treatment of Glabellar Lines [ see Warnings and Precautions (5.5) ] Dry Eye with the Treatment of Glabellar Lines [see Warnings and Precautions (5.6) ] Pre-existing Neuromuscular Disorders [ see Warnings and Precautions (5.7) ] Human Albumin and Transmission of Viral Diseases [ see Warnings and Precautions (5.8) ] Intradermal Immune Reaction [ see Warnings and Precautions (5.9) ] Pre-existing Conditions at the Injection Site [ See Warnings and Precautions (5.10) ]
Drug Interactions
Concomitant use of DYSPORT and aminoglycosides or other agents interfering with neuromuscular transmission or muscle relaxants, should be observed closely because the effect of DYSPORT may be potentiated (7.1, 7.4) Anticholinergic drugs may potentiate systemic anticholinergic effects (7.2) The effect of administering different botulinum neurotoxins during the course of treatment with DYSPORT is unknown (7.3)
Medication Information
Warnings and Precautions
The potency units of DYSPORT are not interchangeable with other preparations of botulinum toxin products ( 5.2 ) Immediate medical attention may be required in cases of respiratory, speech or swallowing difficulties ( 5.4 ) Recommended dose and frequency of administration should not be exceeded ( 5.5 ) Dry eye may occur with glabellar line treatment; if symptoms persist, consider referring patient to an ophthalmologist ( 5.6 ) Concomitant neuromuscular disorder may exacerbate clinical effects of treatment ( 5.7 )
Indications and Usage
DYSPORT is an acetylcholine release inhibitor and neuromuscular blocking agent indicated for: the treatment of cervical dystonia in adults ( 1.1 ) the temporary improvement in the appearance of moderate to severe glabellar lines associated with procerus and corrugator muscle activity in adults < 65 years of age ( 1.2 ) The treatment of spasticity in patients 2 years of age and older ( 1.3 )
Dosage and Administration
Preparation of DYSPORT Solution for Administration ( 2.2 ) Once reconstituted, store in original container in a refrigerator 2°C to 8°C (36°F to 46°F) and use within 24 hours Do not freeze after reconstitution Reconstitution instructions are specific for the 300 Unit and 500 Unit vials Reconstituted DYSPORT is intended for intramuscular injection only. After reconstitution, DYSPORT should be used for only one injection session and for only one patient. Cervical Dystonia ( 2.3 ) • Initial dose is 500 Units given intramuscularly as a divided dose among the affected muscles • Re-treatment every 12 to 16 weeks or longer, as necessary, based on return of clinical symptoms with doses administered between 250 and 1000 Units to optimize clinical benefit • Re-treatment should not occur in intervals of less than 12 weeks • Titrate in 250 Unit steps according to patient's response Glabellar Lines ( 2.4 ) Administer a total dose of 50 Units, divided in five equal aliquots of 10 Units each, intramuscularly to affected muscles to achieve clinical effect Re-treatment should be administered no more frequently than every 3 months Spasticity in Adults ( 2.5 ) Select dose based on muscles affected, severity of spasticity, and treatment and adverse reaction history botulinum toxins Dosing for upper limb spasticity: between 500 Units and 1000 Units Dosing for lower limb spasticity: up to 1500 Units The maximum recommended total dose per treatment session (upper and lower limb combined) in adults is 1500 Units Re-treatment, based on return of clinical symptoms, should not occur in intervals of less than 12 weeks Spasticity in Pediatric Patients ( 2.6 ) Select dose based on the affected muscle, severity of spasticity, and treatment and adverse reaction history with all botulinum toxins. Recommended dosing for upper limb spasticity: 8 Units/kg to 16 Units/kg per limb. The maximum recommended total dose administered per treatment session must not exceed 16 Units/kg or 640 Units, whichever is lower. Recommended dosing for lower limb spasticity: 10 Units/kg to 15 Units/kg per limb. Total dose per treatment session must not exceed 15 Units/kg for unilateral lower limb injections, 30 Units/kg for bilateral injections, or 1000 Units, whichever is lower. The maximum recommended total dose per treatment session is 30 Units/kg or 1000 Units, whichever is lower. Re-treatment, based on return of clinical symptoms, should not occur in intervals of less than 3 months.
Contraindications
DYSPORT is contraindicated in patients with: Known hypersensitivity to any botulinum toxin products, cow's milk protein, or to any of the components in the formulation [ see Warnings and Precautions (5.3) ] . This product may contain trace amounts of cow's milk protein [ see Description (11) ] . Infection at the proposed injection site(s).
Adverse Reactions
The following serious adverse reactions are discussed below and elsewhere in labeling: Spread of Toxin Effect [ see Warning s and Precautions (5.1)] Lack of Interchangeability between Botulinum Toxin Products [ see Warnings and Precautions (5.2) ] Hypersensitivity Reactions [ see Warnings and Precautions (5.3) ] Dysphagia and Breathing Difficulties [ see Warnings and Precautions (5.4) ] Facial Anatomy in the Treatment of Glabellar Lines [ see Warnings and Precautions (5.5) ] Dry Eye with the Treatment of Glabellar Lines [see Warnings and Precautions (5.6) ] Pre-existing Neuromuscular Disorders [ see Warnings and Precautions (5.7) ] Human Albumin and Transmission of Viral Diseases [ see Warnings and Precautions (5.8) ] Intradermal Immune Reaction [ see Warnings and Precautions (5.9) ] Pre-existing Conditions at the Injection Site [ See Warnings and Precautions (5.10) ]
Drug Interactions
Concomitant use of DYSPORT and aminoglycosides or other agents interfering with neuromuscular transmission or muscle relaxants, should be observed closely because the effect of DYSPORT may be potentiated (7.1, 7.4) Anticholinergic drugs may potentiate systemic anticholinergic effects (7.2) The effect of administering different botulinum neurotoxins during the course of treatment with DYSPORT is unknown (7.3)
Description
Postmarketing reports indicate that the effects of DYSPORT and all botulinum toxin products may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These may include asthenia, generalized muscle weakness, diplopia, blurred vision, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence and breathing difficulties. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life threatening and there have been reports of death. The risk of symptoms is probably greatest in children treated for spasticity but symptoms can also occur in adults treated for spasticity and other conditions, particularly in those patients who have underlying conditions that would predispose them to these symptoms. In unapproved uses and in approved indications, cases of spread of effect have been reported at doses comparable to or lower than the maximum recommended total dose [ see Warnings and Precautions (5.1) ].
Section 34077-8
Risk Summary
There are no adequate and well-controlled clinical studies with DYSPORT in pregnant women.
DYSPORT should only be used during pregnancy if the potential benefit justifies the potential risk to the fetus.
DYSPORT produced embryo-fetal toxicity in relation to maternal toxicity when given to pregnant rats and rabbits at doses lower than or similar to the maximum recommended human dose (MRHD) of 1000 Units on a body weight (Units/kg) basis (
see Data).
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. The background risk of major birth defects and miscarriage for the indicated population is unknown.
Data
Animal Data
In a study in which pregnant rats received daily intramuscular injections of DYSPORT (2.2, 6.6, or 22 Units/kg on gestation days 6 through 17 or intermittently 44 Units/kg on gestation days 6 and 12 only) during organogenesis, increased early embryonic death was observed with both schedules at the highest tested doses (22 and 44 Units/kg), which were associated with maternal toxicity. The no-effect dose for embryo-fetal developmental toxicity was 2.2 Units/kg (less than maximum recommended human [MRHD] on a body weight basis).
In a study in which pregnant rabbits received daily intramuscular injections of DYSPORT® (0.3, 3.3 or 6.7 Units/kg) on gestation days 6 through 19 or intermittently (13.3 Units/kg on gestation days 6 and 13 only) during organogenesis, no embryofetal data were available at the highest dose administered daily (6.7 Units/kg) because of premature death in all does at that dose. At the lower daily doses or with intermittent dosing, no adverse developmental effects were observed. All dosed for which data were available are less than the MRHD on a body weight basis.
In a study in which pregnant rats received 6 weekly intramuscular injections of DYSPORT (4.4, 11.1, 22.2, or 44 Units/kg) beginning on day 6 of gestation and continuing through parturition to weaning, an increase in stillbirths was observed at the highest dose tested, which was maternally toxic. The no-effect dose for pre- and post-natal development toxicity was 22.2 Units/kg (similar to the MRHD).
Section 34083-6
Carcinogenesis
Studies to evaluate the carcinogenic potential of DYSPORT have not been conducted.
Section 42229-5
DYSPORT is indicated for the treatment of cervical dystonia in adults.
Section 42231-1
MEDICATION GUIDE
DYSPORT® (
DIS-port)
(abobotulinumtoxinA)
for Injection
What is the most important information I should know about DYSPORT?
DYSPORT may cause serious side effects that can be life threatening including:
• Problems breathing or swallowing
• Spread of toxin effects
These problems can happen within hours, or days to weeks after an injection of DYSPORT. Call your doctor or get medical help right away if you have any of these problems after treatment with DYSPORT:
1. Problems swallowing, speaking, or breathing. These problems can happen within hours, or days to weeks after an injection of DYSPORTusually because the muscles that you use to breathe and swallow can become weak after the injection. Death can happen as a complication if you have severe problems with swallowing or breathing after treatment with DYSPORT.
• People with certain breathing problems may need to use muscles in their neck to help them breathe. These patients may be at greater risk for serious breathing problems with DYSPORT.
• Swallowing problems may last for several weeks. People who cannot swallow well may need a feeding tube to receive food and water. If swallowing problems are severe, food or liquids may go into your lungs. People who already have swallowing or breathing problems before receiving DYSPORT have the highest risk of getting these problems.
2. Spread of toxin effects.In some cases, the effect of botulinum toxin may affect areas of the body away from the injection site and cause symptoms of a serious condition called botulism. The symptoms of botulism include:
• loss of strength and muscle weakness all over the body
• blurred vision and drooping eyelids
• trouble saying words clearly (dysarthria)
• trouble breathing
• double vision
• hoarseness or change or loss of voice (dysphonia)
• loss of bladder control
• trouble swallowing
These symptoms can happen within hours, or days to weeks after you receive an injection of DYSPORT.These problems could make it unsafe for you to drive a car or do other dangerous activities. See "What should I avoid while receiving DYSPORT?"
What is DYSPORT?
DYSPORT is a prescription medicine that is injected into muscles and used:
- to treat cervical dystonia (CD) in adults
- to improve the look of moderate to severe frown lines between the eyebrows (glabellar lines) in adults younger than 65 years of age for a short period of time (temporary)
- to treat increased muscle stiffness in people 2 years of age and older with spasticity
It is not known whether DYSPORT is safe or effective in people younger than:
- 18 years of age for the treatment of cervical dystonia
- 18 years of age for the treatment of glabellar lines
- 2 years of age for the treatment of spasticity
It is not known whether DYSPORT is safe or effective for the treatment of other wrinkles.
Who should not take DYSPORT?
Do not take DYSPORT if you:
- are allergic to DYSPORT or any of the ingredients in DYSPORT. See the end of this Medication Guide for a list of ingredients in DYSPORT
- are allergic to cow's milk protein
- had an allergic reaction to any other botulinum toxin product such as Myobloc® (rimabotulinumtoxinB), Botox® (onabotulinumtoxinA), or Xeomin® (incobotulinumtoxinA)
- have a skin infection at the planned injection site
What should I tell my doctor before taking DYSPORT?
Tell your doctor about all your medical conditions, including if you:
- have a disease that affects your muscles and nerves (such as amyotrophic lateral sclerosis [ALS or Lou Gehrig's disease], myasthenia gravis or Lambert-Eaton syndrome). See "What is the most important information I should know about DYSPORT?"
- have allergies to any botulinum toxin product
- had any side effect from botulinum toxin product in the past
- have or have had a breathing problem, such as asthma or emphysema
- have or have had swallowing problems
- have or have had bleeding problems
- have diabetes
- have or have had a slow heart beat or other problem with your heart rate or rhythm
- have plans to have surgery
- had surgery on your face
- have weakness in or near your muscles being treated
- have drooping eyelids
- experienced dry eye with previous use of botulinum toxin products
- have any other change in the way your face normally looks
- are pregnant or plan to become pregnant. It is not known if DYSPORT can harm your unborn baby
- are breast-feeding or planning to breast-feed. It is not known if DYSPORT passes into breast milk
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal products. Using DYSPORT with certain other medicines may cause serious side effects.
Do not start any new medicines until you have told your doctor that you have received DYSPORT in the past.
Especially tell your doctor if you:
- have received any other botulinum toxin product in the last four months
- have received injections of botulinum toxin, such as Myobloc® (rimabotulinumtoxinB), Botox® (onabotulinumtoxinA) or Xeomin® (incobotulinumtoxinA) in the past; be sure your doctor knows exactly which product you received
- have recently received an antibiotic by injection
- take muscle relaxants
- take an allergy or cold medicine
- take a sleep medicine
Ask your doctor if you are not sure if your medicine is one that is listed above.
Know the medicines you take. Keep a list of your medicines with you to show your doctor and pharmacist each time you get a new medicine.
How will I receive DYSPORT?
- DYSPORT is an injection that your doctor will give you
- DYSPORT is injected into the affected muscles
- If you are and adult, your doctor may give you another dose of DYSPORT after 12 weeks or longer, if it is needed
- If you are an adult being treated for CD or spasticity or you are a child (2 to 17 years of age) being treated for spasticity, your doctor may change your dose of DYSPORT, until you and your doctor find the best dose for you. Children should not be retreated sooner than every 12 - 16 weeks.
- The dose of DYSPORT is not the same as the dose of any other botulinum toxin product
What should I avoid while receiving DYSPORT?
DYSPORT may cause loss of strength or general muscle weakness, blurred vision, or drooping eyelids within hours to weeks of taking DYSPORT. If this happens, do not drive a car, operate machinery, or do other dangerous activities. See "What is the most important information I should know about DYSPORT?"
What are the possible side effects of DYSPORT?
DYSPORT can cause serious side effects. See "What is the most important information I should know about DYSPORT?"
The most common side effects of DYSPORT in people with cervical dystonia include:
- muscle weakness
- dry mouth
- feeling of tiredness
- muscle pain
- problems speaking
- eye problems
- difficulty swallowing
- injection site pain or discomfort
- headache
The most common side effects of DYSPORT in people with glabellar lines include:
- stuffy or runny nose and sore throat
- injection site pain
- upper respiratory infection
- blood in urine
- headache
- injection site reaction
- swelling of eyelids
- drooping eyelids
- sinus infection
- nausea
The most common side effects of DYSPORT in adults with upper limb spasticity include:
• muscle weakness
The most common side effects of DYSPORT in adults with lower limb spasticity include:
- muscle weakness
- pain in your arms or legs
- fall
The most common side effects of DYSPORT in children (2 to 17 years of age) with upper limb spasticity include:
- upper respiratory tract infection
- sore throat
The most common side effects of DYSPORT in children (2 to 17 years of age) with lower limb spasticity include:
- stuffy or runny nose and sore throat
- cough
- fever
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects DYSPORT . For more information, ask your doctor or pharmacist.
Tell your doctor if you have dry eye or changes in vision following use of DYSPORT.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about DYSPORT:
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide.
This Medication Guide summarizes the most important information about DYSPORT. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about DYSPORT that is written for healthcare professionals.
What are the ingredients in DYSPORT?
Active ingredient: (botulinum toxin Type A)
Inactive ingredients: human albumin and lactose. DYSPORT may contain cow's milk protein.
Distributed by: Ipsen Biopharmaceuticals, Inc. Cambridge, MA 02142 and Galderma Laboratories, L.P. Dallas, TX 75201
Manufactured by: Ipsen Biopharma Ltd., Wrexham, LL13 9UF, UK U.S. License No. 1787
For more information about DYSPORT, call 855-463-5127 or go to
www.dysport.comor
www.DysportUSA.com.
This Medication Guide has been approved by the U.S. Food and Drug Administration
Revised 09/2023
DYSPORT is a registered trademark of Ipsen Biopharm Limited. Botox, Xeomin and Myobloc are registered trademarks of their respective owners.
© 2023. All rights reserved.
Section 43683-2
Section 44425-7
DYSPORT® (abobotulinumtoxinA) for Injection is a sterile, lyophilized powder supplied in a single-dose, glass vial. Unopened vials of DYSPORT must be stored refrigerated between 2°C to 8°C (36°F to 46°F). Protect from light.
Do not use after the expiration date on the vial. All vials, including expired vials, or equipment used with DYSPORT should be disposed of carefully as is done with all medical waste.
DYSPORT contains a unique hologram on the carton. If you do not see the hologram, do not use the product. Instead contact 855-463-5127.
10 Overdosage
Excessive doses of DYSPORT may be expected to produce neuromuscular weakness with a variety of symptoms. Respiratory support may be required where excessive doses cause paralysis of respiratory muscles. In the event of overdose, the patient should be medically monitored for symptoms of excessive muscle weakness or muscle paralysis [ see Boxed Warning and Warnings and Precautions (5.2) ]. Symptomatic treatment may be necessary.
Symptoms of overdose are likely not to be present immediately following injection. Should accidental injection or oral ingestion occur, the person should be medically supervised for several weeks for signs and symptoms of excessive muscle weakness or paralysis.
There is no significant information regarding overdose from clinical studies.
In the event of overdose, antitoxin raised against botulinum toxin is available from the Centers for Disease Control and Prevention (CDC) in Atlanta, GA. However, the antitoxin will not reverse any botulinum toxin-induced effects already apparent by the time of antitoxin administration. In the event of suspected or actual cases of botulinum toxin poisoning, please contact your local or state Health Department to process a request for antitoxin through the CDC. If you do not receive a response within 30 minutes, please contact the CDC directly at 770-488-7100. More information can be obtained at https://www.cdc.gov/laboratory/drugservice/index.html .
8.2 Lactation
Risk Summary
There are no data on the presence of DYSPORT in human or animal milk, the effects on the breastfed infant, or the effects on milk production.
The development and health benefits of breastfeeding should be considered along with the mother's clinical need for DYSPORT and any potential adverse effects on the breastfed infant from DYSPORT or from the underlying maternal condition.
1.3 Spasticity
DYSPORT is indicated for the treatment of spasticity in patients 2 years of age and older.
11 Description
Botulinum toxin type A, the active ingredient in DYSPORT, is a purified neurotoxin type A complex produced by fermentation of the bacterium Clostridium botulinumtype A, Hall Strain. It is purified from the culture supernatant by a series of precipitation, dialysis, and chromatography steps. The neurotoxin complex is composed of the neurotoxin, hemagglutinin proteins and non-toxin non-hemagglutinin protein.
DYSPORT® (abobotulinumtoxinA) for injection is a sterile, lyophilized powder supplied in a single-dose vial for reconstitution intended for intramuscular injection. Each vial contains 300 Units or 500 Units of lyophilized abobotulinumtoxinA, human serum albumin (125 mcg) and lactose (2.5 mg). DYSPORT may contain trace amounts of cow's milk proteins [ see Contraindications (4) and Warnings and Precautions (5.3)]. ].
The primary release procedure for DYSPORT uses a cell-based potency assay to determine the potency relative to a reference standard. The assay and reference material are specific to DYSPORT. One unit of DYSPORT corresponds to the calculated median lethal intraperitoneal dose (LD 50) in mice. Due to specific details of the assay system, such as vehicle, dilution scheme and laboratory protocols, Units of biological activity of DYSPORT cannot be converted into Units of any other botulinum toxin or any toxin assessed with any other specific assay method.
8.5 Geriatric Use
Cervical Dystonia
There were insufficient numbers of patients aged 65 years and over in the clinical studies to determine whether they respond differently than younger patients. In general, elderly patients should be observed to evaluate their tolerability of DYSPORT, due to the greater frequency of concomitant disease and other drug therapy [ see Dosage and Administration (2.3) ].
Glabellar Lines
Of the total number of subjects in the placebo-controlled clinical studies of DYSPORT, 8 (1%) were 65 years and over. Efficacy was not observed in subjects aged 65 years and over [ see Clinical Studies (14.2) ]. For the entire safety database of geriatric subjects, although there was no increase in the incidence of eyelid ptosis, geriatric subjects did have an increase in the number of ocular adverse reactions compared to younger subjects (11% vs. 5%) [ see Dosage and Administration (2.4) ].
Adult Spasticity
Upper Limb Spasticity
Of the total number of subjects in placebo-controlled clinical studies of DYSPORT, 30 percent were aged 65 years and over, while 8 percent were aged 75 years and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals can not be ruled out.
Lower Limb Spasticity
Of the total number of subjects in placebo controlled clinical studies of DYSPORT, 18% (n=115) were 65 and over, while 3% (n=20) were 75 and over. Subjects aged 65 years and over who were treated with DYSPORT reported a greater percentage of adverse reactions as compared to younger subjects (46% versus 39%). Fall and asthenia were observed with greater frequency in older subjects, as compared to those younger (10% versus 6% and 4% versus 2%, respectively).
8.6 Ethnic Groups
Exploratory analyses in trials for glabellar lines in African-American subjects with Fitzpatrick skin types IV, V, or VI and in Hispanic subjects suggested that response rates at Day 30 were comparable to and no worse than the overall population.
6.2 Immunogenicity
As with all therapeutic proteins, there is a potential for immunogenicity.
The incidence of antibody formation is highly dependent on the sensitivity and specificity of the assay. In addition, the observed incidence of antibody positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies across products in this class may be misleading.
1.2 Glabellar Lines
DYSPORT is indicated for the temporary improvement in the appearance of moderate to severe glabellar lines associated with procerus and corrugator muscle activity in adults less than 65 years of age.
4 Contraindications
DYSPORT is contraindicated in patients with:
- Known hypersensitivity to any botulinum toxin products, cow's milk protein, or to any of the components in the formulation [ see Warnings and Precautions (5.3)] . This product may contain trace amounts of cow's milk protein [ see Description (11)] .
- Infection at the proposed injection site(s).
6 Adverse Reactions
The following serious adverse reactions are discussed below and elsewhere in labeling:
- Spread of Toxin Effect [ see Warnings and Precautions (5.1)]
- Lack of Interchangeability between Botulinum Toxin Products [ see Warnings and Precautions (5.2)]
- Hypersensitivity Reactions
[
see Warnings and Precautions (5.3)
]
- Dysphagia and Breathing Difficulties [ see Warnings and Precautions (5.4)]
- Facial Anatomy in the Treatment of Glabellar Lines [ see Warnings and Precautions (5.5)]
- Dry Eye with the Treatment of Glabellar Lines [see Warnings and Precautions (5.6)]
- Pre-existing Neuromuscular Disorders [ see Warnings and Precautions (5.7)]
- Human Albumin and Transmission of Viral Diseases [ see Warnings and Precautions (5.8)]
- Intradermal Immune Reaction
[
see Warnings and Precautions (5.9)]
- Pre-existing Conditions at the Injection Site [ See Warnings and Precautions (5.10) ]
7 Drug Interactions
- Concomitant use of DYSPORT and aminoglycosides or other agents interfering with neuromuscular transmission or muscle relaxants, should be observed closely because the effect of DYSPORT may be potentiated (7.1, 7.4)
- Anticholinergic drugs may potentiate systemic anticholinergic effects (7.2)
- The effect of administering different botulinum neurotoxins during the course of treatment with DYSPORT is unknown (7.3)
14.2 Glabellar Lines
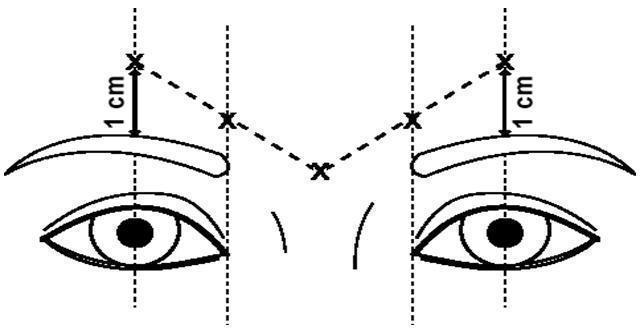
Three double-blind, randomized, placebo-controlled, clinical studies evaluated the efficacy of DYSPORT for use in the temporary improvement of the appearance of moderate to severe glabellar lines. These three studies enrolled healthy adults (ages 19-75) with glabellar lines of at least moderate severity at maximum frown. Subjects were excluded if they had marked ptosis, deep dermal scarring, or a substantial inability to lessen glabellar lines, even by physically spreading them apart. The subjects in these studies received either DYSPORT or placebo. The total dose was delivered in equally divided aliquots to specified injection sites (see Figure 1 ).
Investigators and subjects assessed efficacy at maximum frown by using a 4-point scale (none, mild, moderate, severe).
Overall treatment success was defined as post-treatment glabellar line severity of none or mild with at least 2 grade improvement from Baseline for the combined investigator and subject assessments (composite assessment) on Day 30 (see Table 16). Additional endpoints for each of the studies were post-treatment glabellar line severity of none or mild with at least a 1 grade improvement from Baseline for the separate investigator and subject assessments on Day 30.
After completion of the randomized studies, subjects were offered participation in a two-year, open-label re-treatment study to assess the safety of multiple treatments.
| 2 Grade Improvement | ||
| Study | DYSPORT n/N (%) | Placebo n/N (%) |
| GL-1 | 58/105 (55%) | 0/53 (0%) |
| GL-2 | 37/71 (52%) | 0/71 (0%) |
| GL-3 | 120/200 (60%) | 0/100 (0%) |
Treatment with DYSPORT reduced the severity of glabellar lines for up to four months.
12.2 Pharmacodynamics
The primary pharmacodynamic effect of DYSPORT is due to chemical denervation of the treated muscle resulting in a measurable decrease of the compound muscle action potential, causing a localized reduction of muscle activity.
12.3 Pharmacokinetics
Using currently available analytical technology, it is not possible to detect DYSPORT in the peripheral blood following intramuscular injection at the recommended doses.
7.4 muscle Relaxants
Excessive weakness may also be exaggerated by administration of a muscle relaxant before or after administration of DYSPORT.
14.1 Cervical Dystonia
The efficacy of DYSPORT was evaluated in two randomized, double-blind, placebo-controlled, single-dose, parallel-group studies in treatment-naive cervical dystonia patients. The principal analyses from these trials provide the primary demonstration of efficacy involving 252 patients (121 on DYSPORT, 131 on placebo) with 36% male and 64% female. Ninety-nine percent of the patients were Caucasian.
In both placebo-controlled studies (Study 1 and Study 2), a dose of 500 Units of DYSPORT was given by intramuscular injection divided among two to four affected muscles. These studies were followed by long-term open-label extensions that allowed titration in 250 Unit steps to doses in a range of 250 to 1000 Units, after the initial dose of 500 Units. In the extension studies, re-treatment was determined by clinical need after a minimum of 12 weeks. The median time to re-treatment was 14 weeks and 18 weeks for the 75 thpercentile.
The primary assessment of efficacy was based on the total Toronto Western Spasmodic Torticollis Rating Scale (TWSTRS) change from baseline at Week 4 for both studies. The scale evaluates the severity of dystonia, patient-perceived disability from dystonia, and pain. The adjusted mean change from baseline in the TWSTRS total score was statistically significantly greater for the DYSPORT group than the placebo group at Weeks 4 in both studies (see Table 14) .
| Study 1 | Study 2 | |||
|
DYSPORT
500 Units N=55 |
Placebo
N=61 |
DYSPORT
500 Units N=37 |
Placebo
N=43 |
|
|
Baseline (week 0)
Mean (SD) |
43.8 (8.0) |
45.8 (8.9) |
45.1 (8.7) |
46.2 (9.4) |
|
Week 4
Mean (SD) Change from Baseline Change from baseline is expressed as adjusted least square mean (SE)
|
30.0 (12.7) -15.6 (2.0) |
40.2 (11.8) -6.7 (2.) |
35.2 (13.8) -9.6 (2.0) |
42.4 (12.2) -3.7 (1.8) |
| treatment difference
95% confidence interval |
-8.9
Significant at
p-value < 0.05
[-12.9 to -4.7] |
-5.9
[-10.6 to -1.3] |
||
|
Week 8
Mean (SD) Change from Baseline |
29.2 (11.0) -14.7 (2.0) |
39.6 (13.5) -5.9 (2.0) |
||
| treatment difference
95% confidence interval |
-8.8
[-12.9 to -4.7] |
Analyses by gender, weight, geographic region, underlying pain, cervical dystonia severity at baseline and history of treatment with botulinum toxin did not show any meaningful differences between groups.
Table 15 indicates the average DYSPORT dose, and percentage of total dose, injected into specific muscles in the pivotal clinical trials.
|
Number of patients
injected per muscle Total number of patients in combined studies 2 and 1 who received initial treatment = 121.
|
DYSPORT
Dose Injected |
Percentage of the total
DYSPORT Dose Injected |
|||
|
Median
[DYSPORT Units] (min, max) |
75th percentile
[DYSPORT Units] |
Median
[%] (min, max) |
75th percentile
[%] |
||
| Sternocleidomastoid | 90 | 125 Units
(50, 350) |
150 Units | 26.5%
(10, 70) |
30.0% |
| Splenius capitis | 85 | 200 Units
(75, 450) |
250 Units | 40.0%
(15, 90) |
50.0% |
| Trapezius | 50 | 102.6 Units
(50, 300) |
150 Units | 20.6%
(10, 60) |
30.0% |
| Levator scapulae | 35 | 105.3 Units
(50, 200) |
125 Units | 21.1%
(10, 40) |
25.0% |
| Scalenus (medius and anterior) | 26 | 115.5 Units
(50, 300) |
150 Units | 23.1%
(10,60) |
30.0% |
| Semispinalis capitis | 21 | 131.6 Units
(50, 250) |
175 Units | 29.4%
(10, 50) |
35.0% |
| Longissimus | 3 | 150 Units
(100, 200) |
200 Units | 30.0%
(20, 40) |
40.0% |
1 Indications and Usage
DYSPORT is an acetylcholine release inhibitor and neuromuscular blocking agent indicated for:
12.1 Mechanism of Action
DYSPORT inhibits release of the neurotransmitter, acetylcholine, from peripheral cholinergic nerve endings. Toxin activity occurs in the following sequence: Toxin heavy chain mediated binding to specific surface receptors on nerve endings, internalization of the toxin by receptor mediated endocytosis, pH-induced translocation of the toxin light chain to the cell cytosol and cleavage of SNAP25 leading to intracellular blockage of neurotransmitter exocytosis into the neuromuscular junction. This accounts for the therapeutic utility of the toxin in diseases characterized by excessive efferent activity in motor nerves.
Recovery of transmission occurs gradually as the neuromuscular junction recovers from SNAP25 cleavage and as new nerve endings are formed.
7.2 Anticholinergic Drugs
Use of anticholinergic drugs after administration of DYSPORT may potentiate systemic anticholinergic effects such as blurred vision.
5 Warnings and Precautions
- The potency units of DYSPORT are not interchangeable with other preparations of botulinum toxin products ( 5.2)
- Immediate medical attention may be required in cases of respiratory, speech or swallowing difficulties ( 5.4)
- Recommended dose and frequency of administration should not be exceeded ( 5.5)
- Dry eye may occur with glabellar line treatment; if symptoms persist, consider referring patient to an ophthalmologist ( 5.6)
- Concomitant neuromuscular disorder may exacerbate clinical effects of treatment ( 5.7)
2 Dosage and Administration
Preparation of DYSPORT Solution for Administration ( 2.2)
- Once reconstituted, store in original container in a refrigerator 2°C to 8°C (36°F to 46°F) and use within 24 hours
- Do not freeze after reconstitution
- Reconstitution instructions are specific for the 300 Unit and 500 Unit vials
- Reconstituted DYSPORT is intended for intramuscular injection only. After reconstitution, DYSPORT should be used for only one injection session and for only one patient.
Cervical Dystonia ( 2.3)
• Initial dose is 500 Units given intramuscularly as a divided dose among the affected muscles
• Re-treatment every 12 to 16 weeks or longer, as necessary, based on return of clinical symptoms with doses administered between 250 and 1000 Units to optimize clinical benefit
• Re-treatment should not occur in intervals of less than 12 weeks
• Titrate in 250 Unit steps according to patient's response
Glabellar Lines ( 2.4)
- Administer a total dose of 50 Units, divided in five equal aliquots of 10 Units each, intramuscularly to affected muscles to achieve clinical effect
- Re-treatment should be administered no more frequently than every 3 months
Spasticity in Adults ( 2.5)
- Select dose based on muscles affected, severity of spasticity, and treatment and adverse reaction history botulinum toxins
- Dosing for upper limb spasticity: between 500 Units and 1000 Units
- Dosing for lower limb spasticity: up to 1500 Units
- The maximum recommended total dose per treatment session (upper and lower limb combined) in adults is 1500 Units
- Re-treatment, based on return of clinical symptoms, should not occur in intervals of less than 12 weeks
Spasticity in Pediatric Patients ( 2.6)
- Select dose based on the affected muscle, severity of spasticity, and treatment and adverse reaction history with all botulinum toxins.
- Recommended dosing for upper limb spasticity: 8 Units/kg to 16 Units/kg per limb. The maximum recommended total dose administered per treatment session must not exceed 16 Units/kg or 640 Units, whichever is lower.
- Recommended dosing for lower limb spasticity: 10 Units/kg to 15 Units/kg per limb. Total dose per treatment session must not exceed 15 Units/kg for unilateral lower limb injections, 30 Units/kg for bilateral injections, or 1000 Units, whichever is lower.
- The maximum recommended total dose per treatment session is 30 Units/kg or 1000 Units, whichever is lower. Re-treatment, based on return of clinical symptoms, should not occur in intervals of less than 3 months.
3 Dosage Forms and Strengths
For injection: 300 Units or 500 Units of lyophilized powder in a single-dose vial for reconstitution with preservative-free 0.9% Sodium Chloride Injection, USP.
6.3 Postmarketing Experience
Because adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post-approval use of DYSPORT: vertigo, photophobia, influenza-like illness, amyotrophy, muscle atrophy, burning sensation, facial paresis, hypoesthesia, erythema, dry eye, and excessive granulation tissue. Hypersensitivity reactions including anaphylaxis have been reported.
2.4 Dosing in Glabellar Lines
The dose of DYSPORT for the treatment of glabellar lines in adults is a total of 50 Units given intramuscularly in five equal aliquots of 10 Units each to achieve clinical effect (see Figure 1).
8 Use in Specific Populations
- Administer DYSPORT with care in elderly patients, reflecting the greater frequency of concomitant disease and other drug therapy ( 8.5)
2.1 Instructions for Safe Use
The potency units of DYSPORT are not interchangeable with other preparations of botulinum toxin products and, therefore, units of biological activity of DYSPORT cannot be compared to or converted into units of any other botulinum toxin products assessed with any other specific assay method [ see Warnings and Precautions (5.2) and Description (11) ]. Reconstituted DYSPORT is intended for intramuscular injection only.
2.3 Dosing in Cervical Dystonia
The recommended initial dose of DYSPORT for the treatment of cervical dystonia in adults is 500 Units given intramuscularly as a divided dose among affected muscles in patients with or without a history of prior treatment with botulinum toxin. (A description of the average DYSPORT dose and percentage of total dose injected into specific muscles in the pivotal clinical trials can be found in Table 15 of Section 14.1, Clinical Studies – Cervical Dystonia.) Limiting the dose injected into the sternocleidomastoid muscle may reduce the occurrence of dysphagia. Clinical studies with DYSPORT in cervical dystonia suggest that the peak effect occurs between two and four weeks after injection. Simultaneous guided injection of DYSPORT with EMG and/or ultrasound may be helpful in locating active muscles.
Dose Modification
Where dose modification is necessary for the treatment of cervical dystonia, uncontrolled open-label studies suggest that dose adjustment can be made in 250 Unit steps according to the individual patient's response, with re-treatment every 12 weeks or longer, as necessary, based on return of clinical symptoms. Uncontrolled open-label studies also suggest that the total dose administered in a single treatment should be between 250 Units and 1000 Units. Re-treatment, if needed, should not occur in intervals of less than 12 weeks. Doses above 1000 Units have not been systematically evaluated.
5.9 Intradermal Immune Reaction
The possibility of an immune reaction when injected intradermally is unknown. The safety of DYSPORT for the treatment of hyperhidrosis has not been established. DYSPORT is approved only for intramuscular injection.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Swallowing, Speaking, or Breathing Difficulties, or Other Unusual Symptoms
Advise patients to inform their doctor or pharmacist if they develop any unusual symptoms (including difficulty with swallowing, speaking or breathing), or if any known symptom persists or worsens
[see Warnings and Precautions (5.1, 5.4)].
Dry Eye with the Treatment of Glabellar Lines
Inform patients that DYSPORT injection may cause eye dryness [see Warnings and Precautions (5.6)]. Advise patients to report symptoms of eye dryness (e.g., eye pain, eye irritation, photosensitivity, or changes in vision) to their doctor.
Ability to Operate Machinery or Vehicles
Inform patients that if loss of strength, muscle weakness, blurred vision or drooping eyelids occur, they should avoid driving a car or engaging in other potentially hazardous activities.
Principal Display Panel 300 Unit
GALDERMA
Dysport
®
(AbobotulinumtoxinA)
for injection
For intramuscular use
300 UNITS/SINGLE-DOSE VIAL
1 VIAL
WARNING: Dosing units of botulinum toxins are not interchangeable between commercial products
NDC 0299-5962-30
Rx only
14.4 Spasticity in Pediatric Patients
Upper Limb Spasticity in Pediatric Patients
The efficacy of DYSPORT for the treatment of upper limb spasticity in pediatric patients 2 to 17 years of age was evaluated in a double-blind, low-dose controlled, multicenter study (NCT02106351). A total of 208 patients with spasticity because of cerebral palsy who were toxin naïve or non-naïve (66% had prior treatment with botulinum toxin), weighed at least 10 kgs, and had a baseline Modified Ashworth Score (MAS) of grade 2 or greater (99% patients) at the primary targeted muscle groups (PTMG), were enrolled in the modified Intention to Treat population (mITT). Patients received DYSPORT 16 Units/kg (n=70), DYSPORT 8 Units/kg (n=69), or DYSPORT 2 Units/kg (n=69) injected into the upper limb. The elbow flexors and wrist flexors respectively were the PTMG in 57% and in 43% of patients. The median age of the patients in this study was 9 years (range 2 to 17 years; 57% were between 2 and 9 years of age); 60% of patients were male, and 75% were White.
The primary efficacy endpoint was the mean change from baseline in MAS in the PTMG at Week 6 (see Table 24). The secondary efficacy endpoint was the mean Physician Global Assessment (PGA) score assessed at Week 6 (Table 25). Although PGA scores numerically favored DYSPORT treatment over the low-dose control, the difference was not statistically significant.
| Control Group | Treatment Groups | ||
|
|
DYSPORT
2 U/kg (N=69) |
DYSPORT
8 U/kg (N=69) |
DYSPORT
16 U/kg (N=70) |
| Baseline | |||
| Mean (SD) | 3.1 (0.3) | 3.1 (0.3) | 3.1 (0.5) |
| Week 6 | |||
| LS amean change from baseline in PTMG bon MAS | -1.6 | -2.0 | -2.3 |
| Difference from control in LS ameans | -0.4 | -0.7 | |
| p-value c | 0.0118 d | <0.0001 | |
| Week 16 | |||
| LS amean change from baseline in PGMG bon MAS | -0.9 | -1.2 | -1.5 |
| Difference from control in LS ameans | -0.3 d | -0.6 d |
aLS = Least Square
bPTMG=Primary Targeted Muscle Group
cp-value is derived from ANCOVA on ranked MAS score change from baseline with treatment, baseline score, age range at baseline, prior botulinum toxin treatment status at baseline, and center as explanatory variables
dNominal p-value <0.05
| Control Group | Treatment Group | ||
|
DYSPORT
2 U/kg (N=68) |
DYSPORT
8 U/kg (N=69) |
DYSPORT
16 U/kg (N=70) |
|
| Week 6 | |||
| Mean score (SD) | 1.7 (0.9) | 2.0 (0.9) | 2.0 (0.9) |
| LS amean in PGA | 1.8 | 2.0 | 2.0 |
| Difference from control in LS amean | 0.2 | 0.2 | |
| p-value b | 0.2043 | 0.1880 | |
| Week 16 | |||
| Mean score (SD) | 1.7 (1.0) | 1.6 (1.1) | 1.9 (1.2) |
| LS amean in PGA | 1.8 | 1.7 | 1.9 |
| Difference from contron in LS amean | -0.1 | 0.1 | |
| p-value b | 0.7001 | 0.4041 |
aLS=Least Square
bp-value is derived from ANOVA on ranked PGA score with treatment, age range at baseline, prior botulinum toxin treatment status at baseline, and center as explanatory variables
Lower Limb Spasticity in Pediatric Patients
The efficacy of DYSPORT for the treatment of lower limb spasticity in patients 2 to 17 years of age was evaluated in a double-blind, placebo-controlled, multicenter study. A total of 235 patients with cerebral palsy causing dynamic equinus foot deformity who were toxin-naïve or non-naïve and had a Modified Ashworth Score (MAS) of grade 2 or greater at the ankle plantar flexors were enrolled. Patients received DYSPORT 10 Units/kg/leg (n=79), DYSPORT 15 Units/kg/leg (n=79) or placebo (n=77) injected into the gastrocnemius and soleus muscles (see Table 27). Forty-one percent of patients (n=66) were treated bilaterally and received a total lower limb DYSPORT dose of either 20 Units/kg (n=37) or 30 Units/kg (n=29). The median age of the patients in this study was 5 years (range 2 to 17 years); 60% of patients were male, and 73% were Caucasian.
The primary efficacy endpoint was the mean change from baseline in MAS in ankle plantar flexor at Week 4; a co-primary endpoint was the mean Physician’s Global Assessment (PGA) score at Week 4 (see Table 26).
|
Placebo
(N=77) |
DYSPORT
10 Units/kg/leg (N=79) |
DYSPORT
15 Units/kg/leg (N=79) |
||
| LS Mean Change from
Baseline in Ankle plantar flexor Muscle Tone on the MAS |
Week 4 | -0.5 | -0.9* | -1.0* |
| Week 12 | -0.5 | -0.8* | -1.0* | |
| LS Mean PGA of
Response to Treatment |
Week 4 | 0.7 | 1.5* | 1.5* |
| Week 12 | 0.4 | 0.8* | 1.0* | |
|
LS=Least Square
|
Warning: Distant Spread of Toxin Effect
Postmarketing reports indicate that the effects of DYSPORT and all botulinum toxin products may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These may include asthenia, generalized muscle weakness, diplopia, blurred vision, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence and breathing difficulties. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life threatening and there have been reports of death. The risk of symptoms is probably greatest in children treated for spasticity but symptoms can also occur in adults treated for spasticity and other conditions, particularly in those patients who have underlying conditions that would predispose them to these symptoms. In unapproved uses and in approved indications, cases of spread of effect have been reported at doses comparable to or lower than the maximum recommended total dose [ see Warnings and Precautions (5.1) ].
7.3 other Botulinum Neurotoxin Products
The effect of administering botulinum neurotoxin products including DYSPORT, at the same time or within several months of each other is unknown. Excessive weakness may be exacerbated by another administration of botulinum toxin prior to the resolution of the effects of a previously administered botulinum toxin.
2.6 Dosing in Spasticity in Pediatric Patients
DYSPORT dosing for spasticity in pediatric patients is based on Units per kilogram of body weight. To calculate the total units of DYSPORT required for treatment of one limb, select the dose of DYSPORT in Units/kg and the body weight (kg) of the patient (see Tables 5 and 6). Dosing in initial and sequential treatment sessions should be tailored to the individual patient based on the size, number and location of muscles involved, severity of spasticity, the presence of local muscle weakness, the patient’s response to previous treatment, and/or adverse reaction history with botulinum toxins.
No more than 0.5 mL should generally be administered at any single injection site. The maximum recommended total dose of DYSPORT in a single treatment session for spasticity in pediatric patients 2 years or older is 30 Units/kg or 1000 Units in a 3-month interval.
Although actual location of the injection sites can be determined by palpation, the use of injection guiding technique (e.g., electromyography or electrical stimulation, or ultrasound) is recommended to target the injection sites.
Upper Limb Spasticity in Pediatric Patients 2 Years of Age and Older
In the clinical trial that assessed the efficacy and safety of DYSPORT for treatment of upper limb spasticity in pediatric patients 2 years of age or older with a weight of at least 10 kg [
see Clinical Studies (14.4)], doses of 8 Units/kg or 16 Units/kg were divided among selected muscles of the target upper limb at a given treatment session (see Table 5 and Figure 4).
Table 5 describes the recommended Units/kg dose of DYSPORT per muscle. The maximum recommended total dose of DYSPORT administered for treatment of upper limb spasticity must not exceed 16 Units/kg or 640 Units, whichever is lower.
| Muscle |
Recommended Dose Range
per muscle per upper limb (units/kg Body Weight) |
Number of injection sites
per muscle |
| Brachialis | 3 Units/kg to 6 Units/kg | Up to 2 |
| Brachioradialis | 1.5 Units/kg to 3 Units/kg | 1 |
| Biceps brachii | 3 Units/kg to 6 Units/kg | Up to 2 |
| Pronator teres | 1 Units/kg to 2 Units/kg | 1 |
| Prontator quadratus | 0.5 Unit/kg to 1 Units/kg | 1 |
| Flexor carpi radialis (FCR) | 2 Units/kg to 4 Units/kg | Up to 2 |
| Flexor carpi ulnaris (FCU) | 1.5 Units/kg to 3 Units/kg | 1 |
| Flexor digitorum profundus (FDP) | 1 Units/kg to 2 Units/kg | 1 |
| Flexor digitorum superficialis (FDS) | 1.5 Units/kg to 3 Units/kg | Up to 4 |
| Total dose | 8 Units/kg to 16 Units/kg in upper limbs (and not exceeding 640 Units) |
Figure 4: Muscles for Injection for Upper Limb Spasticity in Pediatric Patients
Repeat DYSPORT treatment should be administered when the effect of a previous injection has diminished but no sooner than 16 weeks after the previous injection. A majority of patients in the clinical study were retreated between 16-28 weeks; however, some patients had a longer duration of response (i.e., 34 weeks or more). The degree and pattern of muscle spasticity at the time of re-injection may necessitate alterations in the dose of DYSPORT and muscles to be injected.
Lower Limb Spasticity in Pediatric Patients 2 Years of Age and Older
In the clinical trial that assessed the efficacy and safety of DYSPORT for treatment of lower limb spasticity in pediatric patients 2 years of age or older
[see Clinical Studies (14.4)], doses of 10 Units/kg to 15 Units/kg were divided among selected muscles of the target lower limb at a given treatment session (see Table 6 and Figure 5).
Table 6 describes the recommended Units/kg dose of DYSPORT per muscle of the Gastrocnemius-Soleus Complex (GSC). The recommended total DYSPORT dose per treatment session is 10 Units/kg to 15 Units/kg for unilateral lower limb injections or 20 Units/kg to 30 Units/kg for bilateral lower limb injections. However, the total dose of DYSPORT administered in a 3-month interval must not exceed 15 Units/kg for unilateral lower limb injections, 30 Units/kg for bilateral lower limb injections, or 1000 units, whichever is lower. The total dose administered should be divided between the affected spastic muscles of the lower limb(s). When possible, the dose should be distributed across more than 1 injection site in any single muscle (see Table 6).
| Muscle Injected |
Recommended DYSPORT Dose Range per
muscle per leg (Units/kg Body Weight) |
Recommended number of
injections per muscle |
| Gastrocnemius | 6 Units/kg to 9 Units/kg
the listed individual doses to be injected in the muscles can be used within the range mentioned without exceeding 15 Units/kg total dose for unilateral injection or 30 Units/kg for bilateral injections or 1000 Units whichever is lower.
|
Up to 4 |
| Soleus | 4 Units/kg to 6 Units/kg | Up to 2 |
| Total | 10 Units/kg to 15 Units/kg divided across both muscles | Up to 6 |
Figure 5: Muscles for Injection for Lower Limb Spasticity in Pediatric Patients
Although actual location of the injection sites can be determined by palpation, the use of injection guiding technique (e.g. electromyography or electrical stimulation or ultrasound) is recommended to target the injection sites.
Repeat DYSPORT treatment should be administered when the effect of a previous injection has diminished but no sooner than 12 weeks after the previous injection. A majority of patients in the clinical studies were retreated between 16-22 weeks, however; some had a longer duration of response. The degree and pattern of muscle spasticity and overall clinical benefit at the time of re-injection may necessitate alterations in the dose of DYSPORT and muscles to be injected.
The safety and effectiveness of DYSPORT injected into proximal muscles of the lower limb for the treatment of spasticity in pediatric patients has not been established.
5.10 Pre Existing Conditions At the Injection Site
Caution should be exercised when DYSPORT is used where the targeted muscle shows excessive weakness or atrophy.
5.8 Human Albumin and Transmission of Viral Diseases
This product contains albumin, a derivative of human blood. Based on effective donor screening and product manufacturing processes, it carries an extremely remote risk for transmission of viral diseases and variant Creutzfeldt-Jakob disease (vCJD). There is a theoretical risk for transmission of Creutzfeldt-Jakob disease (CJD), but if that risk actually exists, the risk of transmission would also be considered extremely remote. No cases of transmission of viral diseases, CJD, or vCJD have ever been identified for licensed albumin or albumin contained in other licensed products.
7.1 Aminoglycosides and Other Agents Interfering With Neuromuscular Transmission
Co-administration of DYSPORT and aminoglycosides or other agents interfering with neuromuscular transmission (e.g., curare-like agents) should only be performed with caution because the effect of the botulinum toxin may be potentiated. If co-administered, observe the patient closely.
Structured Label Content
Section 34077-8 (34077-8)
Risk Summary
There are no adequate and well-controlled clinical studies with DYSPORT in pregnant women.
DYSPORT should only be used during pregnancy if the potential benefit justifies the potential risk to the fetus.
DYSPORT produced embryo-fetal toxicity in relation to maternal toxicity when given to pregnant rats and rabbits at doses lower than or similar to the maximum recommended human dose (MRHD) of 1000 Units on a body weight (Units/kg) basis (
see Data).
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. The background risk of major birth defects and miscarriage for the indicated population is unknown.
Data
Animal Data
In a study in which pregnant rats received daily intramuscular injections of DYSPORT (2.2, 6.6, or 22 Units/kg on gestation days 6 through 17 or intermittently 44 Units/kg on gestation days 6 and 12 only) during organogenesis, increased early embryonic death was observed with both schedules at the highest tested doses (22 and 44 Units/kg), which were associated with maternal toxicity. The no-effect dose for embryo-fetal developmental toxicity was 2.2 Units/kg (less than maximum recommended human [MRHD] on a body weight basis).
In a study in which pregnant rabbits received daily intramuscular injections of DYSPORT® (0.3, 3.3 or 6.7 Units/kg) on gestation days 6 through 19 or intermittently (13.3 Units/kg on gestation days 6 and 13 only) during organogenesis, no embryofetal data were available at the highest dose administered daily (6.7 Units/kg) because of premature death in all does at that dose. At the lower daily doses or with intermittent dosing, no adverse developmental effects were observed. All dosed for which data were available are less than the MRHD on a body weight basis.
In a study in which pregnant rats received 6 weekly intramuscular injections of DYSPORT (4.4, 11.1, 22.2, or 44 Units/kg) beginning on day 6 of gestation and continuing through parturition to weaning, an increase in stillbirths was observed at the highest dose tested, which was maternally toxic. The no-effect dose for pre- and post-natal development toxicity was 22.2 Units/kg (similar to the MRHD).
Section 34083-6 (34083-6)
Carcinogenesis
Studies to evaluate the carcinogenic potential of DYSPORT have not been conducted.
Section 42229-5 (42229-5)
DYSPORT is indicated for the treatment of cervical dystonia in adults.
Section 42231-1 (42231-1)
MEDICATION GUIDE
DYSPORT® (
DIS-port)
(abobotulinumtoxinA)
for Injection
What is the most important information I should know about DYSPORT?
DYSPORT may cause serious side effects that can be life threatening including:
• Problems breathing or swallowing
• Spread of toxin effects
These problems can happen within hours, or days to weeks after an injection of DYSPORT. Call your doctor or get medical help right away if you have any of these problems after treatment with DYSPORT:
1. Problems swallowing, speaking, or breathing. These problems can happen within hours, or days to weeks after an injection of DYSPORTusually because the muscles that you use to breathe and swallow can become weak after the injection. Death can happen as a complication if you have severe problems with swallowing or breathing after treatment with DYSPORT.
• People with certain breathing problems may need to use muscles in their neck to help them breathe. These patients may be at greater risk for serious breathing problems with DYSPORT.
• Swallowing problems may last for several weeks. People who cannot swallow well may need a feeding tube to receive food and water. If swallowing problems are severe, food or liquids may go into your lungs. People who already have swallowing or breathing problems before receiving DYSPORT have the highest risk of getting these problems.
2. Spread of toxin effects.In some cases, the effect of botulinum toxin may affect areas of the body away from the injection site and cause symptoms of a serious condition called botulism. The symptoms of botulism include:
• loss of strength and muscle weakness all over the body
• blurred vision and drooping eyelids
• trouble saying words clearly (dysarthria)
• trouble breathing
• double vision
• hoarseness or change or loss of voice (dysphonia)
• loss of bladder control
• trouble swallowing
These symptoms can happen within hours, or days to weeks after you receive an injection of DYSPORT.These problems could make it unsafe for you to drive a car or do other dangerous activities. See "What should I avoid while receiving DYSPORT?"
What is DYSPORT?
DYSPORT is a prescription medicine that is injected into muscles and used:
- to treat cervical dystonia (CD) in adults
- to improve the look of moderate to severe frown lines between the eyebrows (glabellar lines) in adults younger than 65 years of age for a short period of time (temporary)
- to treat increased muscle stiffness in people 2 years of age and older with spasticity
It is not known whether DYSPORT is safe or effective in people younger than:
- 18 years of age for the treatment of cervical dystonia
- 18 years of age for the treatment of glabellar lines
- 2 years of age for the treatment of spasticity
It is not known whether DYSPORT is safe or effective for the treatment of other wrinkles.
Who should not take DYSPORT?
Do not take DYSPORT if you:
- are allergic to DYSPORT or any of the ingredients in DYSPORT. See the end of this Medication Guide for a list of ingredients in DYSPORT
- are allergic to cow's milk protein
- had an allergic reaction to any other botulinum toxin product such as Myobloc® (rimabotulinumtoxinB), Botox® (onabotulinumtoxinA), or Xeomin® (incobotulinumtoxinA)
- have a skin infection at the planned injection site
What should I tell my doctor before taking DYSPORT?
Tell your doctor about all your medical conditions, including if you:
- have a disease that affects your muscles and nerves (such as amyotrophic lateral sclerosis [ALS or Lou Gehrig's disease], myasthenia gravis or Lambert-Eaton syndrome). See "What is the most important information I should know about DYSPORT?"
- have allergies to any botulinum toxin product
- had any side effect from botulinum toxin product in the past
- have or have had a breathing problem, such as asthma or emphysema
- have or have had swallowing problems
- have or have had bleeding problems
- have diabetes
- have or have had a slow heart beat or other problem with your heart rate or rhythm
- have plans to have surgery
- had surgery on your face
- have weakness in or near your muscles being treated
- have drooping eyelids
- experienced dry eye with previous use of botulinum toxin products
- have any other change in the way your face normally looks
- are pregnant or plan to become pregnant. It is not known if DYSPORT can harm your unborn baby
- are breast-feeding or planning to breast-feed. It is not known if DYSPORT passes into breast milk
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal products. Using DYSPORT with certain other medicines may cause serious side effects.
Do not start any new medicines until you have told your doctor that you have received DYSPORT in the past.
Especially tell your doctor if you:
- have received any other botulinum toxin product in the last four months
- have received injections of botulinum toxin, such as Myobloc® (rimabotulinumtoxinB), Botox® (onabotulinumtoxinA) or Xeomin® (incobotulinumtoxinA) in the past; be sure your doctor knows exactly which product you received
- have recently received an antibiotic by injection
- take muscle relaxants
- take an allergy or cold medicine
- take a sleep medicine
Ask your doctor if you are not sure if your medicine is one that is listed above.
Know the medicines you take. Keep a list of your medicines with you to show your doctor and pharmacist each time you get a new medicine.
How will I receive DYSPORT?
- DYSPORT is an injection that your doctor will give you
- DYSPORT is injected into the affected muscles
- If you are and adult, your doctor may give you another dose of DYSPORT after 12 weeks or longer, if it is needed
- If you are an adult being treated for CD or spasticity or you are a child (2 to 17 years of age) being treated for spasticity, your doctor may change your dose of DYSPORT, until you and your doctor find the best dose for you. Children should not be retreated sooner than every 12 - 16 weeks.
- The dose of DYSPORT is not the same as the dose of any other botulinum toxin product
What should I avoid while receiving DYSPORT?
DYSPORT may cause loss of strength or general muscle weakness, blurred vision, or drooping eyelids within hours to weeks of taking DYSPORT. If this happens, do not drive a car, operate machinery, or do other dangerous activities. See "What is the most important information I should know about DYSPORT?"
What are the possible side effects of DYSPORT?
DYSPORT can cause serious side effects. See "What is the most important information I should know about DYSPORT?"
The most common side effects of DYSPORT in people with cervical dystonia include:
- muscle weakness
- dry mouth
- feeling of tiredness
- muscle pain
- problems speaking
- eye problems
- difficulty swallowing
- injection site pain or discomfort
- headache
The most common side effects of DYSPORT in people with glabellar lines include:
- stuffy or runny nose and sore throat
- injection site pain
- upper respiratory infection
- blood in urine
- headache
- injection site reaction
- swelling of eyelids
- drooping eyelids
- sinus infection
- nausea
The most common side effects of DYSPORT in adults with upper limb spasticity include:
• muscle weakness
The most common side effects of DYSPORT in adults with lower limb spasticity include:
- muscle weakness
- pain in your arms or legs
- fall
The most common side effects of DYSPORT in children (2 to 17 years of age) with upper limb spasticity include:
- upper respiratory tract infection
- sore throat
The most common side effects of DYSPORT in children (2 to 17 years of age) with lower limb spasticity include:
- stuffy or runny nose and sore throat
- cough
- fever
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects DYSPORT . For more information, ask your doctor or pharmacist.
Tell your doctor if you have dry eye or changes in vision following use of DYSPORT.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about DYSPORT:
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide.
This Medication Guide summarizes the most important information about DYSPORT. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about DYSPORT that is written for healthcare professionals.
What are the ingredients in DYSPORT?
Active ingredient: (botulinum toxin Type A)
Inactive ingredients: human albumin and lactose. DYSPORT may contain cow's milk protein.
Distributed by: Ipsen Biopharmaceuticals, Inc. Cambridge, MA 02142 and Galderma Laboratories, L.P. Dallas, TX 75201
Manufactured by: Ipsen Biopharma Ltd., Wrexham, LL13 9UF, UK U.S. License No. 1787
For more information about DYSPORT, call 855-463-5127 or go to
www.dysport.comor
www.DysportUSA.com.
This Medication Guide has been approved by the U.S. Food and Drug Administration
Revised 09/2023
DYSPORT is a registered trademark of Ipsen Biopharm Limited. Botox, Xeomin and Myobloc are registered trademarks of their respective owners.
© 2023. All rights reserved.
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
DYSPORT® (abobotulinumtoxinA) for Injection is a sterile, lyophilized powder supplied in a single-dose, glass vial. Unopened vials of DYSPORT must be stored refrigerated between 2°C to 8°C (36°F to 46°F). Protect from light.
Do not use after the expiration date on the vial. All vials, including expired vials, or equipment used with DYSPORT should be disposed of carefully as is done with all medical waste.
DYSPORT contains a unique hologram on the carton. If you do not see the hologram, do not use the product. Instead contact 855-463-5127.
10 Overdosage (10 OVERDOSAGE)
Excessive doses of DYSPORT may be expected to produce neuromuscular weakness with a variety of symptoms. Respiratory support may be required where excessive doses cause paralysis of respiratory muscles. In the event of overdose, the patient should be medically monitored for symptoms of excessive muscle weakness or muscle paralysis [ see Boxed Warning and Warnings and Precautions (5.2) ]. Symptomatic treatment may be necessary.
Symptoms of overdose are likely not to be present immediately following injection. Should accidental injection or oral ingestion occur, the person should be medically supervised for several weeks for signs and symptoms of excessive muscle weakness or paralysis.
There is no significant information regarding overdose from clinical studies.
In the event of overdose, antitoxin raised against botulinum toxin is available from the Centers for Disease Control and Prevention (CDC) in Atlanta, GA. However, the antitoxin will not reverse any botulinum toxin-induced effects already apparent by the time of antitoxin administration. In the event of suspected or actual cases of botulinum toxin poisoning, please contact your local or state Health Department to process a request for antitoxin through the CDC. If you do not receive a response within 30 minutes, please contact the CDC directly at 770-488-7100. More information can be obtained at https://www.cdc.gov/laboratory/drugservice/index.html .
8.2 Lactation
Risk Summary
There are no data on the presence of DYSPORT in human or animal milk, the effects on the breastfed infant, or the effects on milk production.
The development and health benefits of breastfeeding should be considered along with the mother's clinical need for DYSPORT and any potential adverse effects on the breastfed infant from DYSPORT or from the underlying maternal condition.
1.3 Spasticity
DYSPORT is indicated for the treatment of spasticity in patients 2 years of age and older.
11 Description (11 DESCRIPTION)
Botulinum toxin type A, the active ingredient in DYSPORT, is a purified neurotoxin type A complex produced by fermentation of the bacterium Clostridium botulinumtype A, Hall Strain. It is purified from the culture supernatant by a series of precipitation, dialysis, and chromatography steps. The neurotoxin complex is composed of the neurotoxin, hemagglutinin proteins and non-toxin non-hemagglutinin protein.
DYSPORT® (abobotulinumtoxinA) for injection is a sterile, lyophilized powder supplied in a single-dose vial for reconstitution intended for intramuscular injection. Each vial contains 300 Units or 500 Units of lyophilized abobotulinumtoxinA, human serum albumin (125 mcg) and lactose (2.5 mg). DYSPORT may contain trace amounts of cow's milk proteins [ see Contraindications (4) and Warnings and Precautions (5.3)]. ].
The primary release procedure for DYSPORT uses a cell-based potency assay to determine the potency relative to a reference standard. The assay and reference material are specific to DYSPORT. One unit of DYSPORT corresponds to the calculated median lethal intraperitoneal dose (LD 50) in mice. Due to specific details of the assay system, such as vehicle, dilution scheme and laboratory protocols, Units of biological activity of DYSPORT cannot be converted into Units of any other botulinum toxin or any toxin assessed with any other specific assay method.
8.5 Geriatric Use
Cervical Dystonia
There were insufficient numbers of patients aged 65 years and over in the clinical studies to determine whether they respond differently than younger patients. In general, elderly patients should be observed to evaluate their tolerability of DYSPORT, due to the greater frequency of concomitant disease and other drug therapy [ see Dosage and Administration (2.3) ].
Glabellar Lines
Of the total number of subjects in the placebo-controlled clinical studies of DYSPORT, 8 (1%) were 65 years and over. Efficacy was not observed in subjects aged 65 years and over [ see Clinical Studies (14.2) ]. For the entire safety database of geriatric subjects, although there was no increase in the incidence of eyelid ptosis, geriatric subjects did have an increase in the number of ocular adverse reactions compared to younger subjects (11% vs. 5%) [ see Dosage and Administration (2.4) ].
Adult Spasticity
Upper Limb Spasticity
Of the total number of subjects in placebo-controlled clinical studies of DYSPORT, 30 percent were aged 65 years and over, while 8 percent were aged 75 years and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals can not be ruled out.
Lower Limb Spasticity
Of the total number of subjects in placebo controlled clinical studies of DYSPORT, 18% (n=115) were 65 and over, while 3% (n=20) were 75 and over. Subjects aged 65 years and over who were treated with DYSPORT reported a greater percentage of adverse reactions as compared to younger subjects (46% versus 39%). Fall and asthenia were observed with greater frequency in older subjects, as compared to those younger (10% versus 6% and 4% versus 2%, respectively).
8.6 Ethnic Groups
Exploratory analyses in trials for glabellar lines in African-American subjects with Fitzpatrick skin types IV, V, or VI and in Hispanic subjects suggested that response rates at Day 30 were comparable to and no worse than the overall population.
6.2 Immunogenicity
As with all therapeutic proteins, there is a potential for immunogenicity.
The incidence of antibody formation is highly dependent on the sensitivity and specificity of the assay. In addition, the observed incidence of antibody positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies across products in this class may be misleading.
1.2 Glabellar Lines
DYSPORT is indicated for the temporary improvement in the appearance of moderate to severe glabellar lines associated with procerus and corrugator muscle activity in adults less than 65 years of age.
4 Contraindications (4 CONTRAINDICATIONS)
DYSPORT is contraindicated in patients with:
- Known hypersensitivity to any botulinum toxin products, cow's milk protein, or to any of the components in the formulation [ see Warnings and Precautions (5.3)] . This product may contain trace amounts of cow's milk protein [ see Description (11)] .
- Infection at the proposed injection site(s).
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed below and elsewhere in labeling:
- Spread of Toxin Effect [ see Warnings and Precautions (5.1)]
- Lack of Interchangeability between Botulinum Toxin Products [ see Warnings and Precautions (5.2)]
- Hypersensitivity Reactions
[
see Warnings and Precautions (5.3)
]
- Dysphagia and Breathing Difficulties [ see Warnings and Precautions (5.4)]
- Facial Anatomy in the Treatment of Glabellar Lines [ see Warnings and Precautions (5.5)]
- Dry Eye with the Treatment of Glabellar Lines [see Warnings and Precautions (5.6)]
- Pre-existing Neuromuscular Disorders [ see Warnings and Precautions (5.7)]
- Human Albumin and Transmission of Viral Diseases [ see Warnings and Precautions (5.8)]
- Intradermal Immune Reaction
[
see Warnings and Precautions (5.9)]
- Pre-existing Conditions at the Injection Site [ See Warnings and Precautions (5.10) ]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Concomitant use of DYSPORT and aminoglycosides or other agents interfering with neuromuscular transmission or muscle relaxants, should be observed closely because the effect of DYSPORT may be potentiated (7.1, 7.4)
- Anticholinergic drugs may potentiate systemic anticholinergic effects (7.2)
- The effect of administering different botulinum neurotoxins during the course of treatment with DYSPORT is unknown (7.3)
14.2 Glabellar Lines
Three double-blind, randomized, placebo-controlled, clinical studies evaluated the efficacy of DYSPORT for use in the temporary improvement of the appearance of moderate to severe glabellar lines. These three studies enrolled healthy adults (ages 19-75) with glabellar lines of at least moderate severity at maximum frown. Subjects were excluded if they had marked ptosis, deep dermal scarring, or a substantial inability to lessen glabellar lines, even by physically spreading them apart. The subjects in these studies received either DYSPORT or placebo. The total dose was delivered in equally divided aliquots to specified injection sites (see Figure 1 ).
Investigators and subjects assessed efficacy at maximum frown by using a 4-point scale (none, mild, moderate, severe).
Overall treatment success was defined as post-treatment glabellar line severity of none or mild with at least 2 grade improvement from Baseline for the combined investigator and subject assessments (composite assessment) on Day 30 (see Table 16). Additional endpoints for each of the studies were post-treatment glabellar line severity of none or mild with at least a 1 grade improvement from Baseline for the separate investigator and subject assessments on Day 30.
After completion of the randomized studies, subjects were offered participation in a two-year, open-label re-treatment study to assess the safety of multiple treatments.
| 2 Grade Improvement | ||
| Study | DYSPORT n/N (%) | Placebo n/N (%) |
| GL-1 | 58/105 (55%) | 0/53 (0%) |
| GL-2 | 37/71 (52%) | 0/71 (0%) |
| GL-3 | 120/200 (60%) | 0/100 (0%) |
Treatment with DYSPORT reduced the severity of glabellar lines for up to four months.
12.2 Pharmacodynamics
The primary pharmacodynamic effect of DYSPORT is due to chemical denervation of the treated muscle resulting in a measurable decrease of the compound muscle action potential, causing a localized reduction of muscle activity.
12.3 Pharmacokinetics
Using currently available analytical technology, it is not possible to detect DYSPORT in the peripheral blood following intramuscular injection at the recommended doses.
7.4 muscle Relaxants (7.4 Muscle Relaxants)
Excessive weakness may also be exaggerated by administration of a muscle relaxant before or after administration of DYSPORT.
14.1 Cervical Dystonia
The efficacy of DYSPORT was evaluated in two randomized, double-blind, placebo-controlled, single-dose, parallel-group studies in treatment-naive cervical dystonia patients. The principal analyses from these trials provide the primary demonstration of efficacy involving 252 patients (121 on DYSPORT, 131 on placebo) with 36% male and 64% female. Ninety-nine percent of the patients were Caucasian.
In both placebo-controlled studies (Study 1 and Study 2), a dose of 500 Units of DYSPORT was given by intramuscular injection divided among two to four affected muscles. These studies were followed by long-term open-label extensions that allowed titration in 250 Unit steps to doses in a range of 250 to 1000 Units, after the initial dose of 500 Units. In the extension studies, re-treatment was determined by clinical need after a minimum of 12 weeks. The median time to re-treatment was 14 weeks and 18 weeks for the 75 thpercentile.
The primary assessment of efficacy was based on the total Toronto Western Spasmodic Torticollis Rating Scale (TWSTRS) change from baseline at Week 4 for both studies. The scale evaluates the severity of dystonia, patient-perceived disability from dystonia, and pain. The adjusted mean change from baseline in the TWSTRS total score was statistically significantly greater for the DYSPORT group than the placebo group at Weeks 4 in both studies (see Table 14) .
| Study 1 | Study 2 | |||
|
DYSPORT
500 Units N=55 |
Placebo
N=61 |
DYSPORT
500 Units N=37 |
Placebo
N=43 |
|
|
Baseline (week 0)
Mean (SD) |
43.8 (8.0) |
45.8 (8.9) |
45.1 (8.7) |
46.2 (9.4) |
|
Week 4
Mean (SD) Change from Baseline Change from baseline is expressed as adjusted least square mean (SE)
|
30.0 (12.7) -15.6 (2.0) |
40.2 (11.8) -6.7 (2.) |
35.2 (13.8) -9.6 (2.0) |
42.4 (12.2) -3.7 (1.8) |
| treatment difference
95% confidence interval |
-8.9
Significant at
p-value < 0.05
[-12.9 to -4.7] |
-5.9
[-10.6 to -1.3] |
||
|
Week 8
Mean (SD) Change from Baseline |
29.2 (11.0) -14.7 (2.0) |
39.6 (13.5) -5.9 (2.0) |
||
| treatment difference
95% confidence interval |
-8.8
[-12.9 to -4.7] |
Analyses by gender, weight, geographic region, underlying pain, cervical dystonia severity at baseline and history of treatment with botulinum toxin did not show any meaningful differences between groups.
Table 15 indicates the average DYSPORT dose, and percentage of total dose, injected into specific muscles in the pivotal clinical trials.
|
Number of patients
injected per muscle Total number of patients in combined studies 2 and 1 who received initial treatment = 121.
|
DYSPORT
Dose Injected |
Percentage of the total
DYSPORT Dose Injected |
|||
|
Median
[DYSPORT Units] (min, max) |
75th percentile
[DYSPORT Units] |
Median
[%] (min, max) |
75th percentile
[%] |
||
| Sternocleidomastoid | 90 | 125 Units
(50, 350) |
150 Units | 26.5%
(10, 70) |
30.0% |
| Splenius capitis | 85 | 200 Units
(75, 450) |
250 Units | 40.0%
(15, 90) |
50.0% |
| Trapezius | 50 | 102.6 Units
(50, 300) |
150 Units | 20.6%
(10, 60) |
30.0% |
| Levator scapulae | 35 | 105.3 Units
(50, 200) |
125 Units | 21.1%
(10, 40) |
25.0% |
| Scalenus (medius and anterior) | 26 | 115.5 Units
(50, 300) |
150 Units | 23.1%
(10,60) |
30.0% |
| Semispinalis capitis | 21 | 131.6 Units
(50, 250) |
175 Units | 29.4%
(10, 50) |
35.0% |
| Longissimus | 3 | 150 Units
(100, 200) |
200 Units | 30.0%
(20, 40) |
40.0% |
1 Indications and Usage (1 INDICATIONS AND USAGE)
DYSPORT is an acetylcholine release inhibitor and neuromuscular blocking agent indicated for:
12.1 Mechanism of Action
DYSPORT inhibits release of the neurotransmitter, acetylcholine, from peripheral cholinergic nerve endings. Toxin activity occurs in the following sequence: Toxin heavy chain mediated binding to specific surface receptors on nerve endings, internalization of the toxin by receptor mediated endocytosis, pH-induced translocation of the toxin light chain to the cell cytosol and cleavage of SNAP25 leading to intracellular blockage of neurotransmitter exocytosis into the neuromuscular junction. This accounts for the therapeutic utility of the toxin in diseases characterized by excessive efferent activity in motor nerves.
Recovery of transmission occurs gradually as the neuromuscular junction recovers from SNAP25 cleavage and as new nerve endings are formed.
7.2 Anticholinergic Drugs
Use of anticholinergic drugs after administration of DYSPORT may potentiate systemic anticholinergic effects such as blurred vision.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- The potency units of DYSPORT are not interchangeable with other preparations of botulinum toxin products ( 5.2)
- Immediate medical attention may be required in cases of respiratory, speech or swallowing difficulties ( 5.4)
- Recommended dose and frequency of administration should not be exceeded ( 5.5)
- Dry eye may occur with glabellar line treatment; if symptoms persist, consider referring patient to an ophthalmologist ( 5.6)
- Concomitant neuromuscular disorder may exacerbate clinical effects of treatment ( 5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Preparation of DYSPORT Solution for Administration ( 2.2)
- Once reconstituted, store in original container in a refrigerator 2°C to 8°C (36°F to 46°F) and use within 24 hours
- Do not freeze after reconstitution
- Reconstitution instructions are specific for the 300 Unit and 500 Unit vials
- Reconstituted DYSPORT is intended for intramuscular injection only. After reconstitution, DYSPORT should be used for only one injection session and for only one patient.
Cervical Dystonia ( 2.3)
• Initial dose is 500 Units given intramuscularly as a divided dose among the affected muscles
• Re-treatment every 12 to 16 weeks or longer, as necessary, based on return of clinical symptoms with doses administered between 250 and 1000 Units to optimize clinical benefit
• Re-treatment should not occur in intervals of less than 12 weeks
• Titrate in 250 Unit steps according to patient's response
Glabellar Lines ( 2.4)
- Administer a total dose of 50 Units, divided in five equal aliquots of 10 Units each, intramuscularly to affected muscles to achieve clinical effect
- Re-treatment should be administered no more frequently than every 3 months
Spasticity in Adults ( 2.5)
- Select dose based on muscles affected, severity of spasticity, and treatment and adverse reaction history botulinum toxins
- Dosing for upper limb spasticity: between 500 Units and 1000 Units
- Dosing for lower limb spasticity: up to 1500 Units
- The maximum recommended total dose per treatment session (upper and lower limb combined) in adults is 1500 Units
- Re-treatment, based on return of clinical symptoms, should not occur in intervals of less than 12 weeks
Spasticity in Pediatric Patients ( 2.6)
- Select dose based on the affected muscle, severity of spasticity, and treatment and adverse reaction history with all botulinum toxins.
- Recommended dosing for upper limb spasticity: 8 Units/kg to 16 Units/kg per limb. The maximum recommended total dose administered per treatment session must not exceed 16 Units/kg or 640 Units, whichever is lower.
- Recommended dosing for lower limb spasticity: 10 Units/kg to 15 Units/kg per limb. Total dose per treatment session must not exceed 15 Units/kg for unilateral lower limb injections, 30 Units/kg for bilateral injections, or 1000 Units, whichever is lower.
- The maximum recommended total dose per treatment session is 30 Units/kg or 1000 Units, whichever is lower. Re-treatment, based on return of clinical symptoms, should not occur in intervals of less than 3 months.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: 300 Units or 500 Units of lyophilized powder in a single-dose vial for reconstitution with preservative-free 0.9% Sodium Chloride Injection, USP.
6.3 Postmarketing Experience
Because adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post-approval use of DYSPORT: vertigo, photophobia, influenza-like illness, amyotrophy, muscle atrophy, burning sensation, facial paresis, hypoesthesia, erythema, dry eye, and excessive granulation tissue. Hypersensitivity reactions including anaphylaxis have been reported.
2.4 Dosing in Glabellar Lines
The dose of DYSPORT for the treatment of glabellar lines in adults is a total of 50 Units given intramuscularly in five equal aliquots of 10 Units each to achieve clinical effect (see Figure 1).
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Administer DYSPORT with care in elderly patients, reflecting the greater frequency of concomitant disease and other drug therapy ( 8.5)
2.1 Instructions for Safe Use
The potency units of DYSPORT are not interchangeable with other preparations of botulinum toxin products and, therefore, units of biological activity of DYSPORT cannot be compared to or converted into units of any other botulinum toxin products assessed with any other specific assay method [ see Warnings and Precautions (5.2) and Description (11) ]. Reconstituted DYSPORT is intended for intramuscular injection only.
2.3 Dosing in Cervical Dystonia
The recommended initial dose of DYSPORT for the treatment of cervical dystonia in adults is 500 Units given intramuscularly as a divided dose among affected muscles in patients with or without a history of prior treatment with botulinum toxin. (A description of the average DYSPORT dose and percentage of total dose injected into specific muscles in the pivotal clinical trials can be found in Table 15 of Section 14.1, Clinical Studies – Cervical Dystonia.) Limiting the dose injected into the sternocleidomastoid muscle may reduce the occurrence of dysphagia. Clinical studies with DYSPORT in cervical dystonia suggest that the peak effect occurs between two and four weeks after injection. Simultaneous guided injection of DYSPORT with EMG and/or ultrasound may be helpful in locating active muscles.
Dose Modification
Where dose modification is necessary for the treatment of cervical dystonia, uncontrolled open-label studies suggest that dose adjustment can be made in 250 Unit steps according to the individual patient's response, with re-treatment every 12 weeks or longer, as necessary, based on return of clinical symptoms. Uncontrolled open-label studies also suggest that the total dose administered in a single treatment should be between 250 Units and 1000 Units. Re-treatment, if needed, should not occur in intervals of less than 12 weeks. Doses above 1000 Units have not been systematically evaluated.
5.9 Intradermal Immune Reaction
The possibility of an immune reaction when injected intradermally is unknown. The safety of DYSPORT for the treatment of hyperhidrosis has not been established. DYSPORT is approved only for intramuscular injection.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Swallowing, Speaking, or Breathing Difficulties, or Other Unusual Symptoms
Advise patients to inform their doctor or pharmacist if they develop any unusual symptoms (including difficulty with swallowing, speaking or breathing), or if any known symptom persists or worsens
[see Warnings and Precautions (5.1, 5.4)].
Dry Eye with the Treatment of Glabellar Lines
Inform patients that DYSPORT injection may cause eye dryness [see Warnings and Precautions (5.6)]. Advise patients to report symptoms of eye dryness (e.g., eye pain, eye irritation, photosensitivity, or changes in vision) to their doctor.
Ability to Operate Machinery or Vehicles
Inform patients that if loss of strength, muscle weakness, blurred vision or drooping eyelids occur, they should avoid driving a car or engaging in other potentially hazardous activities.
Principal Display Panel 300 Unit (PRINCIPAL DISPLAY PANEL - 300 UNIT)
GALDERMA
Dysport
®
(AbobotulinumtoxinA)
for injection
For intramuscular use
300 UNITS/SINGLE-DOSE VIAL
1 VIAL
WARNING: Dosing units of botulinum toxins are not interchangeable between commercial products
NDC 0299-5962-30
Rx only
14.4 Spasticity in Pediatric Patients
Upper Limb Spasticity in Pediatric Patients
The efficacy of DYSPORT for the treatment of upper limb spasticity in pediatric patients 2 to 17 years of age was evaluated in a double-blind, low-dose controlled, multicenter study (NCT02106351). A total of 208 patients with spasticity because of cerebral palsy who were toxin naïve or non-naïve (66% had prior treatment with botulinum toxin), weighed at least 10 kgs, and had a baseline Modified Ashworth Score (MAS) of grade 2 or greater (99% patients) at the primary targeted muscle groups (PTMG), were enrolled in the modified Intention to Treat population (mITT). Patients received DYSPORT 16 Units/kg (n=70), DYSPORT 8 Units/kg (n=69), or DYSPORT 2 Units/kg (n=69) injected into the upper limb. The elbow flexors and wrist flexors respectively were the PTMG in 57% and in 43% of patients. The median age of the patients in this study was 9 years (range 2 to 17 years; 57% were between 2 and 9 years of age); 60% of patients were male, and 75% were White.
The primary efficacy endpoint was the mean change from baseline in MAS in the PTMG at Week 6 (see Table 24). The secondary efficacy endpoint was the mean Physician Global Assessment (PGA) score assessed at Week 6 (Table 25). Although PGA scores numerically favored DYSPORT treatment over the low-dose control, the difference was not statistically significant.
| Control Group | Treatment Groups | ||
|
|
DYSPORT
2 U/kg (N=69) |
DYSPORT
8 U/kg (N=69) |
DYSPORT
16 U/kg (N=70) |
| Baseline | |||
| Mean (SD) | 3.1 (0.3) | 3.1 (0.3) | 3.1 (0.5) |
| Week 6 | |||
| LS amean change from baseline in PTMG bon MAS | -1.6 | -2.0 | -2.3 |
| Difference from control in LS ameans | -0.4 | -0.7 | |
| p-value c | 0.0118 d | <0.0001 | |
| Week 16 | |||
| LS amean change from baseline in PGMG bon MAS | -0.9 | -1.2 | -1.5 |
| Difference from control in LS ameans | -0.3 d | -0.6 d |
aLS = Least Square
bPTMG=Primary Targeted Muscle Group
cp-value is derived from ANCOVA on ranked MAS score change from baseline with treatment, baseline score, age range at baseline, prior botulinum toxin treatment status at baseline, and center as explanatory variables
dNominal p-value <0.05
| Control Group | Treatment Group | ||
|
DYSPORT
2 U/kg (N=68) |
DYSPORT
8 U/kg (N=69) |
DYSPORT
16 U/kg (N=70) |
|
| Week 6 | |||
| Mean score (SD) | 1.7 (0.9) | 2.0 (0.9) | 2.0 (0.9) |
| LS amean in PGA | 1.8 | 2.0 | 2.0 |
| Difference from control in LS amean | 0.2 | 0.2 | |
| p-value b | 0.2043 | 0.1880 | |
| Week 16 | |||
| Mean score (SD) | 1.7 (1.0) | 1.6 (1.1) | 1.9 (1.2) |
| LS amean in PGA | 1.8 | 1.7 | 1.9 |
| Difference from contron in LS amean | -0.1 | 0.1 | |
| p-value b | 0.7001 | 0.4041 |
aLS=Least Square
bp-value is derived from ANOVA on ranked PGA score with treatment, age range at baseline, prior botulinum toxin treatment status at baseline, and center as explanatory variables
Lower Limb Spasticity in Pediatric Patients
The efficacy of DYSPORT for the treatment of lower limb spasticity in patients 2 to 17 years of age was evaluated in a double-blind, placebo-controlled, multicenter study. A total of 235 patients with cerebral palsy causing dynamic equinus foot deformity who were toxin-naïve or non-naïve and had a Modified Ashworth Score (MAS) of grade 2 or greater at the ankle plantar flexors were enrolled. Patients received DYSPORT 10 Units/kg/leg (n=79), DYSPORT 15 Units/kg/leg (n=79) or placebo (n=77) injected into the gastrocnemius and soleus muscles (see Table 27). Forty-one percent of patients (n=66) were treated bilaterally and received a total lower limb DYSPORT dose of either 20 Units/kg (n=37) or 30 Units/kg (n=29). The median age of the patients in this study was 5 years (range 2 to 17 years); 60% of patients were male, and 73% were Caucasian.
The primary efficacy endpoint was the mean change from baseline in MAS in ankle plantar flexor at Week 4; a co-primary endpoint was the mean Physician’s Global Assessment (PGA) score at Week 4 (see Table 26).
|
Placebo
(N=77) |
DYSPORT
10 Units/kg/leg (N=79) |
DYSPORT
15 Units/kg/leg (N=79) |
||
| LS Mean Change from
Baseline in Ankle plantar flexor Muscle Tone on the MAS |
Week 4 | -0.5 | -0.9* | -1.0* |
| Week 12 | -0.5 | -0.8* | -1.0* | |
| LS Mean PGA of
Response to Treatment |
Week 4 | 0.7 | 1.5* | 1.5* |
| Week 12 | 0.4 | 0.8* | 1.0* | |
|
LS=Least Square
|
Warning: Distant Spread of Toxin Effect (WARNING: DISTANT SPREAD OF TOXIN EFFECT)
Postmarketing reports indicate that the effects of DYSPORT and all botulinum toxin products may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These may include asthenia, generalized muscle weakness, diplopia, blurred vision, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence and breathing difficulties. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life threatening and there have been reports of death. The risk of symptoms is probably greatest in children treated for spasticity but symptoms can also occur in adults treated for spasticity and other conditions, particularly in those patients who have underlying conditions that would predispose them to these symptoms. In unapproved uses and in approved indications, cases of spread of effect have been reported at doses comparable to or lower than the maximum recommended total dose [ see Warnings and Precautions (5.1) ].
7.3 other Botulinum Neurotoxin Products (7.3 Other Botulinum Neurotoxin Products)
The effect of administering botulinum neurotoxin products including DYSPORT, at the same time or within several months of each other is unknown. Excessive weakness may be exacerbated by another administration of botulinum toxin prior to the resolution of the effects of a previously administered botulinum toxin.
2.6 Dosing in Spasticity in Pediatric Patients
DYSPORT dosing for spasticity in pediatric patients is based on Units per kilogram of body weight. To calculate the total units of DYSPORT required for treatment of one limb, select the dose of DYSPORT in Units/kg and the body weight (kg) of the patient (see Tables 5 and 6). Dosing in initial and sequential treatment sessions should be tailored to the individual patient based on the size, number and location of muscles involved, severity of spasticity, the presence of local muscle weakness, the patient’s response to previous treatment, and/or adverse reaction history with botulinum toxins.
No more than 0.5 mL should generally be administered at any single injection site. The maximum recommended total dose of DYSPORT in a single treatment session for spasticity in pediatric patients 2 years or older is 30 Units/kg or 1000 Units in a 3-month interval.
Although actual location of the injection sites can be determined by palpation, the use of injection guiding technique (e.g., electromyography or electrical stimulation, or ultrasound) is recommended to target the injection sites.
Upper Limb Spasticity in Pediatric Patients 2 Years of Age and Older
In the clinical trial that assessed the efficacy and safety of DYSPORT for treatment of upper limb spasticity in pediatric patients 2 years of age or older with a weight of at least 10 kg [
see Clinical Studies (14.4)], doses of 8 Units/kg or 16 Units/kg were divided among selected muscles of the target upper limb at a given treatment session (see Table 5 and Figure 4).
Table 5 describes the recommended Units/kg dose of DYSPORT per muscle. The maximum recommended total dose of DYSPORT administered for treatment of upper limb spasticity must not exceed 16 Units/kg or 640 Units, whichever is lower.
| Muscle |
Recommended Dose Range
per muscle per upper limb (units/kg Body Weight) |
Number of injection sites
per muscle |
| Brachialis | 3 Units/kg to 6 Units/kg | Up to 2 |
| Brachioradialis | 1.5 Units/kg to 3 Units/kg | 1 |
| Biceps brachii | 3 Units/kg to 6 Units/kg | Up to 2 |
| Pronator teres | 1 Units/kg to 2 Units/kg | 1 |
| Prontator quadratus | 0.5 Unit/kg to 1 Units/kg | 1 |
| Flexor carpi radialis (FCR) | 2 Units/kg to 4 Units/kg | Up to 2 |
| Flexor carpi ulnaris (FCU) | 1.5 Units/kg to 3 Units/kg | 1 |
| Flexor digitorum profundus (FDP) | 1 Units/kg to 2 Units/kg | 1 |
| Flexor digitorum superficialis (FDS) | 1.5 Units/kg to 3 Units/kg | Up to 4 |
| Total dose | 8 Units/kg to 16 Units/kg in upper limbs (and not exceeding 640 Units) |
Figure 4: Muscles for Injection for Upper Limb Spasticity in Pediatric Patients
Repeat DYSPORT treatment should be administered when the effect of a previous injection has diminished but no sooner than 16 weeks after the previous injection. A majority of patients in the clinical study were retreated between 16-28 weeks; however, some patients had a longer duration of response (i.e., 34 weeks or more). The degree and pattern of muscle spasticity at the time of re-injection may necessitate alterations in the dose of DYSPORT and muscles to be injected.
Lower Limb Spasticity in Pediatric Patients 2 Years of Age and Older
In the clinical trial that assessed the efficacy and safety of DYSPORT for treatment of lower limb spasticity in pediatric patients 2 years of age or older
[see Clinical Studies (14.4)], doses of 10 Units/kg to 15 Units/kg were divided among selected muscles of the target lower limb at a given treatment session (see Table 6 and Figure 5).
Table 6 describes the recommended Units/kg dose of DYSPORT per muscle of the Gastrocnemius-Soleus Complex (GSC). The recommended total DYSPORT dose per treatment session is 10 Units/kg to 15 Units/kg for unilateral lower limb injections or 20 Units/kg to 30 Units/kg for bilateral lower limb injections. However, the total dose of DYSPORT administered in a 3-month interval must not exceed 15 Units/kg for unilateral lower limb injections, 30 Units/kg for bilateral lower limb injections, or 1000 units, whichever is lower. The total dose administered should be divided between the affected spastic muscles of the lower limb(s). When possible, the dose should be distributed across more than 1 injection site in any single muscle (see Table 6).
| Muscle Injected |
Recommended DYSPORT Dose Range per
muscle per leg (Units/kg Body Weight) |
Recommended number of
injections per muscle |
| Gastrocnemius | 6 Units/kg to 9 Units/kg
the listed individual doses to be injected in the muscles can be used within the range mentioned without exceeding 15 Units/kg total dose for unilateral injection or 30 Units/kg for bilateral injections or 1000 Units whichever is lower.
|
Up to 4 |
| Soleus | 4 Units/kg to 6 Units/kg | Up to 2 |
| Total | 10 Units/kg to 15 Units/kg divided across both muscles | Up to 6 |
Figure 5: Muscles for Injection for Lower Limb Spasticity in Pediatric Patients
Although actual location of the injection sites can be determined by palpation, the use of injection guiding technique (e.g. electromyography or electrical stimulation or ultrasound) is recommended to target the injection sites.
Repeat DYSPORT treatment should be administered when the effect of a previous injection has diminished but no sooner than 12 weeks after the previous injection. A majority of patients in the clinical studies were retreated between 16-22 weeks, however; some had a longer duration of response. The degree and pattern of muscle spasticity and overall clinical benefit at the time of re-injection may necessitate alterations in the dose of DYSPORT and muscles to be injected.
The safety and effectiveness of DYSPORT injected into proximal muscles of the lower limb for the treatment of spasticity in pediatric patients has not been established.
5.10 Pre Existing Conditions At the Injection Site (5.10 Pre-existing Conditions at the Injection Site)
Caution should be exercised when DYSPORT is used where the targeted muscle shows excessive weakness or atrophy.
5.8 Human Albumin and Transmission of Viral Diseases
This product contains albumin, a derivative of human blood. Based on effective donor screening and product manufacturing processes, it carries an extremely remote risk for transmission of viral diseases and variant Creutzfeldt-Jakob disease (vCJD). There is a theoretical risk for transmission of Creutzfeldt-Jakob disease (CJD), but if that risk actually exists, the risk of transmission would also be considered extremely remote. No cases of transmission of viral diseases, CJD, or vCJD have ever been identified for licensed albumin or albumin contained in other licensed products.
7.1 Aminoglycosides and Other Agents Interfering With Neuromuscular Transmission (7.1 Aminoglycosides and Other Agents Interfering with Neuromuscular Transmission)
Co-administration of DYSPORT and aminoglycosides or other agents interfering with neuromuscular transmission (e.g., curare-like agents) should only be performed with caution because the effect of the botulinum toxin may be potentiated. If co-administered, observe the patient closely.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:30.013034 · Updated: 2026-03-14T22:15:52.273968