Lioresal®
6f3bdcc0-ffc8-4911-8c35-958c8103a2c5
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Abrupt discontinuation of intrathecal baclofen, regardless of the cause, has resulted in sequelae that include high fever, altered mental status, exaggerated rebound spasticity, and muscle rigidity, that in rare cases has advanced to rhabdomyolysis, multiple organ-system failure and death. Prevention of abrupt discontinuation of intrathecal baclofen requires careful attention to programming and monitoring of the infusion system, refill scheduling and procedures, and pump alarms. Patients and caregivers should be advised of the importance of keeping scheduled refill visits and should be educated on the early symptoms of baclofen withdrawal. Special attention should be given to patients at apparent risk (e.g. spinal cord injuries at T-6 or above, communication difficulties, history of withdrawal symptoms from oral or intrathecal baclofen). Consult the technical manual of the implantable infusion system for additional postimplant clinician and patient information (see WARNINGS ).
Indications and Usage
LIORESAL INTRATHECAL (baclofen injection) is indicated for use in the management of severe spasticity. Patients should first respond to a screening dose of intrathecal baclofen prior to consideration for long term infusion via an implantable pump. For spasticity of spinal cord origin, chronic infusion of LIORESAL INTRATHECAL via an implantable pump should be reserved for patients unresponsive to oral baclofen therapy, or those who experience intolerable CNS side effects at effective doses. Patients with spasticity due to traumatic brain injury should wait at least one year after the injury before consideration of long term intrathecal baclofen therapy. LIORESAL INTRATHECAL is intended for use by the intrathecal route in single bolus test doses (via spinal catheter or lumbar puncture) and, for chronic use, only in implantable pumps approved by the FDA specifically for the administration of LIORESAL INTRATHECAL into the intrathecal space.
Dosage and Administration
Refer to the manufacturer's manual for the implantable pump approved for intrathecal infusion for specific instructions and precautions for programming the pump and/or refilling the reservoir. There are various pumps with varying reservoir volumes and there are various refill kits available. It is important to be familiar with all of these products in order to select the appropriate refill kit for the particular pump in use.
Contraindications
Hypersensitivity to baclofen. LIORESAL INTRATHECAL is not recommended for intravenous, intramuscular, subcutaneous or epidural administration.
How Supplied
LIORESAL INTRATHECAL (baclofen injection) is packaged in single use ampules containing 0.05 mg/1 mL (50 mcg/mL), 10 mg/20 mL (500 mcg/mL), 10 mg/5 mL (2000 mcg/mL), or 40 mg/20 mL (2000 mcg/mL) supplied as follows: Screening dose (Model 8563s): five ampules each containing 0.05 mg/1 mL (50 mcg/mL) (NDC 70121-2496-5). LIORESAL INTRATHECAL (baclofen injection) Refill Kits. Each refill kit includes the indicated amount of LIORESAL INTRATHECAL, a drug preparation kit, a pump refill kit with accessories that are compatible with Medtronic SynchroMed ® Infusion Systems, and associated instructions. Model 8561: one ampule containing 10 mg/20 mL (500 mcg/mL) (NDC 70121-2501-1). Model 8562: two ampules, each contains 10 mg/5 mL (2000 mcg/mL) (NDC 70121-2502-2). Model 8564: one ampule containing 40 mg/20 mL (2000 mcg/mL) (NDC 70121-2503-1). Model 8565: two ampules, each contains 10 mg/20 mL (500 mcg/mL) (NDC 70121-2504-2). Model 8566: two ampules, each contains 40 mg/20 mL (2000 mcg/mL) (NDC 70121-2505-2).
Medication Information
Recent Major Changes
Abrupt discontinuation of intrathecal baclofen, regardless of the cause, has resulted in sequelae that include high fever, altered mental status, exaggerated rebound spasticity, and muscle rigidity, that in rare cases has advanced to rhabdomyolysis, multiple organ-system failure and death.
Prevention of abrupt discontinuation of intrathecal baclofen requires careful attention to programming and monitoring of the infusion system, refill scheduling and procedures, and pump alarms. Patients and caregivers should be advised of the importance of keeping scheduled refill visits and should be educated on the early symptoms of baclofen withdrawal. Special attention should be given to patients at apparent risk (e.g. spinal cord injuries at T-6 or above, communication difficulties, history of withdrawal symptoms from oral or intrathecal baclofen). Consult the technical manual of the implantable infusion system for additional postimplant clinician and patient information (see WARNINGS).
Indications and Usage
LIORESAL INTRATHECAL (baclofen injection) is indicated for use in the management of severe spasticity. Patients should first respond to a screening dose of intrathecal baclofen prior to consideration for long term infusion via an implantable pump. For spasticity of spinal cord origin, chronic infusion of LIORESAL INTRATHECAL via an implantable pump should be reserved for patients unresponsive to oral baclofen therapy, or those who experience intolerable CNS side effects at effective doses. Patients with spasticity due to traumatic brain injury should wait at least one year after the injury before consideration of long term intrathecal baclofen therapy. LIORESAL INTRATHECAL is intended for use by the intrathecal route in single bolus test doses (via spinal catheter or lumbar puncture) and, for chronic use, only in implantable pumps approved by the FDA specifically for the administration of LIORESAL INTRATHECAL into the intrathecal space.
Dosage and Administration
Refer to the manufacturer's manual for the implantable pump approved for intrathecal infusion for specific instructions and precautions for programming the pump and/or refilling the reservoir. There are various pumps with varying reservoir volumes and there are various refill kits available. It is important to be familiar with all of these products in order to select the appropriate refill kit for the particular pump in use.
Contraindications
Hypersensitivity to baclofen. LIORESAL INTRATHECAL is not recommended for intravenous, intramuscular, subcutaneous or epidural administration.
How Supplied
LIORESAL INTRATHECAL (baclofen injection) is packaged in single use ampules containing 0.05 mg/1 mL (50 mcg/mL), 10 mg/20 mL (500 mcg/mL), 10 mg/5 mL (2000 mcg/mL), or 40 mg/20 mL (2000 mcg/mL) supplied as follows:
Screening dose (Model 8563s): five ampules each containing 0.05 mg/1 mL (50 mcg/mL)
(NDC 70121-2496-5).
LIORESAL INTRATHECAL (baclofen injection) Refill Kits. Each refill kit includes the indicated amount of LIORESAL INTRATHECAL, a drug preparation kit, a pump refill kit with accessories that are compatible with Medtronic SynchroMed® Infusion Systems, and associated instructions.
Model 8561: one ampule containing 10 mg/20 mL (500 mcg/mL)
(NDC 70121-2501-1).
Model 8562: two ampules, each contains 10 mg/5 mL (2000 mcg/mL)
(NDC 70121-2502-2).
Model 8564: one ampule containing 40 mg/20 mL (2000 mcg/mL)
(NDC 70121-2503-1).
Model 8565: two ampules, each contains 10 mg/20 mL (500 mcg/mL)
(NDC 70121-2504-2).
Model 8566: two ampules, each contains 40 mg/20 mL (2000 mcg/mL)
(NDC 70121-2505-2).
Description
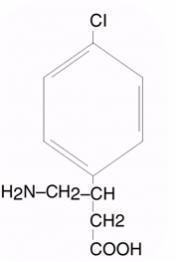
LIORESAL INTRATHECAL (baclofen injection) is a muscle relaxant and antispastic. Its chemical name is 4-amino-3-(4-chlorophenyl) butanoic acid, and its structural formula is:
Baclofen is a white to off-white, odorless or practically odorless crystalline powder, with a molecular weight of 213.66. It is slightly soluble in water, very slightly soluble in methanol, and insoluble in chloroform.
LIORESAL INTRATHECAL is a sterile, pyrogen-free, isotonic solution free of antioxidants, preservatives or other potentially neurotoxic additives indicated only for intrathecal administration. The drug is stable in solution at 37° C and compatible with CSF. Each milliliter of LIORESAL INTRATHECAL contains baclofen U. S. P. 50 mcg, 500 mcg or 2000 mcg and sodium chloride 9 mg in Water for Injection; pH range is 5.0 - 7.0. Each ampule is intended for SINGLE USE ONLY. Discard any unused portion. DO NOT AUTOCLAVE.
Section 34073-7
DRUG INTERACTIONS
There is inadequate systematic experience with the use of LIORESAL INTRATHECAL in combination with other medications to predict specific drug-drug interactions. Interactions attributed to the combined use of LIORESAL INTRATHECAL and epidural morphine include hypotension and dyspnea.
Section 34075-2
LABORATORY TESTS
No specific laboratory tests are deemed essential for the management of patients on LIORESAL INTRATHECAL.
Section 34080-2
NURSING MOTHERS
In mothers treated with oral LIORESAL (baclofen USP) in therapeutic doses, the active substance passes into the milk. It is not known whether detectable levels of drug are present in milk of nursing mothers receiving LIORESAL INTRATHECAL. As a general rule, nursing should be undertaken while a patient is receiving LIORESAL INTRATHECAL only if the potential benefit justifies the potential risks to the infant.
Section 34081-0
PEDIATRIC USE
Children should be of sufficient body mass to accommodate the implantable pump for chronic infusion. Please consult pump manufacturer‘s manual for specific recommendations.
Safety and effectiveness in pediatric patients below the age of 4 have not been established.
Section 34083-6
CARCINOGENESIS, MUTAGENESIS, AND IMPAIRMENT OF FERTILITY
No increase in tumors was seen in rats receiving baclofen orally for two years. Adequate genotoxicity assays of baclofen have not been performed.
Section 42228-7
PREGNANCY
There are no adequate and well-controlled studies in pregnant women. In animal studies, baclofen had adverse effects on embryofetal development when administered orally to pregnant rats. LIORESAL INTRATHECAL should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Baclofen given orally increased the incidence of fetal structural abnormalities (omphaloceles) in rats. Reductions in food intake and body weight gain were observed in the dams. Fetal structural abnormalities were not observed in mice or rabbits
Section 42229-5
Intrathecal Bolus:
Section 43681-6
Pharmacodynamics of LIORESAL INTRATHECAL:
Section 51945-4
Section 59845-8
Drug Preparation Kit
For use with Medtronic ® SynchroMed Implantable Programmable Infusion Pumps
Instructions for Use
The Lioresal® Intrathecal (baclofen injection) Drug Preparation Kit is used for kit models 8561, 8562, 8564, 8565 and 8566 to draw the drug from
the ampule(s). The appropriate ampule(s) is/are placed within the corresponding kit.
In addition, each kit contains:
-Syringe cap(s)
-Filter Straw® tube(s)
-Ampule opener(s)
-Syringe label(s)
-Plastic bag
-Four gauze pads
-20 mL syringe
-Lioresal® Intrathecal (baclofen injection)
prescribing information
-Drug Preparation Kit Instructions for Use
INDICATIONS
The Lioresal® Intrathecal (baclofen injection) Drug Preparation Kit is intended for use with Medtronic SynchroMed implantable programmable infusion pumps. It is not for use with the Medtronic MiniMed® infusion pumps.
Refer to the Lioresal® lntrathecal (baclofen injection) drug Prescribing Information for indications, contraindications, warnings, precautions, dosage and administration information, and screening procedures.
CONTRAINDICATIONS
Refer to the contraindications listed in the appropriate pump technical manual, 856X refill kit instructions for use, and drug labeling.
PRECAUTIONS
If dilution of the drug is required, refer to the drug labeling for proper diluent and procedures.
For emergency procedures, refer to the Refill Kit 856X Instructions For Use that are packaged with the refill kit.
STERILIZATION
All components of the Drug Preparation Kit are sterile. Do not resterilize. Should sterility be in question, discard and use a new kit. The outside of the ampule is sterile. If sterile technique is broken,
the contents of the ampule are still sterile.
INSTRUCTIONS FOR USE
Use sterile technique throughout the procedure to prevent contamination of the components and drug.
Note: The drug ampule is not sterile.
1. Remove the drug ampule from the box. Confirm drug ampule concentration prior to drug administration.
2. Open the ampule as follows:
Note: The entire neck of the ampule is scored. An ampule opener has been provided to ease the opening of the ampule; always exercise caution when opening a glass ampule
Note: If the ampule opener is not used, use gauze pads to hold the top of the ampule when opening.
Figure 1.
a. Hold the ampule in one hand and place opener (fins first) over the neck until it rests on the base portion of the ampule. Grasp opener with forefinger and thumb while holding the ampule base. Hold ampule upright and snap. Dispose of opener and ampule top to prevent potential contamination (Figure 1).
3. Assemble the syringe and Filter Straw® tube (Figure 2), making sure the syringe connection is snug to avoid leakage.
Figure 2. Assemble syringe and Filter Straw® tube
4.Withdraw the drug from the ampule(s) (Figure 3).
Figure 3. Withdraw drug from ampule
5.Remove the Filter Straw® tube from the syringe. Appropriately discard Filter Straw® tube.
6.Purge the air from the syringe.
7. If drug syringe is to be transferred prior to procedure a syringe cap has been provided. Attach the syringe cap, making sure the cap is snug to avoid leakage.
8. For convenience, a syringe label has been provided to capture patient's name, drug name, drug concentration, and date.
9. Peel the backing from the label and attach the label to the syringe, making sure not to cover the volume indicators on the syringe (Figure 4).
Figure 4. Syringe with cap and label
10. If the drug needs to be transported, place the syringe(s) in the plastic bag.
11. If applicable, repeat steps 1 – 10 with the second syringe, Filter Straw® tube, syringe cap, and syringe label supplied in the Model 8562, 8565 and 8566 kits.
12. Refer to the 856X refill kit Instructions for Use for pump refill instructions.
Note: Purge air from syringe prior to drug administration.
Rx Only
Medtronic® and MiniMed® are trademarks of Medtronic® and are registered in the U.S. and possibly other countries.
Lioresal® is a registered trademark of Amneal
Filter Straw® is a registered trademark of B.Braun Medical Inc.
Amneal Pharmaceuticals LLC
Bridgewater, NJ 08807
Rev. 03-2023-00
Storage
Does not require refrigeration. Do not store above 86° F (30° C). Do not freeze. Do not heat sterilize.
Warnings
LIORESAL INTRATHECAL is for use in single bolus intrathecal injections (via a catheter placed in the lumbar intrathecal space or injection by lumbar puncture) and in implantable pumps approved by the FDA specifically for the intrathecal administration of baclofen. Because of the possibility of potentially life-threatening CNS depression, cardiovascular collapse, and/or respiratory failure, physicians must be adequately trained and educated in chronic intrathecal infusion therapy. The pump system should not be implanted until the patient’s response to bolus LIORESAL INTRATHECAL injection is adequately evaluated. Evaluation (consisting of a screening procedure: see Dosage and Administration) requires that LIORESAL INTRATHECAL be administered into the intrathecal space via a catheter or lumbar puncture. Because of the risks associated with the screening procedure and the adjustment of dosage following pump implantation, these phases must be conducted in a medically supervised and adequately equipped environment following the instructions outlined in the Dosage and Administration section. Resuscitative equipment should be available.
Following surgical implantation of the pump, particularly during the initial phases of pump use, the patient should be monitored closely until it is certain that the patient’s response to the infusion is acceptable and reasonably stable. On each occasion that the dosing rate of the pump and/or the concentration of LIORESAL INTRATHECAL (baclofen injection) in the reservoir is adjusted, close medical monitoring is required until it is certain that the patient’s response to the infusion is acceptable and reasonably stable.
It is mandatory that the patient, all patient caregivers, and the physicians responsible for the patient receive adequate information regarding the risks of this mode of treatment. All medical personnel and caregivers should be instructed in 1) the signs and symptoms of overdose, 2) procedures to be followed in the event of overdose and 3) proper home care of the pump and insertion site.
Screening
Patients should be infection-free prior to the screening trial with LIORESAL INTRATHECAL (baclofen injection) because the presence of a systemic infection may interfere with an assessment of the patient’s response to bolus LIORESAL INTRATHECAL.
Stability
Parenteral drug products should be inspected for particulate matter and discoloration prior to administration, whenever solution and container permit.
Overdosage
Special attention must be given to recognizing the signs and symptoms of overdosage, especially during the initial screening and dose-titration phase of treatment, but also during reintroduction of LIORESAL INTRATHECAL after a period of interruption in therapy.
Precautions
Children should be of sufficient body mass to accommodate the implantable pump for chronic infusion. Please consult pump manufacturer‘s manual for specific recommendations.
Safety and effectiveness in pediatric patients below the age of 4 have not been established.
Delivery Regimen:
LIORESAL INTRATHECAL is most often administered in a continuous infusion mode immediately following implant. For those patients implanted with programmable pumps who have achieved relatively satisfactory control on continuous infusion, further benefit may be attained using more complex schedules of LIORESAL INTRATHECAL delivery. For example, patients who have increased spasms at night may require a 20% increase in their hourly infusion rate. Changes in flow rate should be programmed to start two hours before the time of desired clinical effect.
Pump Implantation
Patients should be infection-free prior to pump implantation because the presence of infection may increase the risk of surgical complications. Moreover, a systemic infection may complicate dosing.
Continuous Infusion:
LIORESAL INTRATHECAL'S antispastic action is first seen at 6 to 8 hours after initiation of continuous infusion. Maximum activity is observed in 24 to 48 hours.
Clinical Pharmacology
The precise mechanism of action of baclofen as a muscle relaxant and antispasticity agent is not fully understood. Baclofen inhibits both monosynaptic and polysynaptic reflexes at the spinal level, possibly by decreasing excitatory neurotransmitter release from primary afferent terminals, although actions at supraspinal sites may also occur and contribute to its clinical effect. Baclofen is a structural analog of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA), and may exert its effects by stimulation of the GABAB receptor subtype.
LIORESAL INTRATHECAL when introduced directly into the intrathecal space permits effective CSF concentrations to be achieved with resultant plasma concentrations 100 times less than those occurring with oral administration. In people, as well as in animals, baclofen has been shown to have general CNS depressant properties as indicated by the production of sedation with tolerance, somnolence, ataxia, and respiratory and cardiovascular depression.
Delivery Specifications
The specific concentration that should be used depends upon the total daily dose required as well as the delivery rate of the pump. LIORESAL INTRATHECAL may require dilution when used with certain implantable pumps. Please consult manufacturer’s manual for specific recommendations.
Pump Dose Adjustment and Titration
In most patients, it will be necessary to increase the dose gradually over time to maintain effectiveness; a sudden requirement for substantial dose escalation typically indicates a catheter complication (i. e., catheter kink or dislodgement). Reservoir refilling must be performed by fully trained and qualified personnel following the directions provided by the pump manufacturer. Inadvertent injection into the subcutaneous tissue can occur if the reservoir refill septum is not properly accessed. Subcutaneous injection may result in symptoms of a systemic overdose or early depletion of the reservoir. Refill intervals should be carefully calculated to prevent depletion of the reservoir, as this would result in the return of severe spasticity and possibly symptoms of withdrawal.
Strict aseptic technique in filling is required to avoid bacterial contamination and serious infection. A period of observation appropriate to the clinical situation should follow each refill or manipulation of the drug reservoir.
Extreme caution must be used when filling an FDA approved implantable pump equipped with an injection port that allows direct access to the intrathecal catheter. Direct injection into the catheter through the catheter access port may cause a life-threatening overdose.
Treatment Suggestions for Overdose:
There is no specific antidote for treating overdoses of LIORESAL INTRATHECAL (baclofen injection); however, the following steps should ordinarily be undertaken:
1) Residual LIORESAL INTRATHECAL solution should be removed from the pump as soon as possible.
2) Patients with respiratory depression should be intubated if necessary, until the drug is eliminated.
If lumbar puncture is not contraindicated, consideration should be given to withdrawing 30-40 mL of CSF to reduce CSF baclofen concentration.
Pharmacokinetics of Lioresal Intrathecal:
The pharmacokinetics of CSF clearance of LIORESAL INTRATHECAL calculated from intrathecal bolus or continuous infusion studies approximates CSF turnover, suggesting elimination is by bulk-flow removal of CSF.
Considerations Based On Experience With Oral Lioresal (baclofen Usp)
A dose-related increase in incidence of ovarian cysts was observed in female rats treated chronically with oral LIORESAL. Ovarian cysts have been found by palpation in about 4% of the multiple sclerosis patients who were treated with oral LIORESAL for up to one year. In most cases these cysts disappeared spontaneously while patients continued to receive the drug. Ovarian cysts are estimated to occur spontaneously in approximately 1% to 5% of the normal female population.
Structured Label Content
Recent Major Changes (34066-1)
Abrupt discontinuation of intrathecal baclofen, regardless of the cause, has resulted in sequelae that include high fever, altered mental status, exaggerated rebound spasticity, and muscle rigidity, that in rare cases has advanced to rhabdomyolysis, multiple organ-system failure and death.
Prevention of abrupt discontinuation of intrathecal baclofen requires careful attention to programming and monitoring of the infusion system, refill scheduling and procedures, and pump alarms. Patients and caregivers should be advised of the importance of keeping scheduled refill visits and should be educated on the early symptoms of baclofen withdrawal. Special attention should be given to patients at apparent risk (e.g. spinal cord injuries at T-6 or above, communication difficulties, history of withdrawal symptoms from oral or intrathecal baclofen). Consult the technical manual of the implantable infusion system for additional postimplant clinician and patient information (see WARNINGS).
Section 34073-7 (34073-7)
DRUG INTERACTIONS
There is inadequate systematic experience with the use of LIORESAL INTRATHECAL in combination with other medications to predict specific drug-drug interactions. Interactions attributed to the combined use of LIORESAL INTRATHECAL and epidural morphine include hypotension and dyspnea.
Section 34075-2 (34075-2)
LABORATORY TESTS
No specific laboratory tests are deemed essential for the management of patients on LIORESAL INTRATHECAL.
Section 34080-2 (34080-2)
NURSING MOTHERS
In mothers treated with oral LIORESAL (baclofen USP) in therapeutic doses, the active substance passes into the milk. It is not known whether detectable levels of drug are present in milk of nursing mothers receiving LIORESAL INTRATHECAL. As a general rule, nursing should be undertaken while a patient is receiving LIORESAL INTRATHECAL only if the potential benefit justifies the potential risks to the infant.
Section 34081-0 (34081-0)
PEDIATRIC USE
Children should be of sufficient body mass to accommodate the implantable pump for chronic infusion. Please consult pump manufacturer‘s manual for specific recommendations.
Safety and effectiveness in pediatric patients below the age of 4 have not been established.
Section 34083-6 (34083-6)
CARCINOGENESIS, MUTAGENESIS, AND IMPAIRMENT OF FERTILITY
No increase in tumors was seen in rats receiving baclofen orally for two years. Adequate genotoxicity assays of baclofen have not been performed.
Section 42228-7 (42228-7)
PREGNANCY
There are no adequate and well-controlled studies in pregnant women. In animal studies, baclofen had adverse effects on embryofetal development when administered orally to pregnant rats. LIORESAL INTRATHECAL should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Baclofen given orally increased the incidence of fetal structural abnormalities (omphaloceles) in rats. Reductions in food intake and body weight gain were observed in the dams. Fetal structural abnormalities were not observed in mice or rabbits
Section 42229-5 (42229-5)
Intrathecal Bolus:
Section 43681-6 (43681-6)
Pharmacodynamics of LIORESAL INTRATHECAL:
Section 51945-4 (51945-4)
Section 59845-8 (59845-8)
Drug Preparation Kit
For use with Medtronic ® SynchroMed Implantable Programmable Infusion Pumps
Instructions for Use
The Lioresal® Intrathecal (baclofen injection) Drug Preparation Kit is used for kit models 8561, 8562, 8564, 8565 and 8566 to draw the drug from
the ampule(s). The appropriate ampule(s) is/are placed within the corresponding kit.
In addition, each kit contains:
-Syringe cap(s)
-Filter Straw® tube(s)
-Ampule opener(s)
-Syringe label(s)
-Plastic bag
-Four gauze pads
-20 mL syringe
-Lioresal® Intrathecal (baclofen injection)
prescribing information
-Drug Preparation Kit Instructions for Use
INDICATIONS
The Lioresal® Intrathecal (baclofen injection) Drug Preparation Kit is intended for use with Medtronic SynchroMed implantable programmable infusion pumps. It is not for use with the Medtronic MiniMed® infusion pumps.
Refer to the Lioresal® lntrathecal (baclofen injection) drug Prescribing Information for indications, contraindications, warnings, precautions, dosage and administration information, and screening procedures.
CONTRAINDICATIONS
Refer to the contraindications listed in the appropriate pump technical manual, 856X refill kit instructions for use, and drug labeling.
PRECAUTIONS
If dilution of the drug is required, refer to the drug labeling for proper diluent and procedures.
For emergency procedures, refer to the Refill Kit 856X Instructions For Use that are packaged with the refill kit.
STERILIZATION
All components of the Drug Preparation Kit are sterile. Do not resterilize. Should sterility be in question, discard and use a new kit. The outside of the ampule is sterile. If sterile technique is broken,
the contents of the ampule are still sterile.
INSTRUCTIONS FOR USE
Use sterile technique throughout the procedure to prevent contamination of the components and drug.
Note: The drug ampule is not sterile.
1. Remove the drug ampule from the box. Confirm drug ampule concentration prior to drug administration.
2. Open the ampule as follows:
Note: The entire neck of the ampule is scored. An ampule opener has been provided to ease the opening of the ampule; always exercise caution when opening a glass ampule
Note: If the ampule opener is not used, use gauze pads to hold the top of the ampule when opening.
Figure 1.
a. Hold the ampule in one hand and place opener (fins first) over the neck until it rests on the base portion of the ampule. Grasp opener with forefinger and thumb while holding the ampule base. Hold ampule upright and snap. Dispose of opener and ampule top to prevent potential contamination (Figure 1).
3. Assemble the syringe and Filter Straw® tube (Figure 2), making sure the syringe connection is snug to avoid leakage.
Figure 2. Assemble syringe and Filter Straw® tube
4.Withdraw the drug from the ampule(s) (Figure 3).
Figure 3. Withdraw drug from ampule
5.Remove the Filter Straw® tube from the syringe. Appropriately discard Filter Straw® tube.
6.Purge the air from the syringe.
7. If drug syringe is to be transferred prior to procedure a syringe cap has been provided. Attach the syringe cap, making sure the cap is snug to avoid leakage.
8. For convenience, a syringe label has been provided to capture patient's name, drug name, drug concentration, and date.
9. Peel the backing from the label and attach the label to the syringe, making sure not to cover the volume indicators on the syringe (Figure 4).
Figure 4. Syringe with cap and label
10. If the drug needs to be transported, place the syringe(s) in the plastic bag.
11. If applicable, repeat steps 1 – 10 with the second syringe, Filter Straw® tube, syringe cap, and syringe label supplied in the Model 8562, 8565 and 8566 kits.
12. Refer to the 856X refill kit Instructions for Use for pump refill instructions.
Note: Purge air from syringe prior to drug administration.
Rx Only
Medtronic® and MiniMed® are trademarks of Medtronic® and are registered in the U.S. and possibly other countries.
Lioresal® is a registered trademark of Amneal
Filter Straw® is a registered trademark of B.Braun Medical Inc.
Amneal Pharmaceuticals LLC
Bridgewater, NJ 08807
Rev. 03-2023-00
Storage
Does not require refrigeration. Do not store above 86° F (30° C). Do not freeze. Do not heat sterilize.
Warnings (WARNINGS)
LIORESAL INTRATHECAL is for use in single bolus intrathecal injections (via a catheter placed in the lumbar intrathecal space or injection by lumbar puncture) and in implantable pumps approved by the FDA specifically for the intrathecal administration of baclofen. Because of the possibility of potentially life-threatening CNS depression, cardiovascular collapse, and/or respiratory failure, physicians must be adequately trained and educated in chronic intrathecal infusion therapy. The pump system should not be implanted until the patient’s response to bolus LIORESAL INTRATHECAL injection is adequately evaluated. Evaluation (consisting of a screening procedure: see Dosage and Administration) requires that LIORESAL INTRATHECAL be administered into the intrathecal space via a catheter or lumbar puncture. Because of the risks associated with the screening procedure and the adjustment of dosage following pump implantation, these phases must be conducted in a medically supervised and adequately equipped environment following the instructions outlined in the Dosage and Administration section. Resuscitative equipment should be available.
Following surgical implantation of the pump, particularly during the initial phases of pump use, the patient should be monitored closely until it is certain that the patient’s response to the infusion is acceptable and reasonably stable. On each occasion that the dosing rate of the pump and/or the concentration of LIORESAL INTRATHECAL (baclofen injection) in the reservoir is adjusted, close medical monitoring is required until it is certain that the patient’s response to the infusion is acceptable and reasonably stable.
It is mandatory that the patient, all patient caregivers, and the physicians responsible for the patient receive adequate information regarding the risks of this mode of treatment. All medical personnel and caregivers should be instructed in 1) the signs and symptoms of overdose, 2) procedures to be followed in the event of overdose and 3) proper home care of the pump and insertion site.
Screening
Patients should be infection-free prior to the screening trial with LIORESAL INTRATHECAL (baclofen injection) because the presence of a systemic infection may interfere with an assessment of the patient’s response to bolus LIORESAL INTRATHECAL.
Stability
Parenteral drug products should be inspected for particulate matter and discoloration prior to administration, whenever solution and container permit.
Overdosage (OVERDOSAGE)
Special attention must be given to recognizing the signs and symptoms of overdosage, especially during the initial screening and dose-titration phase of treatment, but also during reintroduction of LIORESAL INTRATHECAL after a period of interruption in therapy.
Description (DESCRIPTION)
LIORESAL INTRATHECAL (baclofen injection) is a muscle relaxant and antispastic. Its chemical name is 4-amino-3-(4-chlorophenyl) butanoic acid, and its structural formula is:
Baclofen is a white to off-white, odorless or practically odorless crystalline powder, with a molecular weight of 213.66. It is slightly soluble in water, very slightly soluble in methanol, and insoluble in chloroform.
LIORESAL INTRATHECAL is a sterile, pyrogen-free, isotonic solution free of antioxidants, preservatives or other potentially neurotoxic additives indicated only for intrathecal administration. The drug is stable in solution at 37° C and compatible with CSF. Each milliliter of LIORESAL INTRATHECAL contains baclofen U. S. P. 50 mcg, 500 mcg or 2000 mcg and sodium chloride 9 mg in Water for Injection; pH range is 5.0 - 7.0. Each ampule is intended for SINGLE USE ONLY. Discard any unused portion. DO NOT AUTOCLAVE.
Precautions (PRECAUTIONS)
Children should be of sufficient body mass to accommodate the implantable pump for chronic infusion. Please consult pump manufacturer‘s manual for specific recommendations.
Safety and effectiveness in pediatric patients below the age of 4 have not been established.
How Supplied (HOW SUPPLIED)
LIORESAL INTRATHECAL (baclofen injection) is packaged in single use ampules containing 0.05 mg/1 mL (50 mcg/mL), 10 mg/20 mL (500 mcg/mL), 10 mg/5 mL (2000 mcg/mL), or 40 mg/20 mL (2000 mcg/mL) supplied as follows:
Screening dose (Model 8563s): five ampules each containing 0.05 mg/1 mL (50 mcg/mL)
(NDC 70121-2496-5).
LIORESAL INTRATHECAL (baclofen injection) Refill Kits. Each refill kit includes the indicated amount of LIORESAL INTRATHECAL, a drug preparation kit, a pump refill kit with accessories that are compatible with Medtronic SynchroMed® Infusion Systems, and associated instructions.
Model 8561: one ampule containing 10 mg/20 mL (500 mcg/mL)
(NDC 70121-2501-1).
Model 8562: two ampules, each contains 10 mg/5 mL (2000 mcg/mL)
(NDC 70121-2502-2).
Model 8564: one ampule containing 40 mg/20 mL (2000 mcg/mL)
(NDC 70121-2503-1).
Model 8565: two ampules, each contains 10 mg/20 mL (500 mcg/mL)
(NDC 70121-2504-2).
Model 8566: two ampules, each contains 40 mg/20 mL (2000 mcg/mL)
(NDC 70121-2505-2).
Contraindications (CONTRAINDICATIONS)
Hypersensitivity to baclofen. LIORESAL INTRATHECAL is not recommended for intravenous, intramuscular, subcutaneous or epidural administration.
Delivery Regimen:
LIORESAL INTRATHECAL is most often administered in a continuous infusion mode immediately following implant. For those patients implanted with programmable pumps who have achieved relatively satisfactory control on continuous infusion, further benefit may be attained using more complex schedules of LIORESAL INTRATHECAL delivery. For example, patients who have increased spasms at night may require a 20% increase in their hourly infusion rate. Changes in flow rate should be programmed to start two hours before the time of desired clinical effect.
Pump Implantation
Patients should be infection-free prior to pump implantation because the presence of infection may increase the risk of surgical complications. Moreover, a systemic infection may complicate dosing.
Continuous Infusion:
LIORESAL INTRATHECAL'S antispastic action is first seen at 6 to 8 hours after initiation of continuous infusion. Maximum activity is observed in 24 to 48 hours.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
The precise mechanism of action of baclofen as a muscle relaxant and antispasticity agent is not fully understood. Baclofen inhibits both monosynaptic and polysynaptic reflexes at the spinal level, possibly by decreasing excitatory neurotransmitter release from primary afferent terminals, although actions at supraspinal sites may also occur and contribute to its clinical effect. Baclofen is a structural analog of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA), and may exert its effects by stimulation of the GABAB receptor subtype.
LIORESAL INTRATHECAL when introduced directly into the intrathecal space permits effective CSF concentrations to be achieved with resultant plasma concentrations 100 times less than those occurring with oral administration. In people, as well as in animals, baclofen has been shown to have general CNS depressant properties as indicated by the production of sedation with tolerance, somnolence, ataxia, and respiratory and cardiovascular depression.
Indications and Usage (INDICATIONS AND USAGE)
LIORESAL INTRATHECAL (baclofen injection) is indicated for use in the management of severe spasticity. Patients should first respond to a screening dose of intrathecal baclofen prior to consideration for long term infusion via an implantable pump. For spasticity of spinal cord origin, chronic infusion of LIORESAL INTRATHECAL via an implantable pump should be reserved for patients unresponsive to oral baclofen therapy, or those who experience intolerable CNS side effects at effective doses. Patients with spasticity due to traumatic brain injury should wait at least one year after the injury before consideration of long term intrathecal baclofen therapy. LIORESAL INTRATHECAL is intended for use by the intrathecal route in single bolus test doses (via spinal catheter or lumbar puncture) and, for chronic use, only in implantable pumps approved by the FDA specifically for the administration of LIORESAL INTRATHECAL into the intrathecal space.
Delivery Specifications
The specific concentration that should be used depends upon the total daily dose required as well as the delivery rate of the pump. LIORESAL INTRATHECAL may require dilution when used with certain implantable pumps. Please consult manufacturer’s manual for specific recommendations.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Refer to the manufacturer's manual for the implantable pump approved for intrathecal infusion for specific instructions and precautions for programming the pump and/or refilling the reservoir. There are various pumps with varying reservoir volumes and there are various refill kits available. It is important to be familiar with all of these products in order to select the appropriate refill kit for the particular pump in use.
Pump Dose Adjustment and Titration
In most patients, it will be necessary to increase the dose gradually over time to maintain effectiveness; a sudden requirement for substantial dose escalation typically indicates a catheter complication (i. e., catheter kink or dislodgement). Reservoir refilling must be performed by fully trained and qualified personnel following the directions provided by the pump manufacturer. Inadvertent injection into the subcutaneous tissue can occur if the reservoir refill septum is not properly accessed. Subcutaneous injection may result in symptoms of a systemic overdose or early depletion of the reservoir. Refill intervals should be carefully calculated to prevent depletion of the reservoir, as this would result in the return of severe spasticity and possibly symptoms of withdrawal.
Strict aseptic technique in filling is required to avoid bacterial contamination and serious infection. A period of observation appropriate to the clinical situation should follow each refill or manipulation of the drug reservoir.
Extreme caution must be used when filling an FDA approved implantable pump equipped with an injection port that allows direct access to the intrathecal catheter. Direct injection into the catheter through the catheter access port may cause a life-threatening overdose.
Treatment Suggestions for Overdose:
There is no specific antidote for treating overdoses of LIORESAL INTRATHECAL (baclofen injection); however, the following steps should ordinarily be undertaken:
1) Residual LIORESAL INTRATHECAL solution should be removed from the pump as soon as possible.
2) Patients with respiratory depression should be intubated if necessary, until the drug is eliminated.
If lumbar puncture is not contraindicated, consideration should be given to withdrawing 30-40 mL of CSF to reduce CSF baclofen concentration.
Pharmacokinetics of Lioresal Intrathecal: (Pharmacokinetics of LIORESAL INTRATHECAL:)
The pharmacokinetics of CSF clearance of LIORESAL INTRATHECAL calculated from intrathecal bolus or continuous infusion studies approximates CSF turnover, suggesting elimination is by bulk-flow removal of CSF.
Considerations Based On Experience With Oral Lioresal (baclofen Usp) (Considerations based on experience with oral LIORESAL (baclofen USP))
A dose-related increase in incidence of ovarian cysts was observed in female rats treated chronically with oral LIORESAL. Ovarian cysts have been found by palpation in about 4% of the multiple sclerosis patients who were treated with oral LIORESAL for up to one year. In most cases these cysts disappeared spontaneously while patients continued to receive the drug. Ovarian cysts are estimated to occur spontaneously in approximately 1% to 5% of the normal female population.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:06.741933 · Updated: 2026-03-14T22:36:30.622003