These Highlights Do Not Include All The Information Needed To Use Clariscan Safely And Effectively. See Full Prescribing Information For Clariscan.
6f8c9fac-c995-4bf4-950d-ab89deb291b5
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS See full prescribing information for complete boxed warning Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. Clariscan is not approved for intrathecal use. ( 5.1 ) GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of Clariscan in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. The risk for NSF appears highest among patients with: Chronic, severe kidney disease (GFR < 30 mL/min/1.73 m 2 ), or Acute kidney injury. Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (for example, age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing ( 5.2 ).
Indications and Usage
Clariscan is a gadolinium-based contrast agent indicated for intravenous use with magnetic resonance imaging (MRI) in brain (intracranial), spine and associated tissues in adult and pediatric patients (including term neonates) to detect and visualize areas with disruption of the blood brain barrier (BBB) and/or abnormal vascularity.
Dosage and Administration
Adult and pediatric patients: The recommended dose of Clariscan is 0.2 mL/kg (0.1 mmol/kg) body weight administered as an intravenous bolus injection at a flow rate of approximately 2 mL/second for adults and 1 to 2 mL/second for pediatric patients (including term neonates). The dose is delivered by manual or power injection. ( 2 )
Warnings and Precautions
Hypersensitivity Reactions: Anaphylactoid/anaphylactic reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have uncommonly occurred. Monitor patients closely for need of emergency cardiorespiratory support. ( 5.3 ) Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs. ( 5.4 )
Contraindications
History of clinically important hypersensitivity reactions to Clariscan [see Warnings and Precautions (5.3) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Nephrogenic systemic fibrosis [see Warnings and Precautions (5.2) ] Hypersensitivity reactions [see Warnings and Precautions (5.3) ] Gadolinium Retention [see Warnings and Precautions (5.4) ]
Drug Interactions
Gadoterate does not interfere with serum and plasma calcium measurements determined by colorimetric assays. Specific drug interaction studies with gadoterate meglumine have not been conducted.
Storage and Handling
Clariscan Injection is a clear, colorless to yellow solution containing 0.5 mmol/mL of gadoterate meglumine. It is supplied in vials and pre-filled syringes. Clariscan Injection is supplied in 10 mL vials containing 5 mL or 10 mL of solution and in 20 mL vials containing 15 mL or 20 mL of solution. Each single-dose vial is closed with a rubber stopper and sealed with an aluminum cap and the contents are sterile. Vials are packaged in a box of 10, in the following configurations: 2.5 mmol per 5 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-06) 5 mmol per 10 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-01) 7.5 mmol per 15 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-02) 10 mmol per 20 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-05) Clariscan Injection is supplied in 20 mL plastic pre-filled syringes containing 10 mL, 15 mL, or 20 mL of solution. Each syringe is sealed with rubber closures and the contents are sterile. Syringes, including plunger rod, are individually packaged in a box of 10, in the following configurations: 5 mmol per 10 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-12) 7.5 mmol per 15 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-17) 10 mmol per 20 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-22)
How Supplied
Clariscan Injection is a clear, colorless to yellow solution containing 0.5 mmol/mL of gadoterate meglumine. It is supplied in vials and pre-filled syringes. Clariscan Injection is supplied in 10 mL vials containing 5 mL or 10 mL of solution and in 20 mL vials containing 15 mL or 20 mL of solution. Each single-dose vial is closed with a rubber stopper and sealed with an aluminum cap and the contents are sterile. Vials are packaged in a box of 10, in the following configurations: 2.5 mmol per 5 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-06) 5 mmol per 10 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-01) 7.5 mmol per 15 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-02) 10 mmol per 20 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-05) Clariscan Injection is supplied in 20 mL plastic pre-filled syringes containing 10 mL, 15 mL, or 20 mL of solution. Each syringe is sealed with rubber closures and the contents are sterile. Syringes, including plunger rod, are individually packaged in a box of 10, in the following configurations: 5 mmol per 10 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-12) 7.5 mmol per 15 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-17) 10 mmol per 20 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-22)
Medication Information
Warnings and Precautions
Hypersensitivity Reactions: Anaphylactoid/anaphylactic reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have uncommonly occurred. Monitor patients closely for need of emergency cardiorespiratory support. ( 5.3 ) Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs. ( 5.4 )
Indications and Usage
Clariscan is a gadolinium-based contrast agent indicated for intravenous use with magnetic resonance imaging (MRI) in brain (intracranial), spine and associated tissues in adult and pediatric patients (including term neonates) to detect and visualize areas with disruption of the blood brain barrier (BBB) and/or abnormal vascularity.
Dosage and Administration
Adult and pediatric patients: The recommended dose of Clariscan is 0.2 mL/kg (0.1 mmol/kg) body weight administered as an intravenous bolus injection at a flow rate of approximately 2 mL/second for adults and 1 to 2 mL/second for pediatric patients (including term neonates). The dose is delivered by manual or power injection. ( 2 )
Contraindications
History of clinically important hypersensitivity reactions to Clariscan [see Warnings and Precautions (5.3) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Nephrogenic systemic fibrosis [see Warnings and Precautions (5.2) ] Hypersensitivity reactions [see Warnings and Precautions (5.3) ] Gadolinium Retention [see Warnings and Precautions (5.4) ]
Drug Interactions
Gadoterate does not interfere with serum and plasma calcium measurements determined by colorimetric assays. Specific drug interaction studies with gadoterate meglumine have not been conducted.
Storage and Handling
Clariscan Injection is a clear, colorless to yellow solution containing 0.5 mmol/mL of gadoterate meglumine. It is supplied in vials and pre-filled syringes. Clariscan Injection is supplied in 10 mL vials containing 5 mL or 10 mL of solution and in 20 mL vials containing 15 mL or 20 mL of solution. Each single-dose vial is closed with a rubber stopper and sealed with an aluminum cap and the contents are sterile. Vials are packaged in a box of 10, in the following configurations: 2.5 mmol per 5 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-06) 5 mmol per 10 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-01) 7.5 mmol per 15 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-02) 10 mmol per 20 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-05) Clariscan Injection is supplied in 20 mL plastic pre-filled syringes containing 10 mL, 15 mL, or 20 mL of solution. Each syringe is sealed with rubber closures and the contents are sterile. Syringes, including plunger rod, are individually packaged in a box of 10, in the following configurations: 5 mmol per 10 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-12) 7.5 mmol per 15 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-17) 10 mmol per 20 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-22)
How Supplied
Clariscan Injection is a clear, colorless to yellow solution containing 0.5 mmol/mL of gadoterate meglumine. It is supplied in vials and pre-filled syringes. Clariscan Injection is supplied in 10 mL vials containing 5 mL or 10 mL of solution and in 20 mL vials containing 15 mL or 20 mL of solution. Each single-dose vial is closed with a rubber stopper and sealed with an aluminum cap and the contents are sterile. Vials are packaged in a box of 10, in the following configurations: 2.5 mmol per 5 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-06) 5 mmol per 10 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-01) 7.5 mmol per 15 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-02) 10 mmol per 20 mL (0.5 mmol per mL) in glass vial (NDC 0407-2943-05) Clariscan Injection is supplied in 20 mL plastic pre-filled syringes containing 10 mL, 15 mL, or 20 mL of solution. Each syringe is sealed with rubber closures and the contents are sterile. Syringes, including plunger rod, are individually packaged in a box of 10, in the following configurations: 5 mmol per 10 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-12) 7.5 mmol per 15 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-17) 10 mmol per 20 mL (0.5 mmol per mL) in plastic pre-filled syringe (NDC 0407-2943-22)
Description
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS See full prescribing information for complete boxed warning Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. Clariscan is not approved for intrathecal use. ( 5.1 ) GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of Clariscan in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. The risk for NSF appears highest among patients with: Chronic, severe kidney disease (GFR < 30 mL/min/1.73 m 2 ), or Acute kidney injury. Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (for example, age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing ( 5.2 ).
Section 42229-5
Risk Associated with Intrathecal Use
Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. Clariscan is not approved for intrathecal use [see Warnings and Precautions (5.1)].
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Rev. 3/2025 |
| MEDICATION GUIDE | |
|
CLARISCAN™ (kla-ri'-skan) (gadoterate meglumine) Injection, for intravenous use |
|
What is the most important information I should know about Clariscan?
|
|
What is Clariscan?
|
|
| Do not receive Clariscan if you have had a severe allergic reaction to Clariscan. | |
Before receiving Clariscan, tell your healthcare provider about all your medical conditions, including if you:
|
|
What are possible side effects of Clariscan?
|
|
|
The most common side effects of Clariscan include: nausea, headache, pain, or cold feeling at the injection site, and rash.
These are not all the possible side effects of Clariscan. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
General information about the safe and effective uses of Clariscan.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your healthcare provider for information about Clariscan that is written for health professionals. |
|
|
What are the ingredients in Clariscan?
Active ingredient: gadoterate meglumine Inactive ingredients: DOTA, water for injection © 2025 GE HealthCare Distributed by GE Healthcare Inc., Arlington Heights, IL 60004 U.S.A. GE is a trademark of General Electric Company used under trademark license. For more information, go to www.GEHealthCare.com or call 1-800-654-0118. |
Section 44425-7
Storage
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP, Controlled Room Temperature].
Pre-filled syringes must not be frozen. Frozen syringes should be discarded.
Should solidification occur in the vial because of exposure to the cold, bring Clariscan to room temperature before use. If allowed to stand at room temperature for a minimum of 90 minutes, Clariscan should return to a clear, colorless to yellow solution. Before use, examine the product to assure that all solids are dissolved and that the container and closure have not been damaged. Discard the vial if solids persist.
10 Overdosage
Clariscan administered to healthy volunteers and to adult patients at cumulative doses up to 0.3 mmol/kg was tolerated in a manner similar to lower doses. Adverse reactions to overdosage with gadoterate meglumine have not been reported. Gadoterate meglumine can be removed from the body by hemodialysis [see Clinical Pharmacology (12.3)].
11 Description
Clariscan (gadoterate meglumine) is a paramagnetic macrocyclic ionic contrast agent administered for magnetic resonance imaging. The chemical name for gadoterate meglumine is D-glucitol, 1-deoxy-1-(methylamino)-, [1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraaceto(4-)-.kappa.N1, .kappa.N4, .kappa.N7, .kappa.N10, .kappa.O1, .kappa.O4, .kappa.O7, .kappa.O10]gadolinate(1-)(1:1); it has a formula weight of 753.9 g/mol and empirical formula of C23H42O13N5Gd (anhydrous basis).
The structural formula of gadoterate meglumine in solution is as follows:
CAS Registry No. 92943-93-6
Clariscan Injection is a sterile, nonpyrogenic, clear, colorless to yellow, aqueous solution of 0.5 mmol/mL of gadoterate meglumine. Each vial and pre-filled syringe contains 2.5 mmol per 5 mL, 5 mmol per 10 mL, 7.5 mmol per 15 mL and 10 mmol per 20 mL. No preservative is added. Each mL of Clariscan contains 376.9 mg of gadoterate meglumine, 0.25 mg of DOTA and water for injection. Clariscan has a pH of 6.5 to 8.0.
The main physiochemical properties of Clariscan are provided below:
| Parameter | Value |
|---|---|
| Density @ 20°C | 1.1753 g/cm3 |
| Viscosity @ 20°C | 3.4 mPa.s |
| Viscosity @ 37°C | 2.4 mPa.s |
| Osmolality | 1350 mOsm/kg water |
The thermodynamic stability constants for gadoterate (log Ktherm and log Kcond at pH 7.4) are 25.6 and 19.3, respectively.
2.2 Drug Handling
- Visually inspect Clariscan for particulate matter prior to administration. Do not use the solution if particulate matter is present or if the container appears damaged. Clariscan should be a clear, colorless to yellow solution.
- Do not mix with other drugs or parenteral nutrition.
- Discard any unused portions of the drug.
8.4 Pediatric Use
The safety and efficacy of gadoterate meglumine at a single dose of 0.1 mmol/kg have been established in pediatric patients from birth (term neonates ≥ 37 weeks gestational age) to 17 years of age based on clinical data in 133 pediatric patients 2 years of age and older, and clinical data in 52 pediatric patients birth to less than 2 years of age that supported extrapolation from adult data [see Clinical Studies (14)]. Adverse reactions in pediatric patients were similar to those reported in adults [see Adverse Reactions (6.1)]. No dosage adjustment according to age is necessary in pediatric patients [see Dosage and Administration (2.1), Pharmacokinetics (12.3)]. The safety of gadoterate meglumine has not been established in preterm neonates.
No cases of NSF associated with gadoterate meglumine or any other GBCA have been identified in pediatric patients age 6 years and younger [see Warnings and Precautions (5.2)]. Normal estimated GFR (eGFR) is approximately 30 mL/minute/1.73 m2 at birth and increases to adult values by 2 years of age.
8.5 Geriatric Use
In clinical studies of gadoterate meglumine, 900 patients were 65 years of age and over, and 304 patients were 75 years of age and over. No overall differences in safety or efficacy were observed between these subjects and younger subjects. In general, use of Clariscan in elderly patients should be cautious, reflecting the greater frequency of impaired renal function and concomitant disease or other drug therapy. No age-related dosage adjustment is necessary.
4 Contraindications
History of clinically important hypersensitivity reactions to Clariscan [see Warnings and Precautions (5.3)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
Nephrogenic systemic fibrosis [see Warnings and Precautions (5.2)]
Hypersensitivity reactions [see Warnings and Precautions (5.3)]
Gadolinium Retention [see Warnings and Precautions (5.4)]
7 Drug Interactions
Gadoterate does not interfere with serum and plasma calcium measurements determined by colorimetric assays. Specific drug interaction studies with gadoterate meglumine have not been conducted.
8.6 Renal Impairment
No Clariscan dosage adjustment is recommended for patients with renal impairment. Gadoterate meglumine can be removed from the body by hemodialysis [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Gadoterate affects proton relaxation times and consequently the MR signal, and the contrast obtained is characterized by the relaxivity of the gadoterate molecule. The relaxivity values for gadoterate are similar across the spectrum of magnetic field strengths used in clinical MRI (0.2 to 1.5 T).
Disruption of the blood-brain barrier or abnormal vascularity allows distribution of gadoterate in lesions such as neoplasms, abscesses, and infarcts.
12.3 Pharmacokinetics
The pharmacokinetics of total gadolinium assessed up to 48 hours following an intravenously administered 0.1 mmol/kg dose of gadoterate meglumine in healthy adult subjects demonstrated a mean elimination half-life (reported as mean ± SD) of about 1.4 ± 0.2 hours and 2.0 ± 0.7 hours in female and male subjects, respectively. Similar pharmacokinetic profile and elimination half-life values were observed after intravenous injection of 0.1 mmol/kg of gadoterate meglumine followed 20 minutes later by a second injection of 0.2 mmol/kg (1.7 ± 0.3 hours and 1.9 ± 0.2 hours in female and male subjects, respectively).
2.1 Dosing Guidelines
For adult and pediatric patients (including term neonates), the recommended dose of Clariscan is 0.2 mL/kg (0.1 mmol/kg) body weight administered as an intravenous bolus injection, manually or by power injector, at a flow rate of approximately 2 mL/second for adults and 1 to 2 mL/second for pediatric patients. Table 1 provides weight-adjusted dose volumes.
| Body Weight | Volume | |
|---|---|---|
| Pounds (lb) | Kilograms (kg) | Milliliters (mL) |
| 5.5 | 2.5 | 0.5 |
| 11 | 5 | 1 |
| 22 | 10 | 2 |
| 44 | 20 | 4 |
| 66 | 30 | 6 |
| 88 | 40 | 8 |
| 110 | 50 | 10 |
| 132 | 60 | 12 |
| 154 | 70 | 14 |
| 176 | 80 | 16 |
| 198 | 90 | 18 |
| 220 | 100 | 20 |
| 242 | 110 | 22 |
| 264 | 120 | 24 |
| 286 | 130 | 26 |
| 308 | 140 | 28 |
| 330 | 150 | 30 |
To ensure complete injection of Clariscan, the injection may be followed by normal saline flush. Contrast MRI can begin immediately following Clariscan injection.
1 Indications and Usage
Clariscan is a gadolinium-based contrast agent indicated for intravenous use with magnetic resonance imaging (MRI) in brain (intracranial), spine and associated tissues in adult and pediatric patients (including term neonates) to detect and visualize areas with disruption of the blood brain barrier (BBB) and/or abnormal vascularity.
5.5 Acute Kidney Injury
In patients with chronically reduced renal function, acute kidney injury requiring dialysis has occurred with the use of GBCAs. The risk of acute kidney injury may increase with increasing dose of the contrast agent; administer the lowest dose necessary for adequate imaging. Screen all patients for renal impairment by obtaining a history and/or laboratory tests. Consider follow-up renal function assessments for patients with a history of renal dysfunction.
12.1 Mechanism of Action
Gadoterate is a paramagnetic molecule that develops a magnetic moment when placed in a magnetic field. The magnetic moment enhances the relaxation rates of water protons in its vicinity, leading to an increase in signal intensity (brightness) of tissues.
In magnetic resonance imaging (MRI), visualization of normal and pathological tissue depends in part on variations in the radiofrequency signal intensity that occurs with:
-
1)differences in proton density
-
2)differences of the spin-lattice or longitudinal relaxation times (T1)
-
3)differences in the spin-spin or transverse relaxation time (T2)
When placed in a magnetic field, gadoterate shortens the T1 and T2 relaxation times in target tissues. At recommended doses, the effect is observed with greatest sensitivity in the T1-weighted sequences.
17.3 General Precautions
- Pregnancy: Advise pregnant women of the potential risk of fetal exposure to gadoterate [see Use in Specific Populations (8.1)].
- Gadolinium Retention: Advise patients that gadolinium is retained for months or years in brain, bone, skin, and other organs in patients with normal renal function. The clinical consequences of retention are unknown. Retention depends on multiple factors and is greater following administration of linear GBCAs than following administration of macrocyclic GBCAs [see Warnings and Precautions (5.4)].
5.4 Gadolinium Retention
Gadolinium is retained for months or years in several organs. The highest concentrations (nanomoles per gram of tissue) have been identified in the bone, followed by other organs (e.g., brain, skin, kidney, liver and spleen). The duration of retention also varies by tissue and is longest in bone. Linear GBCAs cause more retention than macrocyclic GBCAs. At equivalent doses, gadolinium retention varies among the linear agents with Omniscan (gadodiamide) and Optimark (gadoversetamide) causing greater retention than other linear agents [Eovist (gadoxetate disodium), Magnevist (gadopentetate dimeglumine), MultiHance (gadobenate dimeglumine)]. Retention is lowest and similar among the macrocyclic GBCAs [Clariscan (gadoterate meglumine), Dotarem (gadoterate meglumine), Gadavist (gadobutrol), ProHance (gadoteridol)].
Consequences of gadolinium retention in the brain have not been established. Pathologic and clinical consequences of GBCA administration and retention in skin and other organs have been established in patients with impaired renal function [see Warnings and Precautions (5.2)]. There are rare reports of pathologic skin changes in patients with normal renal function. Adverse events involving multiple organ systems have been reported in patients with normal renal function without an established causal link to gadolinium retention [see Adverse Reactions (6.2)].
While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients, and patients with inflammatory conditions. Consider the retention characteristics of the agent when choosing a GBCA for these patients. Minimize repetitive GBCA imaging studies, particularly closely spaced studies when possible.
5 Warnings and Precautions
- Hypersensitivity Reactions: Anaphylactoid/anaphylactic reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have uncommonly occurred. Monitor patients closely for need of emergency cardiorespiratory support. (5.3)
- Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs. (5.4)
2 Dosage and Administration
Adult and pediatric patients: The recommended dose of Clariscan is 0.2 mL/kg (0.1 mmol/kg) body weight administered as an intravenous bolus injection at a flow rate of approximately 2 mL/second for adults and 1 to 2 mL/second for pediatric patients (including term neonates). The dose is delivered by manual or power injection. (2)
3 Dosage Forms and Strengths
Clariscan 0.5 mmol/mL is a sterile, clear, colorless to yellow, aqueous solution for intravenous injection containing 376.9 mg/mL gadoterate meglumine and is available in vials and pre-filled syringes.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during postmarketing use of gadoterate meglumine or other GBCAs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
| System Organ Class | Adverse Reaction |
|---|---|
| Cardiac Disorders | bradycardia, tachycardia, arrhythmia |
| Immune System Disorders | hypersensitivity / anaphylactoid reactions including cardiac arrest, respiratory arrest, cyanosis, pharyngeal edema, laryngospasm, bronchospasm, angioedema, conjunctivitis, ocular hyperemia, eyelid edema, lacrimation increased, hyperhidrosis, urticaria |
| Nervous System Disorders | coma, convulsion, syncope, presyncope, parosmia, tremor |
| Musculoskeletal and Connective Tissue Disorders | muscle contracture, muscle weakness |
| Gastrointestinal Disorders | diarrhea, salivary hypersecretion, acute pancreatitis with onset within 48 hours after GBCA administration |
| General Disorders and Administration Site Conditions | malaise, fever Adverse reactions with variable onset and duration have been reported after GBCA administration. These include fatigue, asthenia, pain syndromes, and heterogeneous clusters of symptoms in the neurological, cutaneous, and musculoskeletal systems. |
| Respiratory, Thoracic and Mediastinal Disorders | Acute respiratory distress syndrome, pulmonary edema |
| Skin and Subcutaneous Tissue Disorders | NSF, in patients whose reports were confounded by the receipt of other GBCAs or in situations where receipt of other GBCAs could not be ruled out. No unconfounded cases of NSF have been reported with gadoterate meglumine. Gadolinium-associated plaques. |
| Vascular Disorders | superficial phlebitis |
17.2 Common Adverse Reactions
Inform patients that they may experience:
- Reactions along the venous injection site, such as mild and transient burning or pain or feeling of warmth or coldness at the injection site.
- Side effects of headache, nausea, abnormal taste and feeling hot.
8 Use in Specific Populations
Pregnancy: Use only if imaging is essential during pregnancy and cannot be delayed. (8.1)
5.3 Hypersensitivity Reactions
Anaphylactic and anaphylactoid reactions have been reported with gadoterate meglumine, involving cardiovascular, respiratory, and/or cutaneous manifestations. Some patients experienced circulatory collapse and died. In most cases, initial symptoms occurred within minutes of gadoterate meglumine administration and resolved with prompt emergency treatment [see Adverse Reactions (6)].
- Before Clariscan administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and/or allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to Clariscan.
- Administer Clariscan only in situations where trained personnel and therapies are promptly available for the treatment of hypersensitivity reactions, including personnel trained in resuscitation.
- During and following Clariscan administration, observe patients for signs and symptoms of hypersensitivity reactions.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data described below reflect gadoterate meglumine exposure in 2867 patients, representing 2682 adults and 185 pediatric patients. Overall, 55% of the patients were men. In clinical trials where ethnicity was recorded, the ethnic distribution was 81% Caucasian, 11% Asian, 4% Black, and 4% others. The average age was 53 years (range from < 1 week to 97 years).
Overall, 4% of patients reported at least one adverse reaction, primarily occurring immediately or within 24 hours following gadoterate meglumine administration. Most adverse reactions were mild or moderate in intensity and transient in nature.
Table 2 lists adverse reactions that occurred in ≥ 0.2% patients who received gadoterate meglumine.
| Reaction | Rate (%) n= 2867 |
|---|---|
| Nausea | 0.6% |
| Headache | 0.4% |
| Injection Site Pain | 0.4% |
| Injection Site Coldness | 0.2% |
| Rash | 0.2% |
Adverse reactions that occurred with a frequency < 0.2% in patients who received gadoterate meglumine include: feeling cold, feeling hot, burning sensation, somnolence, pain, dizziness, dysgeusia, blood creatinine increased, hypotension, hypertension, asthenia, fatigue, injection site reactions (inflammation, extravasation, pruritus, swelling, warmth), paresthesia, pruritus, laryngeal discomfort, pain in extremity, vomiting, anxiety and palpitations.
17 Patient Counseling Information
- Advise the patient to read the FDA-approved patient labeling (Medication Guide)
5.2 Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of Clariscan among these patients unless the diagnostic information is essential and not available with non-contrast MRI or other modalities. The GBCA-associated NSF risk appears highest for patients with chronic, severe kidney disease (GFR < 30 mL/min/1.73 m2) as well as patients with acute kidney injury. The risk appears lower for patients with chronic, moderate kidney disease (GFR 30 to 59 mL/min/1.73 m2) and little, if any, for patients with chronic, mild kidney disease (GFR 60 to 89 mL/min/1.73 m2). NSF may result in fatal or debilitating fibrosis affecting the skin, muscle, and internal organs. Report any diagnosis of NSF following Clariscan administration to GE HealthCare at (1-800-654-0118) or FDA at (1-800-FDA-1088 or www.fda.gov/medwatch).
Screen patients for acute kidney injury and other conditions that may reduce renal function. Features of acute kidney injury consist of rapid (over hours to days), and usually reversible, decrease in kidney function, commonly in the setting of surgery, severe infection, injury or drug-induced kidney toxicity. Serum creatinine levels and estimated GFR may not reliably assess renal function in the setting of acute kidney injury. For patients at risk for chronically reduced renal function (e.g., age > 60 years, diabetes mellitus or chronic hypertension), estimate the GFR through laboratory testing.
The factors that may increase the risk for NSF are repeated or higher than recommended doses of a GBCA, and the degree of renal impairment at the time of exposure. Record the specific GBCA and the dose administered to a patient. For patients at highest risk for NSF, do not exceed the recommended Clariscan dose and allow a sufficient period of time for elimination of the drug prior to re-administration. For patients receiving hemodialysis, physicians may consider the prompt initiation of hemodialysis following the administration of a GBCA in order to enhance the contrast agent's elimination. The usefulness of hemodialysis in the prevention of NSF is unknown [see Dosage and Administration (2) and Clinical Pharmacology (12)].
17.1 Nephrogenic Systemic Fibrosis
Instruct patients to inform their healthcare provider if they:
- have a history of kidney disease, or
- have recently received a GBCA.
GBCAs increase the risk for NSF among patients with impaired elimination of the drugs. To counsel patients at risk for NSF:
- Describe the clinical manifestations of NSF.
- Describe procedures to screen for the detection of renal impairment.
Instruct the patients to contact their physician if they develop signs or symptoms of NSF following Clariscan administration, such as burning, itching, swelling, scaling, hardening and tightening of the skin; red or dark patches on the skin; stiffness in joints with trouble moving, bending or straightening the arms, hands, legs or feet; pain in the hip bones or ribs; or muscle weakness.
16 How Supplied/storage and Handling
Clariscan Injection is a clear, colorless to yellow solution containing 0.5 mmol/mL of gadoterate meglumine. It is supplied in vials and pre-filled syringes.
- Clariscan Injection is supplied in 10 mL vials containing 5 mL or 10 mL of solution and in 20 mL vials containing 15 mL or 20 mL of solution.
Each single-dose vial is closed with a rubber stopper and sealed with an aluminum cap and the contents are sterile. Vials are packaged in a box of 10, in the following configurations:
| 2.5 mmol per 5 mL (0.5 mmol per mL) in glass vial | (NDC 0407-2943-06) |
| 5 mmol per 10 mL (0.5 mmol per mL) in glass vial | (NDC 0407-2943-01) |
| 7.5 mmol per 15 mL (0.5 mmol per mL) in glass vial | (NDC 0407-2943-02) |
| 10 mmol per 20 mL (0.5 mmol per mL) in glass vial | (NDC 0407-2943-05) |
- Clariscan Injection is supplied in 20 mL plastic pre-filled syringes containing 10 mL, 15 mL, or 20 mL of solution.
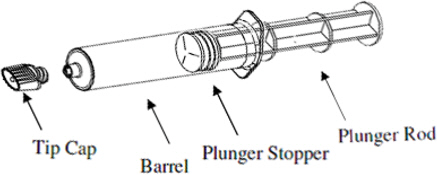
Each syringe is sealed with rubber closures and the contents are sterile. Syringes, including plunger rod, are individually packaged in a box of 10, in the following configurations:
| 5 mmol per 10 mL (0.5 mmol per mL) in plastic pre-filled syringe | (NDC 0407-2943-12) |
| 7.5 mmol per 15 mL (0.5 mmol per mL) in plastic pre-filled syringe | (NDC 0407-2943-17) |
| 10 mmol per 20 mL (0.5 mmol per mL) in plastic pre-filled syringe | (NDC 0407-2943-22) |
5.1 Risk Associated With Intrathecal Use
Intrathecal administration of GBCAs can cause serious adverse reactions including death, coma, encephalopathy, and seizures. The safety and effectiveness of Clariscan have not been established with intrathecal use. Clariscan is not approved for intrathecal use [see Dosage and Administration (2.1)].
13.2 Animal Toxicology And/or Pharmacology
Local intolerance reactions, including moderate irritation associated with infiltration of inflammatory cells were observed after perivenous injection in rabbits suggesting the possibility of local irritation if the contrast medium leaks around the veins in a clinical setting [see Warnings and Precautions (5.5)].
Toxicity of gadoterate meglumine was evaluated in neonatal and juvenile (pre- and post-weaning) rats following a single or repeated intravenous administration at doses 1, 2, and 4 times the MHD based on BSA. Gadoterate meglumine was well tolerated at all dose levels tested and had no effect on growth, pre-weaning development, behavior and sexual maturation.
5.6 Extravasation and Injection Site Reactions
Ensure catheter and venous patency before the injection of Clariscan. Extravasation into tissues during Clariscan administration may result in tissue irritation [see Nonclinical Toxicology (13.2)].
Principal Display Panel 10 Ml Vial Box Label
NDC 0407-2943-01
Rx Only
Clariscan™
(gadoterate meglumine) Injection
5 mmol per 10 mL
(0.5 mmol per mL)
For Intravenous Administration
Sterile Solution
Discard Unused Portion
Dispense the accompanying
Medication Guide to each patient
10 x 10 mL Single-dose Vials
Principal Display Panel 15 Ml Syringe Box Label
NDC 0407-2943-17
Rx Only
Clariscan™
(gadoterate meglumine) Injection
7.5 mmol per 15 mL
(0.5 mmol per mL)
Sterile Solution
For Intravenous Administration
Discard Unused Portion
Dispense the accompanying
Medication Guide to each patient
1 x 15 mL Single-Dose Plastic Syringe
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed to evaluate the carcinogenic potential of gadoterate meglumine.
Gadoterate meglumine did not demonstrate mutagenic potential in in vitro bacterial reverse mutation assays (Ames test) using Salmonella typhimurium, in an in vitro chromosome aberration assay in Chinese hamster ovary cells, in an in vitro gene mutation assay in Chinese hamster lung cells, nor in an in vivo mouse micronucleus assay.
No impairment of male or female fertility and reproductive performance was observed in rats after intravenous administration of gadoterate meglumine at the maximum tested dose of 10 mmol/kg/day (16 times the maximum human dose based on surface area), given during more than 9 weeks in males and more than 4 weeks in females. Sperm counts and sperm motility were not adversely affected by treatment with the drug.
Warning: Risk Associated With Intrathecal Use and Nephrogenic Systemic Fibrosis
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS
See full prescribing information for complete boxed warning
- Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. Clariscan is not approved for intrathecal use. (5.1)
-
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of Clariscan in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. The risk for NSF appears highest among patients with:
- Chronic, severe kidney disease (GFR < 30 mL/min/1.73 m2), or
- Acute kidney injury.
Structured Label Content
Section 42229-5 (42229-5)
Risk Associated with Intrathecal Use
Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. Clariscan is not approved for intrathecal use [see Warnings and Precautions (5.1)].
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Rev. 3/2025 |
| MEDICATION GUIDE | |
|
CLARISCAN™ (kla-ri'-skan) (gadoterate meglumine) Injection, for intravenous use |
|
What is the most important information I should know about Clariscan?
|
|
What is Clariscan?
|
|
| Do not receive Clariscan if you have had a severe allergic reaction to Clariscan. | |
Before receiving Clariscan, tell your healthcare provider about all your medical conditions, including if you:
|
|
What are possible side effects of Clariscan?
|
|
|
The most common side effects of Clariscan include: nausea, headache, pain, or cold feeling at the injection site, and rash.
These are not all the possible side effects of Clariscan. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
General information about the safe and effective uses of Clariscan.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your healthcare provider for information about Clariscan that is written for health professionals. |
|
|
What are the ingredients in Clariscan?
Active ingredient: gadoterate meglumine Inactive ingredients: DOTA, water for injection © 2025 GE HealthCare Distributed by GE Healthcare Inc., Arlington Heights, IL 60004 U.S.A. GE is a trademark of General Electric Company used under trademark license. For more information, go to www.GEHealthCare.com or call 1-800-654-0118. |
Section 44425-7 (44425-7)
Storage
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP, Controlled Room Temperature].
Pre-filled syringes must not be frozen. Frozen syringes should be discarded.
Should solidification occur in the vial because of exposure to the cold, bring Clariscan to room temperature before use. If allowed to stand at room temperature for a minimum of 90 minutes, Clariscan should return to a clear, colorless to yellow solution. Before use, examine the product to assure that all solids are dissolved and that the container and closure have not been damaged. Discard the vial if solids persist.
10 Overdosage (10 OVERDOSAGE)
Clariscan administered to healthy volunteers and to adult patients at cumulative doses up to 0.3 mmol/kg was tolerated in a manner similar to lower doses. Adverse reactions to overdosage with gadoterate meglumine have not been reported. Gadoterate meglumine can be removed from the body by hemodialysis [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
Clariscan (gadoterate meglumine) is a paramagnetic macrocyclic ionic contrast agent administered for magnetic resonance imaging. The chemical name for gadoterate meglumine is D-glucitol, 1-deoxy-1-(methylamino)-, [1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraaceto(4-)-.kappa.N1, .kappa.N4, .kappa.N7, .kappa.N10, .kappa.O1, .kappa.O4, .kappa.O7, .kappa.O10]gadolinate(1-)(1:1); it has a formula weight of 753.9 g/mol and empirical formula of C23H42O13N5Gd (anhydrous basis).
The structural formula of gadoterate meglumine in solution is as follows:
CAS Registry No. 92943-93-6
Clariscan Injection is a sterile, nonpyrogenic, clear, colorless to yellow, aqueous solution of 0.5 mmol/mL of gadoterate meglumine. Each vial and pre-filled syringe contains 2.5 mmol per 5 mL, 5 mmol per 10 mL, 7.5 mmol per 15 mL and 10 mmol per 20 mL. No preservative is added. Each mL of Clariscan contains 376.9 mg of gadoterate meglumine, 0.25 mg of DOTA and water for injection. Clariscan has a pH of 6.5 to 8.0.
The main physiochemical properties of Clariscan are provided below:
| Parameter | Value |
|---|---|
| Density @ 20°C | 1.1753 g/cm3 |
| Viscosity @ 20°C | 3.4 mPa.s |
| Viscosity @ 37°C | 2.4 mPa.s |
| Osmolality | 1350 mOsm/kg water |
The thermodynamic stability constants for gadoterate (log Ktherm and log Kcond at pH 7.4) are 25.6 and 19.3, respectively.
2.2 Drug Handling
- Visually inspect Clariscan for particulate matter prior to administration. Do not use the solution if particulate matter is present or if the container appears damaged. Clariscan should be a clear, colorless to yellow solution.
- Do not mix with other drugs or parenteral nutrition.
- Discard any unused portions of the drug.
8.4 Pediatric Use
The safety and efficacy of gadoterate meglumine at a single dose of 0.1 mmol/kg have been established in pediatric patients from birth (term neonates ≥ 37 weeks gestational age) to 17 years of age based on clinical data in 133 pediatric patients 2 years of age and older, and clinical data in 52 pediatric patients birth to less than 2 years of age that supported extrapolation from adult data [see Clinical Studies (14)]. Adverse reactions in pediatric patients were similar to those reported in adults [see Adverse Reactions (6.1)]. No dosage adjustment according to age is necessary in pediatric patients [see Dosage and Administration (2.1), Pharmacokinetics (12.3)]. The safety of gadoterate meglumine has not been established in preterm neonates.
No cases of NSF associated with gadoterate meglumine or any other GBCA have been identified in pediatric patients age 6 years and younger [see Warnings and Precautions (5.2)]. Normal estimated GFR (eGFR) is approximately 30 mL/minute/1.73 m2 at birth and increases to adult values by 2 years of age.
8.5 Geriatric Use
In clinical studies of gadoterate meglumine, 900 patients were 65 years of age and over, and 304 patients were 75 years of age and over. No overall differences in safety or efficacy were observed between these subjects and younger subjects. In general, use of Clariscan in elderly patients should be cautious, reflecting the greater frequency of impaired renal function and concomitant disease or other drug therapy. No age-related dosage adjustment is necessary.
4 Contraindications (4 CONTRAINDICATIONS)
History of clinically important hypersensitivity reactions to Clariscan [see Warnings and Precautions (5.3)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
Nephrogenic systemic fibrosis [see Warnings and Precautions (5.2)]
Hypersensitivity reactions [see Warnings and Precautions (5.3)]
Gadolinium Retention [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Gadoterate does not interfere with serum and plasma calcium measurements determined by colorimetric assays. Specific drug interaction studies with gadoterate meglumine have not been conducted.
8.6 Renal Impairment
No Clariscan dosage adjustment is recommended for patients with renal impairment. Gadoterate meglumine can be removed from the body by hemodialysis [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Gadoterate affects proton relaxation times and consequently the MR signal, and the contrast obtained is characterized by the relaxivity of the gadoterate molecule. The relaxivity values for gadoterate are similar across the spectrum of magnetic field strengths used in clinical MRI (0.2 to 1.5 T).
Disruption of the blood-brain barrier or abnormal vascularity allows distribution of gadoterate in lesions such as neoplasms, abscesses, and infarcts.
12.3 Pharmacokinetics
The pharmacokinetics of total gadolinium assessed up to 48 hours following an intravenously administered 0.1 mmol/kg dose of gadoterate meglumine in healthy adult subjects demonstrated a mean elimination half-life (reported as mean ± SD) of about 1.4 ± 0.2 hours and 2.0 ± 0.7 hours in female and male subjects, respectively. Similar pharmacokinetic profile and elimination half-life values were observed after intravenous injection of 0.1 mmol/kg of gadoterate meglumine followed 20 minutes later by a second injection of 0.2 mmol/kg (1.7 ± 0.3 hours and 1.9 ± 0.2 hours in female and male subjects, respectively).
2.1 Dosing Guidelines
For adult and pediatric patients (including term neonates), the recommended dose of Clariscan is 0.2 mL/kg (0.1 mmol/kg) body weight administered as an intravenous bolus injection, manually or by power injector, at a flow rate of approximately 2 mL/second for adults and 1 to 2 mL/second for pediatric patients. Table 1 provides weight-adjusted dose volumes.
| Body Weight | Volume | |
|---|---|---|
| Pounds (lb) | Kilograms (kg) | Milliliters (mL) |
| 5.5 | 2.5 | 0.5 |
| 11 | 5 | 1 |
| 22 | 10 | 2 |
| 44 | 20 | 4 |
| 66 | 30 | 6 |
| 88 | 40 | 8 |
| 110 | 50 | 10 |
| 132 | 60 | 12 |
| 154 | 70 | 14 |
| 176 | 80 | 16 |
| 198 | 90 | 18 |
| 220 | 100 | 20 |
| 242 | 110 | 22 |
| 264 | 120 | 24 |
| 286 | 130 | 26 |
| 308 | 140 | 28 |
| 330 | 150 | 30 |
To ensure complete injection of Clariscan, the injection may be followed by normal saline flush. Contrast MRI can begin immediately following Clariscan injection.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Clariscan is a gadolinium-based contrast agent indicated for intravenous use with magnetic resonance imaging (MRI) in brain (intracranial), spine and associated tissues in adult and pediatric patients (including term neonates) to detect and visualize areas with disruption of the blood brain barrier (BBB) and/or abnormal vascularity.
5.5 Acute Kidney Injury
In patients with chronically reduced renal function, acute kidney injury requiring dialysis has occurred with the use of GBCAs. The risk of acute kidney injury may increase with increasing dose of the contrast agent; administer the lowest dose necessary for adequate imaging. Screen all patients for renal impairment by obtaining a history and/or laboratory tests. Consider follow-up renal function assessments for patients with a history of renal dysfunction.
12.1 Mechanism of Action
Gadoterate is a paramagnetic molecule that develops a magnetic moment when placed in a magnetic field. The magnetic moment enhances the relaxation rates of water protons in its vicinity, leading to an increase in signal intensity (brightness) of tissues.
In magnetic resonance imaging (MRI), visualization of normal and pathological tissue depends in part on variations in the radiofrequency signal intensity that occurs with:
-
1)differences in proton density
-
2)differences of the spin-lattice or longitudinal relaxation times (T1)
-
3)differences in the spin-spin or transverse relaxation time (T2)
When placed in a magnetic field, gadoterate shortens the T1 and T2 relaxation times in target tissues. At recommended doses, the effect is observed with greatest sensitivity in the T1-weighted sequences.
17.3 General Precautions
- Pregnancy: Advise pregnant women of the potential risk of fetal exposure to gadoterate [see Use in Specific Populations (8.1)].
- Gadolinium Retention: Advise patients that gadolinium is retained for months or years in brain, bone, skin, and other organs in patients with normal renal function. The clinical consequences of retention are unknown. Retention depends on multiple factors and is greater following administration of linear GBCAs than following administration of macrocyclic GBCAs [see Warnings and Precautions (5.4)].
5.4 Gadolinium Retention
Gadolinium is retained for months or years in several organs. The highest concentrations (nanomoles per gram of tissue) have been identified in the bone, followed by other organs (e.g., brain, skin, kidney, liver and spleen). The duration of retention also varies by tissue and is longest in bone. Linear GBCAs cause more retention than macrocyclic GBCAs. At equivalent doses, gadolinium retention varies among the linear agents with Omniscan (gadodiamide) and Optimark (gadoversetamide) causing greater retention than other linear agents [Eovist (gadoxetate disodium), Magnevist (gadopentetate dimeglumine), MultiHance (gadobenate dimeglumine)]. Retention is lowest and similar among the macrocyclic GBCAs [Clariscan (gadoterate meglumine), Dotarem (gadoterate meglumine), Gadavist (gadobutrol), ProHance (gadoteridol)].
Consequences of gadolinium retention in the brain have not been established. Pathologic and clinical consequences of GBCA administration and retention in skin and other organs have been established in patients with impaired renal function [see Warnings and Precautions (5.2)]. There are rare reports of pathologic skin changes in patients with normal renal function. Adverse events involving multiple organ systems have been reported in patients with normal renal function without an established causal link to gadolinium retention [see Adverse Reactions (6.2)].
While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients, and patients with inflammatory conditions. Consider the retention characteristics of the agent when choosing a GBCA for these patients. Minimize repetitive GBCA imaging studies, particularly closely spaced studies when possible.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions: Anaphylactoid/anaphylactic reactions with cardiovascular, respiratory or cutaneous manifestations, ranging from mild to severe, including death, have uncommonly occurred. Monitor patients closely for need of emergency cardiorespiratory support. (5.3)
- Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs. (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Adult and pediatric patients: The recommended dose of Clariscan is 0.2 mL/kg (0.1 mmol/kg) body weight administered as an intravenous bolus injection at a flow rate of approximately 2 mL/second for adults and 1 to 2 mL/second for pediatric patients (including term neonates). The dose is delivered by manual or power injection. (2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Clariscan 0.5 mmol/mL is a sterile, clear, colorless to yellow, aqueous solution for intravenous injection containing 376.9 mg/mL gadoterate meglumine and is available in vials and pre-filled syringes.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during postmarketing use of gadoterate meglumine or other GBCAs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
| System Organ Class | Adverse Reaction |
|---|---|
| Cardiac Disorders | bradycardia, tachycardia, arrhythmia |
| Immune System Disorders | hypersensitivity / anaphylactoid reactions including cardiac arrest, respiratory arrest, cyanosis, pharyngeal edema, laryngospasm, bronchospasm, angioedema, conjunctivitis, ocular hyperemia, eyelid edema, lacrimation increased, hyperhidrosis, urticaria |
| Nervous System Disorders | coma, convulsion, syncope, presyncope, parosmia, tremor |
| Musculoskeletal and Connective Tissue Disorders | muscle contracture, muscle weakness |
| Gastrointestinal Disorders | diarrhea, salivary hypersecretion, acute pancreatitis with onset within 48 hours after GBCA administration |
| General Disorders and Administration Site Conditions | malaise, fever Adverse reactions with variable onset and duration have been reported after GBCA administration. These include fatigue, asthenia, pain syndromes, and heterogeneous clusters of symptoms in the neurological, cutaneous, and musculoskeletal systems. |
| Respiratory, Thoracic and Mediastinal Disorders | Acute respiratory distress syndrome, pulmonary edema |
| Skin and Subcutaneous Tissue Disorders | NSF, in patients whose reports were confounded by the receipt of other GBCAs or in situations where receipt of other GBCAs could not be ruled out. No unconfounded cases of NSF have been reported with gadoterate meglumine. Gadolinium-associated plaques. |
| Vascular Disorders | superficial phlebitis |
17.2 Common Adverse Reactions
Inform patients that they may experience:
- Reactions along the venous injection site, such as mild and transient burning or pain or feeling of warmth or coldness at the injection site.
- Side effects of headache, nausea, abnormal taste and feeling hot.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Use only if imaging is essential during pregnancy and cannot be delayed. (8.1)
5.3 Hypersensitivity Reactions
Anaphylactic and anaphylactoid reactions have been reported with gadoterate meglumine, involving cardiovascular, respiratory, and/or cutaneous manifestations. Some patients experienced circulatory collapse and died. In most cases, initial symptoms occurred within minutes of gadoterate meglumine administration and resolved with prompt emergency treatment [see Adverse Reactions (6)].
- Before Clariscan administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and/or allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to Clariscan.
- Administer Clariscan only in situations where trained personnel and therapies are promptly available for the treatment of hypersensitivity reactions, including personnel trained in resuscitation.
- During and following Clariscan administration, observe patients for signs and symptoms of hypersensitivity reactions.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data described below reflect gadoterate meglumine exposure in 2867 patients, representing 2682 adults and 185 pediatric patients. Overall, 55% of the patients were men. In clinical trials where ethnicity was recorded, the ethnic distribution was 81% Caucasian, 11% Asian, 4% Black, and 4% others. The average age was 53 years (range from < 1 week to 97 years).
Overall, 4% of patients reported at least one adverse reaction, primarily occurring immediately or within 24 hours following gadoterate meglumine administration. Most adverse reactions were mild or moderate in intensity and transient in nature.
Table 2 lists adverse reactions that occurred in ≥ 0.2% patients who received gadoterate meglumine.
| Reaction | Rate (%) n= 2867 |
|---|---|
| Nausea | 0.6% |
| Headache | 0.4% |
| Injection Site Pain | 0.4% |
| Injection Site Coldness | 0.2% |
| Rash | 0.2% |
Adverse reactions that occurred with a frequency < 0.2% in patients who received gadoterate meglumine include: feeling cold, feeling hot, burning sensation, somnolence, pain, dizziness, dysgeusia, blood creatinine increased, hypotension, hypertension, asthenia, fatigue, injection site reactions (inflammation, extravasation, pruritus, swelling, warmth), paresthesia, pruritus, laryngeal discomfort, pain in extremity, vomiting, anxiety and palpitations.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
- Advise the patient to read the FDA-approved patient labeling (Medication Guide)
5.2 Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of Clariscan among these patients unless the diagnostic information is essential and not available with non-contrast MRI or other modalities. The GBCA-associated NSF risk appears highest for patients with chronic, severe kidney disease (GFR < 30 mL/min/1.73 m2) as well as patients with acute kidney injury. The risk appears lower for patients with chronic, moderate kidney disease (GFR 30 to 59 mL/min/1.73 m2) and little, if any, for patients with chronic, mild kidney disease (GFR 60 to 89 mL/min/1.73 m2). NSF may result in fatal or debilitating fibrosis affecting the skin, muscle, and internal organs. Report any diagnosis of NSF following Clariscan administration to GE HealthCare at (1-800-654-0118) or FDA at (1-800-FDA-1088 or www.fda.gov/medwatch).
Screen patients for acute kidney injury and other conditions that may reduce renal function. Features of acute kidney injury consist of rapid (over hours to days), and usually reversible, decrease in kidney function, commonly in the setting of surgery, severe infection, injury or drug-induced kidney toxicity. Serum creatinine levels and estimated GFR may not reliably assess renal function in the setting of acute kidney injury. For patients at risk for chronically reduced renal function (e.g., age > 60 years, diabetes mellitus or chronic hypertension), estimate the GFR through laboratory testing.
The factors that may increase the risk for NSF are repeated or higher than recommended doses of a GBCA, and the degree of renal impairment at the time of exposure. Record the specific GBCA and the dose administered to a patient. For patients at highest risk for NSF, do not exceed the recommended Clariscan dose and allow a sufficient period of time for elimination of the drug prior to re-administration. For patients receiving hemodialysis, physicians may consider the prompt initiation of hemodialysis following the administration of a GBCA in order to enhance the contrast agent's elimination. The usefulness of hemodialysis in the prevention of NSF is unknown [see Dosage and Administration (2) and Clinical Pharmacology (12)].
17.1 Nephrogenic Systemic Fibrosis
Instruct patients to inform their healthcare provider if they:
- have a history of kidney disease, or
- have recently received a GBCA.
GBCAs increase the risk for NSF among patients with impaired elimination of the drugs. To counsel patients at risk for NSF:
- Describe the clinical manifestations of NSF.
- Describe procedures to screen for the detection of renal impairment.
Instruct the patients to contact their physician if they develop signs or symptoms of NSF following Clariscan administration, such as burning, itching, swelling, scaling, hardening and tightening of the skin; red or dark patches on the skin; stiffness in joints with trouble moving, bending or straightening the arms, hands, legs or feet; pain in the hip bones or ribs; or muscle weakness.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Clariscan Injection is a clear, colorless to yellow solution containing 0.5 mmol/mL of gadoterate meglumine. It is supplied in vials and pre-filled syringes.
- Clariscan Injection is supplied in 10 mL vials containing 5 mL or 10 mL of solution and in 20 mL vials containing 15 mL or 20 mL of solution.
Each single-dose vial is closed with a rubber stopper and sealed with an aluminum cap and the contents are sterile. Vials are packaged in a box of 10, in the following configurations:
| 2.5 mmol per 5 mL (0.5 mmol per mL) in glass vial | (NDC 0407-2943-06) |
| 5 mmol per 10 mL (0.5 mmol per mL) in glass vial | (NDC 0407-2943-01) |
| 7.5 mmol per 15 mL (0.5 mmol per mL) in glass vial | (NDC 0407-2943-02) |
| 10 mmol per 20 mL (0.5 mmol per mL) in glass vial | (NDC 0407-2943-05) |
- Clariscan Injection is supplied in 20 mL plastic pre-filled syringes containing 10 mL, 15 mL, or 20 mL of solution.
Each syringe is sealed with rubber closures and the contents are sterile. Syringes, including plunger rod, are individually packaged in a box of 10, in the following configurations:
| 5 mmol per 10 mL (0.5 mmol per mL) in plastic pre-filled syringe | (NDC 0407-2943-12) |
| 7.5 mmol per 15 mL (0.5 mmol per mL) in plastic pre-filled syringe | (NDC 0407-2943-17) |
| 10 mmol per 20 mL (0.5 mmol per mL) in plastic pre-filled syringe | (NDC 0407-2943-22) |
5.1 Risk Associated With Intrathecal Use (5.1 Risk Associated with Intrathecal Use)
Intrathecal administration of GBCAs can cause serious adverse reactions including death, coma, encephalopathy, and seizures. The safety and effectiveness of Clariscan have not been established with intrathecal use. Clariscan is not approved for intrathecal use [see Dosage and Administration (2.1)].
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Local intolerance reactions, including moderate irritation associated with infiltration of inflammatory cells were observed after perivenous injection in rabbits suggesting the possibility of local irritation if the contrast medium leaks around the veins in a clinical setting [see Warnings and Precautions (5.5)].
Toxicity of gadoterate meglumine was evaluated in neonatal and juvenile (pre- and post-weaning) rats following a single or repeated intravenous administration at doses 1, 2, and 4 times the MHD based on BSA. Gadoterate meglumine was well tolerated at all dose levels tested and had no effect on growth, pre-weaning development, behavior and sexual maturation.
5.6 Extravasation and Injection Site Reactions
Ensure catheter and venous patency before the injection of Clariscan. Extravasation into tissues during Clariscan administration may result in tissue irritation [see Nonclinical Toxicology (13.2)].
Principal Display Panel 10 Ml Vial Box Label (PRINCIPAL DISPLAY PANEL - 10 mL Vial Box Label)
NDC 0407-2943-01
Rx Only
Clariscan™
(gadoterate meglumine) Injection
5 mmol per 10 mL
(0.5 mmol per mL)
For Intravenous Administration
Sterile Solution
Discard Unused Portion
Dispense the accompanying
Medication Guide to each patient
10 x 10 mL Single-dose Vials
Principal Display Panel 15 Ml Syringe Box Label (PRINCIPAL DISPLAY PANEL - 15 mL Syringe Box Label)
NDC 0407-2943-17
Rx Only
Clariscan™
(gadoterate meglumine) Injection
7.5 mmol per 15 mL
(0.5 mmol per mL)
Sterile Solution
For Intravenous Administration
Discard Unused Portion
Dispense the accompanying
Medication Guide to each patient
1 x 15 mL Single-Dose Plastic Syringe
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed to evaluate the carcinogenic potential of gadoterate meglumine.
Gadoterate meglumine did not demonstrate mutagenic potential in in vitro bacterial reverse mutation assays (Ames test) using Salmonella typhimurium, in an in vitro chromosome aberration assay in Chinese hamster ovary cells, in an in vitro gene mutation assay in Chinese hamster lung cells, nor in an in vivo mouse micronucleus assay.
No impairment of male or female fertility and reproductive performance was observed in rats after intravenous administration of gadoterate meglumine at the maximum tested dose of 10 mmol/kg/day (16 times the maximum human dose based on surface area), given during more than 9 weeks in males and more than 4 weeks in females. Sperm counts and sperm motility were not adversely affected by treatment with the drug.
Warning: Risk Associated With Intrathecal Use and Nephrogenic Systemic Fibrosis (WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS)
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS
See full prescribing information for complete boxed warning
- Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. Clariscan is not approved for intrathecal use. (5.1)
-
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of Clariscan in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. The risk for NSF appears highest among patients with:
- Chronic, severe kidney disease (GFR < 30 mL/min/1.73 m2), or
- Acute kidney injury.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:49.037427 · Updated: 2026-03-14T22:28:06.461439