These Highlights Do Not Include All The Information Needed To Use Entyvio®
6e94621c-1a95-4af9-98d1-52b9e6f1949c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration Important Administration Information ( 2.1 ) 2/2026 Warnings and Precautions Infections ( 5.2 ) 2/2026
Indications and Usage
ENTYVIO is indicated in adults for the treatment of: moderately to severely active ulcerative colitis (UC). moderately to severely active Crohn's disease (CD).
Dosage and Administration
Important Administration Information Before Initiating ENTYVIO Consider evaluating patients for tuberculosis (TB) infection. ( 2.1 , 5.2 ) Update immunizations according to current immunization guidelines. ( 2.1 , 5.5 ) Intravenous Administration : ENTYVIO should be administered intravenously by a healthcare provider. ( 2.1 ) Subcutaneous Injection : ENTYVIO prefilled syringe and ENTYVIO PEN are intended for subcutaneous use. A patient may self-inject or caregiver may inject after proper training on correct subcutaneous injection technique. ( 2.1 ) Recommended Dosage ( 2.2 ) Week 0 : 300 mg infused intravenously over approximately 30 minutes. Week 2 : 300 mg infused intravenously over approximately 30 minutes. Week 6 : Patients may remain on ENTYVIO intravenous therapy or switch to subcutaneous injection after receiving two ENTYVIO intravenous doses administered at Week 0 and Week 2. Intravenous Infusion : 300 mg infused over approximately 30 minutes and then every eight weeks thereafter. Subcutaneous Injection : 108 mg subcutaneously once every two weeks. Discontinue ENTYVIO in patients who do not show evidence of therapeutic benefit by Week 14. Patients currently receiving and responding to ENTYVIO intravenous therapy after Week 6 may also be switched to subcutaneous injection. Administer the first subcutaneous dose in place of the next scheduled intravenous infusion and every two weeks thereafter. Preparation and Administration Instructions: See full prescribing information for complete information on reconstitution, dilution, administration, and storage. ( 2.3 , 2.4 )
Warnings and Precautions
Infusion-Related Reactions and Hypersensitivity Reactions : Discontinue ENTYVIO and initiate appropriate treatment if serious reactions occur. ( 5.1 ) Infections : Treatment with ENTYVIO should not be initiated in patients with a clinically important active infection until the infection resolves or is adequately treated. If a serious infection develops, ENTYVIO should not be administered until the infection resolves. ( 5.2 ) Progressive Multifocal Leukoencephalopathy (PML) : Although unlikely, a risk of PML cannot be ruled out. Monitor patients for any new or worsening neurological signs or symptoms. ( 5.3 )
Contraindications
ENTYVIO is contraindicated in patients who have had a known serious or severe hypersensitivity reaction to ENTYVIO or any of its excipients (such as dyspnea, bronchospasm, urticaria, flushing, rash and increased heart rate) [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following topics are also discussed in detail in the Warnings and Precautions section: Infusion-Related Reactions and Hypersensitivity Reactions [see Warnings and Precautions (5.1) ] Infections [see Warnings and Precautions (5.2) ] Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.3) ] Liver Injury [see Warnings and Precautions (5.4) ]
Medication Information
Warnings and Precautions
Infusion-Related Reactions and Hypersensitivity Reactions : Discontinue ENTYVIO and initiate appropriate treatment if serious reactions occur. ( 5.1 ) Infections : Treatment with ENTYVIO should not be initiated in patients with a clinically important active infection until the infection resolves or is adequately treated. If a serious infection develops, ENTYVIO should not be administered until the infection resolves. ( 5.2 ) Progressive Multifocal Leukoencephalopathy (PML) : Although unlikely, a risk of PML cannot be ruled out. Monitor patients for any new or worsening neurological signs or symptoms. ( 5.3 )
Indications and Usage
ENTYVIO is indicated in adults for the treatment of: moderately to severely active ulcerative colitis (UC). moderately to severely active Crohn's disease (CD).
Dosage and Administration
Important Administration Information Before Initiating ENTYVIO Consider evaluating patients for tuberculosis (TB) infection. ( 2.1 , 5.2 ) Update immunizations according to current immunization guidelines. ( 2.1 , 5.5 ) Intravenous Administration : ENTYVIO should be administered intravenously by a healthcare provider. ( 2.1 ) Subcutaneous Injection : ENTYVIO prefilled syringe and ENTYVIO PEN are intended for subcutaneous use. A patient may self-inject or caregiver may inject after proper training on correct subcutaneous injection technique. ( 2.1 ) Recommended Dosage ( 2.2 ) Week 0 : 300 mg infused intravenously over approximately 30 minutes. Week 2 : 300 mg infused intravenously over approximately 30 minutes. Week 6 : Patients may remain on ENTYVIO intravenous therapy or switch to subcutaneous injection after receiving two ENTYVIO intravenous doses administered at Week 0 and Week 2. Intravenous Infusion : 300 mg infused over approximately 30 minutes and then every eight weeks thereafter. Subcutaneous Injection : 108 mg subcutaneously once every two weeks. Discontinue ENTYVIO in patients who do not show evidence of therapeutic benefit by Week 14. Patients currently receiving and responding to ENTYVIO intravenous therapy after Week 6 may also be switched to subcutaneous injection. Administer the first subcutaneous dose in place of the next scheduled intravenous infusion and every two weeks thereafter. Preparation and Administration Instructions: See full prescribing information for complete information on reconstitution, dilution, administration, and storage. ( 2.3 , 2.4 )
Contraindications
ENTYVIO is contraindicated in patients who have had a known serious or severe hypersensitivity reaction to ENTYVIO or any of its excipients (such as dyspnea, bronchospasm, urticaria, flushing, rash and increased heart rate) [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following topics are also discussed in detail in the Warnings and Precautions section: Infusion-Related Reactions and Hypersensitivity Reactions [see Warnings and Precautions (5.1) ] Infections [see Warnings and Precautions (5.2) ] Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.3) ] Liver Injury [see Warnings and Precautions (5.4) ]
Description
Dosage and Administration Important Administration Information ( 2.1 ) 2/2026 Warnings and Precautions Infections ( 5.2 ) 2/2026
Section 42229-5
Intravenous Administration
- ENTYVIO should be administered by a healthcare provider prepared to manage hypersensitivity reactions including anaphylaxis, if they occur [see Warnings and Precautions (5.1)]. Appropriate monitoring and medical support measures should be available for immediate use. Observe patients during infusion and until the infusion is complete.
- Reconstitute and dilute ENTYVIO lyophilized powder prior to administration as a 30-minute intravenous infusion [see Dosage and Administration (2.3)].
Section 42231-1
| MEDICATION GUIDE | ||
|---|---|---|
| ENTYVIO® (en ti' vee oh) (vedolizumab) for injection, for intravenous use |
ENTYVIO® (en ti' vee oh) (vedolizumab) injection, for subcutaneous use |
ENTYVIO® (en ti' vee oh) PEN (vedolizumab) injection, for subcutaneous use |
| This Medication Guide has been approved by the U.S. Food and Drug Administration | VMB245 R12 Revised: 2/2026 | |
|
What is the most important information I should know about ENTYVIO? ENTYVIO may cause serious side effects, including:
|
||
|
What is ENTYVIO?
ENTYVIO is a prescription medicine used in adults for the treatment of:
|
||
|
Who should not receive ENTYVIO?
Do not receive ENTYVIO if you have had an allergic reaction to ENTYVIO or any of the ingredients in ENTYVIO. See the end of this Medication Guide for a complete list of ingredients in ENTYVIO. |
||
Before receiving ENTYVIO, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take or have recently taken Tysabri (natalizumab), Tyruko (natalizumab-sztn), a Tumor Necrosis Factor (TNF) blocker medicine, a medicine that weakens your immune system (immunosuppressant), or corticosteroid medicine. |
||
|
How should I use ENTYVIO? When given in a vein (intravenously):
|
||
|
What are the possible side effects of ENTYVIO?
ENTYVIO may cause serious side effects, see "What is the most important information I should know about ENTYVIO?" The most common side effects of ENTYVIO include: common cold, headache, joint pain, nausea, fever, infections of the nose and throat, tiredness, cough, bronchitis, flu, back pain, rash, itching, sinus infection, throat pain, pain in extremities, and with injections under the skin: pain, swelling, itching, hives, bruising, rash, or redness at the injection site. These are not all of the possible side effects of ENTYVIO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store ENTYVIO?
|
||
|
General information about the safe and effective use of ENTYVIO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ENTYVIO for a condition for which it was not prescribed. Do not give ENTYVIO to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ENTYVIO that is written for health professionals. |
||
|
What are the ingredients in ENTYVIO?
Active ingredient: vedolizumab Inactive ingredients in vial for intravenous infusion: arginine hydrochloride, histidine, histidine monohydrochloride, polysorbate 80 and sucrose Inactive ingredients in prefilled syringe or prefilled pen for subcutaneous injection: arginine hydrochloride, citric acid monohydrate, histidine, histidine monohydrochloride, polysorbate 80, sodium citrate dihydrate, and Sterile Water for Injection Manufactured by: Takeda Pharmaceuticals U.S.A., Inc. Cambridge, MA 02142 U.S. License No. 1898 ENTYVIO is a registered trademark of Millennium Pharmaceuticals Inc. and is used under license by Takeda Pharmaceuticals U.S.A., Inc. All other trademark names are the property of their respective owners. ©2026 Takeda Pharmaceuticals U.S.A., Inc. For more information, go to www.ENTYVIO.com or call 1-877-TAKEDA7 (1-877-825-3327). |
Section 43683-2
Section 44425-7
Storage and Handling
- Refrigerate ENTYVIO unopened vials, prefilled syringes, and prefilled pens at 2°C to 8°C (36° to 46°F).
- If needed, the ENTYVIO prefilled syringe or ENTYVIO PEN can be left out of the refrigerator in the original package at room temperature up to 25°C (77°F) for up to 7 days (for example, when traveling). Do not use ENTYVIO prefilled syringe or ENTYVIO PEN if left out of the refrigerator for more than 7 days.
- Do not freeze ENTYVIO vial, prefilled syringe, or prefilled pen. Do not use ENTYVIO vial, prefilled syringe, or prefilled pen if it has been frozen.
- Do not shake the ENTYVIO prefilled syringe or ENTYVIO PEN.
- Retain in original package to protect from light until the time of use.
11 Description
Vedolizumab, an integrin receptor antagonist, is a humanized IgG1 monoclonal antibody produced in Chinese hamster ovary cells that binds to the human α4β7 integrin. ENTYVIO has an approximate molecular weight of 147 kilodaltons.
5.2 Infections
Patients treated with ENTYVIO are at increased risk for developing infections [see Adverse Reactions (6.1)]. Serious infections reported in clinical trials include anal abscess, sepsis (some fatal), tuberculosis (TB), salmonella sepsis, Listeria meningitis, giardiasis, and cytomegaloviral colitis. Postmarketing cases of systemic bacterial, fungal, viral, and parasitic opportunistic infections have been reported.
Treatment with ENTYVIO should not be initiated in patients with a clinically important active infection until the infection resolves or is adequately treated. In patients with a chronic infection or a history of recurrent infection, consider the risks and benefits prior to prescribing ENTYVIO. During treatment with ENTYVIO, instruct patients to seek medical advice if signs or symptoms of clinically important acute or chronic infection occur. If a serious infection develops or an infection is not responding to standard therapy, monitor the patient closely. ENTYVIO should not be administered until the infection resolves.
Tuberculosis
Consider evaluating patients for TB infection prior to initiating treatment with ENTYVIO. Treatment with Entyvio should not be administered to patients with active TB infection. Initiate treatment of latent TB prior to administering ENTYVIO. Consider anti-TB therapy prior to initiation of ENTYVIO in patients with a past history of latent or active TB in whom an adequate course of treatment cannot be confirmed. Monitor patients for signs and symptoms of active TB during and after ENTYVIO treatment.
5.4 Liver Injury
There have been reports of elevations of transaminase and/or bilirubin in patients receiving ENTYVIO. In general, the combination of transaminase elevations and elevated bilirubin without evidence of obstruction is generally recognized as an important predictor of severe liver injury that may lead to death or the need for a liver transplant in some patients. ENTYVIO should be discontinued in patients with jaundice or other evidence of significant liver injury [see Adverse Reactions (6.1)].
7.2 Tnf Blockers
Because of the potential for increased risk of infections, avoid the concomitant use of ENTYVIO with TNF blockers.
5.5 Immunizations
Prior to initiating treatment with ENTYVIO, all patients should be brought up to date with all immunizations according to current immunization guidelines [see Dosage and Administration (2.1)] . Patients receiving ENTYVIO may receive non-live vaccines (e.g., influenza vaccine injection) and may receive live vaccines if the benefits outweigh the risks. There are no data on the secondary transmission of infection by live vaccines in patients receiving ENTYVIO [see Adverse Reactions (6.1)].
8.4 Pediatric Use
Safety and effectiveness of ENTYVIO in pediatric patients have not been established.
8.5 Geriatric Use
Clinical trials of ENTYVIO did not include sufficient numbers of patients aged 65 and over (72 patients with Crohn's disease or ulcerative colitis aged 65 and over were treated with ENTYVIO during controlled Phase 3 trials) to determine whether they respond differently from younger adult patients. However, no overall differences in safety or effectiveness were observed between these patients and younger adult patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of ENTYVIO or of other vedolizumab products.
4 Contraindications
ENTYVIO is contraindicated in patients who have had a known serious or severe hypersensitivity reaction to ENTYVIO or any of its excipients (such as dyspnea, bronchospasm, urticaria, flushing, rash and increased heart rate) [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The following topics are also discussed in detail in the Warnings and Precautions section:
- Infusion-Related Reactions and Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Infections [see Warnings and Precautions (5.2)]
- Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.3)]
- Liver Injury [see Warnings and Precautions (5.4)]
Instructions for Use
ENTYVIO
®
(en ti' vee oh) PEN
(vedolizumab)
injection, for subcutaneous use
Single-dose prefilled pen
This Instructions for Use contains information on how to inject ENTYVIO.
Your ENTYVIO single-dose prefilled pen
Important information you need to know before injecting ENTYVIO:
|
Storing ENTYVIO
- Store your prefilled pen in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Your prefilled pen can be left in its box at room temperature up to 77°F (25°C) for up to 7 days (for example, when traveling). Do not use the prefilled pen if it is left out of the refrigerator for more than 7 days.
- Do not freeze the prefilled pen.
- Do not leave the prefilled pen in direct sunlight.
- Throw away the prefilled pen in a FDA-cleared sharps disposal container if it has been left out of the refrigerator for more than 7 days, frozen, or left in direct sunlight. See Step 14 for instructions on how to throw away (dispose of) the prefilled pen.
- Always keep ENTYVIO PENs, the sharps disposal container, and all medicines out of the reach of children.
| U.S. License No. 1898 | ||
| Getting Your Supplies Ready | ||
| Step 1. Remove the ENTYVIO PEN box from the refrigerator | ||
Take 1 prefilled pen box from the refrigerator and check the expiration date on the box (see
Figure A
).
|
Figure A | |
| Step 2. Wait 30 minutes | ||
Wait 30 minutes and let the prefilled pen come to room temperature (see
Figure B
).
|
Figure B | |
| Step 3. Gather supplies | ||
Find a clean, flat surface like a table. Gather supplies that are not in the prefilled pen box (see
Figure C
).
|
Figure C | |
| Preparing to Inject ENTYVIO | ||
| Step 4. Wash hands | ||
| Wash your hands with soap and clean water (see Figure D ). | Figure D | |
| Step 5. Remove the prefilled pen from the tray | ||
Peel off the paper on the tray and lift the prefilled pen straight out (see
Figure E
).
|
Figure E | |
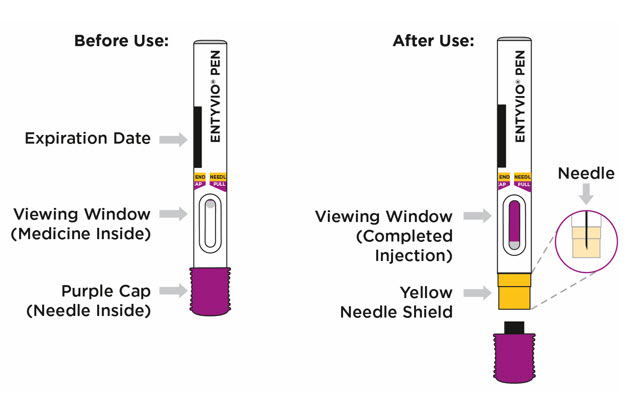
| Step 6. Inspect the prefilled pen | ||
Check the expiration date (EXP) printed on the prefilled pen and the medicine in the prefilled pen viewing window (see
Figure F
). The medicine should be colorless to pale yellow. It is normal to see air bubbles. Inspect the prefilled pen for any damage.
|
||
| Step 7. Choose injection site | ||
Choose an injection site on your bare skin from one of the following (see
Figure G
):
|
Figure G | |
| Step 8. Clean the injection site | ||
Clean the injection site with an alcohol pad (see
Figure H
). Let your skin dry.
|
Figure H | |
| Continue to Step 9 ➔ | ||
| Injecting ENTYVIO | ||
| Step 9. Remove the purple cap and throw it away | ||
When you are ready to inject, pull the purple cap straight off and throw it right away in the sharps disposal container (see
Figure I
).
|
Figure I | |
| Step 10. Place the prefilled pen on the injection site | ||
|
Figure J | |
| Step 11. Start injecting ENTYVIO | Step 12. Complete injecting ENTYVIO | |
Push the prefilled pen straight down and hold for at least 10 seconds (see
Figure K
).
|
Continue holding the prefilled pen with constant pressure until the viewing window has filled with purple to make sure you have received your full dose (see
Figure L
).
|
|
| Step 13. Lift prefilled pen from skin | ||
Lift the prefilled pen from the injection site (see
Figure M
). The yellow needle shield will drop down and lock over the needle.
|
Figure M | |
| Step 14. Throw away (dispose of) the prefilled pen | ||
Throw away (dispose of) the used prefilled pen in a FDA-cleared sharps disposal container right away after use (see
Figure N
). Do not recycle or throw away the prefilled pen in your household trash.
Manufactured by: Takeda Pharmaceuticals U.S.A., Inc. Cambridge, MA 02142 |
Figure N |
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 5/2024
12.2 Pharmacodynamics
In clinical trials with intravenous ENTYVIO at doses ranging from 0.2 to 10 mg/kg (which includes doses outside of the recommended dose), saturation of α4β7 receptors on subsets of circulating lymphocytes involved in gut-immune surveillance was observed.
In clinical trials with intravenous ENTYVIO at doses ranging from 0.2 to 10 mg/kg and 180 to 750 mg (which include doses outside of the recommended dose) in healthy subjects and in patients with ulcerative colitis or Crohn's disease, vedolizumab did not elevate neutrophils, basophils, eosinophils, B-helper and cytotoxic T-lymphocytes, total memory helper T-lymphocytes, monocytes or natural killer cells.
A reduction in gastrointestinal inflammation was observed in rectal biopsy specimens from Phase 2 ulcerative colitis patients exposed to ENTYVIO for four or six weeks compared to placebo control as assessed by histopathology.
In a study of 14 healthy subjects, ENTYVIO did not affect the CD4+ lymphocyte cell counts, CD8+ lymphocyte cell counts, or the CD4+:CD8+ ratios in the CSF [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
Similar pharmacokinetics were observed in ulcerative colitis and Crohn's disease patients administered 300 mg ENTYVIO as a 30-minute intravenous infusion on Weeks 0, 2, and 6, and then every eight weeks up to Week 52 (Table 3).
| Patient Population | Weeks 0, 2, and 6 ENTYVIO 300 mg Intravenously | After Week 6 to 52 ENTYVIO 300 mg Intravenously Every 8 Weeks |
|---|---|---|
| Trough Serum Concentration at Week 6 (mcg/mL) | Trough Serum Concentration at Week 46 Steady-state trough serum concentration. (mcg/mL) |
|
| Ulcerative Colitis | 26.3 ± 12.9 (N=210) |
11.2 ± 7.2 (N=77) |
| Crohn's Disease | 27.4 ± 19.2 (N=198) |
13.0 ± 9.1 (N=72) |
In ulcerative colitis and Crohn’s disease patients, administered 300 mg ENTYVIO as a 30-minute intravenous infusion on Weeks 0 and 2, followed by 108 mg ENTYVIO as a subcutaneous injection every 2 weeks starting from Week 6, the mean steady state serum trough concentrations were 35.8 mcg/mL (SD ± 15.2) and 31.4 mcg/mL (SD ± 14.7), respectively.
The bioavailability of vedolizumab following a 108 mg single-dose subcutaneous injection relative to a 300 mg single-dose intravenous infusion in healthy subjects was approximately 75%. Following a 108 mg single-dose subcutaneous injection in healthy subjects, the median Tmax was 7 days with a range of 3 to 14 days and the mean Cmax was 15.4 mcg/mL (SD ± 3.2).
Vedolizumab clearance depends on both linear and nonlinear pathways; the nonlinear clearance decreases with increasing concentrations. Population pharmacokinetic analyses indicated that the linear clearance was approximately 0.16 L/day, the serum half-life was approximately 26 days, and the distribution volume was approximately 5 L.
Vedolizumab was not detected in the cerebrospinal fluid (CSF) of 14 healthy subjects at five weeks after a single intravenous administration of 450 mg ENTYVIO (1.5 times the recommended dosage).
7.3 Cyp450 Substrates
The formation of CYP450 enzymes may be suppressed by increased levels of certain cytokines (e.g., IL-6, IL-10, TNFα, IFN) during chronic inflammation. Therefore, use of ENTYVIO may normalize the formation of CYP450 enzymes by modulating the underlying disease. Upon initiation or discontinuation of ENTYVIO in patients treated with CYP450 substrates, monitor drug concentrations or other therapeutic parameters, and adjust the dosage of the CYP substrate as needed. See the prescribing information of specific CYP substrates.
1 Indications and Usage
ENTYVIO is indicated in adults for the treatment of:
- moderately to severely active ulcerative colitis (UC).
- moderately to severely active Crohn's disease (CD).
12.1 Mechanism of Action
Vedolizumab is a humanized monoclonal antibody that specifically binds to the α4β7 integrin and blocks the interaction of α4β7 integrin with mucosal addressin cell adhesion molecule-1 (MAdCAM-1) and inhibits the migration of memory T-lymphocytes across the endothelium into inflamed gastrointestinal parenchymal tissue. Vedolizumab does not bind to or inhibit function of the α4β1 and αEβ7 integrins and does not antagonize the interaction of α4 integrins with vascular cell adhesion molecule-1 (VCAM-1).
The α4β7 integrin is expressed on the surface of a discrete subset of memory T-lymphocytes that preferentially migrate into the gastrointestinal tract. MAdCAM-1 is mainly expressed on gut endothelial cells and plays a critical role in the homing of T-lymphocytes to gut lymph tissue. The interaction of the α4β7 integrin with MAdCAM-1 has been implicated as an important contributor to the chronic inflammation that is a hallmark of ulcerative colitis and Crohn's disease.
7.1 Natalizumab Products
Because of the potential for increased risk of PML and other infections, avoid the concomitant use of ENTYVIO with natalizumab products.
5 Warnings and Precautions
- Infusion-Related Reactions and Hypersensitivity Reactions: Discontinue ENTYVIO and initiate appropriate treatment if serious reactions occur. (5.1)
- Infections: Treatment with ENTYVIO should not be initiated in patients with a clinically important active infection until the infection resolves or is adequately treated. If a serious infection develops, ENTYVIO should not be administered until the infection resolves. (5.2)
- Progressive Multifocal Leukoencephalopathy (PML): Although unlikely, a risk of PML cannot be ruled out. Monitor patients for any new or worsening neurological signs or symptoms. (5.3)
2 Dosage and Administration
Important Administration Information
- Before Initiating ENTYVIO
- Intravenous Administration: ENTYVIO should be administered intravenously by a healthcare provider. (2.1)
- Subcutaneous Injection: ENTYVIO prefilled syringe and ENTYVIO PEN are intended for subcutaneous use. A patient may self-inject or caregiver may inject after proper training on correct subcutaneous injection technique. (2.1)
Recommended Dosage (2.2)
- Week 0: 300 mg infused intravenously over approximately 30 minutes.
- Week 2: 300 mg infused intravenously over approximately 30 minutes.
-
Week 6: Patients may remain on ENTYVIO intravenous therapy or switch to subcutaneous injection after receiving two ENTYVIO intravenous doses administered at Week 0 and Week 2.
- Intravenous Infusion: 300 mg infused over approximately 30 minutes and then every eight weeks thereafter.
- Subcutaneous Injection: 108 mg subcutaneously once every two weeks.
- Discontinue ENTYVIO in patients who do not show evidence of therapeutic benefit by Week 14.
- Patients currently receiving and responding to ENTYVIO intravenous therapy after Week 6 may also be switched to subcutaneous injection. Administer the first subcutaneous dose in place of the next scheduled intravenous infusion and every two weeks thereafter.
Preparation and Administration Instructions:
3 Dosage Forms and Strengths
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ENTYVIO. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: Anaphylaxis [see Warnings and Precautions (5.1)].
Gastrointestinal system disorders: Acute pancreatitis.
Respiratory, thoracic, and mediastinal disorders: Interstitial lung disease, pneumonitis.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to intravenous ENTYVIO in 3,326 patients and healthy volunteers in clinical trials, including 1,396 exposed for greater than one year, and 835 exposed for greater than two years.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
14.2 Clinical Studies in Crohn's Disease
Intravenous Administration
The safety and efficacy of intravenous ENTYVIO were evaluated in three randomized, double-blind, placebo-controlled clinical trials (CD Trials I, II, and III) in adult patients with moderately to severely active Crohn's disease (CD) (Crohn's Disease Activity Index [CDAI] score of 220 to 450).
Enrolled patients in the U.S. had over the previous five-year period an inadequate response or intolerance to immunomodulator therapy (i.e., azathioprine, 6-mercaptopurine, or methotrexate) and/or an inadequate response, loss of response, or intolerance to one or more TNF blockers. Outside the U.S., prior treatment with corticosteroids was sufficient for entry if over the previous five-year period the patients were corticosteroid dependent (i.e., unable to successfully taper corticosteroids without a return of the symptoms of CD) or had an inadequate response or intolerance to corticosteroids.
Patients that had received natalizumab ever in the past, and patients that had received a TNF blocker in the past 30 to 60 days were excluded from enrollment. Concomitant use of natalizumab or a TNF blocker was not allowed.
2.1 Important Administration Information
Before Initiating ENTYVIO
- Consider evaluating patients for tuberculosis (TB) infection prior to initiating treatment with ENTYVIO [see Warnings and Precautions (5.2)].
- Update immunizations according to current immunization guidelines [see Warnings and Precautions (5.5)].
Principal Display Panel 300 Mg Vial Label
NDC 64764-300-20 Rx only
Entyvio
vedolizumab
for injection
300 mg per vial*
For Intravenous Use Only
Must be reconstituted
and diluted prior to use
Single-Dose Vial - Discard Unused
Portion
Principal Display Panel 300 Mg Vial Carton
NDC 64764-300-20 Rx only
Entyvio
vedolizumab
for injection
300 mg per vial*
ATTENTION: Each patient is required to
receive the enclosed Medication Guide.
For Intravenous Use Only
Must be reconstituted and diluted prior
to use
Single-Dose Vial - Discard Unused Portion
Takeda
5.3 Progressive Multifocal Leukoencephalopathy
PML, a rare and often fatal opportunistic infection of the central nervous system (CNS), has been reported with systemic immunosuppressants, including another integrin receptor antagonist. PML is caused by the John Cunningham (JC) virus and typically only occurs in patients who are immunocompromised. One case of PML in an ENTYVIO-treated patient with multiple contributory factors has been reported in the postmarketing setting (e.g., human immunodeficiency virus [HIV] infection with a CD4 count of 300 cells/mm3 and prior and concomitant immunosuppression). Although unlikely, a risk of PML cannot be ruled out.
Monitor patients on ENTYVIO for any new onset, or worsening, of neurological signs and symptoms. Typical signs and symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes. The progression of deficits usually leads to death or severe disability over weeks or months. If PML is suspected, withhold dosing with ENTYVIO and refer to a neurologist; if confirmed, discontinue dosing permanently.
Principal Display Panel 108 Mg/0.68 Ml Pen Tray Carton
NDC 64764-108-21 Rx only
ENTYVIO® PEN
(vedolizumab)
injection
108 mg/0.68 mL
For Subcutaneous Use Only
1 Single-Dose Prefilled Pen
ATTENTION PHARMACIST: Each
patient is required to receive the
enclosed Medication Guide.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of vedolizumab. Studies to evaluate the possible impairment of fertility or mutagenic potential of vedolizumab have not been performed.
Principal Display Panel 108 Mg/0.68 Ml Syringe Tray Carton
NDC 64764-107-11 Rx only
ENTYVIO®
(vedolizumab)
injection
108 mg/0.68 mL
For Subcutaneous Use Only
1 Single-Dose Prefilled Syringe
ATTENTION PHARMACIST: Each patient
is required to receive the enclosed
Medication Guide.
5.1 Infusion Related Reactions and Hypersensitivity Reactions
Infusion-related reactions and hypersensitivity reactions have been reported, including anaphylaxis, dyspnea, bronchospasm, urticaria, flushing, rash, and increased blood pressure and heart rate [see Adverse Reactions (6.1, 6.2)]. These reactions may occur with the first or subsequent infusions of ENTYVIO and may vary in their time of onset from during infusion or up to several hours post-infusion.
If anaphylaxis or other serious infusion-related or hypersensitivity reactions occur, discontinue administration of ENTYVIO immediately and initiate appropriate treatment.
2.4 Preparation and Administration Instructions for Subcutaneous Injection
- Inspect the solution visually for particulate matter and discoloration prior to administration. ENTYVIO in prefilled syringe or ENTYVIO PEN should be a clear to moderately opalescent, colorless to slightly yellow solution. Do not use ENTYVIO prefilled syringes or ENTYVIO PENs with visible particulate matter or discoloration.
- Administer each subcutaneous injection at a different anatomic location (such as thighs, any quadrant of abdomen, or upper arms) than the previous injection. Administration of ENTYVIO in the back of upper arm may only be performed by a healthcare professional or caregiver. Do not inject into moles, scars, bruises, or areas where the skin is tender, erythematous, or indurated.
Missed Subcutaneous Dose
If treatment with subcutaneous ENTYVIO is interrupted or if a scheduled dose(s) of subcutaneous ENTYVIO is missed, inject the next subcutaneous dose as soon as possible and then every 2 weeks thereafter.
In the event of incomplete dose administration (i.e., patient attempts administration of dose with ENTYVIO PEN, however it is uncertain if a full dose was administered), instruct the patient to call their pharmacy or healthcare provider.
Structured Label Content
Section 42229-5 (42229-5)
Intravenous Administration
- ENTYVIO should be administered by a healthcare provider prepared to manage hypersensitivity reactions including anaphylaxis, if they occur [see Warnings and Precautions (5.1)]. Appropriate monitoring and medical support measures should be available for immediate use. Observe patients during infusion and until the infusion is complete.
- Reconstitute and dilute ENTYVIO lyophilized powder prior to administration as a 30-minute intravenous infusion [see Dosage and Administration (2.3)].
Section 42231-1 (42231-1)
| MEDICATION GUIDE | ||
|---|---|---|
| ENTYVIO® (en ti' vee oh) (vedolizumab) for injection, for intravenous use |
ENTYVIO® (en ti' vee oh) (vedolizumab) injection, for subcutaneous use |
ENTYVIO® (en ti' vee oh) PEN (vedolizumab) injection, for subcutaneous use |
| This Medication Guide has been approved by the U.S. Food and Drug Administration | VMB245 R12 Revised: 2/2026 | |
|
What is the most important information I should know about ENTYVIO? ENTYVIO may cause serious side effects, including:
|
||
|
What is ENTYVIO?
ENTYVIO is a prescription medicine used in adults for the treatment of:
|
||
|
Who should not receive ENTYVIO?
Do not receive ENTYVIO if you have had an allergic reaction to ENTYVIO or any of the ingredients in ENTYVIO. See the end of this Medication Guide for a complete list of ingredients in ENTYVIO. |
||
Before receiving ENTYVIO, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take or have recently taken Tysabri (natalizumab), Tyruko (natalizumab-sztn), a Tumor Necrosis Factor (TNF) blocker medicine, a medicine that weakens your immune system (immunosuppressant), or corticosteroid medicine. |
||
|
How should I use ENTYVIO? When given in a vein (intravenously):
|
||
|
What are the possible side effects of ENTYVIO?
ENTYVIO may cause serious side effects, see "What is the most important information I should know about ENTYVIO?" The most common side effects of ENTYVIO include: common cold, headache, joint pain, nausea, fever, infections of the nose and throat, tiredness, cough, bronchitis, flu, back pain, rash, itching, sinus infection, throat pain, pain in extremities, and with injections under the skin: pain, swelling, itching, hives, bruising, rash, or redness at the injection site. These are not all of the possible side effects of ENTYVIO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store ENTYVIO?
|
||
|
General information about the safe and effective use of ENTYVIO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ENTYVIO for a condition for which it was not prescribed. Do not give ENTYVIO to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ENTYVIO that is written for health professionals. |
||
|
What are the ingredients in ENTYVIO?
Active ingredient: vedolizumab Inactive ingredients in vial for intravenous infusion: arginine hydrochloride, histidine, histidine monohydrochloride, polysorbate 80 and sucrose Inactive ingredients in prefilled syringe or prefilled pen for subcutaneous injection: arginine hydrochloride, citric acid monohydrate, histidine, histidine monohydrochloride, polysorbate 80, sodium citrate dihydrate, and Sterile Water for Injection Manufactured by: Takeda Pharmaceuticals U.S.A., Inc. Cambridge, MA 02142 U.S. License No. 1898 ENTYVIO is a registered trademark of Millennium Pharmaceuticals Inc. and is used under license by Takeda Pharmaceuticals U.S.A., Inc. All other trademark names are the property of their respective owners. ©2026 Takeda Pharmaceuticals U.S.A., Inc. For more information, go to www.ENTYVIO.com or call 1-877-TAKEDA7 (1-877-825-3327). |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and Handling
- Refrigerate ENTYVIO unopened vials, prefilled syringes, and prefilled pens at 2°C to 8°C (36° to 46°F).
- If needed, the ENTYVIO prefilled syringe or ENTYVIO PEN can be left out of the refrigerator in the original package at room temperature up to 25°C (77°F) for up to 7 days (for example, when traveling). Do not use ENTYVIO prefilled syringe or ENTYVIO PEN if left out of the refrigerator for more than 7 days.
- Do not freeze ENTYVIO vial, prefilled syringe, or prefilled pen. Do not use ENTYVIO vial, prefilled syringe, or prefilled pen if it has been frozen.
- Do not shake the ENTYVIO prefilled syringe or ENTYVIO PEN.
- Retain in original package to protect from light until the time of use.
11 Description (11 DESCRIPTION)
Vedolizumab, an integrin receptor antagonist, is a humanized IgG1 monoclonal antibody produced in Chinese hamster ovary cells that binds to the human α4β7 integrin. ENTYVIO has an approximate molecular weight of 147 kilodaltons.
5.2 Infections
Patients treated with ENTYVIO are at increased risk for developing infections [see Adverse Reactions (6.1)]. Serious infections reported in clinical trials include anal abscess, sepsis (some fatal), tuberculosis (TB), salmonella sepsis, Listeria meningitis, giardiasis, and cytomegaloviral colitis. Postmarketing cases of systemic bacterial, fungal, viral, and parasitic opportunistic infections have been reported.
Treatment with ENTYVIO should not be initiated in patients with a clinically important active infection until the infection resolves or is adequately treated. In patients with a chronic infection or a history of recurrent infection, consider the risks and benefits prior to prescribing ENTYVIO. During treatment with ENTYVIO, instruct patients to seek medical advice if signs or symptoms of clinically important acute or chronic infection occur. If a serious infection develops or an infection is not responding to standard therapy, monitor the patient closely. ENTYVIO should not be administered until the infection resolves.
Tuberculosis
Consider evaluating patients for TB infection prior to initiating treatment with ENTYVIO. Treatment with Entyvio should not be administered to patients with active TB infection. Initiate treatment of latent TB prior to administering ENTYVIO. Consider anti-TB therapy prior to initiation of ENTYVIO in patients with a past history of latent or active TB in whom an adequate course of treatment cannot be confirmed. Monitor patients for signs and symptoms of active TB during and after ENTYVIO treatment.
5.4 Liver Injury
There have been reports of elevations of transaminase and/or bilirubin in patients receiving ENTYVIO. In general, the combination of transaminase elevations and elevated bilirubin without evidence of obstruction is generally recognized as an important predictor of severe liver injury that may lead to death or the need for a liver transplant in some patients. ENTYVIO should be discontinued in patients with jaundice or other evidence of significant liver injury [see Adverse Reactions (6.1)].
7.2 Tnf Blockers (7.2 TNF Blockers)
Because of the potential for increased risk of infections, avoid the concomitant use of ENTYVIO with TNF blockers.
5.5 Immunizations
Prior to initiating treatment with ENTYVIO, all patients should be brought up to date with all immunizations according to current immunization guidelines [see Dosage and Administration (2.1)] . Patients receiving ENTYVIO may receive non-live vaccines (e.g., influenza vaccine injection) and may receive live vaccines if the benefits outweigh the risks. There are no data on the secondary transmission of infection by live vaccines in patients receiving ENTYVIO [see Adverse Reactions (6.1)].
8.4 Pediatric Use
Safety and effectiveness of ENTYVIO in pediatric patients have not been established.
8.5 Geriatric Use
Clinical trials of ENTYVIO did not include sufficient numbers of patients aged 65 and over (72 patients with Crohn's disease or ulcerative colitis aged 65 and over were treated with ENTYVIO during controlled Phase 3 trials) to determine whether they respond differently from younger adult patients. However, no overall differences in safety or effectiveness were observed between these patients and younger adult patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of ENTYVIO or of other vedolizumab products.
4 Contraindications (4 CONTRAINDICATIONS)
ENTYVIO is contraindicated in patients who have had a known serious or severe hypersensitivity reaction to ENTYVIO or any of its excipients (such as dyspnea, bronchospasm, urticaria, flushing, rash and increased heart rate) [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following topics are also discussed in detail in the Warnings and Precautions section:
- Infusion-Related Reactions and Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Infections [see Warnings and Precautions (5.2)]
- Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.3)]
- Liver Injury [see Warnings and Precautions (5.4)]
Instructions for Use (INSTRUCTIONS FOR USE)
ENTYVIO
®
(en ti' vee oh) PEN
(vedolizumab)
injection, for subcutaneous use
Single-dose prefilled pen
This Instructions for Use contains information on how to inject ENTYVIO.
Your ENTYVIO single-dose prefilled pen
Important information you need to know before injecting ENTYVIO:
|
Storing ENTYVIO
- Store your prefilled pen in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Your prefilled pen can be left in its box at room temperature up to 77°F (25°C) for up to 7 days (for example, when traveling). Do not use the prefilled pen if it is left out of the refrigerator for more than 7 days.
- Do not freeze the prefilled pen.
- Do not leave the prefilled pen in direct sunlight.
- Throw away the prefilled pen in a FDA-cleared sharps disposal container if it has been left out of the refrigerator for more than 7 days, frozen, or left in direct sunlight. See Step 14 for instructions on how to throw away (dispose of) the prefilled pen.
- Always keep ENTYVIO PENs, the sharps disposal container, and all medicines out of the reach of children.
| U.S. License No. 1898 | ||
| Getting Your Supplies Ready | ||
| Step 1. Remove the ENTYVIO PEN box from the refrigerator | ||
Take 1 prefilled pen box from the refrigerator and check the expiration date on the box (see
Figure A
).
|
Figure A | |
| Step 2. Wait 30 minutes | ||
Wait 30 minutes and let the prefilled pen come to room temperature (see
Figure B
).
|
Figure B | |
| Step 3. Gather supplies | ||
Find a clean, flat surface like a table. Gather supplies that are not in the prefilled pen box (see
Figure C
).
|
Figure C | |
| Preparing to Inject ENTYVIO | ||
| Step 4. Wash hands | ||
| Wash your hands with soap and clean water (see Figure D ). | Figure D | |
| Step 5. Remove the prefilled pen from the tray | ||
Peel off the paper on the tray and lift the prefilled pen straight out (see
Figure E
).
|
Figure E | |
| Step 6. Inspect the prefilled pen | ||
Check the expiration date (EXP) printed on the prefilled pen and the medicine in the prefilled pen viewing window (see
Figure F
). The medicine should be colorless to pale yellow. It is normal to see air bubbles. Inspect the prefilled pen for any damage.
|
||
| Step 7. Choose injection site | ||
Choose an injection site on your bare skin from one of the following (see
Figure G
):
|
Figure G | |
| Step 8. Clean the injection site | ||
Clean the injection site with an alcohol pad (see
Figure H
). Let your skin dry.
|
Figure H | |
| Continue to Step 9 ➔ | ||
| Injecting ENTYVIO | ||
| Step 9. Remove the purple cap and throw it away | ||
When you are ready to inject, pull the purple cap straight off and throw it right away in the sharps disposal container (see
Figure I
).
|
Figure I | |
| Step 10. Place the prefilled pen on the injection site | ||
|
Figure J | |
| Step 11. Start injecting ENTYVIO | Step 12. Complete injecting ENTYVIO | |
Push the prefilled pen straight down and hold for at least 10 seconds (see
Figure K
).
|
Continue holding the prefilled pen with constant pressure until the viewing window has filled with purple to make sure you have received your full dose (see
Figure L
).
|
|
| Step 13. Lift prefilled pen from skin | ||
Lift the prefilled pen from the injection site (see
Figure M
). The yellow needle shield will drop down and lock over the needle.
|
Figure M | |
| Step 14. Throw away (dispose of) the prefilled pen | ||
Throw away (dispose of) the used prefilled pen in a FDA-cleared sharps disposal container right away after use (see
Figure N
). Do not recycle or throw away the prefilled pen in your household trash.
Manufactured by: Takeda Pharmaceuticals U.S.A., Inc. Cambridge, MA 02142 |
Figure N |
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 5/2024
12.2 Pharmacodynamics
In clinical trials with intravenous ENTYVIO at doses ranging from 0.2 to 10 mg/kg (which includes doses outside of the recommended dose), saturation of α4β7 receptors on subsets of circulating lymphocytes involved in gut-immune surveillance was observed.
In clinical trials with intravenous ENTYVIO at doses ranging from 0.2 to 10 mg/kg and 180 to 750 mg (which include doses outside of the recommended dose) in healthy subjects and in patients with ulcerative colitis or Crohn's disease, vedolizumab did not elevate neutrophils, basophils, eosinophils, B-helper and cytotoxic T-lymphocytes, total memory helper T-lymphocytes, monocytes or natural killer cells.
A reduction in gastrointestinal inflammation was observed in rectal biopsy specimens from Phase 2 ulcerative colitis patients exposed to ENTYVIO for four or six weeks compared to placebo control as assessed by histopathology.
In a study of 14 healthy subjects, ENTYVIO did not affect the CD4+ lymphocyte cell counts, CD8+ lymphocyte cell counts, or the CD4+:CD8+ ratios in the CSF [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
Similar pharmacokinetics were observed in ulcerative colitis and Crohn's disease patients administered 300 mg ENTYVIO as a 30-minute intravenous infusion on Weeks 0, 2, and 6, and then every eight weeks up to Week 52 (Table 3).
| Patient Population | Weeks 0, 2, and 6 ENTYVIO 300 mg Intravenously | After Week 6 to 52 ENTYVIO 300 mg Intravenously Every 8 Weeks |
|---|---|---|
| Trough Serum Concentration at Week 6 (mcg/mL) | Trough Serum Concentration at Week 46 Steady-state trough serum concentration. (mcg/mL) |
|
| Ulcerative Colitis | 26.3 ± 12.9 (N=210) |
11.2 ± 7.2 (N=77) |
| Crohn's Disease | 27.4 ± 19.2 (N=198) |
13.0 ± 9.1 (N=72) |
In ulcerative colitis and Crohn’s disease patients, administered 300 mg ENTYVIO as a 30-minute intravenous infusion on Weeks 0 and 2, followed by 108 mg ENTYVIO as a subcutaneous injection every 2 weeks starting from Week 6, the mean steady state serum trough concentrations were 35.8 mcg/mL (SD ± 15.2) and 31.4 mcg/mL (SD ± 14.7), respectively.
The bioavailability of vedolizumab following a 108 mg single-dose subcutaneous injection relative to a 300 mg single-dose intravenous infusion in healthy subjects was approximately 75%. Following a 108 mg single-dose subcutaneous injection in healthy subjects, the median Tmax was 7 days with a range of 3 to 14 days and the mean Cmax was 15.4 mcg/mL (SD ± 3.2).
Vedolizumab clearance depends on both linear and nonlinear pathways; the nonlinear clearance decreases with increasing concentrations. Population pharmacokinetic analyses indicated that the linear clearance was approximately 0.16 L/day, the serum half-life was approximately 26 days, and the distribution volume was approximately 5 L.
Vedolizumab was not detected in the cerebrospinal fluid (CSF) of 14 healthy subjects at five weeks after a single intravenous administration of 450 mg ENTYVIO (1.5 times the recommended dosage).
7.3 Cyp450 Substrates (7.3 CYP450 Substrates)
The formation of CYP450 enzymes may be suppressed by increased levels of certain cytokines (e.g., IL-6, IL-10, TNFα, IFN) during chronic inflammation. Therefore, use of ENTYVIO may normalize the formation of CYP450 enzymes by modulating the underlying disease. Upon initiation or discontinuation of ENTYVIO in patients treated with CYP450 substrates, monitor drug concentrations or other therapeutic parameters, and adjust the dosage of the CYP substrate as needed. See the prescribing information of specific CYP substrates.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ENTYVIO is indicated in adults for the treatment of:
- moderately to severely active ulcerative colitis (UC).
- moderately to severely active Crohn's disease (CD).
12.1 Mechanism of Action
Vedolizumab is a humanized monoclonal antibody that specifically binds to the α4β7 integrin and blocks the interaction of α4β7 integrin with mucosal addressin cell adhesion molecule-1 (MAdCAM-1) and inhibits the migration of memory T-lymphocytes across the endothelium into inflamed gastrointestinal parenchymal tissue. Vedolizumab does not bind to or inhibit function of the α4β1 and αEβ7 integrins and does not antagonize the interaction of α4 integrins with vascular cell adhesion molecule-1 (VCAM-1).
The α4β7 integrin is expressed on the surface of a discrete subset of memory T-lymphocytes that preferentially migrate into the gastrointestinal tract. MAdCAM-1 is mainly expressed on gut endothelial cells and plays a critical role in the homing of T-lymphocytes to gut lymph tissue. The interaction of the α4β7 integrin with MAdCAM-1 has been implicated as an important contributor to the chronic inflammation that is a hallmark of ulcerative colitis and Crohn's disease.
7.1 Natalizumab Products
Because of the potential for increased risk of PML and other infections, avoid the concomitant use of ENTYVIO with natalizumab products.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Infusion-Related Reactions and Hypersensitivity Reactions: Discontinue ENTYVIO and initiate appropriate treatment if serious reactions occur. (5.1)
- Infections: Treatment with ENTYVIO should not be initiated in patients with a clinically important active infection until the infection resolves or is adequately treated. If a serious infection develops, ENTYVIO should not be administered until the infection resolves. (5.2)
- Progressive Multifocal Leukoencephalopathy (PML): Although unlikely, a risk of PML cannot be ruled out. Monitor patients for any new or worsening neurological signs or symptoms. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Important Administration Information
- Before Initiating ENTYVIO
- Intravenous Administration: ENTYVIO should be administered intravenously by a healthcare provider. (2.1)
- Subcutaneous Injection: ENTYVIO prefilled syringe and ENTYVIO PEN are intended for subcutaneous use. A patient may self-inject or caregiver may inject after proper training on correct subcutaneous injection technique. (2.1)
Recommended Dosage (2.2)
- Week 0: 300 mg infused intravenously over approximately 30 minutes.
- Week 2: 300 mg infused intravenously over approximately 30 minutes.
-
Week 6: Patients may remain on ENTYVIO intravenous therapy or switch to subcutaneous injection after receiving two ENTYVIO intravenous doses administered at Week 0 and Week 2.
- Intravenous Infusion: 300 mg infused over approximately 30 minutes and then every eight weeks thereafter.
- Subcutaneous Injection: 108 mg subcutaneously once every two weeks.
- Discontinue ENTYVIO in patients who do not show evidence of therapeutic benefit by Week 14.
- Patients currently receiving and responding to ENTYVIO intravenous therapy after Week 6 may also be switched to subcutaneous injection. Administer the first subcutaneous dose in place of the next scheduled intravenous infusion and every two weeks thereafter.
Preparation and Administration Instructions:
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ENTYVIO. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: Anaphylaxis [see Warnings and Precautions (5.1)].
Gastrointestinal system disorders: Acute pancreatitis.
Respiratory, thoracic, and mediastinal disorders: Interstitial lung disease, pneumonitis.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to intravenous ENTYVIO in 3,326 patients and healthy volunteers in clinical trials, including 1,396 exposed for greater than one year, and 835 exposed for greater than two years.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
14.2 Clinical Studies in Crohn's Disease
Intravenous Administration
The safety and efficacy of intravenous ENTYVIO were evaluated in three randomized, double-blind, placebo-controlled clinical trials (CD Trials I, II, and III) in adult patients with moderately to severely active Crohn's disease (CD) (Crohn's Disease Activity Index [CDAI] score of 220 to 450).
Enrolled patients in the U.S. had over the previous five-year period an inadequate response or intolerance to immunomodulator therapy (i.e., azathioprine, 6-mercaptopurine, or methotrexate) and/or an inadequate response, loss of response, or intolerance to one or more TNF blockers. Outside the U.S., prior treatment with corticosteroids was sufficient for entry if over the previous five-year period the patients were corticosteroid dependent (i.e., unable to successfully taper corticosteroids without a return of the symptoms of CD) or had an inadequate response or intolerance to corticosteroids.
Patients that had received natalizumab ever in the past, and patients that had received a TNF blocker in the past 30 to 60 days were excluded from enrollment. Concomitant use of natalizumab or a TNF blocker was not allowed.
2.1 Important Administration Information
Before Initiating ENTYVIO
- Consider evaluating patients for tuberculosis (TB) infection prior to initiating treatment with ENTYVIO [see Warnings and Precautions (5.2)].
- Update immunizations according to current immunization guidelines [see Warnings and Precautions (5.5)].
Principal Display Panel 300 Mg Vial Label (PRINCIPAL DISPLAY PANEL - 300 mg Vial Label)
NDC 64764-300-20 Rx only
Entyvio
vedolizumab
for injection
300 mg per vial*
For Intravenous Use Only
Must be reconstituted
and diluted prior to use
Single-Dose Vial - Discard Unused
Portion
Principal Display Panel 300 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 300 mg Vial Carton)
NDC 64764-300-20 Rx only
Entyvio
vedolizumab
for injection
300 mg per vial*
ATTENTION: Each patient is required to
receive the enclosed Medication Guide.
For Intravenous Use Only
Must be reconstituted and diluted prior
to use
Single-Dose Vial - Discard Unused Portion
Takeda
5.3 Progressive Multifocal Leukoencephalopathy
PML, a rare and often fatal opportunistic infection of the central nervous system (CNS), has been reported with systemic immunosuppressants, including another integrin receptor antagonist. PML is caused by the John Cunningham (JC) virus and typically only occurs in patients who are immunocompromised. One case of PML in an ENTYVIO-treated patient with multiple contributory factors has been reported in the postmarketing setting (e.g., human immunodeficiency virus [HIV] infection with a CD4 count of 300 cells/mm3 and prior and concomitant immunosuppression). Although unlikely, a risk of PML cannot be ruled out.
Monitor patients on ENTYVIO for any new onset, or worsening, of neurological signs and symptoms. Typical signs and symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes. The progression of deficits usually leads to death or severe disability over weeks or months. If PML is suspected, withhold dosing with ENTYVIO and refer to a neurologist; if confirmed, discontinue dosing permanently.
Principal Display Panel 108 Mg/0.68 Ml Pen Tray Carton (PRINCIPAL DISPLAY PANEL - 108 mg/0.68 mL Pen Tray Carton)
NDC 64764-108-21 Rx only
ENTYVIO® PEN
(vedolizumab)
injection
108 mg/0.68 mL
For Subcutaneous Use Only
1 Single-Dose Prefilled Pen
ATTENTION PHARMACIST: Each
patient is required to receive the
enclosed Medication Guide.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the carcinogenic potential of vedolizumab. Studies to evaluate the possible impairment of fertility or mutagenic potential of vedolizumab have not been performed.
Principal Display Panel 108 Mg/0.68 Ml Syringe Tray Carton (PRINCIPAL DISPLAY PANEL - 108 mg/0.68 mL Syringe Tray Carton)
NDC 64764-107-11 Rx only
ENTYVIO®
(vedolizumab)
injection
108 mg/0.68 mL
For Subcutaneous Use Only
1 Single-Dose Prefilled Syringe
ATTENTION PHARMACIST: Each patient
is required to receive the enclosed
Medication Guide.
5.1 Infusion Related Reactions and Hypersensitivity Reactions (5.1 Infusion-Related Reactions and Hypersensitivity Reactions)
Infusion-related reactions and hypersensitivity reactions have been reported, including anaphylaxis, dyspnea, bronchospasm, urticaria, flushing, rash, and increased blood pressure and heart rate [see Adverse Reactions (6.1, 6.2)]. These reactions may occur with the first or subsequent infusions of ENTYVIO and may vary in their time of onset from during infusion or up to several hours post-infusion.
If anaphylaxis or other serious infusion-related or hypersensitivity reactions occur, discontinue administration of ENTYVIO immediately and initiate appropriate treatment.
2.4 Preparation and Administration Instructions for Subcutaneous Injection
- Inspect the solution visually for particulate matter and discoloration prior to administration. ENTYVIO in prefilled syringe or ENTYVIO PEN should be a clear to moderately opalescent, colorless to slightly yellow solution. Do not use ENTYVIO prefilled syringes or ENTYVIO PENs with visible particulate matter or discoloration.
- Administer each subcutaneous injection at a different anatomic location (such as thighs, any quadrant of abdomen, or upper arms) than the previous injection. Administration of ENTYVIO in the back of upper arm may only be performed by a healthcare professional or caregiver. Do not inject into moles, scars, bruises, or areas where the skin is tender, erythematous, or indurated.
Missed Subcutaneous Dose
If treatment with subcutaneous ENTYVIO is interrupted or if a scheduled dose(s) of subcutaneous ENTYVIO is missed, inject the next subcutaneous dose as soon as possible and then every 2 weeks thereafter.
In the event of incomplete dose administration (i.e., patient attempts administration of dose with ENTYVIO PEN, however it is uncertain if a full dose was administered), instruct the patient to call their pharmacy or healthcare provider.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:36.978459 · Updated: 2026-03-14T22:51:34.930520