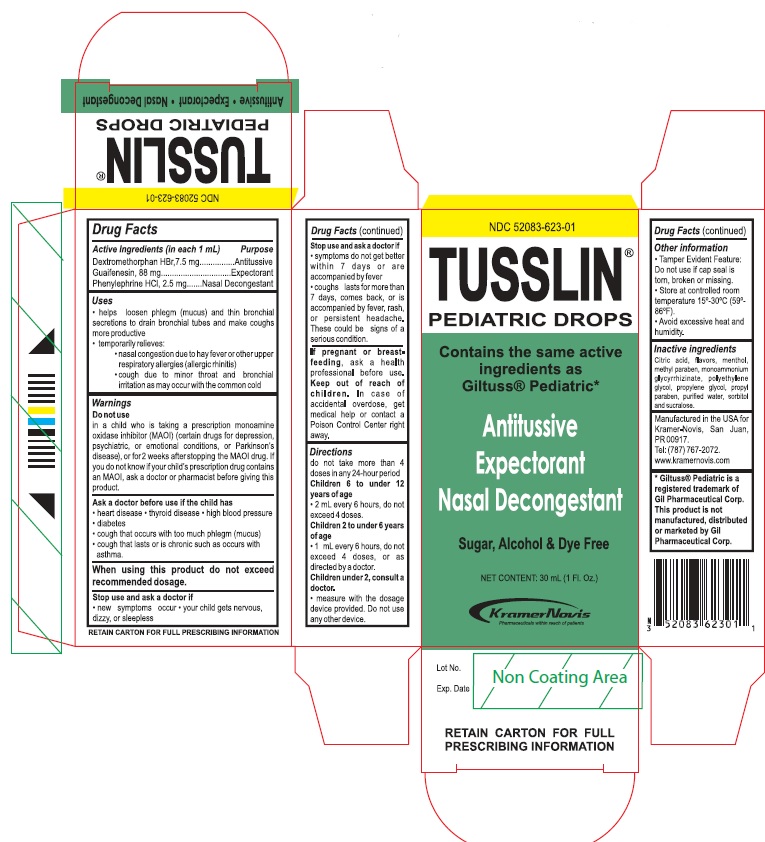
Drug Facts
6fe91407-08c1-465b-9c00-251c94b1f7f0
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients (in each 1 mL) Purpose Dextromethorphan HBr, 7.5 mg..............................Antitussive Guaifenesin, 88 mg..............................................Expectorant Phenylephrine HCl, 2.5 mg................................Nasal decongestant
Medication Information
Warnings and Precautions
Warnings
Do not use
in child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your child's prescription drug contains a MAOI, ask a doctor or pharmacist before giving this product.
Indications and Usage
Other information
Tamper Evident Feature: Do not use if cap seal is torn, broken or missing.
Store at controlled room temperature 15-30°C (59-86°F).
Avoid excessive heat and humidity.
Dosage and Administration
Directions
do not use more than 4 doses in any 24-hour period
Children 6 to under 12 years of age
- 2 mL every 6 hours, do not exceed 4 doses.
Children 2 to under 6 years of age
- 1 mL every 6 hours, do not exceed 4 doses, or as directed by a doctor.
Children under 2, consult a doctor
- measure with the dosage device provided. Do not use any other device.
Description
Active Ingredients (in each 1 mL) Purpose Dextromethorphan HBr, 7.5 mg..............................Antitussive Guaifenesin, 88 mg..............................................Expectorant Phenylephrine HCl, 2.5 mg................................Nasal decongestant
Section 50565-1
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Section 50570-1
Ask a doctor before use if your child has
- heart disease
- thyroid disease
- high blood pressure
- diabetes
- a cough that occurs with too much phlegm (mucus)
- cough that lasts or is chronic such as occurs with asthma
When using this product do not exceed recommended dosage
Stop use and ask a doctor if
- new symptoms occur
- your child gets nervous, dizzy or sleepless
- symptoms do not get better within 7 days or are accompanied by fever
- cough last more than 7 days, comes back, or is accompanied by fever, rash or persistent headache. These could be a signs of a serious condition.
Section 51727-6
Inactive ingredients Citric acid, flavors, menthol, methyl paraben, monoammonium glycyrrhizinate, polyethylene glycol, propylene glycol, propyl paraben, purified water, sorbitol and sucralose.
Section 53413-1
Manufactured in the USA for Kramer-Novis, San Juan, PR 00917.
Tel: (787) 767-2072
www.kramernovis.com
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
- helps loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes and make coughs more productive
- temporarily relieves:
- nasal congestion due to the common cold
- cough due to minor throat and bronchial irritation as may occur with the common cold
Section 55106-9
Active Ingredients (in each 1 mL) Purpose
Dextromethorphan HBr, 7.5 mg..............................Antitussive
Guaifenesin, 88 mg..............................................Expectorant
Phenylephrine HCl, 2.5 mg................................Nasal decongestant
Principal Display Panel
TUSSLIN®
PEDIATRIC DROPS
Contains the same active ingredients as Glituss® Pediatric
Antitussive
Expectorant
Nasal Decongestant
Sugar, Alcohol & Dye Free
NET CONTENT: 30 mL (1 Fl. Oz.)
KRAMER-NOVIS
Structured Label Content
Indications and Usage (34067-9)
Other information
Tamper Evident Feature: Do not use if cap seal is torn, broken or missing.
Store at controlled room temperature 15-30°C (59-86°F).
Avoid excessive heat and humidity.
Dosage and Administration (34068-7)
Directions
do not use more than 4 doses in any 24-hour period
Children 6 to under 12 years of age
- 2 mL every 6 hours, do not exceed 4 doses.
Children 2 to under 6 years of age
- 1 mL every 6 hours, do not exceed 4 doses, or as directed by a doctor.
Children under 2, consult a doctor
- measure with the dosage device provided. Do not use any other device.
Warnings and Precautions (34071-1)
Warnings
Do not use
in child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your child's prescription drug contains a MAOI, ask a doctor or pharmacist before giving this product.
Section 50565-1 (50565-1)
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Section 50570-1 (50570-1)
Ask a doctor before use if your child has
- heart disease
- thyroid disease
- high blood pressure
- diabetes
- a cough that occurs with too much phlegm (mucus)
- cough that lasts or is chronic such as occurs with asthma
When using this product do not exceed recommended dosage
Stop use and ask a doctor if
- new symptoms occur
- your child gets nervous, dizzy or sleepless
- symptoms do not get better within 7 days or are accompanied by fever
- cough last more than 7 days, comes back, or is accompanied by fever, rash or persistent headache. These could be a signs of a serious condition.
Section 51727-6 (51727-6)
Inactive ingredients Citric acid, flavors, menthol, methyl paraben, monoammonium glycyrrhizinate, polyethylene glycol, propylene glycol, propyl paraben, purified water, sorbitol and sucralose.
Section 53413-1 (53413-1)
Manufactured in the USA for Kramer-Novis, San Juan, PR 00917.
Tel: (787) 767-2072
www.kramernovis.com
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
- helps loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes and make coughs more productive
- temporarily relieves:
- nasal congestion due to the common cold
- cough due to minor throat and bronchial irritation as may occur with the common cold
Section 55106-9 (55106-9)
Active Ingredients (in each 1 mL) Purpose
Dextromethorphan HBr, 7.5 mg..............................Antitussive
Guaifenesin, 88 mg..............................................Expectorant
Phenylephrine HCl, 2.5 mg................................Nasal decongestant
Principal Display Panel
TUSSLIN®
PEDIATRIC DROPS
Contains the same active ingredients as Glituss® Pediatric
Antitussive
Expectorant
Nasal Decongestant
Sugar, Alcohol & Dye Free
NET CONTENT: 30 mL (1 Fl. Oz.)
KRAMER-NOVIS
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:52.248629 · Updated: 2026-03-14T22:55:37.343071