higeen®
6fc03798-142e-4104-b961-6c49eeeaf32d
34390-5
HUMAN OTC DRUG LABEL
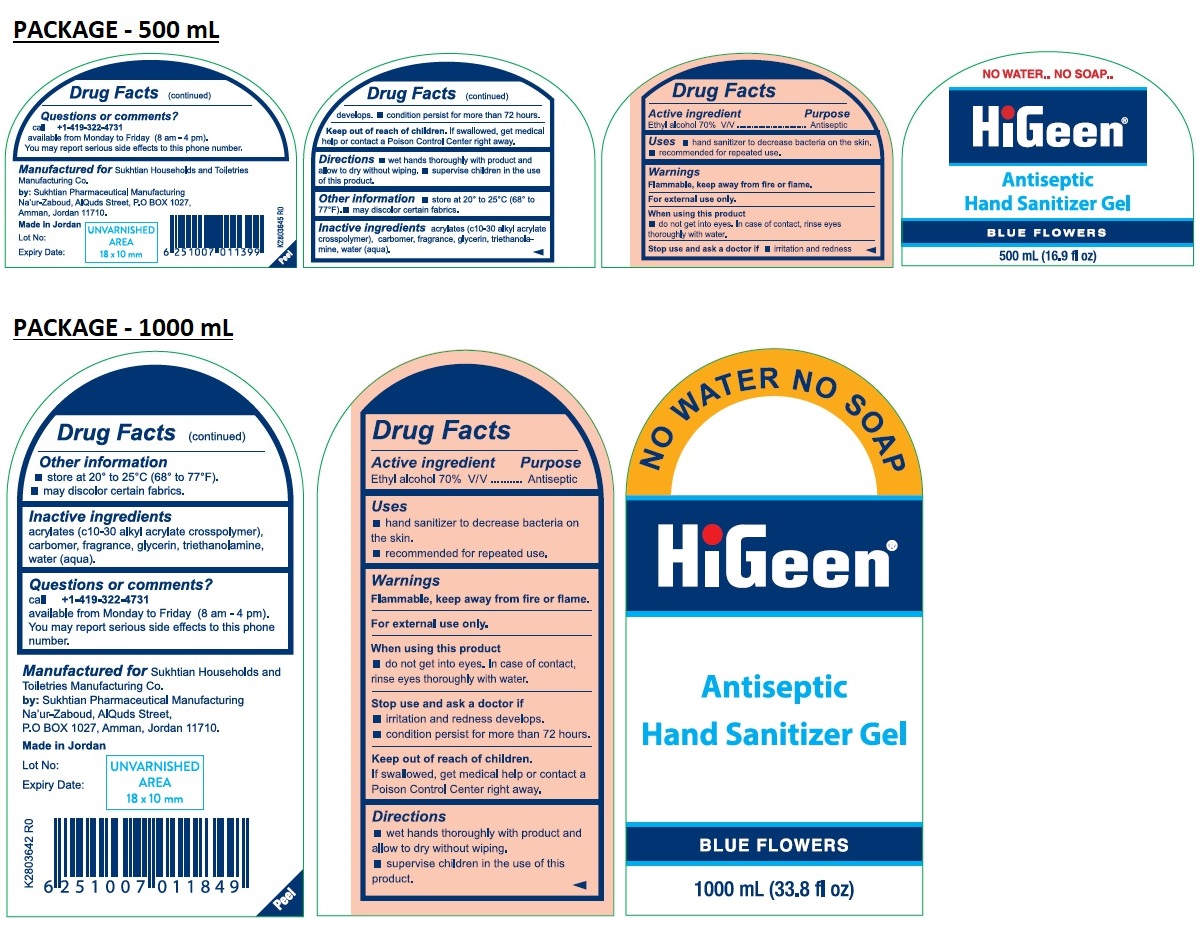
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antiseptic
Description
Ethyl alcohol 70% V/V
Medication Information
Warnings
Flammable, keep away from fire or flame.
For external use only.
When using this product
• do not get into eyes. In case of contact, rinse eyes thoroughly with water.
Stop use and ask a doctor if
• irritation and redness develops.
• condition persist for more than 72 hours.
Uses
• hand sanitizer to decrease bacteria on the skin.
• recommended for repeated use.
Purpose
Antiseptic
Directions
• wet hands thoroughly with product and allow to dry without wiping.
• supervise children in the use of this product.
Other Information
• store at 20° to 25°C (68° to 77°F).
• may discolor certain fabrics.
Inactive Ingredients
acrylates (c10-30 alkyl acrylate crosspolymer), carbomer, fragrance, glycerin, triethanolamine, water (aqua).
Description
Ethyl alcohol 70% V/V
Section 42229-5
NO WATER
NO SOAP
Manufactured for
Sukhtian Households and Toiletries Manufacturing Co.
by: Sukhtian Pharmaceutical Manufacturing
Na'ur-Zaboud, AlQuds street,
P.O BOX 1027, Amman, Jordan 11710.
Made in Jordan
Section 50565-1
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Packaging
Drug Facts
Active Ingredient
Ethyl alcohol 70% V/V
Questions Or Comments?
call +1-419-322-4731
available from Monday to Friday (8 am - 4 pm).
You may report serious side effects to this phone number.
Structured Label Content
Warnings
Flammable, keep away from fire or flame.
For external use only.
When using this product
• do not get into eyes. In case of contact, rinse eyes thoroughly with water.
Stop use and ask a doctor if
• irritation and redness develops.
• condition persist for more than 72 hours.
Uses
• hand sanitizer to decrease bacteria on the skin.
• recommended for repeated use.
Purpose
Antiseptic
Directions
• wet hands thoroughly with product and allow to dry without wiping.
• supervise children in the use of this product.
Other Information (Other information)
• store at 20° to 25°C (68° to 77°F).
• may discolor certain fabrics.
Inactive Ingredients (Inactive ingredients)
acrylates (c10-30 alkyl acrylate crosspolymer), carbomer, fragrance, glycerin, triethanolamine, water (aqua).
Section 42229-5 (42229-5)
NO WATER
NO SOAP
Manufactured for
Sukhtian Households and Toiletries Manufacturing Co.
by: Sukhtian Pharmaceutical Manufacturing
Na'ur-Zaboud, AlQuds street,
P.O BOX 1027, Amman, Jordan 11710.
Made in Jordan
Section 50565-1 (50565-1)
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Packaging
Drug Facts
Active Ingredient (Active ingredient)
Ethyl alcohol 70% V/V
Questions Or Comments? (Questions or comments?)
call +1-419-322-4731
available from Monday to Friday (8 am - 4 pm).
You may report serious side effects to this phone number.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:01:02.222308 · Updated: 2026-03-14T23:01:14.422395