These Highlights Do Not Include All The Information Needed To Use Zerviate ®
6fc5ef4e-4aea-4c34-8d51-0c07ab9aad59
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2 ) 2/2020 Warnings and Precautions ( 5.1 ) 2/2020
Indications and Usage
ZERVIATE ® (cetirizine ophthalmic solution) 0.24% is indicated for the treatment of ocular itching associated with allergic conjunctivitis.
Dosage and Administration
The recommended dosage of ZERVIATE ® is to instill one drop in each affected eye twice daily (approximately 8 hours apart). The single-use containers are to be used immediately after opening and can be used to dose both eyes. Discard the single-use container and any remaining contents after administration. The single-use containers should be stored in the original foil pouch until ready to use.
Warnings and Precautions
Contamination of Tip and Solution. To prevent contaminating the dropper tip and solution, advise patients not to touch the eyelids or surrounding areas with the dropper tip of the bottle or tip of the single-use container. ( 5.1 )
Contraindications
None.
Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trial of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates in practice. In seven clinical trials, patients with allergic conjunctivitis or those at a risk of developing allergic conjunctivitis received one drop of either cetirizine (N=511) or vehicle (N=329) in one or both eyes. The most commonly reported adverse reactions occurred in approximately 1–7% of patients treated with either ZERVIATE ® or vehicle. These reactions were ocular hyperemia, instillation site pain, and visual acuity reduced.
Storage and Handling
ZERVIATE ® is a sterile, buffered, clear, colorless aqueous solution containing cetirizine 0.24% (equivalent to cetirizine hydrochloride 0.29%) supplied in a white low-density polyethylene multi-dose ophthalmic bottle with a low-density polyethylene dropper tip and a white polypropylene cap. ZERVIATE ® is supplied in a 7.5 mL bottle that contains 5 mL and a 10 mL bottle that contains 7.5 mL cetirizine ophthalmic solution, 2.40 mg [equivalent to 2.85 mg cetirizine hydrochloride in one mL solution]. ZERVIATE ® is also supplied in 5 low-density polyethylene 0.2 mL single-use containers within a foil pouch. Carton of 30 single-use containers NDC 82667-015-24
How Supplied
ZERVIATE ® is a sterile, buffered, clear, colorless aqueous solution containing cetirizine 0.24% (equivalent to cetirizine hydrochloride 0.29%) supplied in a white low-density polyethylene multi-dose ophthalmic bottle with a low-density polyethylene dropper tip and a white polypropylene cap. ZERVIATE ® is supplied in a 7.5 mL bottle that contains 5 mL and a 10 mL bottle that contains 7.5 mL cetirizine ophthalmic solution, 2.40 mg [equivalent to 2.85 mg cetirizine hydrochloride in one mL solution]. ZERVIATE ® is also supplied in 5 low-density polyethylene 0.2 mL single-use containers within a foil pouch. Carton of 30 single-use containers NDC 82667-015-24
Medication Information
Warnings and Precautions
Contamination of Tip and Solution. To prevent contaminating the dropper tip and solution, advise patients not to touch the eyelids or surrounding areas with the dropper tip of the bottle or tip of the single-use container. ( 5.1 )
Indications and Usage
ZERVIATE ® (cetirizine ophthalmic solution) 0.24% is indicated for the treatment of ocular itching associated with allergic conjunctivitis.
Dosage and Administration
The recommended dosage of ZERVIATE ® is to instill one drop in each affected eye twice daily (approximately 8 hours apart). The single-use containers are to be used immediately after opening and can be used to dose both eyes. Discard the single-use container and any remaining contents after administration. The single-use containers should be stored in the original foil pouch until ready to use.
Contraindications
None.
Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trial of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates in practice. In seven clinical trials, patients with allergic conjunctivitis or those at a risk of developing allergic conjunctivitis received one drop of either cetirizine (N=511) or vehicle (N=329) in one or both eyes. The most commonly reported adverse reactions occurred in approximately 1–7% of patients treated with either ZERVIATE ® or vehicle. These reactions were ocular hyperemia, instillation site pain, and visual acuity reduced.
Storage and Handling
ZERVIATE ® is a sterile, buffered, clear, colorless aqueous solution containing cetirizine 0.24% (equivalent to cetirizine hydrochloride 0.29%) supplied in a white low-density polyethylene multi-dose ophthalmic bottle with a low-density polyethylene dropper tip and a white polypropylene cap. ZERVIATE ® is supplied in a 7.5 mL bottle that contains 5 mL and a 10 mL bottle that contains 7.5 mL cetirizine ophthalmic solution, 2.40 mg [equivalent to 2.85 mg cetirizine hydrochloride in one mL solution]. ZERVIATE ® is also supplied in 5 low-density polyethylene 0.2 mL single-use containers within a foil pouch. Carton of 30 single-use containers NDC 82667-015-24
How Supplied
ZERVIATE ® is a sterile, buffered, clear, colorless aqueous solution containing cetirizine 0.24% (equivalent to cetirizine hydrochloride 0.29%) supplied in a white low-density polyethylene multi-dose ophthalmic bottle with a low-density polyethylene dropper tip and a white polypropylene cap. ZERVIATE ® is supplied in a 7.5 mL bottle that contains 5 mL and a 10 mL bottle that contains 7.5 mL cetirizine ophthalmic solution, 2.40 mg [equivalent to 2.85 mg cetirizine hydrochloride in one mL solution]. ZERVIATE ® is also supplied in 5 low-density polyethylene 0.2 mL single-use containers within a foil pouch. Carton of 30 single-use containers NDC 82667-015-24
Description
Dosage and Administration ( 2 ) 2/2020 Warnings and Precautions ( 5.1 ) 2/2020
Section 42229-5
Risk Summary
There were no adequate or well-controlled studies with ZERVIATE ®in pregnant women.
Cetirizine should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus.
Section 44425-7
Storage:Store at 15°C to 25°C (59°F to 77°F).
Single-use containers should be stored in the original foil pouch.
11 Description
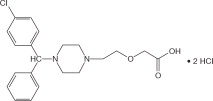
ZERVIATE ®is a sterile ophthalmic solution containing cetirizine, which is a histamine-1 (H1) receptor antagonist, for topical administration to the eyes. Cetirizine hydrochloride is a white, crystalline, water-soluble powder with a molecular weight of 461.8 and a molecular formula of C 21H 25ClN 2O 3•2HCl. The chemical structure is presented below:
Chemical Name:
( RS)-2-[2-[4-[(4-Chlorophenyl) phenylmethyl] piperazin-1-yl] ethoxy] acetic acid, dihydrochloride
Each mL of ZERVIATE ®contains an active ingredient [cetirizine 2.40 mg (equivalent to 2.85 mg of cetirizine hydrochloride)] and the following inactive ingredients: benzalkonium chloride 0.010% (preservative); glycerin; sodium phosphate, dibasic; edetate disodium; polyethylene glycol 400; polysorbate 80; hypromellose; hydrochloric acid/sodiumhydroxide (to adjust pH); and water for injection. ZERVIATE ®solution has a pH of approximately 7.0 and osmolality of approximately 300 mOsm/kg.
8.4 Pediatric Use
The safety and effectiveness of ZERVIATE ®(cetirizine ophthalmic solution) 0.24% has been established in pediatric patients two years of age and older. Use of ZERVIATE ®in these pediatric patients is supported by evidence from adequate and well-controlled studies of ZERVIATE ®in pediatric and adult patients.
8.5 Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and younger patients.
14 Clinical Studies
The efficacy of ZERVIATE ®(cetirizine ophthalmic solution) 0.24% was established in three randomized, double-masked, placebocontrolled, conjunctival allergen challenge (CAC) clinical trials in patients with a history of allergic conjunctivitis. Onset and duration of action were evaluated in two of these trials in which patients were randomized to receive ZERVIATE ®or vehicle ophthalmic solutions. Patients were evaluated with an ocular itching severity score ranging from 0 (no itching) to 4 (incapacitating itch) at several time points after CAC administration. Table 1displays data from the mean ocular itching severity scores after ocular administration of an antigen using the CAC model. A one unit difference compared to vehicle is considered a clinically meaningful change in the ocular itching severity score.
Patients treated with ZERVIATE ®demonstrated statistically and clinically significantly less ocular itching compared to vehicle at 15 minutes and 8 hours after treatment.
|
1Treatment difference values shown are the group mean active minus the group mean vehicle at each post-CAC time point. |
||||||||
|
* p<0.05 |
||||||||
| Study 1 | Study 2 | |||||||
| Statistics | 15 minutes post-treatment | 8 hours post-treatment | 15 minutes post-treatment | 8 hours post-treatment | ||||
| ZERVIATE N=50 |
Vehicle
N=50 |
ZERVIATE N=50 |
Vehicle
N=50 |
ZERVIATE N=51 |
Vehicle
N=50 |
ZERVIATE N=51 |
Vehicle
N=50 |
|
| 3 Minute Post-CAC | ||||||||
| Mean | 1.00 | 2.38 | 1.76 | 2.69 | 1.01 | 2.54 | 1.94 | 2.86 |
| Treatment
Difference (95% CI) 1 |
-1.38
(-1.72, -1.05)* |
-0.93
(-1.26, -0.61)* |
-1.53
(-1.92, -1.15)* |
-0.92
(-1.25, -0.58)* |
||||
| 5 Minute Post-CAC | ||||||||
| Mean | 1.18 | 2.43 | 1.85 | 2.74 | 1.17 | 2.51 | 2.03 | 2.94 |
| Treatment
Difference (95% CI) 1 |
-1.25
(-1.58, -0.91)* |
-0.89
(-1.24, -0.54)* |
-1.34
(-1.71, -0.97)* |
-0.90
(-1.23, -0.57)* |
||||
| 7 Minute Post-CAC | ||||||||
| Mean | 1.11 | 2.11 | 1.54 | 2.53 | 1.15 | 2.23 | 1.82 | 2.66 |
| Treatment
Difference (95% CI) 1 |
-1.00
(-1.35, -0.65)* |
-0.99
(-1.40, -0.59)* |
-1.07
(-1.46, -0.69)* |
-0.84
(-1.21, -0.48)* |
4 Contraindications
None.
6 Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trial of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates in practice.
In seven clinical trials, patients with allergic conjunctivitis or those at a risk of developing allergic conjunctivitis received one drop of either cetirizine (N=511) or vehicle (N=329) in one or both eyes. The most commonly reported adverse reactions occurred in approximately 1–7% of patients treated with either ZERVIATE ®or vehicle. These reactions were ocular hyperemia, instillation site pain, and visual acuity reduced.
12.3 Pharmacokinetics
In healthy subjects, bilateral topical ocular dosing of one drop of ZERVIATE ®resulted in a mean cetirizine plasma C maxof 1.7 ng/mL following a single dose and 3.1 ng/mL after twice-daily dosing for one week. The observed mean terminal half-life of cetirizine was 8.6 hours following a single dose and 8.2 hours after twice-daily dosing of ZERVIATE ®for one week.
5.2 Contact Lens Wear
Patients should be advised not to wear a contact lens if their eye is red.
ZERVIATE ®should not be instilled while wearing contact lenses. Remove contact lenses prior to instillation of ZERVIATE ®. The preservative in ZERVIATE ®, benzalkonium chloride, may be absorbed by soft contact lenses. Lenses may be reinserted 10 minutes following administration of ZERVIATE ®.
1 Indications and Usage
ZERVIATE ®(cetirizine ophthalmic solution) 0.24% is indicated for the treatment of ocular itching associated with allergic conjunctivitis.
12.1 Mechanism of Action
ZERVIATE ®, an antihistamine, is a histamine-1 (H1) receptor antagonist. Its effects are mediated via selective inhibition of H1 histamine receptors. The antihistaminic activity of cetirizine has been documented in a variety of animal and human models. In vivoand ex vivoanimal models have shown negligible anticholinergic and antiserotonergic activity. In vitroreceptor binding studies have shown no measurable affinity for other than H1 receptors.
5 Warnings and Precautions
Contamination of Tip and Solution. To prevent contaminating the dropper tip and solution, advise patients not to touch the eyelids or surrounding areas with the dropper tip of the bottle or tip of the single-use container. ( 5.1)
2 Dosage and Administration
The recommended dosage of ZERVIATE ® is to instill one drop in each affected eye twice daily (approximately 8 hours apart).
The single-use containers are to be used immediately after opening and can be used to dose both eyes. Discard the single-use container and any remaining contents after administration. The single-use containers should be stored in the original foil pouch until ready to use.
3 Dosage Forms and Strengths
Cetirizine ophthalmic solution, 0.24% is a sterile, buffered, clear, colorless aqueous solution containing cetirizine 0.24% (equivalent to cetirizine hydrochloride 0.29%).
17 Patient Counseling Information
- Risk of Contamination: Advise patients not to touch dropper tip to eyelids or surrounding areas, as this may contaminate the dropper tip and ophthalmic solution. Advise patients to keep the bottle closed when not in use. Advise patients to discard single-use containers after each use.
- Concomitant Use of Contact Lenses: Advise patients not to wear contact lenses if their eyes are red. Advise patients that ZERVIATE ®should not be used to treat contact lens-related irritation. Advise patients to remove contact lenses prior to instillation of ZERVIATE ®. The preserative in ZERVIATE ®solution, benzalkonium chloride, may be absorbed by soft contact lenses. Lenses may be reinserted 10 minutes following administration of ZERVIATE ®.
- Administration: Advise patients that the solution from one single-use container is to be used immediately after opening. Advise patients that the single-use container can be used to dose both eyes. Discard the single-use container and remaining contents immediately after administration.
- Storage of Single-use Containers: Instruct patients to store single-use containers in the original foil pouch until ready to use.
Rev 11/2024
Manufactured for:
Harrow Eye, LLC
TM
Nashville, TN USA
U.S. Patents: 8,829,005; 9,254,286; 9,750,684; 9,993,471
16 How Supplied/storage and Handling
ZERVIATE ®is a sterile, buffered, clear, colorless aqueous solution containing cetirizine 0.24% (equivalent to cetirizine hydrochloride 0.29%) supplied in a white low-density polyethylene multi-dose ophthalmic bottle with a low-density polyethylene dropper tip and a white polypropylene cap. ZERVIATE ®is supplied in a 7.5 mL bottle that contains 5 mL and a 10 mL bottle that contains 7.5 mL cetirizine ophthalmic solution, 2.40 mg [equivalent to 2.85 mg cetirizine hydrochloride in one mL solution]. ZERVIATE ®is also supplied in 5 low-density polyethylene 0.2 mL single-use containers within a foil pouch.
Carton of 30 single-use containers NDC 82667-015-24
5.1 Contamination of Tip and Solution
As with any eye drop, care should be taken not to touch the eyelids or surrounding areas with the dropper tip of the bottle or tip of the single-use container in order to avoid injury to the eye and to prevent contaminating the tip and solution. Keep the multi-dose bottle closed when not in use. Discard the single-use container after using in each eye.
Principal Display Panel – 2.5 Ml Carton Label
NDC 82667-015-24
ZERVIATE®
(cetirizine ophthalmic
solution) 0.24%
30 single use containers
6 pouches x 5 single-use containers
(0.2 mL each)
FOR TOPICAL
OPHTHALMIC USE
Rx Only HARROW® Sterile
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
There were no adequate or well-controlled studies with ZERVIATE ®in pregnant women.
Cetirizine should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus.
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage:Store at 15°C to 25°C (59°F to 77°F).
Single-use containers should be stored in the original foil pouch.
11 Description (11 DESCRIPTION)
ZERVIATE ®is a sterile ophthalmic solution containing cetirizine, which is a histamine-1 (H1) receptor antagonist, for topical administration to the eyes. Cetirizine hydrochloride is a white, crystalline, water-soluble powder with a molecular weight of 461.8 and a molecular formula of C 21H 25ClN 2O 3•2HCl. The chemical structure is presented below:
Chemical Name:
( RS)-2-[2-[4-[(4-Chlorophenyl) phenylmethyl] piperazin-1-yl] ethoxy] acetic acid, dihydrochloride
Each mL of ZERVIATE ®contains an active ingredient [cetirizine 2.40 mg (equivalent to 2.85 mg of cetirizine hydrochloride)] and the following inactive ingredients: benzalkonium chloride 0.010% (preservative); glycerin; sodium phosphate, dibasic; edetate disodium; polyethylene glycol 400; polysorbate 80; hypromellose; hydrochloric acid/sodiumhydroxide (to adjust pH); and water for injection. ZERVIATE ®solution has a pH of approximately 7.0 and osmolality of approximately 300 mOsm/kg.
8.4 Pediatric Use
The safety and effectiveness of ZERVIATE ®(cetirizine ophthalmic solution) 0.24% has been established in pediatric patients two years of age and older. Use of ZERVIATE ®in these pediatric patients is supported by evidence from adequate and well-controlled studies of ZERVIATE ®in pediatric and adult patients.
8.5 Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and younger patients.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of ZERVIATE ®(cetirizine ophthalmic solution) 0.24% was established in three randomized, double-masked, placebocontrolled, conjunctival allergen challenge (CAC) clinical trials in patients with a history of allergic conjunctivitis. Onset and duration of action were evaluated in two of these trials in which patients were randomized to receive ZERVIATE ®or vehicle ophthalmic solutions. Patients were evaluated with an ocular itching severity score ranging from 0 (no itching) to 4 (incapacitating itch) at several time points after CAC administration. Table 1displays data from the mean ocular itching severity scores after ocular administration of an antigen using the CAC model. A one unit difference compared to vehicle is considered a clinically meaningful change in the ocular itching severity score.
Patients treated with ZERVIATE ®demonstrated statistically and clinically significantly less ocular itching compared to vehicle at 15 minutes and 8 hours after treatment.
|
1Treatment difference values shown are the group mean active minus the group mean vehicle at each post-CAC time point. |
||||||||
|
* p<0.05 |
||||||||
| Study 1 | Study 2 | |||||||
| Statistics | 15 minutes post-treatment | 8 hours post-treatment | 15 minutes post-treatment | 8 hours post-treatment | ||||
| ZERVIATE N=50 |
Vehicle
N=50 |
ZERVIATE N=50 |
Vehicle
N=50 |
ZERVIATE N=51 |
Vehicle
N=50 |
ZERVIATE N=51 |
Vehicle
N=50 |
|
| 3 Minute Post-CAC | ||||||||
| Mean | 1.00 | 2.38 | 1.76 | 2.69 | 1.01 | 2.54 | 1.94 | 2.86 |
| Treatment
Difference (95% CI) 1 |
-1.38
(-1.72, -1.05)* |
-0.93
(-1.26, -0.61)* |
-1.53
(-1.92, -1.15)* |
-0.92
(-1.25, -0.58)* |
||||
| 5 Minute Post-CAC | ||||||||
| Mean | 1.18 | 2.43 | 1.85 | 2.74 | 1.17 | 2.51 | 2.03 | 2.94 |
| Treatment
Difference (95% CI) 1 |
-1.25
(-1.58, -0.91)* |
-0.89
(-1.24, -0.54)* |
-1.34
(-1.71, -0.97)* |
-0.90
(-1.23, -0.57)* |
||||
| 7 Minute Post-CAC | ||||||||
| Mean | 1.11 | 2.11 | 1.54 | 2.53 | 1.15 | 2.23 | 1.82 | 2.66 |
| Treatment
Difference (95% CI) 1 |
-1.00
(-1.35, -0.65)* |
-0.99
(-1.40, -0.59)* |
-1.07
(-1.46, -0.69)* |
-0.84
(-1.21, -0.48)* |
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trial of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates in practice.
In seven clinical trials, patients with allergic conjunctivitis or those at a risk of developing allergic conjunctivitis received one drop of either cetirizine (N=511) or vehicle (N=329) in one or both eyes. The most commonly reported adverse reactions occurred in approximately 1–7% of patients treated with either ZERVIATE ®or vehicle. These reactions were ocular hyperemia, instillation site pain, and visual acuity reduced.
12.3 Pharmacokinetics
In healthy subjects, bilateral topical ocular dosing of one drop of ZERVIATE ®resulted in a mean cetirizine plasma C maxof 1.7 ng/mL following a single dose and 3.1 ng/mL after twice-daily dosing for one week. The observed mean terminal half-life of cetirizine was 8.6 hours following a single dose and 8.2 hours after twice-daily dosing of ZERVIATE ®for one week.
5.2 Contact Lens Wear
Patients should be advised not to wear a contact lens if their eye is red.
ZERVIATE ®should not be instilled while wearing contact lenses. Remove contact lenses prior to instillation of ZERVIATE ®. The preservative in ZERVIATE ®, benzalkonium chloride, may be absorbed by soft contact lenses. Lenses may be reinserted 10 minutes following administration of ZERVIATE ®.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZERVIATE ®(cetirizine ophthalmic solution) 0.24% is indicated for the treatment of ocular itching associated with allergic conjunctivitis.
12.1 Mechanism of Action
ZERVIATE ®, an antihistamine, is a histamine-1 (H1) receptor antagonist. Its effects are mediated via selective inhibition of H1 histamine receptors. The antihistaminic activity of cetirizine has been documented in a variety of animal and human models. In vivoand ex vivoanimal models have shown negligible anticholinergic and antiserotonergic activity. In vitroreceptor binding studies have shown no measurable affinity for other than H1 receptors.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Contamination of Tip and Solution. To prevent contaminating the dropper tip and solution, advise patients not to touch the eyelids or surrounding areas with the dropper tip of the bottle or tip of the single-use container. ( 5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dosage of ZERVIATE ® is to instill one drop in each affected eye twice daily (approximately 8 hours apart).
The single-use containers are to be used immediately after opening and can be used to dose both eyes. Discard the single-use container and any remaining contents after administration. The single-use containers should be stored in the original foil pouch until ready to use.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Cetirizine ophthalmic solution, 0.24% is a sterile, buffered, clear, colorless aqueous solution containing cetirizine 0.24% (equivalent to cetirizine hydrochloride 0.29%).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
- Risk of Contamination: Advise patients not to touch dropper tip to eyelids or surrounding areas, as this may contaminate the dropper tip and ophthalmic solution. Advise patients to keep the bottle closed when not in use. Advise patients to discard single-use containers after each use.
- Concomitant Use of Contact Lenses: Advise patients not to wear contact lenses if their eyes are red. Advise patients that ZERVIATE ®should not be used to treat contact lens-related irritation. Advise patients to remove contact lenses prior to instillation of ZERVIATE ®. The preserative in ZERVIATE ®solution, benzalkonium chloride, may be absorbed by soft contact lenses. Lenses may be reinserted 10 minutes following administration of ZERVIATE ®.
- Administration: Advise patients that the solution from one single-use container is to be used immediately after opening. Advise patients that the single-use container can be used to dose both eyes. Discard the single-use container and remaining contents immediately after administration.
- Storage of Single-use Containers: Instruct patients to store single-use containers in the original foil pouch until ready to use.
Rev 11/2024
Manufactured for:
Harrow Eye, LLC
TM
Nashville, TN USA
U.S. Patents: 8,829,005; 9,254,286; 9,750,684; 9,993,471
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ZERVIATE ®is a sterile, buffered, clear, colorless aqueous solution containing cetirizine 0.24% (equivalent to cetirizine hydrochloride 0.29%) supplied in a white low-density polyethylene multi-dose ophthalmic bottle with a low-density polyethylene dropper tip and a white polypropylene cap. ZERVIATE ®is supplied in a 7.5 mL bottle that contains 5 mL and a 10 mL bottle that contains 7.5 mL cetirizine ophthalmic solution, 2.40 mg [equivalent to 2.85 mg cetirizine hydrochloride in one mL solution]. ZERVIATE ®is also supplied in 5 low-density polyethylene 0.2 mL single-use containers within a foil pouch.
Carton of 30 single-use containers NDC 82667-015-24
5.1 Contamination of Tip and Solution
As with any eye drop, care should be taken not to touch the eyelids or surrounding areas with the dropper tip of the bottle or tip of the single-use container in order to avoid injury to the eye and to prevent contaminating the tip and solution. Keep the multi-dose bottle closed when not in use. Discard the single-use container after using in each eye.
Principal Display Panel – 2.5 Ml Carton Label (Principal Display Panel – 2.5 mL Carton Label)
NDC 82667-015-24
ZERVIATE®
(cetirizine ophthalmic
solution) 0.24%
30 single use containers
6 pouches x 5 single-use containers
(0.2 mL each)
FOR TOPICAL
OPHTHALMIC USE
Rx Only HARROW® Sterile
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:53.110223 · Updated: 2026-03-14T22:25:56.100461