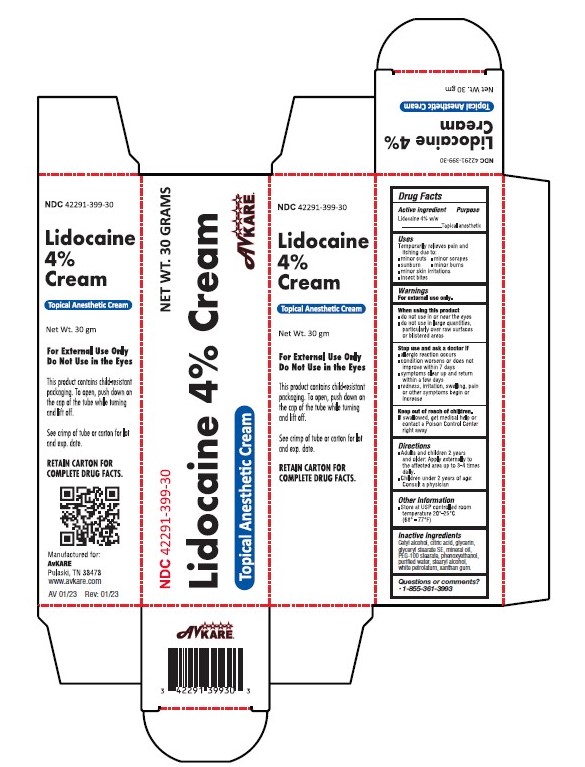
Lidocaine 4% Cream
70aace2f-47b4-e961-e053-2991aa0a6367
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine 4% w/w
Purpose
Topical anesthetic
Medication Information
Purpose
Topical anesthetic
Description
Lidocaine 4% w/w
Uses
temporarily relieves pain and itching due to:
- minor cuts
- minor scrapes
- sunburn
- minor skin irritations
- minor burns
- insect bites
Section 51945-4
Warnings
For external use only.
Directions
- Adults and children 2 years and older: Apply externally to the affected area up to 3-4 times daily.
- Children under 2 years of age: Consult a doctor.
Other Information
Store at USP controlled room temperature 20-25 degC (68-77 deg.F).
Active Ingredient:
Lidocaine 4% w/w
Inactive Ingredients
Cetyl alcohol, citric acid, glycerin, glyceryl stearate SE, mineral oil, PEG-100 stearate, phenoxyethanol, purified water, stearyl alcohol, white petrolatum, xanthan gum
When Using This Product
- do not use in or near the eyes
- do not use in large quantities, particularly over raw surfaces or blistered areas
Stop Use and Ask Doctor If
- Allergic reaction occurs
- condition worsens or does not improve within 7 days
- symptoms clear up and return within a few days
- redness, irritation, swelling, pain or other symptoms begin or increase
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away
Structured Label Content
Uses
temporarily relieves pain and itching due to:
- minor cuts
- minor scrapes
- sunburn
- minor skin irritations
- minor burns
- insect bites
Section 51945-4 (51945-4)
Purpose
Topical anesthetic
Warnings (WARNINGS)
For external use only.
Directions
- Adults and children 2 years and older: Apply externally to the affected area up to 3-4 times daily.
- Children under 2 years of age: Consult a doctor.
Other Information
Store at USP controlled room temperature 20-25 degC (68-77 deg.F).
Active Ingredient:
Lidocaine 4% w/w
Inactive Ingredients (Inactive ingredients)
Cetyl alcohol, citric acid, glycerin, glyceryl stearate SE, mineral oil, PEG-100 stearate, phenoxyethanol, purified water, stearyl alcohol, white petrolatum, xanthan gum
When Using This Product (When using this product)
- do not use in or near the eyes
- do not use in large quantities, particularly over raw surfaces or blistered areas
Stop Use and Ask Doctor If (Stop use and ask doctor if)
- Allergic reaction occurs
- condition worsens or does not improve within 7 days
- symptoms clear up and return within a few days
- redness, irritation, swelling, pain or other symptoms begin or increase
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:14.720117 · Updated: 2026-03-14T23:12:25.780176