These Highlights Do Not Include All The Information Needed To Use Tecentriq Safely And Effectively. See Full Prescribing Information For Tecentriq.
6fa682c9-a312-4932-9831-f286908660ee
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions ( 5.2 ) 08/2025 Indications and Usage ( 1.2 ) 10/2025
Indications and Usage
TECENTRIQ is a programmed death-ligand 1 (PD-L1) blocking antibody indicated: Non-Small Cell Lung Cancer (NSCLC) as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage II to IIIA NSCLC whose tumors have PD-L1 expression on ≥ 1% of tumor cells, as determined by an FDA-approved test. ( 1.1 , 14.1 ) for the first-line treatment of adult patients with metastatic NSCLC whose tumors have high PD-L1 expression (PD-L1 stained ≥ 50% of tumor cells [TC ≥ 50%] or PD-L1 stained tumor-infiltrating immune cells [IC] covering ≥ 10% of the tumor area [IC ≥ 10%] ), as determined by an FDA-approved test, with no EGFR or ALK genomic tumor aberrations. ( 1.1 ) in combination with bevacizumab, paclitaxel, and carboplatin, for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations. ( 1.1 ) in combination with paclitaxel protein-bound and carboplatin for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations ( 1.1 ) for the treatment of adult patients with metastatic NSCLC who have disease progression during or following platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for NSCLC harboring these aberrations prior to receiving TECENTRIQ. ( 1.1 ) Small Cell Lung Cancer (SCLC) in combination with carboplatin and etoposide, for the first-line treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC). ( 1.2 ) in combination with lurbinectedin, for the maintenance treatment of adult patients with ES-SCLC whose disease has not progressed after first-line induction therapy with TECENTRIQ or atezolizumab and hyaluronidase-tqjs, carboplatin and etoposide. ( 1.2 ) Hepatocellular Carcinoma (HCC) in combination with bevacizumab for the treatment of adult patients with unresectable or metastatic HCC who have not received prior systemic therapy. ( 1.3 ) Melanoma in combination with cobimetinib and vemurafenib for the treatment of adult patients with BRAF V600 mutation-positive unresectable or metastatic melanoma. ( 1.4 ) Alveolar Soft Part Sarcoma (ASPS) for the treatment of adult and pediatric patients 2 years of age and older with unresectable or metastatic ASPS. ( 1.5 )
Dosage and Administration
Administer TECENTRIQ intravenously over 60 minutes. If the first infusion is tolerated, all subsequent infusions may be delivered over 30 minutes. NSCLC In the adjuvant setting, administer TECENTRIQ following resection and up to 4 cycles of platinum-based chemotherapy as 840 mg every 2 weeks, 1200 mg every 3 weeks or 1680 mg every 4 weeks for up to 1 year. ( 2.2 ) In the metastatic setting, administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. ( 2.2 ) When administering with chemotherapy with or without bevacizumab, administer TECENTRIQ prior to chemotherapy and bevacizumab when given on the same day. ( 2.2 ) Small Cell Lung Cancer Administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. Administer TECENTRIQ prior to chemotherapy when given on the same day. ( 2.2 ) Hepatocellular Carcinoma Administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. Administer TECENTRIQ prior to bevacizumab when given on the same day. Bevacizumab is administered at 15 mg/kg every 3 weeks. ( 2.2 ) Melanoma Following completion of a 28-day cycle of cobimetinib and vemurafenib, administer TECENTRIQ 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks with cobimetinib 60 mg orally once daily (21 days on /7 days off) and vemurafenib 720 mg orally twice daily. ( 2.2 ) ASPS Adults: Administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. ( 2.2 ) Pediatric patients 2 years of age and older: 15 mg/kg (up to a maximum of 1200 mg), every 3 weeks ( 2.2 )
Warnings and Precautions
Immune-Mediated Adverse Reactions Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated dermatologic adverse reactions, immune-mediated nephritis and renal dysfunction, and solid organ transplant rejection. ( 5.1 ) Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. ( 5.1 ) Withhold or permanently discontinue based on severity and type of reaction. ( 5.1 ). Infusion-Related Reactions : Interrupt, slow the rate of infusion, or permanently discontinue based on severity of infusion reactions. ( 5.2 ) Complications of Allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. ( 5.3 ) Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. ( 5.4 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
No dose reduction for TECENTRIQ is recommended. In general, withhold TECENTRIQ for severe (Grade 3) immune-mediated adverse reactions. Permanently discontinue TECENTRIQ for life-threatening (Grade 4) immune-mediated adverse reactions, recurrent severe (Grade 3) immune-mediated reactions that require systemic immunosuppressive treatment, or an inability to reduce corticosteroid dose to 10 mg or less of prednisone or equivalent per day within 12 weeks of initiating steroids. Dosage modifications for TECENTRIQ for adverse reactions that require management different from these general guidelines are summarized in Table 3 . Table 3: Recommended Dosage Modifications for Adverse Reactions Adverse Reaction Severity Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4 Dosage Modification ALT = alanine aminotransferase, AST = aspartate aminotransferase, ULN = upper limit normal, DRESS = Drug Rash with Eosinophilia and Systemic Symptoms, SJS = Stevens Johnson syndrome, TEN = toxic epidermal necrolysis Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.1) ] Pneumonitis Grade 2 Withhold Resume in patients with complete or partial resolution (Grade 0 to 1) after corticosteroid taper. Permanently discontinue if no complete or partial resolution within 12 weeks of initiating steroids or inability to reduce prednisone to 10 mg per day or less (or equivalent) within 12 weeks of initiating steroids Grades 3 or 4 Permanently discontinue Colitis Grades 2 or 3 Withhold Grade 4 Permanently discontinue Hepatitis with no tumor involvement of the liver AST or ALT increases to more than 3 and up to 8 times ULN or Total bilirubin increases to more than 1.5 and up to 3 times ULN Withhold AST or ALT increases to more than 8 times ULN or Total bilirubin increases to more than 3 times ULN Permanently discontinue Hepatitis with tumor involvement of the liver If AST and ALT are less than or equal to ULN at baseline, withhold or permanently discontinue TECENTRIQ based on recommendations for hepatitis with no liver involvement Baseline AST or ALT is more than 1 and up to 3 times ULN and increases to more than 5 and up to 10 times ULN or Baseline AST or ALT is more than 3 and up to 5 times ULN and increases to more than 8 and up to 10 times ULN Withhold AST or ALT increases to more than 10 times ULN or Total bilirubin increases to more than 3 times ULN Permanently discontinue Endocrinopathies Grades 3 or 4 Withhold until clinically stable or permanently discontinue depending on severity Nephritis with Renal Dysfunction Grades 2 or 3 increased blood creatinine Withhold Grade 4 increased blood creatinine Permanently discontinue Exfoliative Dermatologic Conditions Suspected SJS, TEN, or DRESS Withhold Confirmed SJS, TEN, or DRESS Permanently discontinue Myocarditis or Pericarditis Grades 2, 3, or 4 Permanently discontinue Neurological Toxicities Grade 2 Withhold Grades 3 or 4 Permanently discontinue Other Adverse Reactions Infusion-Related Reactions [see Warnings and Precautions (5.2) ] Grades 1 or 2 Interrupt or slow the rate of infusion Grades 3 or 4 Permanently discontinue
Storage and Handling
TECENTRIQ injection is a sterile, preservative-free, and colorless to slightly yellow solution for intravenous infusion supplied as a carton containing one 840 mg/14 mL single-dose vial (NDC 50242-918-01) or 1,200 mg/20 mL single-dose vial (NDC 50242-917-01).
How Supplied
TECENTRIQ injection is a sterile, preservative-free, and colorless to slightly yellow solution for intravenous infusion supplied as a carton containing one 840 mg/14 mL single-dose vial (NDC 50242-918-01) or 1,200 mg/20 mL single-dose vial (NDC 50242-917-01).
Medication Information
Warnings and Precautions
Immune-Mediated Adverse Reactions Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated dermatologic adverse reactions, immune-mediated nephritis and renal dysfunction, and solid organ transplant rejection. ( 5.1 ) Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. ( 5.1 ) Withhold or permanently discontinue based on severity and type of reaction. ( 5.1 ). Infusion-Related Reactions : Interrupt, slow the rate of infusion, or permanently discontinue based on severity of infusion reactions. ( 5.2 ) Complications of Allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. ( 5.3 ) Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. ( 5.4 , 8.1 , 8.3 )
Indications and Usage
TECENTRIQ is a programmed death-ligand 1 (PD-L1) blocking antibody indicated: Non-Small Cell Lung Cancer (NSCLC) as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage II to IIIA NSCLC whose tumors have PD-L1 expression on ≥ 1% of tumor cells, as determined by an FDA-approved test. ( 1.1 , 14.1 ) for the first-line treatment of adult patients with metastatic NSCLC whose tumors have high PD-L1 expression (PD-L1 stained ≥ 50% of tumor cells [TC ≥ 50%] or PD-L1 stained tumor-infiltrating immune cells [IC] covering ≥ 10% of the tumor area [IC ≥ 10%] ), as determined by an FDA-approved test, with no EGFR or ALK genomic tumor aberrations. ( 1.1 ) in combination with bevacizumab, paclitaxel, and carboplatin, for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations. ( 1.1 ) in combination with paclitaxel protein-bound and carboplatin for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations ( 1.1 ) for the treatment of adult patients with metastatic NSCLC who have disease progression during or following platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for NSCLC harboring these aberrations prior to receiving TECENTRIQ. ( 1.1 ) Small Cell Lung Cancer (SCLC) in combination with carboplatin and etoposide, for the first-line treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC). ( 1.2 ) in combination with lurbinectedin, for the maintenance treatment of adult patients with ES-SCLC whose disease has not progressed after first-line induction therapy with TECENTRIQ or atezolizumab and hyaluronidase-tqjs, carboplatin and etoposide. ( 1.2 ) Hepatocellular Carcinoma (HCC) in combination with bevacizumab for the treatment of adult patients with unresectable or metastatic HCC who have not received prior systemic therapy. ( 1.3 ) Melanoma in combination with cobimetinib and vemurafenib for the treatment of adult patients with BRAF V600 mutation-positive unresectable or metastatic melanoma. ( 1.4 ) Alveolar Soft Part Sarcoma (ASPS) for the treatment of adult and pediatric patients 2 years of age and older with unresectable or metastatic ASPS. ( 1.5 )
Dosage and Administration
Administer TECENTRIQ intravenously over 60 minutes. If the first infusion is tolerated, all subsequent infusions may be delivered over 30 minutes. NSCLC In the adjuvant setting, administer TECENTRIQ following resection and up to 4 cycles of platinum-based chemotherapy as 840 mg every 2 weeks, 1200 mg every 3 weeks or 1680 mg every 4 weeks for up to 1 year. ( 2.2 ) In the metastatic setting, administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. ( 2.2 ) When administering with chemotherapy with or without bevacizumab, administer TECENTRIQ prior to chemotherapy and bevacizumab when given on the same day. ( 2.2 ) Small Cell Lung Cancer Administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. Administer TECENTRIQ prior to chemotherapy when given on the same day. ( 2.2 ) Hepatocellular Carcinoma Administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. Administer TECENTRIQ prior to bevacizumab when given on the same day. Bevacizumab is administered at 15 mg/kg every 3 weeks. ( 2.2 ) Melanoma Following completion of a 28-day cycle of cobimetinib and vemurafenib, administer TECENTRIQ 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks with cobimetinib 60 mg orally once daily (21 days on /7 days off) and vemurafenib 720 mg orally twice daily. ( 2.2 ) ASPS Adults: Administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. ( 2.2 ) Pediatric patients 2 years of age and older: 15 mg/kg (up to a maximum of 1200 mg), every 3 weeks ( 2.2 )
Contraindications
None.
Adverse Reactions
No dose reduction for TECENTRIQ is recommended. In general, withhold TECENTRIQ for severe (Grade 3) immune-mediated adverse reactions. Permanently discontinue TECENTRIQ for life-threatening (Grade 4) immune-mediated adverse reactions, recurrent severe (Grade 3) immune-mediated reactions that require systemic immunosuppressive treatment, or an inability to reduce corticosteroid dose to 10 mg or less of prednisone or equivalent per day within 12 weeks of initiating steroids. Dosage modifications for TECENTRIQ for adverse reactions that require management different from these general guidelines are summarized in Table 3 . Table 3: Recommended Dosage Modifications for Adverse Reactions Adverse Reaction Severity Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4 Dosage Modification ALT = alanine aminotransferase, AST = aspartate aminotransferase, ULN = upper limit normal, DRESS = Drug Rash with Eosinophilia and Systemic Symptoms, SJS = Stevens Johnson syndrome, TEN = toxic epidermal necrolysis Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.1) ] Pneumonitis Grade 2 Withhold Resume in patients with complete or partial resolution (Grade 0 to 1) after corticosteroid taper. Permanently discontinue if no complete or partial resolution within 12 weeks of initiating steroids or inability to reduce prednisone to 10 mg per day or less (or equivalent) within 12 weeks of initiating steroids Grades 3 or 4 Permanently discontinue Colitis Grades 2 or 3 Withhold Grade 4 Permanently discontinue Hepatitis with no tumor involvement of the liver AST or ALT increases to more than 3 and up to 8 times ULN or Total bilirubin increases to more than 1.5 and up to 3 times ULN Withhold AST or ALT increases to more than 8 times ULN or Total bilirubin increases to more than 3 times ULN Permanently discontinue Hepatitis with tumor involvement of the liver If AST and ALT are less than or equal to ULN at baseline, withhold or permanently discontinue TECENTRIQ based on recommendations for hepatitis with no liver involvement Baseline AST or ALT is more than 1 and up to 3 times ULN and increases to more than 5 and up to 10 times ULN or Baseline AST or ALT is more than 3 and up to 5 times ULN and increases to more than 8 and up to 10 times ULN Withhold AST or ALT increases to more than 10 times ULN or Total bilirubin increases to more than 3 times ULN Permanently discontinue Endocrinopathies Grades 3 or 4 Withhold until clinically stable or permanently discontinue depending on severity Nephritis with Renal Dysfunction Grades 2 or 3 increased blood creatinine Withhold Grade 4 increased blood creatinine Permanently discontinue Exfoliative Dermatologic Conditions Suspected SJS, TEN, or DRESS Withhold Confirmed SJS, TEN, or DRESS Permanently discontinue Myocarditis or Pericarditis Grades 2, 3, or 4 Permanently discontinue Neurological Toxicities Grade 2 Withhold Grades 3 or 4 Permanently discontinue Other Adverse Reactions Infusion-Related Reactions [see Warnings and Precautions (5.2) ] Grades 1 or 2 Interrupt or slow the rate of infusion Grades 3 or 4 Permanently discontinue
Storage and Handling
TECENTRIQ injection is a sterile, preservative-free, and colorless to slightly yellow solution for intravenous infusion supplied as a carton containing one 840 mg/14 mL single-dose vial (NDC 50242-918-01) or 1,200 mg/20 mL single-dose vial (NDC 50242-917-01).
How Supplied
TECENTRIQ injection is a sterile, preservative-free, and colorless to slightly yellow solution for intravenous infusion supplied as a carton containing one 840 mg/14 mL single-dose vial (NDC 50242-918-01) or 1,200 mg/20 mL single-dose vial (NDC 50242-917-01).
Description
Warnings and Precautions ( 5.2 ) 08/2025 Indications and Usage ( 1.2 ) 10/2025
Section 42229-5
Preparation
Visually inspect drug product for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard the vial if the solution is cloudy, discolored, or visible particles are observed. Do not shake the vial.
Prepare the solution for infusion as follows:
- Select the appropriate vial(s) based on the prescribed dose.
- Withdraw the required volume of TECENTRIQ from the vial(s) using sterile needle and syringe.
- Dilute to a final concentration between 3.2 mg/mL and 16.8 mg/mL in a polyvinyl chloride (PVC), polyethylene (PE), or polyolefin (PO) infusion bag containing 0.9% Sodium Chloride Injection, USP.
- Dilute with only 0.9% Sodium Chloride Injection, USP.
- Mix diluted solution by gentle inversion. Do not shake.
- Discard used or empty vials of TECENTRIQ.
Section 42231-1
| MEDICATION GUIDE TECENTRIQ® (te-SEN-trik) (atezolizumab) Injection |
|||||
|---|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 10/2025 | ||||
| What is the most important information I should know about TECENTRIQ? | |||||
| TECENTRIQ is a medicine that may treat certain cancers by working with your immune system. TECENTRIQ can cause your immune system to attack normal organs and tissues in any area of your body and can affect the way they work. These problems can sometimes become severe or life-threatening and can lead to death. You can have more than one of these problems at the same time. These problems may happen anytime during your treatment or even after your treatment has ended. | |||||
| Call or see your healthcare provider right away if you develop any new or worse signs or symptoms, including: Lung problems. | |||||
|
|
|
|||
| Intestinal problems. | |||||
|
|||||
| Liver problems. | |||||
|
|
||||
| Hormone gland problems. | |||||
|
|
||||
| Kidney problems. | |||||
|
|
||||
| Skin problems. | |||||
|
|
||||
|
Problems can also happen in other organs. These are not all of the signs and symptoms of immune system problems that can happen with TECENTRIQ. Call or see your healthcare provider right away for any new or worse signs or symptoms, including: |
|||||
|
|||||
| Infusion reactions that can sometimes be severe or life-threatening. Signs and symptoms of infusion reactions may include: | |||||
|
|
||||
| Complications, including graft-versus-host disease (GVHD), in people who have received a bone marrow (stem cell) transplant that uses donor stem cells (allogeneic). These complications can be serious and can lead to death. These complications may happen if you underwent transplantation either before or after being treated with TECENTRIQ. Your healthcare provider will monitor you for these complications. | |||||
|
Getting medical treatment right away may help keep these problems from becoming more serious.
Your healthcare provider will check you for these problems during your treatment with TECENTRIQ. Your healthcare provider may treat you with corticosteroid or hormone replacement medicines. Your healthcare provider may also need to delay or completely stop treatment with TECENTRIQ if you have severe side effects. |
|||||
| What is TECENTRIQ? | |||||
| TECENTRIQ is a prescription medicine used to treat: | |||||
|
|||||
It is not known if TECENTRIQ is safe and effective when used:
|
|||||
| Before receiving TECENTRIQ, tell your healthcare provider about all of your medical conditions, including if you: | |||||
|
|||||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||||
| How will I receive TECENTRIQ? | |||||
|
|||||
| What are the possible side effects of TECENTRIQ? | |||||
| TECENTRIQ can cause serious side effects, including: | |||||
| The most common side effects of TECENTRIQ when used alone include: | |||||
|
|
|
|||
| The most common side effects of TECENTRIQ when used in lung cancer with other anti-cancer medicines include: | |||||
|
|
|
|||
| The most common side effects of TECENTRIQ when used in hepatocellular carcinoma with bevacizumab include: | |||||
|
|
|
|||
| The most common side effects of TECENTRIQ when used in melanoma with cobimetinib and vemurafenib include: | |||||
|
|
|
|||
| TECENTRIQ may cause fertility problems in females, which may affect the ability to have children. Talk to your healthcare provider if you have concerns about fertility. | |||||
| These are not all the possible side effects of TECENTRIQ. | |||||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||||
| General information about the safe and effective use of TECENTRIQ. | |||||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about TECENTRIQ that is written for health professionals. | |||||
| What are the ingredients in TECENTRIQ? | |||||
| Active ingredient: atezolizumab | |||||
| Inactive ingredients: glacial acetic acid, L-histidine, polysorbate 20 and sucrose | |||||
| Manufactured by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 USA | |||||
| U.S. License No.: 1048 TECENTRIQ is a registered trademark of Genentech, Inc. | |||||
| For more information, call 1-844-832-3687 or go to www.TECENTRIQ.com. |
Section 44425-7
Store vials under refrigeration at 2°C to 8°C (36°F to 46°F) in original carton to protect from light. Do not freeze. Do not shake.
1.4 Melanoma
TECENTRIQ, in combination with cobimetinib and vemurafenib, is indicated for the treatment of adult patients with BRAF V600 mutation-positive unresectable or metastatic melanoma [see Dosage and Administration (2.1)].
14.4 Melanoma
The efficacy of TECENTRIQ in combination with cobimetinib and vemurafenib was evaluated in a double-blind, randomized (1:1), placebo-controlled, multicenter trial (IMspire150; NCT02908672) conducted in 514 patients. Randomization was stratified by geographic location (North America vs. Europe vs. Australia, New Zealand, and others) and baseline lactate dehydrogenase (LDH) [less than or equal to upper limit of normal (ULN) vs. greater than ULN]. Eligible patients were required to have previously untreated unresectable or metastatic BRAF V600 mutation-positive melanoma as detected by a locally available test and centrally confirmed with the FoundationOne™ assay. Patients were excluded if they had history of autoimmune disease; administration of a live, attenuated vaccine within 28 days prior to randomization; administration of systemic immunostimulatory agents within 4 weeks or systemic immunosuppressive medications within 2 weeks prior to randomization; and active or untreated CNS metastases.
TECENTRIQ was initiated after patients received a 28-day treatment cycle of cobimetinib 60 mg orally once daily (21 days on / 7 days off) and vemurafenib 960 mg orally twice daily Days 1-21 and 720 mg orally twice daily Days 22-28. Patients received TECENTRIQ 840 mg intravenous infusion over 60 minutes every 2 weeks in combination with cobimetinib 60 mg orally once daily and vemurafenib 720 mg orally twice daily, or placebo in combination with cobimetinib 60 mg orally once daily and vemurafenib 960 mg orally twice daily. Treatment continued until disease progression or unacceptable toxicity. There was no crossover at the time of disease progression. Tumor assessments were performed every 8 weeks (± 1 week) for the first 24 months and every 12 weeks (± 1 week) thereafter.
The major efficacy outcome measure was investigator-assessed progression-free survival (PFS) per RECIST v1.1. Additional efficacy outcomes included PFS assessed by an independent central review, investigator-assessed ORR, OS, and DOR.
The median age of the study population was 54 years (range: 22-88), 58% of patients were male, 95% were White, a baseline ECOG performance status of 0 (77%) or 1 (23%), 33% had elevated LDH, 94% had metastatic disease, 60% were Stage IV (M1C), 56% had less than three metastatic sites at baseline, 3% had prior treatment for brain metastases, 30% had liver metastases at baseline, and 14% had received prior adjuvant systemic therapy. Based on central testing, 74% were identified as having a V600E mutation, 11% as having V600K mutation, and 1% as having V600D or V600R mutations.
Efficacy results are summarized in Table 32 and Figure 10. Patients had a median survival follow up time of 18.9 months.
| TECENTRIQ + Cobimetinib + Vemurafenib N=256 |
Placebo + Cobimetinib + Vemurafenib N=258 |
|
|---|---|---|
|
Progression-Free Survival
As determined by investigator assessment with Response Evaluation Criteria in Solid Tumors v1.1.; CI=confidence interval;
|
||
| Number of events (%) | 148 (58) | 179 (69) |
| Median, months | 15.1 | 10.6 |
| (95% CI) | (11.4, 18.4) | (9.3, 12.7) |
| Hazard ratio Stratified by baseline LDH (95% CI) |
0.78 (0.63, 0.97) | |
| p-value Based on the stratified log-rank test
|
0.0249 | |
|
Overall Response Rate
,
Confirmed Responses
|
||
| Number of responders (%) | 170 (66) | 168 (65) |
| (95% CI) | (60, 72) | (59, 71) |
| Complete responses, n (%) | 41 (16) | 46 (18) |
| Partial response, n (%) | 129 (50) | 122 (47) |
| Duration of Response , | n=170 | n=168 |
| Median, months | 20.4 | 12.5 |
| (95% CI) | (15.1, NE) | (10.7, 16.6) |
Figure 10: Kaplan-Meier Plot for Progression-Free Survival in IMspire150
At a pre-specified analysis at the time of the primary analysis of PFS, the OS data were not mature. The median OS was 28.8 months with 93 (36%) deaths in the TECENTRIQ plus cobimetinib and vemurafenib arm, and 25.1 months with 112 (43%) deaths in the placebo plus cobimetinib and vemurafenib arm. The hazard ratio for OS was 0.85 (95% CI: 0.64, 1.11) and the p-value was 0.2310.
11 Description
Atezolizumab is a programmed cell death ligand 1 (PD-L1) blocking antibody. Atezolizumab is an Fc-engineered, humanized, non-glycosylated IgG1 kappa immunoglobulin that has a calculated molecular mass of 145 kDa.
TECENTRIQ (atezolizumab) injection for intravenous use is a sterile, preservative-free, colorless to slightly yellow solution in single-dose vials. Each 20 mL vial contains 1200 mg of atezolizumab and is formulated in glacial acetic acid (16.5 mg), L-histidine (62 mg), polysorbate 20 (8 mg), and sucrose (821.6 mg), with a pH of 5.8. Each 14 mL vial contains 840 mg of atezolizumab and is formulated in glacial acetic acid (11.5 mg), L-histidine (43.4 mg), polysorbate 20 (5.6 mg), and sucrose (575.1 mg) with a pH of 5.8.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of ADA in other products.
During the first year of treatment with TECENTRIQ across 8 clinical studies, 13% to 36% of patients developed anti-atezolizumab antibodies. Median atezolizumab clearance in patients who tested positive for ADA was 19% (minimum 18%, maximum 49%) higher as compared to atezolizumab clearance in patients who tested negative for ADA; this change in clearance is not expected to be clinically significant.
In OAK and IMbrave150, exploratory analyses showed that the subset of patients who were ADA-positive appeared to have less efficacy (effect on overall survival) as compared to patients who tested negative for ADA [see Clinical Studies (14.1, 14.3)]. In study IMpower150 and IMforte, the impact of ADA on efficacy did not appear to be clinically significant [see Clinical Studies (14.1)]. In the remaining studies, there is insufficient information to characterize the effect of ADA on efficacy.
The presence of ADA did not have a clinically significant effect on the incidence or severity of adverse reactions.
Across clinical studies, the incidence of neutralizing antibodies (NAb) in ADA-positive patients was 24% to 83%. The effect of NAb on atezolizumab exposure and safety did not appear to be clinically significant. The effect of NAb on key efficacy endpoints is uncertain due to small sample sizes.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Severe and Fatal Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.1)]
- Infusion-Related Reactions [see Warnings and Precautions (5.2)]
- Complications of Allogeneic HSCT after PD-1/PD-L1 Inhibitors [see Warnings and Precautions (5.3)]
12.2 Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of atezolizumab have not been fully characterized.
12.3 Pharmacokinetics
Atezolizumab exposure increased dose proportionally over the dose range of 1 mg/kg to 20 mg/kg (0.07 to 1.33 times of the approved recommended doses), including a dose of 1200 mg administered every 3 weeks. Steady state was achieved after 6 to 9 weeks following multiple doses. The systemic accumulation ratio for every 2 weeks administration and every 3 weeks administration is 3.3- and 1.9- fold, respectively.
2.2 Recommended Dosage
The recommended dosages of TECENTRIQ are presented in Table 1. Administer TECENTRIQ as an intravenous infusion over 60 minutes. If the first infusion is tolerated, all subsequent infusions may be delivered over 30 minutes.
| Indication | Recommended Dosage of TECENTRIQ | Duration of Therapy |
|---|---|---|
| Metastatic NSCLC |
|
Until disease progression or unacceptable toxicity |
| Adjuvant Treatment of NSCLC |
|
Up to one year, unless there is disease recurrence or unacceptable toxicity |
| ASPS (adult) |
|
Until disease progression or unacceptable toxicity |
| ASPS (pediatric, 2 years of age and older) | 15 mg/kg (up to a maximum 1200 mg) every 3 weeks |
The recommended intravenous dosages of TECENTRIQ in combination with other therapeutic agents are presented in Table 2. Refer to the respective Prescribing Information for each therapeutic agent administered in combination with TECENTRIQ for the recommended dosage information, as appropriate.
| Indication | Recommended Dosage of TECENTRIQ | Duration of Therapy |
|---|---|---|
| NSCLC |
|
Until disease progression or unacceptable toxicity |
| SCLC |
|
|
| HCC |
|
|
| Melanoma |
Prior to initiating TECENTRIQ, patients should receive a 28-day treatment cycle of cobimetinib 60 mg orally once daily (21 days on and 7 days off) and vemurafenib 960 mg orally twice daily from Days 1-21 and vemurafenib 720 mg orally twice daily from Days 22-28. |
1 Indications and Usage
TECENTRIQ is a programmed death-ligand 1 (PD-L1) blocking antibody indicated:
Non-Small Cell Lung Cancer (NSCLC)
- as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage II to IIIA NSCLC whose tumors have PD-L1 expression on ≥ 1% of tumor cells, as determined by an FDA-approved test. (1.1, 14.1)
- for the first-line treatment of adult patients with metastatic NSCLC whose tumors have high PD-L1 expression (PD-L1 stained ≥ 50% of tumor cells [TC ≥ 50%] or PD-L1 stained tumor-infiltrating immune cells [IC] covering ≥ 10% of the tumor area [IC ≥ 10%] ), as determined by an FDA-approved test, with no EGFR or ALK genomic tumor aberrations. (1.1)
- in combination with bevacizumab, paclitaxel, and carboplatin, for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations. (1.1)
- in combination with paclitaxel protein-bound and carboplatin for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations (1.1)
- for the treatment of adult patients with metastatic NSCLC who have disease progression during or following platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for NSCLC harboring these aberrations prior to receiving TECENTRIQ. (1.1)
Small Cell Lung Cancer (SCLC)
- in combination with carboplatin and etoposide, for the first-line treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC). (1.2)
- in combination with lurbinectedin, for the maintenance treatment of adult patients with ES-SCLC whose disease has not progressed after first-line induction therapy with TECENTRIQ or atezolizumab and hyaluronidase-tqjs, carboplatin and etoposide. (1.2)
Hepatocellular Carcinoma (HCC)
- in combination with bevacizumab for the treatment of adult patients with unresectable or metastatic HCC who have not received prior systemic therapy. ( 1.3)
Melanoma
- in combination with cobimetinib and vemurafenib for the treatment of adult patients with BRAF V600 mutation-positive unresectable or metastatic melanoma. (1.4)
Alveolar Soft Part Sarcoma (ASPS)
- for the treatment of adult and pediatric patients 2 years of age and older with unresectable or metastatic ASPS. (1.5)
12.1 Mechanism of Action
PD L1 may be expressed on tumor cells and/or tumor infiltrating immune cells and can contribute to the inhibition of the anti-tumor immune response in the tumor microenvironment. Binding of PD L1 to the PD 1 and B7.1 receptors found on T cells and antigen presenting cells suppresses cytotoxic T-cell activity, T-cell proliferation and cytokine production.
Atezolizumab is a monoclonal antibody that binds to PD L1 and blocks its interactions with both PD 1 and B7.1 receptors. This releases the PD L1/PD 1 mediated inhibition of the immune response, including activation of the anti-tumor immune response without inducing antibody-dependent cellular cytotoxicity. In syngeneic mouse tumor models, blocking PD L1 activity resulted in decreased tumor growth.
In mouse models of cancer, dual inhibition of the PD-1/PD-L1 and MAPK pathways suppresses tumor growth and improves tumor immunogenicity through increased antigen presentation and T cell infiltration and activation compared to targeted therapy alone.
5.4 Embryo Fetal Toxicity
Based on its mechanism of action, TECENTRIQ can cause fetal harm when administered to a pregnant woman. There are no available data on the use of TECENTRIQ in pregnant women. Animal studies have demonstrated that inhibition of the PD-L1/PD-1 pathway can lead to increased risk of immune-related rejection of the developing fetus resulting in fetal death.
Verify pregnancy status of females of reproductive potential prior to initiating TECENTRIQ. Advise females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with TECENTRIQ and for at least 5 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
1.2 Small Cell Lung Cancer
- TECENTRIQ, in combination with carboplatin and etoposide, is indicated for the first-line treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC).
- TECENTRIQ, in combination with lurbinectedin, is indicated for the maintenance treatment of adult patients with ES-SCLC whose disease has not progressed after first-line induction therapy with TECENTRIQ or atezolizumab and hyaluronidase-tqjs, carboplatin and etoposide.
5 Warnings and Precautions
-
Immune-Mediated Adverse Reactions
- Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated dermatologic adverse reactions, immune-mediated nephritis and renal dysfunction, and solid organ transplant rejection. (5.1)
- Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. (5.1)
- Withhold or permanently discontinue based on severity and type of reaction. (5.1).
- Infusion-Related Reactions: Interrupt, slow the rate of infusion, or permanently discontinue based on severity of infusion reactions. (5.2)
- Complications of Allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. (5.3)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. (5.4, 8.1, 8.3)
2 Dosage and Administration
Administer TECENTRIQ intravenously over 60 minutes. If the first infusion is tolerated, all subsequent infusions may be delivered over 30 minutes.
NSCLC
- In the adjuvant setting, administer TECENTRIQ following resection and up to 4 cycles of platinum-based chemotherapy as 840 mg every 2 weeks, 1200 mg every 3 weeks or 1680 mg every 4 weeks for up to 1 year. (2.2)
- In the metastatic setting, administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. (2.2)
- When administering with chemotherapy with or without bevacizumab, administer TECENTRIQ prior to chemotherapy and bevacizumab when given on the same day. (2.2)
Small Cell Lung Cancer
- Administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. Administer TECENTRIQ prior to chemotherapy when given on the same day. (2.2)
Hepatocellular Carcinoma
- Administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. Administer TECENTRIQ prior to bevacizumab when given on the same day. Bevacizumab is administered at 15 mg/kg every 3 weeks. (2.2)
Melanoma
- Following completion of a 28-day cycle of cobimetinib and vemurafenib, administer TECENTRIQ 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks with cobimetinib 60 mg orally once daily (21 days on /7 days off) and vemurafenib 720 mg orally twice daily. (2.2)
ASPS
1.3 Hepatocellular Carcinoma
TECENTRIQ, in combination with bevacizumab, is indicated for the treatment of adult patients with unresectable or metastatic hepatocellular carcinoma (HCC) who have not received prior systemic therapy.
3 Dosage Forms and Strengths
Injection: 840 mg/14 mL (60 mg/mL) and 1200 mg/20 mL (60 mg/mL) colorless to slightly yellow solution in a single-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of TECENTRIQ. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Cardiac: pericarditis, pericardial effusion, cardiac tamponade
- Musculoskeletal and Connective Tissue: tenosynovitis
14.3 Hepatocellular Carcinoma
The efficacy of TECENTRIQ in combination with bevacizumab was investigated in IMbrave150 (NCT03434379), a multicenter, international, open-label, randomized trial in patients with locally advanced unresectable and/or metastatic hepatocellular carcinoma who have not received prior systemic therapy. Randomization was stratified by geographic region (Asia excluding Japan vs. rest of world), macrovascular invasion and/or extrahepatic spread (presence vs. absence), baseline AFP (<400 vs. ≥400 ng/mL), and by ECOG performance status (0 vs. 1).
A total of 501 patients were randomized (2:1) to receive either TECENTRIQ as an intravenous infusion of 1200 mg, followed by 15 mg/kg bevacizumab, on the same day every 3 weeks or sorafenib 400 mg given orally twice daily, until disease progression or unacceptable toxicity. Patients could discontinue either TECENTRIQ or bevacizumab (e.g., due to adverse events) and continue on single-agent therapy until disease progression or unacceptable toxicity associated with the single-agent.
The study enrolled patients who were ECOG performance score 0 or 1 and who had not received prior systemic treatment. Patients were required to be evaluated for the presence of varices within 6 months prior to treatment, and were excluded if they had variceal bleeding within 6 months prior to treatment, untreated or incompletely treated varices with bleeding, or high risk of bleeding. Patients with Child-Pugh B or C cirrhosis, moderate or severe ascites; history of hepatic encephalopathy; a history of autoimmune disease; administration of a live, attenuated vaccine within 4 weeks prior to randomization; administration of systemic immunostimulatory agents within 4 weeks or systemic immunosuppressive medications within 2 weeks prior to randomization; or untreated or corticosteroid-dependent brain metastases were excluded. Tumor assessments were performed every 6 weeks for the first 54 weeks and every 9 weeks thereafter.
The demographics and baseline disease characteristics of the study population were balanced between the treatment arms. The median age was 65 years (range: 26 to 88) and 83% of patients were male. The majority of patients were Asian (57%) or White (35%); 40% were from Asia (excluding Japan). Approximately 75% of patients presented with macrovascular invasion and/or extrahepatic spread and 37% had a baseline AFP ≥400 ng/mL. Baseline ECOG performance status was 0 (62%) or 1 (38%). HCC risk factors were Hepatitis B in 48% of patients, Hepatitis C in 22%, and 31% of patients had non-viral liver disease. The majority of patients had BCLC stage C (82%) disease at baseline, while 16% had stage B, and 3% had stage A.
The major efficacy outcome measures were overall survival (OS) and independent review facility (IRF)-assessed progression free survival (PFS) per RECIST v1.1. Additional efficacy outcome measures were IRF-assessed overall response rate (ORR) per RECIST and mRECIST.
Efficacy results are presented in Table 31 and Figure 9.
| TECENTRIQ in combination with Bevacizumab (N= 336) |
Sorafenib (N=165) |
|
|---|---|---|
| CI=confidence interval; HCC mRECIST=Modified RECIST Assessment for Hepatocellular Carcinoma; NE=not estimable; RECIST 1.1=Response Evaluation Criteria in Solid Tumors v1.1 | ||
| Overall Survival | ||
| Number of deaths (%) | 96 (29) | 65 (39) |
| Median OS in months (95% CI) | NE (NE, NE) |
13.2 (10.4, NE) |
| Hazard ratio Stratified by geographic region (Asia excluding Japan vs. rest of world), macrovascular invasion and/or extrahepatic spread (presence vs. absence), and baseline AFP (<400 vs. ≥400 ng/mL) (95% CI) |
0.58 (0.42, 0.79) | |
| p-value Based on two-sided stratified log-rank test; as compared to significance level 0.004 (2-sided) based on 161/312=52% information using the OBF method
|
0.0006 | |
|
Progression-Free Survival Per independent radiology review
|
||
| Number of events (%) | 197 (59) | 109 (66) |
| Median PFS in months (95% CI) | 6.8 (5.8, 8.3) | 4.3 (4.0, 5.6) |
| Hazard ratio (95% CI) | 0.59 (0.47, 0.76) | |
| p-value | <0.0001 | |
|
Overall Response Rate
,
Confirmed responses (ORR), RECIST 1.1
|
||
| Number of responders (%) | 93 (28) | 19 (12) |
| (95% CI) | (23, 33) | (7,17) |
| p-value Based on two-sided Cochran-Mantel-Haesnszel test
|
<0.0001 | |
| Complete responses, n (%) | 22 (7) | 0 |
| Partial responses, n (%) | 71 (21) | 19 (12) |
| Duration of Response , (DOR) RECIST 1.1 | ||
| (n=93) | (n=19) | |
| Median DOR in months (95% CI) | NE (NE, NE) |
6.3 (4.7, NE) |
| Range (months) | (1.3 Denotes a censored value , 13.4) |
(1.4, 9.1) |
| Overall Response Rate , (ORR), HCC mRECIST | ||
| Number of responders (%) | 112 (33) | 21 (13) |
| (95% CI) | (28, 39) | (8, 19) |
| p-value | <0.0001 | |
| Complete responses, n (%) | 37 (11) | 3 (1.8) |
| Partial responses, n (%) | 75 (22) | 18 (11) |
| Duration of Response , (DOR) HCC mRECIST | ||
| (n=112) | (n=21) | |
| Median DOR in months (95% CI) |
NE (NE, NE) |
6.3 (4.9, NE) |
| Range (months) | (1.3, 13.4) | (1.4, 9.1) |
Figure 9: Kaplan-Meier Plot of Overall Survival in IMbrave150
Exploratory analyses showed that the subset of patients (20%) who were ADA-positive by week 6 appeared to have reduced efficacy (effect on OS) as compared to patients (80%) who tested negative for treatment-emergent ADA by week 6 [see Clinical Pharmacology (12.6)]. ADA-positive patients by week 6 appeared to have similar overall survival compared to sorafenib-treated patients. In an exploratory analysis, inverse probability weighting was conducted to compare ADA-positive patients and ADA-negative patients in the TECENTRIQ and bevacizumab arm to the sorafenib arm. Inverse probability weighting factors were: baseline sum of longest tumor size (BSLD), baseline ECOG, baseline albumin, baseline LDH, sex, age, race, geographic region, weight, neutrophil-to-lymphocyte ratio, AFP (<400 ng/mL vs ≥400 ng/mL), number of metastatic sites, MVI and/or EHS present at study entry, etiology (HBV vs. HCV vs. non-viral) and Child-Pugh Score (A5 vs. A6). The OS hazard ratio comparing the ADA-positive subgroup of the TECENTRIQ and bevacizumab arm to sorafenib was 0.93 (95% CI: 0.57, 1.53). The OS hazard ratio comparing the ADA-negative subgroup to sorafenib was 0.39 (95% CI: 0.26, 0.60).
8 Use in Specific Populations
Lactation: Advise not to breastfeed. (8.2)
1.1 Non Small Cell Lung Cancer
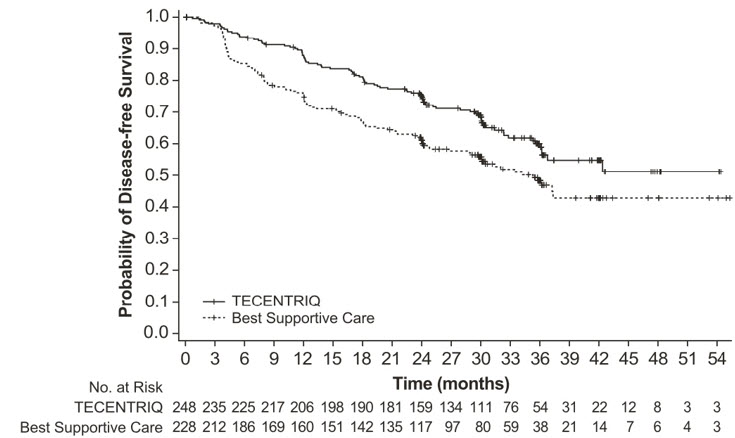
- TECENTRIQ, as a single-agent, is indicated as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with stage II to IIIA [see Clinical Studies (14.1)] non-small cell lung cancer (NSCLC) whose tumors have PD-L1 expression on ≥ 1% of tumor cells, as determined by an FDA-approved test [see Dosage and Administration (2.1)].
- TECENTRIQ, as a single agent, is indicated for the first-line treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors have high PD-L1 expression (PD-L1 stained ≥ 50% of tumor cells [TC ≥ 50%] or PD-L1 stained tumor-infiltrating immune cells [IC] covering ≥ 10% of the tumor area [IC ≥ 10%]), as determined by an FDA-approved test, with no EGFR or ALK genomic tumor aberrations [see Dosage and Administration (2.1)].
- TECENTRIQ, in combination with bevacizumab, paclitaxel, and carboplatin, is indicated for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations.
- TECENTRIQ, in combination with paclitaxel protein-bound and carboplatin, is indicated for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations.
- TECENTRIQ, as a single-agent, is indicated for the treatment of adult patients with metastatic NSCLC who have disease progression during or following platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for NSCLC harboring these aberrations prior to receiving TECENTRIQ.
1.5 Alveolar Soft Part Sarcoma
TECENTRIQ, as a single agent, is indicated for the treatment of adult and pediatric patients 2 years of age and older with unresectable or metastatic alveolar soft part sarcoma (ASPS).
5.2 Infusion Related Reactions
TECENTRIQ can cause severe or life-threatening infusion-related reactions, including anaphylaxis. Monitor for signs and symptoms of infusion-related reactions. Interrupt, slow the rate of, or permanently discontinue TECENTRIQ based on the severity [see Dosage and Administration (2.3)]. For Grade 1 or 2 infusion-related reactions, consider using pre-medications with subsequent doses.
In clinical studies enrolling 2616 patients with various cancers who received TECENTRIQ as a single-agent [see Adverse Reactions (6.1)], infusion-related reactions occurred in 1.3% of patients, including Grade 3 (0.2%). The frequency and severity of infusion-related reactions were similar whether TECENTRIQ was given as a single-agent in patients with various cancers, in combination with other antineoplastic drugs in NSCLC and SCLC, and across the recommended dose range (840 mg Q2W to 1680 mg Q4W).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described in WARNINGS AND PRECAUTIONS reflect exposure to TECENTRIQ as a single-agent in 2616 patients in two randomized, active-controlled studies (POPLAR, OAK) and three open-label, single arm studies (PCD4989g, BIRCH, FIR) which enrolled 1636 patients with metastatic NSCLC, and 980 patients with other tumor types. TECENTRIQ was administered at a dose of 1200 mg intravenously every 3 weeks in all studies except PCD4989g. Among the 2616 patients who received a single-agent TECENTRIQ, 36% were exposed for longer than 6 months and 20% were exposed for longer than 12 months. Using the dataset described for patients who received TECENTRIQ as a single-agent, the most common adverse reactions in ≥ 20% of patients were fatigue/asthenia (48%), decreased appetite (25%), nausea (24%), cough (22%), and dyspnea (22%). In addition, the data reflect exposure to TECENTRIQ as a single agent as adjuvant therapy in 495 patients with early-stage NSCLC enrolled in a randomized study (IMpower010).
In addition, the data reflect exposure to TECENTRIQ in combination with other antineoplastic drugs in 2421 patients with NSCLC (N = 2223) or SCLC (N = 198) enrolled in five randomized, active-controlled trials, including IMpower150, IMpower130 and IMpower133. Among the 2421 patients, 53% were exposed to TECENTRIQ for longer than 6 months and 29% were exposed to TECENTRIQ for longer than 12 months. Among the 2421 patients with NSCLC and SCLC who received TECENTRIQ in combination with other antineoplastic drugs, the most common adverse reactions in ≥20% of patients were fatigue/asthenia (49%), nausea (38%), alopecia (35%), constipation (29%), diarrhea (28%) and decreased appetite (27%).
The data also reflect exposure to TECENTRIQ administered in combination with cobimetinib and vemurafenib in 230 patients enrolled in IMspire150. Among the 230 patients, 62% were exposed to TECENTRIQ for longer than 6 months and 42% were exposed to TECENTRIQ for longer than 12 months.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
TECENTRIQ injection is a sterile, preservative-free, and colorless to slightly yellow solution for intravenous infusion supplied as a carton containing one 840 mg/14 mL single-dose vial (NDC 50242-918-01) or 1,200 mg/20 mL single-dose vial (NDC 50242-917-01).
14.5 Alveolar Soft Part Sarcoma (asps)
The efficacy of TECENTRIQ was evaluated in study ML39345 (NCT03141684), an open-label, single-arm study, in 49 adult and pediatric patients aged 2 years and older with unresectable or metastatic ASPS. Eligible patients were required to have histologically or cytologically confirmed ASPS that was not curable by surgery, and an ECOG performance status of ≤ 2.
Patients were excluded if they had known primary central nervous system (CNS) malignancy or symptomatic CNS metastases, known clinically significant liver disease, or history of idiopathic pulmonary fibrosis, pneumonitis, organizing pneumonia, or evidence of active pneumonitis on screening chest computed tomography (CT) scan.
Adult patients received 1200 mg intravenously and pediatric patients received 15 mg/kg (up to a maximum of 1200 mg) intravenously once every 21 days until disease progression or unacceptable toxicity.
The major efficacy outcomes were Overall Response Rate (ORR) and Duration of Response (DOR) by Independent Review Committee according to Response Evaluation Criteria in Solid Tumors (RECIST) v1.1.
A total of 49 patients were enrolled. The median age of patients was 31 years (range: 12-70); 2% of adult patients (n=47) were ≥65 years of age and the pediatric patients (n=2) were ≥12 years of age; 51% of patients were female, 55% White, 29% Black or African American, 10% Asian; 53% had an ECOG performance status of 0 and 45% had an ECOG performance status of 1. All patients had prior surgery for ASPS and 55% received at least one prior line of treatment for ASPS; 55% received radiotherapy and 53% received chemotherapy. Of the patients who reported staging at initial diagnosis, all were Stage IV.
Efficacy results of this study are summarized in Table 33.
| Endpoint | All Patients (N=49) |
|---|---|
| CI: confidence interval; N: number of patients; +: Censored | |
|
Overall response rate (95% CI) 95% CI based on Clopper–Pearson exact method.
|
24% (13, 39) |
| Complete Responses, n | 0 |
| Partial Responses, n (%) | 12 (24) |
| Duration of response | |
| Median, month | NE |
| (95% CI) | (17.0, NE) |
| Range | 1+, 41+ |
| Durability of Response | |
| ≥6 months, n (%) | 8 (67%) |
| ≥12 months, n (%) | 5 (42%) |
13.2 Animal Toxicology And/or Pharmacology
In animal models, inhibition of PD-L1/PD-1 signaling increased the severity of some infections and enhanced inflammatory responses. M. tuberculosis-infected PD-1 knockout mice exhibit markedly decreased survival compared with wild-type controls, which correlated with increased bacterial proliferation and inflammatory responses in these animals. PD-L1 and PD-1 knockout mice and mice receiving PD-L1 blocking antibody have also shown decreased survival following infection with lymphocytic choriomeningitis virus.
Principal Display Panel 14 Ml Vial Carton
NDC 50242-918-01
Tecentriq®
(atezolizumab)
Injection
840 mg/14 mL
(60 mg/mL)
For Intravenous Infusion After Dilution
Single-Dose Vial
Discard Unused Portion
No preservative.
Attention Pharmacist: Dispense the
accompanying Medication Guide
to each patient.
1 vial
Rx only
Genentech
10198487
Principal Display Panel 20 Ml Vial Carton
NDC 50242-917-01
Tecentriq®
(atezolizumab)
Injection
1200 mg/20 mL
(60 mg/mL)
For Intravenous Infusion After Dilution
Single-Dose Vial
Discard Unused Portion
No preservative.
Attention Pharmacist: Dispense the
accompanying Medication Guide
to each patient.
1 vial
Rx only
Genentech
10199144
2.3 Dosage Modifications for Adverse Reactions
No dose reduction for TECENTRIQ is recommended. In general, withhold TECENTRIQ for severe (Grade 3) immune-mediated adverse reactions. Permanently discontinue TECENTRIQ for life-threatening (Grade 4) immune-mediated adverse reactions, recurrent severe (Grade 3) immune-mediated reactions that require systemic immunosuppressive treatment, or an inability to reduce corticosteroid dose to 10 mg or less of prednisone or equivalent per day within 12 weeks of initiating steroids.
Dosage modifications for TECENTRIQ for adverse reactions that require management different from these general guidelines are summarized in Table 3.
| Adverse Reaction | Severity Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4
|
Dosage Modification |
|---|---|---|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase, ULN = upper limit normal, DRESS = Drug Rash with Eosinophilia and Systemic Symptoms, SJS = Stevens Johnson syndrome, TEN = toxic epidermal necrolysis | ||
| Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.1)] | ||
| Pneumonitis | Grade 2 | Withhold Resume in patients with complete or partial resolution (Grade 0 to 1) after corticosteroid taper. Permanently discontinue if no complete or partial resolution within 12 weeks of initiating steroids or inability to reduce prednisone to 10 mg per day or less (or equivalent) within 12 weeks of initiating steroids
|
| Grades 3 or 4 | Permanently discontinue | |
| Colitis | Grades 2 or 3 | Withhold |
| Grade 4 | Permanently discontinue | |
| Hepatitis with no tumor involvement of the liver | AST or ALT increases to more than 3 and up to 8 times ULN or Total bilirubin increases to more than 1.5 and up to 3 times ULN |
Withhold |
| AST or ALT increases to more than 8 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue | |
| Hepatitis with tumor involvement of the liver If AST and ALT are less than or equal to ULN at baseline, withhold or permanently discontinue TECENTRIQ based on recommendations for hepatitis with no liver involvement
|
Baseline AST or ALT is more than 1 and up to 3 times ULN and increases to more than 5 and up to 10 times ULN or Baseline AST or ALT is more than 3 and up to 5 times ULN and increases to more than 8 and up to 10 times ULN |
Withhold |
| AST or ALT increases to more than 10 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue | |
| Endocrinopathies | Grades 3 or 4 | Withhold until clinically stable or permanently discontinue depending on severity |
| Nephritis with Renal Dysfunction | Grades 2 or 3 increased blood creatinine | Withhold |
| Grade 4 increased blood creatinine | Permanently discontinue | |
| Exfoliative Dermatologic Conditions | Suspected SJS, TEN, or DRESS | Withhold |
| Confirmed SJS, TEN, or DRESS | Permanently discontinue | |
| Myocarditis or Pericarditis | Grades 2, 3, or 4 | Permanently discontinue |
| Neurological Toxicities | Grade 2 | Withhold |
| Grades 3 or 4 | Permanently discontinue | |
| Other Adverse Reactions | ||
| Infusion-Related Reactions [see Warnings and Precautions (5.2)] | Grades 1 or 2 | Interrupt or slow the rate of infusion |
| Grades 3 or 4 | Permanently discontinue |
5.1 Severe and Fatal Immune Mediated Adverse Reactions
TECENTRIQ is a monoclonal antibody that belongs to a class of drugs that bind to either the programmed death-receptor 1 (PD-1) or the PD-ligand 1 (PD-L1), blocking the PD-1/PD-L1 pathway, thereby removing inhibition of the immune response, potentially breaking peripheral tolerance and inducing immune-mediated adverse reactions. Important immune-mediated adverse reactions listed under Warnings and Precautions may not include all possible severe and fatal immune-mediated reactions.
Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue. Immune-mediated adverse reactions can occur at any time after starting a PD-1/PD-L1 blocking antibody. While immune-mediated adverse reactions usually manifest during treatment with PD-1/PD-L1 blocking antibodies, immune-mediated adverse reactions can also manifest after discontinuation of PD-1/PD-L1 blocking antibodies.
Early identification and management of immune-mediated adverse reactions are essential to ensure safe use of PD-1/PD-L1 blocking antibodies. Monitor patients closely for symptoms and signs that may be clinical manifestations of underlying immune-mediated adverse reactions. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. In cases of suspected immune-mediated adverse reactions, initiate appropriate workup to exclude alternative etiologies, including infection. Institute medical management promptly, including specialty consultation as appropriate.
Withhold or permanently discontinue TECENTRIQ depending on severity [see Dosage and Administration (2.3)]. In general, if TECENTRIQ requires interruption or discontinuation, administer systemic corticosteroid therapy (1 to 2 mg/kg/day prednisone or equivalent) until improvement to Grade 1 or less. Upon improvement to Grade 1 or less, initiate corticosteroid taper and continue to taper over at least 1 month. Consider administration of other systemic immunosuppressants in patients whose immune-mediated adverse reactions are not controlled with corticosteroid therapy.
Toxicity management guidelines for adverse reactions that do not necessarily require systemic steroids (e.g., endocrinopathies and dermatologic reactions) are discussed below.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been performed to test the potential of atezolizumab for carcinogenicity or genotoxicity.
Animal fertility studies have not been conducted with atezolizumab; however, an assessment of the male and female reproductive organs was included in a 26-week, repeat-dose toxicity study in cynomolgus monkeys. Weekly administration of atezolizumab to female monkeys at the highest dose tested caused an irregular menstrual cycle pattern and a lack of newly formed corpora lutea in the ovaries. This effect occurred at an estimated AUC approximately 6 times the AUC in patients receiving the recommended dose and was reversible. There was no effect on the male monkey reproductive organs.
5.3 Complications of Allogeneic Hsct After Pd 1/pd L1 Inhibitors
Fatal and other serious complications can occur in patients who receive allogeneic hematopoietic stem cell transplantation (HSCT) before or after being treated with a PD-1/PD-L1 blocking antibody. Transplant-related complications include hyperacute graft-versus-host disease (GVHD), acute GVHD, chronic GVHD, hepatic veno-occlusive disease (VOD) after reduced intensity conditioning, and steroid-requiring febrile syndrome (without an identified infectious cause). These complications may occur despite intervening therapy between PD-1/PD-L1 blockage and allogeneic HSCT.
Follow patients closely for evidence of transplant-related complications and intervene promptly. Consider the benefits versus risks of treatment with a PD-1/PD-L1 blocking antibody prior to or after an allogeneic HSCT.
2.1 Patient Selection for Treatment of Non Small Cell Lung Cancer and Melanoma
Select patients with Stage II to IIIA non-small cell lung cancer for treatment with TECENTRIQ as a single agent based on PD-L1 expression on tumor cells [see Clinical Studies (14.1)].
Select patients with first-line metastatic non-small cell lung cancer for treatment with TECENTRIQ as a single agent based on the PD-L1 expression on tumor cells or on tumor-infiltrating immune cells [see Clinical Studies (14.1)].
Information on FDA-approved tests for the determination of PD-L1 expression in metastatic non-small cell lung cancer are available at: http://www.fda.gov/CompanionDiagnostics.
Select patients with unresectable or metastatic melanoma for treatment with TECENTRIQ in combination with cobimetinib and vemurafenib after confirming the presence of a BRAF V600 mutation [see Clinical Studies (14.4)]. Information on FDA-approved tests for the detection of BRAF V600 mutations in melanoma is available at: http://www.fda.gov/CompanionDiagnostics.
Structured Label Content
Section 42229-5 (42229-5)
Preparation
Visually inspect drug product for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard the vial if the solution is cloudy, discolored, or visible particles are observed. Do not shake the vial.
Prepare the solution for infusion as follows:
- Select the appropriate vial(s) based on the prescribed dose.
- Withdraw the required volume of TECENTRIQ from the vial(s) using sterile needle and syringe.
- Dilute to a final concentration between 3.2 mg/mL and 16.8 mg/mL in a polyvinyl chloride (PVC), polyethylene (PE), or polyolefin (PO) infusion bag containing 0.9% Sodium Chloride Injection, USP.
- Dilute with only 0.9% Sodium Chloride Injection, USP.
- Mix diluted solution by gentle inversion. Do not shake.
- Discard used or empty vials of TECENTRIQ.
Section 42231-1 (42231-1)
| MEDICATION GUIDE TECENTRIQ® (te-SEN-trik) (atezolizumab) Injection |
|||||
|---|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 10/2025 | ||||
| What is the most important information I should know about TECENTRIQ? | |||||
| TECENTRIQ is a medicine that may treat certain cancers by working with your immune system. TECENTRIQ can cause your immune system to attack normal organs and tissues in any area of your body and can affect the way they work. These problems can sometimes become severe or life-threatening and can lead to death. You can have more than one of these problems at the same time. These problems may happen anytime during your treatment or even after your treatment has ended. | |||||
| Call or see your healthcare provider right away if you develop any new or worse signs or symptoms, including: Lung problems. | |||||
|
|
|
|||
| Intestinal problems. | |||||
|
|||||
| Liver problems. | |||||
|
|
||||
| Hormone gland problems. | |||||
|
|
||||
| Kidney problems. | |||||
|
|
||||
| Skin problems. | |||||
|
|
||||
|
Problems can also happen in other organs. These are not all of the signs and symptoms of immune system problems that can happen with TECENTRIQ. Call or see your healthcare provider right away for any new or worse signs or symptoms, including: |
|||||
|
|||||
| Infusion reactions that can sometimes be severe or life-threatening. Signs and symptoms of infusion reactions may include: | |||||
|
|
||||
| Complications, including graft-versus-host disease (GVHD), in people who have received a bone marrow (stem cell) transplant that uses donor stem cells (allogeneic). These complications can be serious and can lead to death. These complications may happen if you underwent transplantation either before or after being treated with TECENTRIQ. Your healthcare provider will monitor you for these complications. | |||||
|
Getting medical treatment right away may help keep these problems from becoming more serious.
Your healthcare provider will check you for these problems during your treatment with TECENTRIQ. Your healthcare provider may treat you with corticosteroid or hormone replacement medicines. Your healthcare provider may also need to delay or completely stop treatment with TECENTRIQ if you have severe side effects. |
|||||
| What is TECENTRIQ? | |||||
| TECENTRIQ is a prescription medicine used to treat: | |||||
|
|||||
It is not known if TECENTRIQ is safe and effective when used:
|
|||||
| Before receiving TECENTRIQ, tell your healthcare provider about all of your medical conditions, including if you: | |||||
|
|||||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||||
| How will I receive TECENTRIQ? | |||||
|
|||||
| What are the possible side effects of TECENTRIQ? | |||||
| TECENTRIQ can cause serious side effects, including: | |||||
| The most common side effects of TECENTRIQ when used alone include: | |||||
|
|
|
|||
| The most common side effects of TECENTRIQ when used in lung cancer with other anti-cancer medicines include: | |||||
|
|
|
|||
| The most common side effects of TECENTRIQ when used in hepatocellular carcinoma with bevacizumab include: | |||||
|
|
|
|||
| The most common side effects of TECENTRIQ when used in melanoma with cobimetinib and vemurafenib include: | |||||
|
|
|
|||
| TECENTRIQ may cause fertility problems in females, which may affect the ability to have children. Talk to your healthcare provider if you have concerns about fertility. | |||||
| These are not all the possible side effects of TECENTRIQ. | |||||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||||
| General information about the safe and effective use of TECENTRIQ. | |||||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about TECENTRIQ that is written for health professionals. | |||||
| What are the ingredients in TECENTRIQ? | |||||
| Active ingredient: atezolizumab | |||||
| Inactive ingredients: glacial acetic acid, L-histidine, polysorbate 20 and sucrose | |||||
| Manufactured by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 USA | |||||
| U.S. License No.: 1048 TECENTRIQ is a registered trademark of Genentech, Inc. | |||||
| For more information, call 1-844-832-3687 or go to www.TECENTRIQ.com. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store vials under refrigeration at 2°C to 8°C (36°F to 46°F) in original carton to protect from light. Do not freeze. Do not shake.
1.4 Melanoma
TECENTRIQ, in combination with cobimetinib and vemurafenib, is indicated for the treatment of adult patients with BRAF V600 mutation-positive unresectable or metastatic melanoma [see Dosage and Administration (2.1)].
14.4 Melanoma
The efficacy of TECENTRIQ in combination with cobimetinib and vemurafenib was evaluated in a double-blind, randomized (1:1), placebo-controlled, multicenter trial (IMspire150; NCT02908672) conducted in 514 patients. Randomization was stratified by geographic location (North America vs. Europe vs. Australia, New Zealand, and others) and baseline lactate dehydrogenase (LDH) [less than or equal to upper limit of normal (ULN) vs. greater than ULN]. Eligible patients were required to have previously untreated unresectable or metastatic BRAF V600 mutation-positive melanoma as detected by a locally available test and centrally confirmed with the FoundationOne™ assay. Patients were excluded if they had history of autoimmune disease; administration of a live, attenuated vaccine within 28 days prior to randomization; administration of systemic immunostimulatory agents within 4 weeks or systemic immunosuppressive medications within 2 weeks prior to randomization; and active or untreated CNS metastases.
TECENTRIQ was initiated after patients received a 28-day treatment cycle of cobimetinib 60 mg orally once daily (21 days on / 7 days off) and vemurafenib 960 mg orally twice daily Days 1-21 and 720 mg orally twice daily Days 22-28. Patients received TECENTRIQ 840 mg intravenous infusion over 60 minutes every 2 weeks in combination with cobimetinib 60 mg orally once daily and vemurafenib 720 mg orally twice daily, or placebo in combination with cobimetinib 60 mg orally once daily and vemurafenib 960 mg orally twice daily. Treatment continued until disease progression or unacceptable toxicity. There was no crossover at the time of disease progression. Tumor assessments were performed every 8 weeks (± 1 week) for the first 24 months and every 12 weeks (± 1 week) thereafter.
The major efficacy outcome measure was investigator-assessed progression-free survival (PFS) per RECIST v1.1. Additional efficacy outcomes included PFS assessed by an independent central review, investigator-assessed ORR, OS, and DOR.
The median age of the study population was 54 years (range: 22-88), 58% of patients were male, 95% were White, a baseline ECOG performance status of 0 (77%) or 1 (23%), 33% had elevated LDH, 94% had metastatic disease, 60% were Stage IV (M1C), 56% had less than three metastatic sites at baseline, 3% had prior treatment for brain metastases, 30% had liver metastases at baseline, and 14% had received prior adjuvant systemic therapy. Based on central testing, 74% were identified as having a V600E mutation, 11% as having V600K mutation, and 1% as having V600D or V600R mutations.
Efficacy results are summarized in Table 32 and Figure 10. Patients had a median survival follow up time of 18.9 months.
| TECENTRIQ + Cobimetinib + Vemurafenib N=256 |
Placebo + Cobimetinib + Vemurafenib N=258 |
|
|---|---|---|
|
Progression-Free Survival
As determined by investigator assessment with Response Evaluation Criteria in Solid Tumors v1.1.; CI=confidence interval;
|
||
| Number of events (%) | 148 (58) | 179 (69) |
| Median, months | 15.1 | 10.6 |
| (95% CI) | (11.4, 18.4) | (9.3, 12.7) |
| Hazard ratio Stratified by baseline LDH (95% CI) |
0.78 (0.63, 0.97) | |
| p-value Based on the stratified log-rank test
|
0.0249 | |
|
Overall Response Rate
,
Confirmed Responses
|
||
| Number of responders (%) | 170 (66) | 168 (65) |
| (95% CI) | (60, 72) | (59, 71) |
| Complete responses, n (%) | 41 (16) | 46 (18) |
| Partial response, n (%) | 129 (50) | 122 (47) |
| Duration of Response , | n=170 | n=168 |
| Median, months | 20.4 | 12.5 |
| (95% CI) | (15.1, NE) | (10.7, 16.6) |
Figure 10: Kaplan-Meier Plot for Progression-Free Survival in IMspire150
At a pre-specified analysis at the time of the primary analysis of PFS, the OS data were not mature. The median OS was 28.8 months with 93 (36%) deaths in the TECENTRIQ plus cobimetinib and vemurafenib arm, and 25.1 months with 112 (43%) deaths in the placebo plus cobimetinib and vemurafenib arm. The hazard ratio for OS was 0.85 (95% CI: 0.64, 1.11) and the p-value was 0.2310.
11 Description (11 DESCRIPTION)
Atezolizumab is a programmed cell death ligand 1 (PD-L1) blocking antibody. Atezolizumab is an Fc-engineered, humanized, non-glycosylated IgG1 kappa immunoglobulin that has a calculated molecular mass of 145 kDa.
TECENTRIQ (atezolizumab) injection for intravenous use is a sterile, preservative-free, colorless to slightly yellow solution in single-dose vials. Each 20 mL vial contains 1200 mg of atezolizumab and is formulated in glacial acetic acid (16.5 mg), L-histidine (62 mg), polysorbate 20 (8 mg), and sucrose (821.6 mg), with a pH of 5.8. Each 14 mL vial contains 840 mg of atezolizumab and is formulated in glacial acetic acid (11.5 mg), L-histidine (43.4 mg), polysorbate 20 (5.6 mg), and sucrose (575.1 mg) with a pH of 5.8.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of ADA in other products.
During the first year of treatment with TECENTRIQ across 8 clinical studies, 13% to 36% of patients developed anti-atezolizumab antibodies. Median atezolizumab clearance in patients who tested positive for ADA was 19% (minimum 18%, maximum 49%) higher as compared to atezolizumab clearance in patients who tested negative for ADA; this change in clearance is not expected to be clinically significant.
In OAK and IMbrave150, exploratory analyses showed that the subset of patients who were ADA-positive appeared to have less efficacy (effect on overall survival) as compared to patients who tested negative for ADA [see Clinical Studies (14.1, 14.3)]. In study IMpower150 and IMforte, the impact of ADA on efficacy did not appear to be clinically significant [see Clinical Studies (14.1)]. In the remaining studies, there is insufficient information to characterize the effect of ADA on efficacy.
The presence of ADA did not have a clinically significant effect on the incidence or severity of adverse reactions.
Across clinical studies, the incidence of neutralizing antibodies (NAb) in ADA-positive patients was 24% to 83%. The effect of NAb on atezolizumab exposure and safety did not appear to be clinically significant. The effect of NAb on key efficacy endpoints is uncertain due to small sample sizes.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Severe and Fatal Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.1)]
- Infusion-Related Reactions [see Warnings and Precautions (5.2)]
- Complications of Allogeneic HSCT after PD-1/PD-L1 Inhibitors [see Warnings and Precautions (5.3)]
12.2 Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of atezolizumab have not been fully characterized.
12.3 Pharmacokinetics
Atezolizumab exposure increased dose proportionally over the dose range of 1 mg/kg to 20 mg/kg (0.07 to 1.33 times of the approved recommended doses), including a dose of 1200 mg administered every 3 weeks. Steady state was achieved after 6 to 9 weeks following multiple doses. The systemic accumulation ratio for every 2 weeks administration and every 3 weeks administration is 3.3- and 1.9- fold, respectively.
2.2 Recommended Dosage
The recommended dosages of TECENTRIQ are presented in Table 1. Administer TECENTRIQ as an intravenous infusion over 60 minutes. If the first infusion is tolerated, all subsequent infusions may be delivered over 30 minutes.
| Indication | Recommended Dosage of TECENTRIQ | Duration of Therapy |
|---|---|---|
| Metastatic NSCLC |
|
Until disease progression or unacceptable toxicity |
| Adjuvant Treatment of NSCLC |
|
Up to one year, unless there is disease recurrence or unacceptable toxicity |
| ASPS (adult) |
|
Until disease progression or unacceptable toxicity |
| ASPS (pediatric, 2 years of age and older) | 15 mg/kg (up to a maximum 1200 mg) every 3 weeks |
The recommended intravenous dosages of TECENTRIQ in combination with other therapeutic agents are presented in Table 2. Refer to the respective Prescribing Information for each therapeutic agent administered in combination with TECENTRIQ for the recommended dosage information, as appropriate.
| Indication | Recommended Dosage of TECENTRIQ | Duration of Therapy |
|---|---|---|
| NSCLC |
|
Until disease progression or unacceptable toxicity |
| SCLC |
|
|
| HCC |
|
|
| Melanoma |
Prior to initiating TECENTRIQ, patients should receive a 28-day treatment cycle of cobimetinib 60 mg orally once daily (21 days on and 7 days off) and vemurafenib 960 mg orally twice daily from Days 1-21 and vemurafenib 720 mg orally twice daily from Days 22-28. |
1 Indications and Usage (1 INDICATIONS AND USAGE)
TECENTRIQ is a programmed death-ligand 1 (PD-L1) blocking antibody indicated:
Non-Small Cell Lung Cancer (NSCLC)
- as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage II to IIIA NSCLC whose tumors have PD-L1 expression on ≥ 1% of tumor cells, as determined by an FDA-approved test. (1.1, 14.1)
- for the first-line treatment of adult patients with metastatic NSCLC whose tumors have high PD-L1 expression (PD-L1 stained ≥ 50% of tumor cells [TC ≥ 50%] or PD-L1 stained tumor-infiltrating immune cells [IC] covering ≥ 10% of the tumor area [IC ≥ 10%] ), as determined by an FDA-approved test, with no EGFR or ALK genomic tumor aberrations. (1.1)
- in combination with bevacizumab, paclitaxel, and carboplatin, for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations. (1.1)
- in combination with paclitaxel protein-bound and carboplatin for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations (1.1)
- for the treatment of adult patients with metastatic NSCLC who have disease progression during or following platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for NSCLC harboring these aberrations prior to receiving TECENTRIQ. (1.1)
Small Cell Lung Cancer (SCLC)
- in combination with carboplatin and etoposide, for the first-line treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC). (1.2)
- in combination with lurbinectedin, for the maintenance treatment of adult patients with ES-SCLC whose disease has not progressed after first-line induction therapy with TECENTRIQ or atezolizumab and hyaluronidase-tqjs, carboplatin and etoposide. (1.2)
Hepatocellular Carcinoma (HCC)
- in combination with bevacizumab for the treatment of adult patients with unresectable or metastatic HCC who have not received prior systemic therapy. ( 1.3)
Melanoma
- in combination with cobimetinib and vemurafenib for the treatment of adult patients with BRAF V600 mutation-positive unresectable or metastatic melanoma. (1.4)
Alveolar Soft Part Sarcoma (ASPS)
- for the treatment of adult and pediatric patients 2 years of age and older with unresectable or metastatic ASPS. (1.5)
12.1 Mechanism of Action
PD L1 may be expressed on tumor cells and/or tumor infiltrating immune cells and can contribute to the inhibition of the anti-tumor immune response in the tumor microenvironment. Binding of PD L1 to the PD 1 and B7.1 receptors found on T cells and antigen presenting cells suppresses cytotoxic T-cell activity, T-cell proliferation and cytokine production.
Atezolizumab is a monoclonal antibody that binds to PD L1 and blocks its interactions with both PD 1 and B7.1 receptors. This releases the PD L1/PD 1 mediated inhibition of the immune response, including activation of the anti-tumor immune response without inducing antibody-dependent cellular cytotoxicity. In syngeneic mouse tumor models, blocking PD L1 activity resulted in decreased tumor growth.
In mouse models of cancer, dual inhibition of the PD-1/PD-L1 and MAPK pathways suppresses tumor growth and improves tumor immunogenicity through increased antigen presentation and T cell infiltration and activation compared to targeted therapy alone.
5.4 Embryo Fetal Toxicity (5.4 Embryo-Fetal Toxicity)
Based on its mechanism of action, TECENTRIQ can cause fetal harm when administered to a pregnant woman. There are no available data on the use of TECENTRIQ in pregnant women. Animal studies have demonstrated that inhibition of the PD-L1/PD-1 pathway can lead to increased risk of immune-related rejection of the developing fetus resulting in fetal death.
Verify pregnancy status of females of reproductive potential prior to initiating TECENTRIQ. Advise females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with TECENTRIQ and for at least 5 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
1.2 Small Cell Lung Cancer
- TECENTRIQ, in combination with carboplatin and etoposide, is indicated for the first-line treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC).
- TECENTRIQ, in combination with lurbinectedin, is indicated for the maintenance treatment of adult patients with ES-SCLC whose disease has not progressed after first-line induction therapy with TECENTRIQ or atezolizumab and hyaluronidase-tqjs, carboplatin and etoposide.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
Immune-Mediated Adverse Reactions
- Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated dermatologic adverse reactions, immune-mediated nephritis and renal dysfunction, and solid organ transplant rejection. (5.1)
- Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. (5.1)
- Withhold or permanently discontinue based on severity and type of reaction. (5.1).
- Infusion-Related Reactions: Interrupt, slow the rate of infusion, or permanently discontinue based on severity of infusion reactions. (5.2)
- Complications of Allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. (5.3)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and use of effective contraception. (5.4, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Administer TECENTRIQ intravenously over 60 minutes. If the first infusion is tolerated, all subsequent infusions may be delivered over 30 minutes.
NSCLC
- In the adjuvant setting, administer TECENTRIQ following resection and up to 4 cycles of platinum-based chemotherapy as 840 mg every 2 weeks, 1200 mg every 3 weeks or 1680 mg every 4 weeks for up to 1 year. (2.2)
- In the metastatic setting, administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. (2.2)
- When administering with chemotherapy with or without bevacizumab, administer TECENTRIQ prior to chemotherapy and bevacizumab when given on the same day. (2.2)
Small Cell Lung Cancer
- Administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. Administer TECENTRIQ prior to chemotherapy when given on the same day. (2.2)
Hepatocellular Carcinoma
- Administer TECENTRIQ as 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. Administer TECENTRIQ prior to bevacizumab when given on the same day. Bevacizumab is administered at 15 mg/kg every 3 weeks. (2.2)
Melanoma
- Following completion of a 28-day cycle of cobimetinib and vemurafenib, administer TECENTRIQ 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks with cobimetinib 60 mg orally once daily (21 days on /7 days off) and vemurafenib 720 mg orally twice daily. (2.2)
ASPS
1.3 Hepatocellular Carcinoma
TECENTRIQ, in combination with bevacizumab, is indicated for the treatment of adult patients with unresectable or metastatic hepatocellular carcinoma (HCC) who have not received prior systemic therapy.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 840 mg/14 mL (60 mg/mL) and 1200 mg/20 mL (60 mg/mL) colorless to slightly yellow solution in a single-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of TECENTRIQ. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Cardiac: pericarditis, pericardial effusion, cardiac tamponade
- Musculoskeletal and Connective Tissue: tenosynovitis
14.3 Hepatocellular Carcinoma
The efficacy of TECENTRIQ in combination with bevacizumab was investigated in IMbrave150 (NCT03434379), a multicenter, international, open-label, randomized trial in patients with locally advanced unresectable and/or metastatic hepatocellular carcinoma who have not received prior systemic therapy. Randomization was stratified by geographic region (Asia excluding Japan vs. rest of world), macrovascular invasion and/or extrahepatic spread (presence vs. absence), baseline AFP (<400 vs. ≥400 ng/mL), and by ECOG performance status (0 vs. 1).
A total of 501 patients were randomized (2:1) to receive either TECENTRIQ as an intravenous infusion of 1200 mg, followed by 15 mg/kg bevacizumab, on the same day every 3 weeks or sorafenib 400 mg given orally twice daily, until disease progression or unacceptable toxicity. Patients could discontinue either TECENTRIQ or bevacizumab (e.g., due to adverse events) and continue on single-agent therapy until disease progression or unacceptable toxicity associated with the single-agent.
The study enrolled patients who were ECOG performance score 0 or 1 and who had not received prior systemic treatment. Patients were required to be evaluated for the presence of varices within 6 months prior to treatment, and were excluded if they had variceal bleeding within 6 months prior to treatment, untreated or incompletely treated varices with bleeding, or high risk of bleeding. Patients with Child-Pugh B or C cirrhosis, moderate or severe ascites; history of hepatic encephalopathy; a history of autoimmune disease; administration of a live, attenuated vaccine within 4 weeks prior to randomization; administration of systemic immunostimulatory agents within 4 weeks or systemic immunosuppressive medications within 2 weeks prior to randomization; or untreated or corticosteroid-dependent brain metastases were excluded. Tumor assessments were performed every 6 weeks for the first 54 weeks and every 9 weeks thereafter.
The demographics and baseline disease characteristics of the study population were balanced between the treatment arms. The median age was 65 years (range: 26 to 88) and 83% of patients were male. The majority of patients were Asian (57%) or White (35%); 40% were from Asia (excluding Japan). Approximately 75% of patients presented with macrovascular invasion and/or extrahepatic spread and 37% had a baseline AFP ≥400 ng/mL. Baseline ECOG performance status was 0 (62%) or 1 (38%). HCC risk factors were Hepatitis B in 48% of patients, Hepatitis C in 22%, and 31% of patients had non-viral liver disease. The majority of patients had BCLC stage C (82%) disease at baseline, while 16% had stage B, and 3% had stage A.
The major efficacy outcome measures were overall survival (OS) and independent review facility (IRF)-assessed progression free survival (PFS) per RECIST v1.1. Additional efficacy outcome measures were IRF-assessed overall response rate (ORR) per RECIST and mRECIST.
Efficacy results are presented in Table 31 and Figure 9.
| TECENTRIQ in combination with Bevacizumab (N= 336) |
Sorafenib (N=165) |
|
|---|---|---|
| CI=confidence interval; HCC mRECIST=Modified RECIST Assessment for Hepatocellular Carcinoma; NE=not estimable; RECIST 1.1=Response Evaluation Criteria in Solid Tumors v1.1 | ||
| Overall Survival | ||
| Number of deaths (%) | 96 (29) | 65 (39) |
| Median OS in months (95% CI) | NE (NE, NE) |
13.2 (10.4, NE) |
| Hazard ratio Stratified by geographic region (Asia excluding Japan vs. rest of world), macrovascular invasion and/or extrahepatic spread (presence vs. absence), and baseline AFP (<400 vs. ≥400 ng/mL) (95% CI) |
0.58 (0.42, 0.79) | |
| p-value Based on two-sided stratified log-rank test; as compared to significance level 0.004 (2-sided) based on 161/312=52% information using the OBF method
|
0.0006 | |
|
Progression-Free Survival Per independent radiology review
|
||
| Number of events (%) | 197 (59) | 109 (66) |
| Median PFS in months (95% CI) | 6.8 (5.8, 8.3) | 4.3 (4.0, 5.6) |
| Hazard ratio (95% CI) | 0.59 (0.47, 0.76) | |
| p-value | <0.0001 | |
|
Overall Response Rate
,
Confirmed responses (ORR), RECIST 1.1
|
||
| Number of responders (%) | 93 (28) | 19 (12) |
| (95% CI) | (23, 33) | (7,17) |
| p-value Based on two-sided Cochran-Mantel-Haesnszel test
|
<0.0001 | |
| Complete responses, n (%) | 22 (7) | 0 |
| Partial responses, n (%) | 71 (21) | 19 (12) |
| Duration of Response , (DOR) RECIST 1.1 | ||
| (n=93) | (n=19) | |
| Median DOR in months (95% CI) | NE (NE, NE) |
6.3 (4.7, NE) |
| Range (months) | (1.3 Denotes a censored value , 13.4) |
(1.4, 9.1) |
| Overall Response Rate , (ORR), HCC mRECIST | ||
| Number of responders (%) | 112 (33) | 21 (13) |
| (95% CI) | (28, 39) | (8, 19) |
| p-value | <0.0001 | |
| Complete responses, n (%) | 37 (11) | 3 (1.8) |
| Partial responses, n (%) | 75 (22) | 18 (11) |
| Duration of Response , (DOR) HCC mRECIST | ||
| (n=112) | (n=21) | |
| Median DOR in months (95% CI) |
NE (NE, NE) |
6.3 (4.9, NE) |
| Range (months) | (1.3, 13.4) | (1.4, 9.1) |
Figure 9: Kaplan-Meier Plot of Overall Survival in IMbrave150
Exploratory analyses showed that the subset of patients (20%) who were ADA-positive by week 6 appeared to have reduced efficacy (effect on OS) as compared to patients (80%) who tested negative for treatment-emergent ADA by week 6 [see Clinical Pharmacology (12.6)]. ADA-positive patients by week 6 appeared to have similar overall survival compared to sorafenib-treated patients. In an exploratory analysis, inverse probability weighting was conducted to compare ADA-positive patients and ADA-negative patients in the TECENTRIQ and bevacizumab arm to the sorafenib arm. Inverse probability weighting factors were: baseline sum of longest tumor size (BSLD), baseline ECOG, baseline albumin, baseline LDH, sex, age, race, geographic region, weight, neutrophil-to-lymphocyte ratio, AFP (<400 ng/mL vs ≥400 ng/mL), number of metastatic sites, MVI and/or EHS present at study entry, etiology (HBV vs. HCV vs. non-viral) and Child-Pugh Score (A5 vs. A6). The OS hazard ratio comparing the ADA-positive subgroup of the TECENTRIQ and bevacizumab arm to sorafenib was 0.93 (95% CI: 0.57, 1.53). The OS hazard ratio comparing the ADA-negative subgroup to sorafenib was 0.39 (95% CI: 0.26, 0.60).
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. (8.2)
1.1 Non Small Cell Lung Cancer (1.1 Non-Small Cell Lung Cancer)
- TECENTRIQ, as a single-agent, is indicated as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with stage II to IIIA [see Clinical Studies (14.1)] non-small cell lung cancer (NSCLC) whose tumors have PD-L1 expression on ≥ 1% of tumor cells, as determined by an FDA-approved test [see Dosage and Administration (2.1)].
- TECENTRIQ, as a single agent, is indicated for the first-line treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors have high PD-L1 expression (PD-L1 stained ≥ 50% of tumor cells [TC ≥ 50%] or PD-L1 stained tumor-infiltrating immune cells [IC] covering ≥ 10% of the tumor area [IC ≥ 10%]), as determined by an FDA-approved test, with no EGFR or ALK genomic tumor aberrations [see Dosage and Administration (2.1)].
- TECENTRIQ, in combination with bevacizumab, paclitaxel, and carboplatin, is indicated for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations.
- TECENTRIQ, in combination with paclitaxel protein-bound and carboplatin, is indicated for the first-line treatment of adult patients with metastatic non-squamous NSCLC with no EGFR or ALK genomic tumor aberrations.
- TECENTRIQ, as a single-agent, is indicated for the treatment of adult patients with metastatic NSCLC who have disease progression during or following platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for NSCLC harboring these aberrations prior to receiving TECENTRIQ.
1.5 Alveolar Soft Part Sarcoma
TECENTRIQ, as a single agent, is indicated for the treatment of adult and pediatric patients 2 years of age and older with unresectable or metastatic alveolar soft part sarcoma (ASPS).
5.2 Infusion Related Reactions (5.2 Infusion-Related Reactions)
TECENTRIQ can cause severe or life-threatening infusion-related reactions, including anaphylaxis. Monitor for signs and symptoms of infusion-related reactions. Interrupt, slow the rate of, or permanently discontinue TECENTRIQ based on the severity [see Dosage and Administration (2.3)]. For Grade 1 or 2 infusion-related reactions, consider using pre-medications with subsequent doses.
In clinical studies enrolling 2616 patients with various cancers who received TECENTRIQ as a single-agent [see Adverse Reactions (6.1)], infusion-related reactions occurred in 1.3% of patients, including Grade 3 (0.2%). The frequency and severity of infusion-related reactions were similar whether TECENTRIQ was given as a single-agent in patients with various cancers, in combination with other antineoplastic drugs in NSCLC and SCLC, and across the recommended dose range (840 mg Q2W to 1680 mg Q4W).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described in WARNINGS AND PRECAUTIONS reflect exposure to TECENTRIQ as a single-agent in 2616 patients in two randomized, active-controlled studies (POPLAR, OAK) and three open-label, single arm studies (PCD4989g, BIRCH, FIR) which enrolled 1636 patients with metastatic NSCLC, and 980 patients with other tumor types. TECENTRIQ was administered at a dose of 1200 mg intravenously every 3 weeks in all studies except PCD4989g. Among the 2616 patients who received a single-agent TECENTRIQ, 36% were exposed for longer than 6 months and 20% were exposed for longer than 12 months. Using the dataset described for patients who received TECENTRIQ as a single-agent, the most common adverse reactions in ≥ 20% of patients were fatigue/asthenia (48%), decreased appetite (25%), nausea (24%), cough (22%), and dyspnea (22%). In addition, the data reflect exposure to TECENTRIQ as a single agent as adjuvant therapy in 495 patients with early-stage NSCLC enrolled in a randomized study (IMpower010).
In addition, the data reflect exposure to TECENTRIQ in combination with other antineoplastic drugs in 2421 patients with NSCLC (N = 2223) or SCLC (N = 198) enrolled in five randomized, active-controlled trials, including IMpower150, IMpower130 and IMpower133. Among the 2421 patients, 53% were exposed to TECENTRIQ for longer than 6 months and 29% were exposed to TECENTRIQ for longer than 12 months. Among the 2421 patients with NSCLC and SCLC who received TECENTRIQ in combination with other antineoplastic drugs, the most common adverse reactions in ≥20% of patients were fatigue/asthenia (49%), nausea (38%), alopecia (35%), constipation (29%), diarrhea (28%) and decreased appetite (27%).
The data also reflect exposure to TECENTRIQ administered in combination with cobimetinib and vemurafenib in 230 patients enrolled in IMspire150. Among the 230 patients, 62% were exposed to TECENTRIQ for longer than 6 months and 42% were exposed to TECENTRIQ for longer than 12 months.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
TECENTRIQ injection is a sterile, preservative-free, and colorless to slightly yellow solution for intravenous infusion supplied as a carton containing one 840 mg/14 mL single-dose vial (NDC 50242-918-01) or 1,200 mg/20 mL single-dose vial (NDC 50242-917-01).
14.5 Alveolar Soft Part Sarcoma (asps) (14.5 Alveolar soft part sarcoma (ASPS))
The efficacy of TECENTRIQ was evaluated in study ML39345 (NCT03141684), an open-label, single-arm study, in 49 adult and pediatric patients aged 2 years and older with unresectable or metastatic ASPS. Eligible patients were required to have histologically or cytologically confirmed ASPS that was not curable by surgery, and an ECOG performance status of ≤ 2.
Patients were excluded if they had known primary central nervous system (CNS) malignancy or symptomatic CNS metastases, known clinically significant liver disease, or history of idiopathic pulmonary fibrosis, pneumonitis, organizing pneumonia, or evidence of active pneumonitis on screening chest computed tomography (CT) scan.
Adult patients received 1200 mg intravenously and pediatric patients received 15 mg/kg (up to a maximum of 1200 mg) intravenously once every 21 days until disease progression or unacceptable toxicity.
The major efficacy outcomes were Overall Response Rate (ORR) and Duration of Response (DOR) by Independent Review Committee according to Response Evaluation Criteria in Solid Tumors (RECIST) v1.1.
A total of 49 patients were enrolled. The median age of patients was 31 years (range: 12-70); 2% of adult patients (n=47) were ≥65 years of age and the pediatric patients (n=2) were ≥12 years of age; 51% of patients were female, 55% White, 29% Black or African American, 10% Asian; 53% had an ECOG performance status of 0 and 45% had an ECOG performance status of 1. All patients had prior surgery for ASPS and 55% received at least one prior line of treatment for ASPS; 55% received radiotherapy and 53% received chemotherapy. Of the patients who reported staging at initial diagnosis, all were Stage IV.
Efficacy results of this study are summarized in Table 33.
| Endpoint | All Patients (N=49) |
|---|---|
| CI: confidence interval; N: number of patients; +: Censored | |
|
Overall response rate (95% CI) 95% CI based on Clopper–Pearson exact method.
|
24% (13, 39) |
| Complete Responses, n | 0 |
| Partial Responses, n (%) | 12 (24) |
| Duration of response | |
| Median, month | NE |
| (95% CI) | (17.0, NE) |
| Range | 1+, 41+ |
| Durability of Response | |
| ≥6 months, n (%) | 8 (67%) |
| ≥12 months, n (%) | 5 (42%) |
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In animal models, inhibition of PD-L1/PD-1 signaling increased the severity of some infections and enhanced inflammatory responses. M. tuberculosis-infected PD-1 knockout mice exhibit markedly decreased survival compared with wild-type controls, which correlated with increased bacterial proliferation and inflammatory responses in these animals. PD-L1 and PD-1 knockout mice and mice receiving PD-L1 blocking antibody have also shown decreased survival following infection with lymphocytic choriomeningitis virus.
Principal Display Panel 14 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 14 mL Vial Carton)
NDC 50242-918-01
Tecentriq®
(atezolizumab)
Injection
840 mg/14 mL
(60 mg/mL)
For Intravenous Infusion After Dilution
Single-Dose Vial
Discard Unused Portion
No preservative.
Attention Pharmacist: Dispense the
accompanying Medication Guide
to each patient.
1 vial
Rx only
Genentech
10198487
Principal Display Panel 20 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 20 mL Vial Carton)
NDC 50242-917-01
Tecentriq®
(atezolizumab)
Injection
1200 mg/20 mL
(60 mg/mL)
For Intravenous Infusion After Dilution
Single-Dose Vial
Discard Unused Portion
No preservative.
Attention Pharmacist: Dispense the
accompanying Medication Guide
to each patient.
1 vial
Rx only
Genentech
10199144
2.3 Dosage Modifications for Adverse Reactions
No dose reduction for TECENTRIQ is recommended. In general, withhold TECENTRIQ for severe (Grade 3) immune-mediated adverse reactions. Permanently discontinue TECENTRIQ for life-threatening (Grade 4) immune-mediated adverse reactions, recurrent severe (Grade 3) immune-mediated reactions that require systemic immunosuppressive treatment, or an inability to reduce corticosteroid dose to 10 mg or less of prednisone or equivalent per day within 12 weeks of initiating steroids.
Dosage modifications for TECENTRIQ for adverse reactions that require management different from these general guidelines are summarized in Table 3.
| Adverse Reaction | Severity Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4
|
Dosage Modification |
|---|---|---|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase, ULN = upper limit normal, DRESS = Drug Rash with Eosinophilia and Systemic Symptoms, SJS = Stevens Johnson syndrome, TEN = toxic epidermal necrolysis | ||
| Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.1)] | ||
| Pneumonitis | Grade 2 | Withhold Resume in patients with complete or partial resolution (Grade 0 to 1) after corticosteroid taper. Permanently discontinue if no complete or partial resolution within 12 weeks of initiating steroids or inability to reduce prednisone to 10 mg per day or less (or equivalent) within 12 weeks of initiating steroids
|
| Grades 3 or 4 | Permanently discontinue | |
| Colitis | Grades 2 or 3 | Withhold |
| Grade 4 | Permanently discontinue | |
| Hepatitis with no tumor involvement of the liver | AST or ALT increases to more than 3 and up to 8 times ULN or Total bilirubin increases to more than 1.5 and up to 3 times ULN |
Withhold |
| AST or ALT increases to more than 8 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue | |
| Hepatitis with tumor involvement of the liver If AST and ALT are less than or equal to ULN at baseline, withhold or permanently discontinue TECENTRIQ based on recommendations for hepatitis with no liver involvement
|
Baseline AST or ALT is more than 1 and up to 3 times ULN and increases to more than 5 and up to 10 times ULN or Baseline AST or ALT is more than 3 and up to 5 times ULN and increases to more than 8 and up to 10 times ULN |
Withhold |
| AST or ALT increases to more than 10 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue | |
| Endocrinopathies | Grades 3 or 4 | Withhold until clinically stable or permanently discontinue depending on severity |
| Nephritis with Renal Dysfunction | Grades 2 or 3 increased blood creatinine | Withhold |
| Grade 4 increased blood creatinine | Permanently discontinue | |
| Exfoliative Dermatologic Conditions | Suspected SJS, TEN, or DRESS | Withhold |
| Confirmed SJS, TEN, or DRESS | Permanently discontinue | |
| Myocarditis or Pericarditis | Grades 2, 3, or 4 | Permanently discontinue |
| Neurological Toxicities | Grade 2 | Withhold |
| Grades 3 or 4 | Permanently discontinue | |
| Other Adverse Reactions | ||
| Infusion-Related Reactions [see Warnings and Precautions (5.2)] | Grades 1 or 2 | Interrupt or slow the rate of infusion |
| Grades 3 or 4 | Permanently discontinue |
5.1 Severe and Fatal Immune Mediated Adverse Reactions (5.1 Severe and Fatal Immune-Mediated Adverse Reactions)
TECENTRIQ is a monoclonal antibody that belongs to a class of drugs that bind to either the programmed death-receptor 1 (PD-1) or the PD-ligand 1 (PD-L1), blocking the PD-1/PD-L1 pathway, thereby removing inhibition of the immune response, potentially breaking peripheral tolerance and inducing immune-mediated adverse reactions. Important immune-mediated adverse reactions listed under Warnings and Precautions may not include all possible severe and fatal immune-mediated reactions.
Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue. Immune-mediated adverse reactions can occur at any time after starting a PD-1/PD-L1 blocking antibody. While immune-mediated adverse reactions usually manifest during treatment with PD-1/PD-L1 blocking antibodies, immune-mediated adverse reactions can also manifest after discontinuation of PD-1/PD-L1 blocking antibodies.
Early identification and management of immune-mediated adverse reactions are essential to ensure safe use of PD-1/PD-L1 blocking antibodies. Monitor patients closely for symptoms and signs that may be clinical manifestations of underlying immune-mediated adverse reactions. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. In cases of suspected immune-mediated adverse reactions, initiate appropriate workup to exclude alternative etiologies, including infection. Institute medical management promptly, including specialty consultation as appropriate.
Withhold or permanently discontinue TECENTRIQ depending on severity [see Dosage and Administration (2.3)]. In general, if TECENTRIQ requires interruption or discontinuation, administer systemic corticosteroid therapy (1 to 2 mg/kg/day prednisone or equivalent) until improvement to Grade 1 or less. Upon improvement to Grade 1 or less, initiate corticosteroid taper and continue to taper over at least 1 month. Consider administration of other systemic immunosuppressants in patients whose immune-mediated adverse reactions are not controlled with corticosteroid therapy.
Toxicity management guidelines for adverse reactions that do not necessarily require systemic steroids (e.g., endocrinopathies and dermatologic reactions) are discussed below.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been performed to test the potential of atezolizumab for carcinogenicity or genotoxicity.
Animal fertility studies have not been conducted with atezolizumab; however, an assessment of the male and female reproductive organs was included in a 26-week, repeat-dose toxicity study in cynomolgus monkeys. Weekly administration of atezolizumab to female monkeys at the highest dose tested caused an irregular menstrual cycle pattern and a lack of newly formed corpora lutea in the ovaries. This effect occurred at an estimated AUC approximately 6 times the AUC in patients receiving the recommended dose and was reversible. There was no effect on the male monkey reproductive organs.
5.3 Complications of Allogeneic Hsct After Pd 1/pd L1 Inhibitors (5.3 Complications of Allogeneic HSCT after PD-1/PD-L1 Inhibitors)
Fatal and other serious complications can occur in patients who receive allogeneic hematopoietic stem cell transplantation (HSCT) before or after being treated with a PD-1/PD-L1 blocking antibody. Transplant-related complications include hyperacute graft-versus-host disease (GVHD), acute GVHD, chronic GVHD, hepatic veno-occlusive disease (VOD) after reduced intensity conditioning, and steroid-requiring febrile syndrome (without an identified infectious cause). These complications may occur despite intervening therapy between PD-1/PD-L1 blockage and allogeneic HSCT.
Follow patients closely for evidence of transplant-related complications and intervene promptly. Consider the benefits versus risks of treatment with a PD-1/PD-L1 blocking antibody prior to or after an allogeneic HSCT.
2.1 Patient Selection for Treatment of Non Small Cell Lung Cancer and Melanoma (2.1 Patient Selection for Treatment of Non-Small Cell Lung Cancer and Melanoma)
Select patients with Stage II to IIIA non-small cell lung cancer for treatment with TECENTRIQ as a single agent based on PD-L1 expression on tumor cells [see Clinical Studies (14.1)].
Select patients with first-line metastatic non-small cell lung cancer for treatment with TECENTRIQ as a single agent based on the PD-L1 expression on tumor cells or on tumor-infiltrating immune cells [see Clinical Studies (14.1)].
Information on FDA-approved tests for the determination of PD-L1 expression in metastatic non-small cell lung cancer are available at: http://www.fda.gov/CompanionDiagnostics.
Select patients with unresectable or metastatic melanoma for treatment with TECENTRIQ in combination with cobimetinib and vemurafenib after confirming the presence of a BRAF V600 mutation [see Clinical Studies (14.4)]. Information on FDA-approved tests for the detection of BRAF V600 mutations in melanoma is available at: http://www.fda.gov/CompanionDiagnostics.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:06.223362 · Updated: 2026-03-14T22:37:51.668409