Baricitinib Tablets, For Oral Use
7052c055-fedf-453b-86bc-01f5465eb157
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
FACT SHEET FOR HEALTHCARE PROVIDERS EMERGENCY USE AUTHORIZATION (EUA) OF BARICITINIB The U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) to permit the emergency use of baricitinib for treatment of coronavirus disease 2019 (COVID-19) in hospitalized pediatric patients 2 to less than 18 years of age requiring supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO). Baricitinib has been authorized by FDA for the emergency uses described above. Baricitinib is not FDA-approved for these uses. Baricitinib is authorized only for the duration of the declaration that circumstances exist justifying the authorization of the emergency use of baricitinib under section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization revoked sooner.
Medication Information
Description
FACT SHEET FOR HEALTHCARE PROVIDERS EMERGENCY USE AUTHORIZATION (EUA) OF BARICITINIB The U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) to permit the emergency use of baricitinib for treatment of coronavirus disease 2019 (COVID-19) in hospitalized pediatric patients 2 to less than 18 years of age requiring supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO). Baricitinib has been authorized by FDA for the emergency uses described above. Baricitinib is not FDA-approved for these uses. Baricitinib is authorized only for the duration of the declaration that circumstances exist justifying the authorization of the emergency use of baricitinib under section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization revoked sooner.
Section 42229-5
FACT SHEET FOR HEALTHCARE PROVIDERS
EMERGENCY USE AUTHORIZATION (EUA) OF BARICITINIB
The U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) to permit the emergency use of baricitinib for treatment of coronavirus disease 2019 (COVID-19) in hospitalized pediatric patients 2 to less than 18 years of age requiring supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO).
Baricitinib has been authorized by FDA for the emergency uses described above. Baricitinib is not FDA-approved for these uses.
Baricitinib is authorized only for the duration of the declaration that circumstances exist justifying the authorization of the emergency use of baricitinib under section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization revoked sooner.
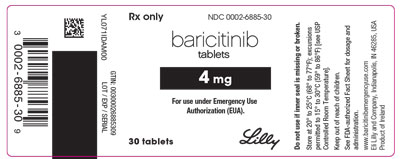
Package Label – Baricitinib 4 Mg 30ct Bottle
Rx Only
NDC 0002-6885-30
baricitinib
tablets
4 mg
For use under Emergency Use Authorization (EUA).
30 tablets
Lilly
Structured Label Content
Section 42229-5 (42229-5)
FACT SHEET FOR HEALTHCARE PROVIDERS
EMERGENCY USE AUTHORIZATION (EUA) OF BARICITINIB
The U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) to permit the emergency use of baricitinib for treatment of coronavirus disease 2019 (COVID-19) in hospitalized pediatric patients 2 to less than 18 years of age requiring supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO).
Baricitinib has been authorized by FDA for the emergency uses described above. Baricitinib is not FDA-approved for these uses.
Baricitinib is authorized only for the duration of the declaration that circumstances exist justifying the authorization of the emergency use of baricitinib under section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization revoked sooner.
Package Label – Baricitinib 4 Mg 30ct Bottle (PACKAGE LABEL – BARICITINIB 4 mg 30ct Bottle)
Rx Only
NDC 0002-6885-30
baricitinib
tablets
4 mg
For use under Emergency Use Authorization (EUA).
30 tablets
Lilly
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:00.642415 · Updated: 2026-03-14T21:48:23.957578