These Highlights Do Not Include All The Information Needed To Use Vyvgart Hytrulo Safely And Effectively. See Full Prescribing Information For Vyvgart Hytrulo.
7104d9e5-9910-47f5-9a93-741ac50f43b6
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.3 ) 10/2025 Dosage and Administration ( 2.2 , 2.5 ) 4/2025 Dosage and Administration ( 2.4 ) 2/2026
Indications and Usage
VYVGART HYTRULO is indicated for the treatment of adult patients with: generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody positive chronic inflammatory demyelinating polyneuropathy (CIDP)
Dosage and Administration
VYVGART HYTRULO is for subcutaneous use only. Do not administer intravenously. Do not dilute VYVGART HYTRULO.
Warnings and Precautions
Infections: Delay administration of VYVGART HYTRULO to patients with an active infection. Monitor for signs and symptoms of infection in patients treated with VYVGART HYTRULO. If serious infection occurs, administer appropriate treatment and consider withholding VYVGART HYTRULO until the infection has resolved. ( 5.1 ) Hypersensitivity Reactions: Anaphylaxis, hypotension leading to syncope, angioedema, dyspnea, rash, and urticaria have occurred in patients treated with VYVGART HYTRULO or intravenous efgartigimod alfa-fcab product. If a hypersensitivity reaction occurs, the healthcare professional should institute appropriate measures if needed or the patient should seek medical attention. ( 4 , 5.2 ) Infusion/injection-Related Reactions: If a severe infusion/injection-related reaction occurs, initiate appropriate therapy; consider the risks and benefits of readministering. If a mild to moderate infusion/injection-related reaction occurs, may rechallenge with close clinical observation, slower infusion/injection rates, and pre-medications. ( 5.3 )
Contraindications
VYVGART HYTRULO is contraindicated in patients with serious hypersensitivity to efgartigimod alfa products, to hyaluronidase, or to any of the excipients of VYVGART HYTRULO. Reactions have included anaphylaxis and hypotension leading to syncope [see Warnings and Precautions (5.2) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Infections [see Warnings and Precautions (5.1) ] Hypersensitivity Reactions [see Warnings and Precautions (5.2) ] Infusion/Injection-Related Reactions [see Warnings and Precautions (5.3) ]
Drug Interactions
Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing VYVGART HYTRULO and using alternative therapies. ( 7 )
Storage and Handling
VYVGART HYTRULO (efgartigimod alfa and hyaluronidase-qvfc) injection is a preservative free, sterile, yellowish, clear to opalescent solution supplied as a single-dose prefilled syringe or vial.
How Supplied
VYVGART HYTRULO (efgartigimod alfa and hyaluronidase-qvfc) injection is a preservative free, sterile, yellowish, clear to opalescent solution supplied as a single-dose prefilled syringe or vial.
Medication Information
Warnings and Precautions
Infections: Delay administration of VYVGART HYTRULO to patients with an active infection. Monitor for signs and symptoms of infection in patients treated with VYVGART HYTRULO. If serious infection occurs, administer appropriate treatment and consider withholding VYVGART HYTRULO until the infection has resolved. ( 5.1 ) Hypersensitivity Reactions: Anaphylaxis, hypotension leading to syncope, angioedema, dyspnea, rash, and urticaria have occurred in patients treated with VYVGART HYTRULO or intravenous efgartigimod alfa-fcab product. If a hypersensitivity reaction occurs, the healthcare professional should institute appropriate measures if needed or the patient should seek medical attention. ( 4 , 5.2 ) Infusion/injection-Related Reactions: If a severe infusion/injection-related reaction occurs, initiate appropriate therapy; consider the risks and benefits of readministering. If a mild to moderate infusion/injection-related reaction occurs, may rechallenge with close clinical observation, slower infusion/injection rates, and pre-medications. ( 5.3 )
Indications and Usage
VYVGART HYTRULO is indicated for the treatment of adult patients with: generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody positive chronic inflammatory demyelinating polyneuropathy (CIDP)
Dosage and Administration
VYVGART HYTRULO is for subcutaneous use only. Do not administer intravenously. Do not dilute VYVGART HYTRULO.
Contraindications
VYVGART HYTRULO is contraindicated in patients with serious hypersensitivity to efgartigimod alfa products, to hyaluronidase, or to any of the excipients of VYVGART HYTRULO. Reactions have included anaphylaxis and hypotension leading to syncope [see Warnings and Precautions (5.2) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Infections [see Warnings and Precautions (5.1) ] Hypersensitivity Reactions [see Warnings and Precautions (5.2) ] Infusion/Injection-Related Reactions [see Warnings and Precautions (5.3) ]
Drug Interactions
Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing VYVGART HYTRULO and using alternative therapies. ( 7 )
Storage and Handling
VYVGART HYTRULO (efgartigimod alfa and hyaluronidase-qvfc) injection is a preservative free, sterile, yellowish, clear to opalescent solution supplied as a single-dose prefilled syringe or vial.
How Supplied
VYVGART HYTRULO (efgartigimod alfa and hyaluronidase-qvfc) injection is a preservative free, sterile, yellowish, clear to opalescent solution supplied as a single-dose prefilled syringe or vial.
Description
Dosage and Administration ( 2.3 ) 10/2025 Dosage and Administration ( 2.2 , 2.5 ) 4/2025 Dosage and Administration ( 2.4 ) 2/2026
Section 42229-5
Single-Dose Prefilled Syringe
VYVGART HYTRULO prefilled syringe may be administered by patients and/or caregivers after proper instruction in subcutaneous injection technique [see Instructions for Use].
Section 42230-3
| PATIENT INFORMATION VYVGART HYTRULO® [viv' gart hye troo' loe] (efgartigimod alfa and hyaluronidase-qvfc) injection, for subcutaneous use |
||||
|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: 2/2026 | |||
|
What is VYVGART HYTRULO?
VYVGART HYTRULO is a prescription medicine used to treat adults with:
|
||||
Do not take VYVGART HYTRULO if you:
|
||||
Before taking VYVGART HYTRULO, tell your healthcare provider about all of your medical conditions, including if you:
|
||||
How should I take VYVGART HYTRULO?
|
||||
|
What are the possible side effects of VYVGART HYTRULO? VYVGART HYTRULO can cause side effects which can be serious, including:
|
||||
|
|
|
||
|
||||
|
|
|||
|
||||
|
|
|||
| The most common side effects of VYVGART HYTRULO include: | ||||
|
|
|
||
| These are not all the possible side effects of VYVGART HYTRULO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
|
How should I store VYVGART HYTRULO?
Refrigerated
|
||||
|
|
|||
|
||||
|
General information about the safe and effective use of VYVGART HYTRULO.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use VYVGART HYTRULO for a condition for which it was not prescribed. Do not give VYVGART HYTRULO to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about VYVGART HYTRULO that is written for health professionals. |
||||
|
What are the ingredients in VYVGART HYTRULO? Active ingredients: efgartigimod alfa and hyaluronidase (human recombinant) Inactive ingredients:
VYVGART HYTRULO is a registered trademark of argenx BV. © 2026 argenx BV |
Section 43683-2
Section 44425-7
Store VYVGART HYTRULO vials refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light until time of use. Do not freeze. Do not shake.
If needed, unopened vials may be stored in the original carton for up to 3 days at room temperature at 20°C to 25°C (68°F to 77°F) for a single period before administration or returned to refrigeration. Do not store the vial at room temperature more than one time. Record the date removed from and the date returned to the refrigerator on the carton.
11 Description
VYVGART HYTRULO is a fixed-combination drug product containing efgartigimod alfa and hyaluronidase (human recombinant).
Efgartigimod alfa, a neonatal Fc receptor blocker, is a human immunoglobulin G1 (IgG1) -derived Fc fragment (fragment, crystallized) of the za allotype, produced in Chinese hamster ovary (CHO) cells. The efgartigimod alfa Fc fragment is a homodimer consisting of two identical peptide chains each consisting of 227 amino acids linked together by two interchain disulfide bonds with affinity for FcRn. The molecular weight of efgartigimod alfa is approximately 54 kDa.
Hyaluronidase (human recombinant) is an endoglycosidase used to increase the dispersion and absorption of co-administered drugs when administered subcutaneously. Hyaluronidase (human recombinant) is a glycosylated single-chain protein produced by Chinese hamster ovary cells containing a DNA plasmid encoding for a soluble fragment of human hyaluronidase (PH20). Hyaluronidase (human recombinant) has a molecular weight of approximately 61 kDa.
VYVGART HYTRULO (efgartigimod alfa and hyaluronidase-qvfc) injection is a sterile, preservative free, yellowish, clear to opalescent solution supplied in a single-dose prefilled syringe or vial for subcutaneous injection.
Each 5 mL single-dose prefilled syringe contains 1,000 mg of efgartigimod alfa and 10,000 units of hyaluronidase (human recombinant). Each mL contains 200 mg of efgartigimod alfa, 2,000 units of hyaluronidase (human recombinant) and arginine hydrochloride (10.5 mg), histidine (1.4 mg), L-histidine hydrochloride monohydrate (2.2 mg), methionine (1.5 mg), polysorbate 80 (0.4 mg), sodium chloride (4.1 mg), sucrose (20.5 mg), and Water for Injection, USP, at a pH of 6.0.
Each 5.6 mL single-dose vial contains 1,008 mg of efgartigimod alfa and 11,200 units of hyaluronidase (human recombinant). Each mL contains 180 mg of efgartigimod alfa, 2,000 units of hyaluronidase (human recombinant) and
-
-histidine (1.4 mg), L- histidine hydrochloride monohydrate (2.2 mg), methionine (1.5 mg), polysorbate 20 (0.4 mg), sodium chloride (5.8 mg), sucrose (20.5 mg), and Water for Injection, USP, at a pH of 6.0 or
-
-arginine hydrochloride (10.5 mg), histidine (1.4 mg), L- histidine hydrochloride monohydrate (2.2 mg), methionine (1.5 mg), polysorbate 80 (0.4 mg), sodium chloride (4.1 mg), sucrose (20.5 mg), and Water for Injection, USP, at a pH of 6.0.
5.1 Infections
VYVGART HYTRULO may increase the risk of infection. The most common infections observed in Study 1 were urinary tract infection (10% of efgartigimod alfa-fcab-treated patients compared to 5% of placebo-treated patients) and respiratory tract infections (33% of efgartigimod alfa-fcab- treated patients compared to 29% of placebo-treated patients) [see Adverse Reactions (6.1) and Clinical Studies (14)]. A higher frequency of patients who received efgartigimod alfa-fcab compared to placebo were observed to have below normal levels for white blood cell counts (12% versus 5%, respectively), lymphocyte counts (28% versus 19%, respectively), and neutrophil counts (13% versus 6%, respectively). The majority of infections and hematologic abnormalities were mild to moderate in severity. Delay VYVGART HYTRULO administration in patients with an active infection until the infection is resolved. During treatment with VYVGART HYTRULO, monitor for clinical signs and symptoms of infections. If serious infection occurs, administer appropriate treatment and consider withholding VYVGART HYTRULO until the infection has resolved.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of VYVGART HYTRULO did not include sufficient numbers of patients aged 65 and older to determine whether they respond differently from younger adult patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of VYVGART HYTRULO or of other efgartigimod products.
In Study 2, in up to 10 weeks following the initiation of a treatment period with 4 weekly administrations, the incidence of anti-efgartigimod alfa antibodies was 35% (19/55) following treatment with VYVGART HYTRULO and 20% (11/55) in patients receiving intravenous efgartigimod alfa-fcab. For both IV and SC arms, neutralizing anti-efgartigimod alfa antibodies were detected in 4% (2/55) of patients.
In Study 3, in up to 12 weeks of treatment in stage A and 48 weeks in stage B, the incidence of anti-efgartigimod alfa antibodies was 6% (20/317) in stage A and 2% (2/111) in stage B, following treatment with VYVGART HYTRULO. Neutralizing anti-efgartigimod alfa antibodies were detected in 0.3% (1/317) of patients in stage A and in no patient in stage B.
Some neutralizing antibodies may not be detected by the assay. The available data are too limited to make definitive conclusions regarding immunogenicity and the effect on pharmacokinetics, safety, or efficacy of VYVGART HYTRULO.
4 Contraindications
VYVGART HYTRULO is contraindicated in patients with serious hypersensitivity to efgartigimod alfa products, to hyaluronidase, or to any of the excipients of VYVGART HYTRULO. Reactions have included anaphylaxis and hypotension leading to syncope [see Warnings and Precautions (5.2)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Infections [see Warnings and Precautions (5.1)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
- Infusion/Injection-Related Reactions [see Warnings and Precautions (5.3)]
7 Drug Interactions
Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing VYVGART HYTRULO and using alternative therapies. (7)
8.6 Renal Impairment
No dose adjustment of VYVGART HYTRULO is needed for patients with mild renal impairment. There are insufficient data to evaluate the impact of moderate renal impairment (eGFR 30-59 mL/min/1.73 m2) and severe renal impairment (eGFR <30 mL/min/1.73 m2) on pharmacokinetic parameters of VYVGART HYTRULO [see Clinical Pharmacology (12.3)].
Instructions for Use
VYVGART HYTRULO [viv' gart hye troo' loe]
(efgartigimod alfa and hyaluronidase-qvfc)
injection, for subcutaneous use
1,000 mg and 10,000 units/5 mL
This Instructions for Use contains information on how to inject VYVGART HYTRULO.
Important Information You Need to Know Before Injecting VYVGART HYTRULO
- VYVGART HYTRULO is for under the skin (subcutaneous) injection only.
- Be sure to read and understand this Instructions for Use before injecting VYVGART HYTRULO. Your healthcare provider should show you or your caregiver how to prepare and inject VYVGART HYTRULO the correct way before using it for the first time. Ask your healthcare provider if you have any questions.
- The prefilled syringe is for one-time-use only and cannot be reused.
- Do not use VYVGART HYTRULO if it has been at room temperature for longer than 30 days.
- Do not use the prefilled syringe if it is expired.
- Do not use the prefilled syringe if it is cracked, broken, or damaged. Return damaged prefilled syringes to the specialty pharmacy.
- Do not use a prefilled syringe if the liquid medicine is discolored or contains particles. The liquid medicine should look clear to yellowish in color. A little cloudiness is normal.
- Do not shake the prefilled syringe.
- You will need supplies that are not provided with VYVGART HYTRULO (see Figure J).
Storing VYVGART HYTRULO Prefilled Syringe
|
||
|
Do not freeze the prefilled syringes or store them in direct sunlight. Do not use a prefilled syringe that has been frozen or left in direct sunlight. |
Prefilled Syringe Parts
| Safety Needle (not provided with VYVGART HYTRULO prefilled syringe) that is 25G, 5/8 inch length, thin wall |
Gather and Check the Prefilled Syringe
| 1 Remove the carton from the refrigerator | |
| 1.1 Remove the carton containing the prefilled syringe from the refrigerator (see Figure C). | |
| 1.2 Remove 1 prefilled syringe from the carton (see Figure D) and place any remaining prefilled syringes back into the refrigerator for later use. | |
| 1.3 Remove the prefilled syringe from the tray (see Figure E). | |
| 2 Check the prefilled syringe before use | |
| 2.1 Check the expiration date on the prefilled syringe label (see Figure F). | |
| Do not use the prefilled syringe if the expiration date has passed. | |
| 2.2 Check the condition of the prefilled syringe and the prefilled syringe cap (see Figure G). | |
| Do not use the prefilled syringe if it is cracked, broken, damaged, or if the prefilled syringe cap is missing. | |
|
2.3 Check the appearance of the liquid medicine in the prefilled syringe (see Figure H). The liquid medicine should look clear to yellowish in color. A little cloudiness is normal. |
|
| Do not use the prefilled syringe if the liquid medicine is discolored or contains particles. |
Prepare for the Injection
| 3 Allow the prefilled syringe to warm to room temperature | ||
| 3.1 Place the prefilled syringe on a clean flat surface and let it sit for at least 30 minutes, to allow it to warm to room temperature (see Figure I). | ||
|
Do not attempt to warm the prefilled syringe in any other way. Do not use the VYVGART HYTRULO prefilled syringe if it has been at room temperature for longer than 30 days. |
||
| 4 Gather supplies and wash your hands | ||
| 4.1 Gather the following supplies that are not provided with the prefilled syringe (see Figure J). | ||
| 4.2 Wash your hands with soap and water (see Figure K). | ||
| 5 Snap off the prefilled syringe cap and attach the safety needle | ||
|
5.1 Carefully open the safety needle package and remove the needle (see Figure L). Throw away the packaging into household trash. |
||
|
5.2 Bend the prefilled syringe cap to one side to snap it off and remove it from the prefilled syringe (see Figure M). Throw away the prefilled syringe cap into the household trash. |
||
| Do not touch the tip of the prefilled syringe after the prefilled syringe cap has been removed. | ||
| 5.3 Hold the prefilled syringe by the syringe body in one hand, and attach the safety needle to the prefilled syringe by twisting it to the right (clockwise) until you feel resistance (see Figure N). | ||
| The safety needle is now attached to the prefilled syringe (see Figure O). | ||
| 6 Choose and clean the injection site on the stomach (abdomen) | ||
|
6.1 Choose an injection site on the stomach (abdomen) at least 2 inches away from the belly button (navel) (see Figure P). Change (rotate) the injection site the next time you inject VYVGART HYTRULO. |
||
|
Do not inject into skin that is irritated, red, bruised, infected, or tender. Do not inject into skin that is hard, scarred, or has moles. Do not inject into a vein. VYVGART HYTRULO is for under the skin (subcutaneous) injection only. |
||
| 6.2 Clean the chosen injection site with an alcohol swab and let it air dry (see Figure Q). | ||
| Do not blow on or touch the injection site after it has been cleaned. |
Inject VYVGART HYTRULO
| 7 Pull back the needle shield and remove the clear needle cap | ||
|
7.1 Pull the needle shield back (see Figure R). Note: The needle shield will be used after the injection to cover the needle and protect from needle-stick injuries. |
||
|
7.2 Hold the prefilled syringe body and remove the clear needle cap by pulling it straight off of the needle (see Figure S). Throw away the clear needle cap into the household trash. |
||
| Do not recap the needle. | ||
| 8 Give the injection | ||
| 8.1 Pinch the cleaned injection site (Figure T). | ||
| While pinching the skin, insert the needle at a 45 to 90 degree angle all the way into the pinched skin (see Figure U). Then release the pinched skin. |
||
| Do not pinch the skin too tightly as this can cause bruising. | ||
|
8.2 Slowly press the thumb pad down all the way until it stops to inject the liquid medicine (see Figure V). It will take about 20 to 30 seconds to inject all of the liquid medicine. You will feel resistance as you press down. Inject more slowly in case there is discomfort. It is ok if you need to pause or change your grip during the injection. |
||
| Do not try to force the thumb pad down quickly as this will make the thumb pad harder to press. | ||
| 8.3 After all the liquid medicine is injected, remove the needle from the skin by pulling it straight out without changing the angle that it was inserted (see Figure W). |
Throw away (dispose of) the Used Prefilled Syringe
| 9 Cover the needle and throw away the used prefilled syringe | ||
|
9.1 Carefully push the needle shield over the needle until it snaps into place and covers the needle (see Figure X). This helps to prevent needle-stick injuries. |
||
| Do not recap the needle using the clear needle cap. Only use the needle shield to cover the needle. | ||
| 9.2 Throw away the used prefilled syringe, with the needle still attached, into an FDA-cleared sharps disposal container right away after use (see Figure Y). | ||
| Do not throw away (dispose of) loose needles and syringes in your household trash. | ||
| 10 Treat the injection site | ||
|
10.1 If there is a small amount of blood or liquid at the injection site, press a sterile gauze over the injection site until the bleeding stops (see Figure Z). If needed, you may apply a small adhesive bandage. |
Additional Disposal Information
If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal. Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container. Always keep the sharps disposal container out of the reach of children. |
Additional Information
| This Instructions for Use has been approved by the U.S. Food and Drug Administration | Issued: 4/2025 |
| For more information, go to www.vyvgart.com or call 1-833-898-4278. Manufactured by: argenx BV Industriepark 7 9052 Zwijnaarde, Belgium U.S. License No. 2217 Halozyme, Inc. 12390 El Camino Real San Diego, CA 92130 U.S. License No. 2187 Distributed by: argenx US, Inc. 33 Arch Street Boston, MA 02110 |
12.2 Pharmacodynamics
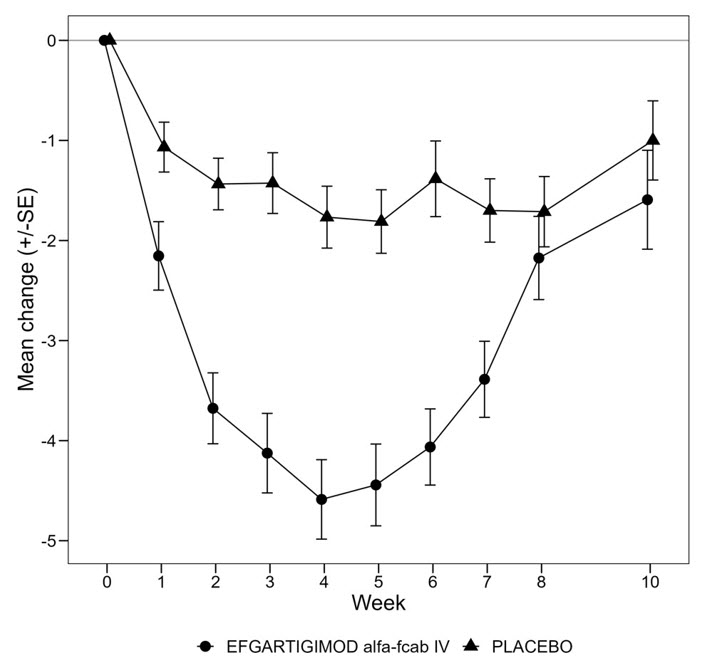
In Study 1 [see Clinical Studies (14)], the pharmacological effect of efgartigimod alfa-fcab was assessed by measuring the decrease in serum IgG levels and AChR autoantibody levels. In patients testing positive for AChR antibodies and who were treated with efgartigimod alfa-fcab intravenous, there was a reduction in total IgG levels relative to baseline. Decrease in AChR autoantibody levels followed a similar pattern. A decrease in AChR-Ab was associated with a clinical response in AChR-Ab positive patients, as measured by the change from baseline in MG- ADL total score.
In Study 2, the pharmacological effect of VYVGART HYTRULO administered subcutaneously (SC) at 1,008 mg / 11,200 Units was compared to efgartigimod alfa-fcab administered intravenously at 10 mg/kg (EFG IV) in gMG patients. The maximum mean reduction in AChR-Ab level was observed at week 4, with a mean reduction of 62.2% and 59.7% in the VYVGART HYTRULO SC and efgartigimod alfa-fcab IV arm, respectively. The decrease in total IgG levels followed a similar pattern. The 90% confidence intervals for the geometric mean ratios of AChR-Ab reduction at day 29 and AUEC0-4w (area under the effect-time curve from time 0 to 4 weeks post dose) were within the range of 80% to 125%, indicating no clinically significant difference between the two formulations.
12.3 Pharmacokinetics
Efgartigimod alfa exposures were approximately dose-proportional up to the highest subcutaneously tested dose of VYVGART HYTRULO (1750 mg, 1.75 times the recommended dosage).
1 Indications and Usage
VYVGART HYTRULO is indicated for the treatment of adult patients with:
- generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody positive
- chronic inflammatory demyelinating polyneuropathy (CIDP)
12.1 Mechanism of Action
VYVGART HYTRULO is a coformulation of efgartigimod alfa and hyaluronidase.
Efgartigimod alfa is a human IgG1 antibody fragment that binds to the neonatal Fc receptor (FcRn), resulting in the reduction of circulating IgG.
Hyaluronidase increases permeability of the subcutaneous tissue by depolymerizing hyaluronan. This effect is transient, and permeability of the subcutaneous tissue is restored within 24 to 48 hours.
5 Warnings and Precautions
- Infections: Delay administration of VYVGART HYTRULO to patients with an active infection. Monitor for signs and symptoms of infection in patients treated with VYVGART HYTRULO. If serious infection occurs, administer appropriate treatment and consider withholding VYVGART HYTRULO until the infection has resolved. (5.1)
- Hypersensitivity Reactions: Anaphylaxis, hypotension leading to syncope, angioedema, dyspnea, rash, and urticaria have occurred in patients treated with VYVGART HYTRULO or intravenous efgartigimod alfa-fcab product. If a hypersensitivity reaction occurs, the healthcare professional should institute appropriate measures if needed or the patient should seek medical attention. (4, 5.2)
- Infusion/injection-Related Reactions: If a severe infusion/injection-related reaction occurs, initiate appropriate therapy; consider the risks and benefits of readministering. If a mild to moderate infusion/injection-related reaction occurs, may rechallenge with close clinical observation, slower infusion/injection rates, and pre-medications. (5.3)
2 Dosage and Administration
- See Full Prescribing Information for instructions on dosage, preparation, and administration. (2.1, 2.2, 2.3, 2.4, 2.5)
- Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of a new treatment cycle with VYVGART HYTRULO. (2.1)
- Important Administration Information
- gMG: recommended dose and dose schedule
- Administer in cycles of once weekly injections for 4 weeks. (2.3)
- Prefilled syringe: 1,000 mg efgartigimod alfa and 10,000 units hyaluronidase administered over 20 to 30 seconds. (2.3)
- Vial: 1,008 mg efgartigimod alfa and 11,200 units hyaluronidase over 30 to 90 seconds. (2.3)
- Administer subsequent treatment cycles based on clinical evaluation. (2.3)
- CIDP: recommended dose and dose schedule
2.1 Recommended Vaccination
Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of a new treatment cycle with VYVGART HYTRULO. Because VYVGART HYTRULO causes transient reduction in IgG levels, vaccination with live vaccines is not recommended during treatment with VYVGART HYTRULO [see Dosage and Administration (2.3) and Warnings and Precautions (5.1)].
3 Dosage Forms and Strengths
Injection: 1,000 mg efgartigimod alfa and 10,000 units hyaluronidase per 5 mL (200 mg/2,000 units per mL) as yellowish, clear to opalescent solution, in a single-dose prefilled syringe.
Injection: 1,008 mg efgartigimod alfa and 11,200 units hyaluronidase per 5.6 mL (180 mg/2,000 units per mL) as yellowish, clear to opalescent solution, in a single-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of efgartigimod alfa products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Hypersensitivity reactions including anaphylaxis and hypotension, and infusion/injection-related reactions [see Warnings and Precautions (5.2, 5.3)].
Neurological: There have been reports of worsening of symptoms and signs of CIDP when transitioning from intravenous immunoglobulin treatments to VYVGART HYTRULO [see Drug Interactions (7.1)].
5.2 Hypersensitivity Reactions
In clinical trials, hypersensitivity reactions, including rash, angioedema, and dyspnea were observed in patients treated with VYVGART HYTRULO or intravenous efgartigimod alfa-fcab. Urticaria was also observed in patients treated with VYVGART HYTRULO. Hypersensitivity reactions were mild or moderate, occurred within one hour to three weeks of administration.
Anaphylaxis and hypotension leading to syncope have been reported in postmarketing experience with intravenous efgartigimod alfa-fcab. Anaphylaxis and hypotension occurred during or within an hour of administration and led to infusion discontinuation and in some cases to permanent treatment discontinuation.
Monitor for clinical signs and symptoms of hypersensitivity reactions for at least 30 minutes after administration [see Dosage and Administration (2.4)]. If a hypersensitivity reaction occurs, the healthcare professional should institute appropriate measures if needed or the patient should seek medical attention. VYVGART HYTRULO is contraindicated in patients with a history of serious hypersensitivity to efgartigimod alfa products, to hyaluronidase, or to any of the excipients of VYVGART HYTRULO [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient and/or caregiver to read FDA-approved patient labeling (Patient Information and Instructions for Use).
14.1 Generalized Myasthenia Gravis
Study 1 (described below) which established the effectiveness of efgartigimod alfa-fcab for the treatment of generalized myasthenia gravis (gMG) in adults who are AChR antibody positive was conducted with efgartigimod alfa-fcab intravenous formulation. In Study 2, VYVGART HYTRULO demonstrated a comparable pharmacodynamic effect on AChR antibody reduction as compared to the efgartigimod alfa-fcab intravenous formulation, which established the efficacy of VYVGART HYTRULO [see Clinical Pharmacology (12.2)].
16 How Supplied/storage and Handling
VYVGART HYTRULO (efgartigimod alfa and hyaluronidase-qvfc) injection is a preservative free, sterile, yellowish, clear to opalescent solution supplied as a single-dose prefilled syringe or vial.
5.3 Infusion/injection Related Reactions
Infusion-related reactions have been reported with intravenous efgartigimod alfa-fcab in postmarketing experience. The most frequent symptoms and signs were hypertension, chills, shivering, and thoracic, abdominal, and back pain. Infusion-related reactions occurred during or within an hour of administration and led to infusion discontinuation. If a severe infusion/injection-related reaction occurs, initiate appropriate therapy. Consider the risks and benefits of readministering VYVGART HYTRULO following a severe infusion/injection-related reaction. If a mild to moderate infusion/injection-related reaction occurs, patients may be rechallenged with close clinical observation, slower infusion/injection rates, and pre-medications.
7.1 Effect of Vyvgart Hytrulo On Other Drugs
Concomitant use of VYVGART HYTRULO with medications that bind to the human neonatal Fc receptor (FcRn) (e.g., immunoglobulin products, monoclonal antibodies, or antibody derivates containing the human Fc domain of the IgG subclass) may lower systemic exposures and reduce effectiveness of such medications. Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing VYVGART HYTRULO and using alternative therapies.
Principal Display Panel 5.6 Ml Vial Carton
NDC 73475-3102-3
VYVGART® Hytrulo
(efgartigimod alfa and
hyaluronidase-qvfc)
INJECTION
1,008 mg and 11,200 units/5.6 mL
(180 mg and 2,000 units/mL)
For subcutaneous injection
over 30 to 90 seconds
One single-dose vial
Discard unused portion
Must be administered by
a healthcare provider
Rx only
Principal Display Panel 5 Ml Syringe Carton
NDC 73475-1221-1
VYVGART Hytrulo®
(efgartigimod alfa and hyaluronidase-qvfc)
INJECTION
1,000 mg and 10,000 units/5 mL
(200 mg and 2,000 units/mL)
For subcutaneous injection over 20 to 30 seconds
One single-dose prefilled syringe
Rx only
2.2 Important Dosage and Administration Instructions
VYVGART HYTRULO is for subcutaneous use only. Do not administer intravenously. Do not dilute VYVGART HYTRULO.
14.2 Chronic Inflammatory Demyelinating Polyneuropathy
The efficacy of VYVGART HYTRULO for the treatment of adults with chronic inflammatory demyelinating polyneuropathy (CIDP) was established in a two stage, multicenter study (Study 3; NCT04281472). Study 3 included a run-in period of up to 12 weeks after withdrawal of existing treatment for CIDP in order to identify patients with active disease, followed by an open-label period to identify VYVGART HYTRULO responders (stage A) who then entered a randomized, double-blind, placebo-controlled, withdrawal period (stage B).
Study 3 enrolled male and female patients age 18 years and older, who at the time of screening, had a documented diagnosis of definite or probable CIDP using the European Federation of Neurological Societies/Peripheral Nerve Society (EFNS/PNS; 2010) criteria for progressing or relapsing forms.
The Inflammatory Neuropathy Cause and Treatment disability score (INCAT) is a scale used to assess the impact of CIDP on daily upper and lower limb function, and is composed of the arm score and leg score (0 to 5 points for each). A total score on the INCAT ranges from 0 to 10 points with a higher number representing more disability. The adjusted INCAT (aINCAT) disability score, identical to the INCAT disability score but with changes in the upper limb function from 0 (normal) to 1 (minor symptoms) excluded, was used to assess efficacy for VYVGART HYTRULO for the treatment of CIDP.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
VYVGART HYTRULO for subcutaneous injection contains efgartigimod alfa and hyaluronidase [see Description (11)].
Principal Display Panel 1,008 Mg and 11,2000 Units/5.6 Ml Vial Carton
NDC 73475-3102-3
VYVGART® Hytrulo
(efgartigimod alfa and
hyaluronidase-qvfc)
INJECTION
1,008 mg and 11,200 units/5.6 mL
(180 mg and 2,000 units/mL)
For subcutaneous injection
over 30 to 90 seconds
One single-dose vial
Discard unused portion
Must be administered by
a healthcare provider
Rx only
Structured Label Content
Section 42229-5 (42229-5)
Single-Dose Prefilled Syringe
VYVGART HYTRULO prefilled syringe may be administered by patients and/or caregivers after proper instruction in subcutaneous injection technique [see Instructions for Use].
Section 42230-3 (42230-3)
| PATIENT INFORMATION VYVGART HYTRULO® [viv' gart hye troo' loe] (efgartigimod alfa and hyaluronidase-qvfc) injection, for subcutaneous use |
||||
|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: 2/2026 | |||
|
What is VYVGART HYTRULO?
VYVGART HYTRULO is a prescription medicine used to treat adults with:
|
||||
Do not take VYVGART HYTRULO if you:
|
||||
Before taking VYVGART HYTRULO, tell your healthcare provider about all of your medical conditions, including if you:
|
||||
How should I take VYVGART HYTRULO?
|
||||
|
What are the possible side effects of VYVGART HYTRULO? VYVGART HYTRULO can cause side effects which can be serious, including:
|
||||
|
|
|
||
|
||||
|
|
|||
|
||||
|
|
|||
| The most common side effects of VYVGART HYTRULO include: | ||||
|
|
|
||
| These are not all the possible side effects of VYVGART HYTRULO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
|
How should I store VYVGART HYTRULO?
Refrigerated
|
||||
|
|
|||
|
||||
|
General information about the safe and effective use of VYVGART HYTRULO.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use VYVGART HYTRULO for a condition for which it was not prescribed. Do not give VYVGART HYTRULO to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about VYVGART HYTRULO that is written for health professionals. |
||||
|
What are the ingredients in VYVGART HYTRULO? Active ingredients: efgartigimod alfa and hyaluronidase (human recombinant) Inactive ingredients:
VYVGART HYTRULO is a registered trademark of argenx BV. © 2026 argenx BV |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store VYVGART HYTRULO vials refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light until time of use. Do not freeze. Do not shake.
If needed, unopened vials may be stored in the original carton for up to 3 days at room temperature at 20°C to 25°C (68°F to 77°F) for a single period before administration or returned to refrigeration. Do not store the vial at room temperature more than one time. Record the date removed from and the date returned to the refrigerator on the carton.
11 Description (11 DESCRIPTION)
VYVGART HYTRULO is a fixed-combination drug product containing efgartigimod alfa and hyaluronidase (human recombinant).
Efgartigimod alfa, a neonatal Fc receptor blocker, is a human immunoglobulin G1 (IgG1) -derived Fc fragment (fragment, crystallized) of the za allotype, produced in Chinese hamster ovary (CHO) cells. The efgartigimod alfa Fc fragment is a homodimer consisting of two identical peptide chains each consisting of 227 amino acids linked together by two interchain disulfide bonds with affinity for FcRn. The molecular weight of efgartigimod alfa is approximately 54 kDa.
Hyaluronidase (human recombinant) is an endoglycosidase used to increase the dispersion and absorption of co-administered drugs when administered subcutaneously. Hyaluronidase (human recombinant) is a glycosylated single-chain protein produced by Chinese hamster ovary cells containing a DNA plasmid encoding for a soluble fragment of human hyaluronidase (PH20). Hyaluronidase (human recombinant) has a molecular weight of approximately 61 kDa.
VYVGART HYTRULO (efgartigimod alfa and hyaluronidase-qvfc) injection is a sterile, preservative free, yellowish, clear to opalescent solution supplied in a single-dose prefilled syringe or vial for subcutaneous injection.
Each 5 mL single-dose prefilled syringe contains 1,000 mg of efgartigimod alfa and 10,000 units of hyaluronidase (human recombinant). Each mL contains 200 mg of efgartigimod alfa, 2,000 units of hyaluronidase (human recombinant) and arginine hydrochloride (10.5 mg), histidine (1.4 mg), L-histidine hydrochloride monohydrate (2.2 mg), methionine (1.5 mg), polysorbate 80 (0.4 mg), sodium chloride (4.1 mg), sucrose (20.5 mg), and Water for Injection, USP, at a pH of 6.0.
Each 5.6 mL single-dose vial contains 1,008 mg of efgartigimod alfa and 11,200 units of hyaluronidase (human recombinant). Each mL contains 180 mg of efgartigimod alfa, 2,000 units of hyaluronidase (human recombinant) and
-
-histidine (1.4 mg), L- histidine hydrochloride monohydrate (2.2 mg), methionine (1.5 mg), polysorbate 20 (0.4 mg), sodium chloride (5.8 mg), sucrose (20.5 mg), and Water for Injection, USP, at a pH of 6.0 or
-
-arginine hydrochloride (10.5 mg), histidine (1.4 mg), L- histidine hydrochloride monohydrate (2.2 mg), methionine (1.5 mg), polysorbate 80 (0.4 mg), sodium chloride (4.1 mg), sucrose (20.5 mg), and Water for Injection, USP, at a pH of 6.0.
5.1 Infections
VYVGART HYTRULO may increase the risk of infection. The most common infections observed in Study 1 were urinary tract infection (10% of efgartigimod alfa-fcab-treated patients compared to 5% of placebo-treated patients) and respiratory tract infections (33% of efgartigimod alfa-fcab- treated patients compared to 29% of placebo-treated patients) [see Adverse Reactions (6.1) and Clinical Studies (14)]. A higher frequency of patients who received efgartigimod alfa-fcab compared to placebo were observed to have below normal levels for white blood cell counts (12% versus 5%, respectively), lymphocyte counts (28% versus 19%, respectively), and neutrophil counts (13% versus 6%, respectively). The majority of infections and hematologic abnormalities were mild to moderate in severity. Delay VYVGART HYTRULO administration in patients with an active infection until the infection is resolved. During treatment with VYVGART HYTRULO, monitor for clinical signs and symptoms of infections. If serious infection occurs, administer appropriate treatment and consider withholding VYVGART HYTRULO until the infection has resolved.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of VYVGART HYTRULO did not include sufficient numbers of patients aged 65 and older to determine whether they respond differently from younger adult patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of VYVGART HYTRULO or of other efgartigimod products.
In Study 2, in up to 10 weeks following the initiation of a treatment period with 4 weekly administrations, the incidence of anti-efgartigimod alfa antibodies was 35% (19/55) following treatment with VYVGART HYTRULO and 20% (11/55) in patients receiving intravenous efgartigimod alfa-fcab. For both IV and SC arms, neutralizing anti-efgartigimod alfa antibodies were detected in 4% (2/55) of patients.
In Study 3, in up to 12 weeks of treatment in stage A and 48 weeks in stage B, the incidence of anti-efgartigimod alfa antibodies was 6% (20/317) in stage A and 2% (2/111) in stage B, following treatment with VYVGART HYTRULO. Neutralizing anti-efgartigimod alfa antibodies were detected in 0.3% (1/317) of patients in stage A and in no patient in stage B.
Some neutralizing antibodies may not be detected by the assay. The available data are too limited to make definitive conclusions regarding immunogenicity and the effect on pharmacokinetics, safety, or efficacy of VYVGART HYTRULO.
4 Contraindications (4 CONTRAINDICATIONS)
VYVGART HYTRULO is contraindicated in patients with serious hypersensitivity to efgartigimod alfa products, to hyaluronidase, or to any of the excipients of VYVGART HYTRULO. Reactions have included anaphylaxis and hypotension leading to syncope [see Warnings and Precautions (5.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Infections [see Warnings and Precautions (5.1)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
- Infusion/Injection-Related Reactions [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing VYVGART HYTRULO and using alternative therapies. (7)
8.6 Renal Impairment
No dose adjustment of VYVGART HYTRULO is needed for patients with mild renal impairment. There are insufficient data to evaluate the impact of moderate renal impairment (eGFR 30-59 mL/min/1.73 m2) and severe renal impairment (eGFR <30 mL/min/1.73 m2) on pharmacokinetic parameters of VYVGART HYTRULO [see Clinical Pharmacology (12.3)].
Instructions for Use (INSTRUCTIONS FOR USE)
VYVGART HYTRULO [viv' gart hye troo' loe]
(efgartigimod alfa and hyaluronidase-qvfc)
injection, for subcutaneous use
1,000 mg and 10,000 units/5 mL
This Instructions for Use contains information on how to inject VYVGART HYTRULO.
Important Information You Need to Know Before Injecting VYVGART HYTRULO
- VYVGART HYTRULO is for under the skin (subcutaneous) injection only.
- Be sure to read and understand this Instructions for Use before injecting VYVGART HYTRULO. Your healthcare provider should show you or your caregiver how to prepare and inject VYVGART HYTRULO the correct way before using it for the first time. Ask your healthcare provider if you have any questions.
- The prefilled syringe is for one-time-use only and cannot be reused.
- Do not use VYVGART HYTRULO if it has been at room temperature for longer than 30 days.
- Do not use the prefilled syringe if it is expired.
- Do not use the prefilled syringe if it is cracked, broken, or damaged. Return damaged prefilled syringes to the specialty pharmacy.
- Do not use a prefilled syringe if the liquid medicine is discolored or contains particles. The liquid medicine should look clear to yellowish in color. A little cloudiness is normal.
- Do not shake the prefilled syringe.
- You will need supplies that are not provided with VYVGART HYTRULO (see Figure J).
Storing VYVGART HYTRULO Prefilled Syringe
|
||
|
Do not freeze the prefilled syringes or store them in direct sunlight. Do not use a prefilled syringe that has been frozen or left in direct sunlight. |
Prefilled Syringe Parts
| Safety Needle (not provided with VYVGART HYTRULO prefilled syringe) that is 25G, 5/8 inch length, thin wall |
Gather and Check the Prefilled Syringe
| 1 Remove the carton from the refrigerator | |
| 1.1 Remove the carton containing the prefilled syringe from the refrigerator (see Figure C). | |
| 1.2 Remove 1 prefilled syringe from the carton (see Figure D) and place any remaining prefilled syringes back into the refrigerator for later use. | |
| 1.3 Remove the prefilled syringe from the tray (see Figure E). | |
| 2 Check the prefilled syringe before use | |
| 2.1 Check the expiration date on the prefilled syringe label (see Figure F). | |
| Do not use the prefilled syringe if the expiration date has passed. | |
| 2.2 Check the condition of the prefilled syringe and the prefilled syringe cap (see Figure G). | |
| Do not use the prefilled syringe if it is cracked, broken, damaged, or if the prefilled syringe cap is missing. | |
|
2.3 Check the appearance of the liquid medicine in the prefilled syringe (see Figure H). The liquid medicine should look clear to yellowish in color. A little cloudiness is normal. |
|
| Do not use the prefilled syringe if the liquid medicine is discolored or contains particles. |
Prepare for the Injection
| 3 Allow the prefilled syringe to warm to room temperature | ||
| 3.1 Place the prefilled syringe on a clean flat surface and let it sit for at least 30 minutes, to allow it to warm to room temperature (see Figure I). | ||
|
Do not attempt to warm the prefilled syringe in any other way. Do not use the VYVGART HYTRULO prefilled syringe if it has been at room temperature for longer than 30 days. |
||
| 4 Gather supplies and wash your hands | ||
| 4.1 Gather the following supplies that are not provided with the prefilled syringe (see Figure J). | ||
| 4.2 Wash your hands with soap and water (see Figure K). | ||
| 5 Snap off the prefilled syringe cap and attach the safety needle | ||
|
5.1 Carefully open the safety needle package and remove the needle (see Figure L). Throw away the packaging into household trash. |
||
|
5.2 Bend the prefilled syringe cap to one side to snap it off and remove it from the prefilled syringe (see Figure M). Throw away the prefilled syringe cap into the household trash. |
||
| Do not touch the tip of the prefilled syringe after the prefilled syringe cap has been removed. | ||
| 5.3 Hold the prefilled syringe by the syringe body in one hand, and attach the safety needle to the prefilled syringe by twisting it to the right (clockwise) until you feel resistance (see Figure N). | ||
| The safety needle is now attached to the prefilled syringe (see Figure O). | ||
| 6 Choose and clean the injection site on the stomach (abdomen) | ||
|
6.1 Choose an injection site on the stomach (abdomen) at least 2 inches away from the belly button (navel) (see Figure P). Change (rotate) the injection site the next time you inject VYVGART HYTRULO. |
||
|
Do not inject into skin that is irritated, red, bruised, infected, or tender. Do not inject into skin that is hard, scarred, or has moles. Do not inject into a vein. VYVGART HYTRULO is for under the skin (subcutaneous) injection only. |
||
| 6.2 Clean the chosen injection site with an alcohol swab and let it air dry (see Figure Q). | ||
| Do not blow on or touch the injection site after it has been cleaned. |
Inject VYVGART HYTRULO
| 7 Pull back the needle shield and remove the clear needle cap | ||
|
7.1 Pull the needle shield back (see Figure R). Note: The needle shield will be used after the injection to cover the needle and protect from needle-stick injuries. |
||
|
7.2 Hold the prefilled syringe body and remove the clear needle cap by pulling it straight off of the needle (see Figure S). Throw away the clear needle cap into the household trash. |
||
| Do not recap the needle. | ||
| 8 Give the injection | ||
| 8.1 Pinch the cleaned injection site (Figure T). | ||
| While pinching the skin, insert the needle at a 45 to 90 degree angle all the way into the pinched skin (see Figure U). Then release the pinched skin. |
||
| Do not pinch the skin too tightly as this can cause bruising. | ||
|
8.2 Slowly press the thumb pad down all the way until it stops to inject the liquid medicine (see Figure V). It will take about 20 to 30 seconds to inject all of the liquid medicine. You will feel resistance as you press down. Inject more slowly in case there is discomfort. It is ok if you need to pause or change your grip during the injection. |
||
| Do not try to force the thumb pad down quickly as this will make the thumb pad harder to press. | ||
| 8.3 After all the liquid medicine is injected, remove the needle from the skin by pulling it straight out without changing the angle that it was inserted (see Figure W). |
Throw away (dispose of) the Used Prefilled Syringe
| 9 Cover the needle and throw away the used prefilled syringe | ||
|
9.1 Carefully push the needle shield over the needle until it snaps into place and covers the needle (see Figure X). This helps to prevent needle-stick injuries. |
||
| Do not recap the needle using the clear needle cap. Only use the needle shield to cover the needle. | ||
| 9.2 Throw away the used prefilled syringe, with the needle still attached, into an FDA-cleared sharps disposal container right away after use (see Figure Y). | ||
| Do not throw away (dispose of) loose needles and syringes in your household trash. | ||
| 10 Treat the injection site | ||
|
10.1 If there is a small amount of blood or liquid at the injection site, press a sterile gauze over the injection site until the bleeding stops (see Figure Z). If needed, you may apply a small adhesive bandage. |
Additional Disposal Information
If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal. Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container. Always keep the sharps disposal container out of the reach of children. |
Additional Information
| This Instructions for Use has been approved by the U.S. Food and Drug Administration | Issued: 4/2025 |
| For more information, go to www.vyvgart.com or call 1-833-898-4278. Manufactured by: argenx BV Industriepark 7 9052 Zwijnaarde, Belgium U.S. License No. 2217 Halozyme, Inc. 12390 El Camino Real San Diego, CA 92130 U.S. License No. 2187 Distributed by: argenx US, Inc. 33 Arch Street Boston, MA 02110 |
12.2 Pharmacodynamics
In Study 1 [see Clinical Studies (14)], the pharmacological effect of efgartigimod alfa-fcab was assessed by measuring the decrease in serum IgG levels and AChR autoantibody levels. In patients testing positive for AChR antibodies and who were treated with efgartigimod alfa-fcab intravenous, there was a reduction in total IgG levels relative to baseline. Decrease in AChR autoantibody levels followed a similar pattern. A decrease in AChR-Ab was associated with a clinical response in AChR-Ab positive patients, as measured by the change from baseline in MG- ADL total score.
In Study 2, the pharmacological effect of VYVGART HYTRULO administered subcutaneously (SC) at 1,008 mg / 11,200 Units was compared to efgartigimod alfa-fcab administered intravenously at 10 mg/kg (EFG IV) in gMG patients. The maximum mean reduction in AChR-Ab level was observed at week 4, with a mean reduction of 62.2% and 59.7% in the VYVGART HYTRULO SC and efgartigimod alfa-fcab IV arm, respectively. The decrease in total IgG levels followed a similar pattern. The 90% confidence intervals for the geometric mean ratios of AChR-Ab reduction at day 29 and AUEC0-4w (area under the effect-time curve from time 0 to 4 weeks post dose) were within the range of 80% to 125%, indicating no clinically significant difference between the two formulations.
12.3 Pharmacokinetics
Efgartigimod alfa exposures were approximately dose-proportional up to the highest subcutaneously tested dose of VYVGART HYTRULO (1750 mg, 1.75 times the recommended dosage).
1 Indications and Usage (1 INDICATIONS AND USAGE)
VYVGART HYTRULO is indicated for the treatment of adult patients with:
- generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) antibody positive
- chronic inflammatory demyelinating polyneuropathy (CIDP)
12.1 Mechanism of Action
VYVGART HYTRULO is a coformulation of efgartigimod alfa and hyaluronidase.
Efgartigimod alfa is a human IgG1 antibody fragment that binds to the neonatal Fc receptor (FcRn), resulting in the reduction of circulating IgG.
Hyaluronidase increases permeability of the subcutaneous tissue by depolymerizing hyaluronan. This effect is transient, and permeability of the subcutaneous tissue is restored within 24 to 48 hours.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Infections: Delay administration of VYVGART HYTRULO to patients with an active infection. Monitor for signs and symptoms of infection in patients treated with VYVGART HYTRULO. If serious infection occurs, administer appropriate treatment and consider withholding VYVGART HYTRULO until the infection has resolved. (5.1)
- Hypersensitivity Reactions: Anaphylaxis, hypotension leading to syncope, angioedema, dyspnea, rash, and urticaria have occurred in patients treated with VYVGART HYTRULO or intravenous efgartigimod alfa-fcab product. If a hypersensitivity reaction occurs, the healthcare professional should institute appropriate measures if needed or the patient should seek medical attention. (4, 5.2)
- Infusion/injection-Related Reactions: If a severe infusion/injection-related reaction occurs, initiate appropriate therapy; consider the risks and benefits of readministering. If a mild to moderate infusion/injection-related reaction occurs, may rechallenge with close clinical observation, slower infusion/injection rates, and pre-medications. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- See Full Prescribing Information for instructions on dosage, preparation, and administration. (2.1, 2.2, 2.3, 2.4, 2.5)
- Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of a new treatment cycle with VYVGART HYTRULO. (2.1)
- Important Administration Information
- gMG: recommended dose and dose schedule
- Administer in cycles of once weekly injections for 4 weeks. (2.3)
- Prefilled syringe: 1,000 mg efgartigimod alfa and 10,000 units hyaluronidase administered over 20 to 30 seconds. (2.3)
- Vial: 1,008 mg efgartigimod alfa and 11,200 units hyaluronidase over 30 to 90 seconds. (2.3)
- Administer subsequent treatment cycles based on clinical evaluation. (2.3)
- CIDP: recommended dose and dose schedule
2.1 Recommended Vaccination
Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of a new treatment cycle with VYVGART HYTRULO. Because VYVGART HYTRULO causes transient reduction in IgG levels, vaccination with live vaccines is not recommended during treatment with VYVGART HYTRULO [see Dosage and Administration (2.3) and Warnings and Precautions (5.1)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 1,000 mg efgartigimod alfa and 10,000 units hyaluronidase per 5 mL (200 mg/2,000 units per mL) as yellowish, clear to opalescent solution, in a single-dose prefilled syringe.
Injection: 1,008 mg efgartigimod alfa and 11,200 units hyaluronidase per 5.6 mL (180 mg/2,000 units per mL) as yellowish, clear to opalescent solution, in a single-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of efgartigimod alfa products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Hypersensitivity reactions including anaphylaxis and hypotension, and infusion/injection-related reactions [see Warnings and Precautions (5.2, 5.3)].
Neurological: There have been reports of worsening of symptoms and signs of CIDP when transitioning from intravenous immunoglobulin treatments to VYVGART HYTRULO [see Drug Interactions (7.1)].
5.2 Hypersensitivity Reactions
In clinical trials, hypersensitivity reactions, including rash, angioedema, and dyspnea were observed in patients treated with VYVGART HYTRULO or intravenous efgartigimod alfa-fcab. Urticaria was also observed in patients treated with VYVGART HYTRULO. Hypersensitivity reactions were mild or moderate, occurred within one hour to three weeks of administration.
Anaphylaxis and hypotension leading to syncope have been reported in postmarketing experience with intravenous efgartigimod alfa-fcab. Anaphylaxis and hypotension occurred during or within an hour of administration and led to infusion discontinuation and in some cases to permanent treatment discontinuation.
Monitor for clinical signs and symptoms of hypersensitivity reactions for at least 30 minutes after administration [see Dosage and Administration (2.4)]. If a hypersensitivity reaction occurs, the healthcare professional should institute appropriate measures if needed or the patient should seek medical attention. VYVGART HYTRULO is contraindicated in patients with a history of serious hypersensitivity to efgartigimod alfa products, to hyaluronidase, or to any of the excipients of VYVGART HYTRULO [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient and/or caregiver to read FDA-approved patient labeling (Patient Information and Instructions for Use).
14.1 Generalized Myasthenia Gravis
Study 1 (described below) which established the effectiveness of efgartigimod alfa-fcab for the treatment of generalized myasthenia gravis (gMG) in adults who are AChR antibody positive was conducted with efgartigimod alfa-fcab intravenous formulation. In Study 2, VYVGART HYTRULO demonstrated a comparable pharmacodynamic effect on AChR antibody reduction as compared to the efgartigimod alfa-fcab intravenous formulation, which established the efficacy of VYVGART HYTRULO [see Clinical Pharmacology (12.2)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
VYVGART HYTRULO (efgartigimod alfa and hyaluronidase-qvfc) injection is a preservative free, sterile, yellowish, clear to opalescent solution supplied as a single-dose prefilled syringe or vial.
5.3 Infusion/injection Related Reactions (5.3 Infusion/Injection-Related Reactions)
Infusion-related reactions have been reported with intravenous efgartigimod alfa-fcab in postmarketing experience. The most frequent symptoms and signs were hypertension, chills, shivering, and thoracic, abdominal, and back pain. Infusion-related reactions occurred during or within an hour of administration and led to infusion discontinuation. If a severe infusion/injection-related reaction occurs, initiate appropriate therapy. Consider the risks and benefits of readministering VYVGART HYTRULO following a severe infusion/injection-related reaction. If a mild to moderate infusion/injection-related reaction occurs, patients may be rechallenged with close clinical observation, slower infusion/injection rates, and pre-medications.
7.1 Effect of Vyvgart Hytrulo On Other Drugs (7.1 Effect of VYVGART HYTRULO on Other Drugs)
Concomitant use of VYVGART HYTRULO with medications that bind to the human neonatal Fc receptor (FcRn) (e.g., immunoglobulin products, monoclonal antibodies, or antibody derivates containing the human Fc domain of the IgG subclass) may lower systemic exposures and reduce effectiveness of such medications. Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing VYVGART HYTRULO and using alternative therapies.
Principal Display Panel 5.6 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 5.6 mL Vial Carton)
NDC 73475-3102-3
VYVGART® Hytrulo
(efgartigimod alfa and
hyaluronidase-qvfc)
INJECTION
1,008 mg and 11,200 units/5.6 mL
(180 mg and 2,000 units/mL)
For subcutaneous injection
over 30 to 90 seconds
One single-dose vial
Discard unused portion
Must be administered by
a healthcare provider
Rx only
Principal Display Panel 5 Ml Syringe Carton (PRINCIPAL DISPLAY PANEL - 5 mL Syringe Carton)
NDC 73475-1221-1
VYVGART Hytrulo®
(efgartigimod alfa and hyaluronidase-qvfc)
INJECTION
1,000 mg and 10,000 units/5 mL
(200 mg and 2,000 units/mL)
For subcutaneous injection over 20 to 30 seconds
One single-dose prefilled syringe
Rx only
2.2 Important Dosage and Administration Instructions
VYVGART HYTRULO is for subcutaneous use only. Do not administer intravenously. Do not dilute VYVGART HYTRULO.
14.2 Chronic Inflammatory Demyelinating Polyneuropathy
The efficacy of VYVGART HYTRULO for the treatment of adults with chronic inflammatory demyelinating polyneuropathy (CIDP) was established in a two stage, multicenter study (Study 3; NCT04281472). Study 3 included a run-in period of up to 12 weeks after withdrawal of existing treatment for CIDP in order to identify patients with active disease, followed by an open-label period to identify VYVGART HYTRULO responders (stage A) who then entered a randomized, double-blind, placebo-controlled, withdrawal period (stage B).
Study 3 enrolled male and female patients age 18 years and older, who at the time of screening, had a documented diagnosis of definite or probable CIDP using the European Federation of Neurological Societies/Peripheral Nerve Society (EFNS/PNS; 2010) criteria for progressing or relapsing forms.
The Inflammatory Neuropathy Cause and Treatment disability score (INCAT) is a scale used to assess the impact of CIDP on daily upper and lower limb function, and is composed of the arm score and leg score (0 to 5 points for each). A total score on the INCAT ranges from 0 to 10 points with a higher number representing more disability. The adjusted INCAT (aINCAT) disability score, identical to the INCAT disability score but with changes in the upper limb function from 0 (normal) to 1 (minor symptoms) excluded, was used to assess efficacy for VYVGART HYTRULO for the treatment of CIDP.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
VYVGART HYTRULO for subcutaneous injection contains efgartigimod alfa and hyaluronidase [see Description (11)].
Principal Display Panel 1,008 Mg and 11,2000 Units/5.6 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 1,008 mg and 11,2000 units/5.6 mL Vial Carton)
NDC 73475-3102-3
VYVGART® Hytrulo
(efgartigimod alfa and
hyaluronidase-qvfc)
INJECTION
1,008 mg and 11,200 units/5.6 mL
(180 mg and 2,000 units/mL)
For subcutaneous injection
over 30 to 90 seconds
One single-dose vial
Discard unused portion
Must be administered by
a healthcare provider
Rx only
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:43.083711 · Updated: 2026-03-14T22:50:26.086884