These Highlights Do Not Include All The Information Needed To Use Acyclovir Cream Safely And Effectively. See Full Prescribing Information For Acyclovir Cream.
6e3b04a8-47a4-425e-931e-9a4fa260b303
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Acyclovir cream is a herpes simplex virus (HSV) deoxynucleoside analogue DNA polymerase inhibitor indicated for the treatment of recurrent herpes labialis (cold sores) in immunocompetent adults and adolescents 12 years of age and older.
Indications and Usage
Acyclovir cream is a herpes simplex virus (HSV) deoxynucleoside analogue DNA polymerase inhibitor indicated for the treatment of recurrent herpes labialis (cold sores) in immunocompetent adults and adolescents 12 years of age and older.
Dosage and Administration
Acyclovir cream should be applied 5 times per day for 4 days. Therapy should be initiated as early as possible following the onset of signs or symptoms of herpes labialis, i.e., during the prodrome or when lesions appear. For adolescents 12 years of age and older, the dosage is the same as in adults.
Warnings and Precautions
Only for topical use of recurrent HSV lesions on the external aspect of lips and the face. Acyclovir cream should not be applied on mucous membranes including in the eye or inside the mouth or nose. ( 5.1 ) There is a potential for irritation and contact sensitization. ( 5.2 )
Contraindications
Acyclovir cream is contraindicated in patients with known hypersensitivity to acyclovir, valacyclovir, or any component of the formulation.
Adverse Reactions
The most common adverse reactions reported were local skin reactions at the application site. ( 6.1 ) Angioedema, anaphylaxis, contact dermatitis and eczema have been reported. ( 6.2 ) To report SUSPECTED ADVERSE REACTIONS, contact Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Clinical experience has identified no interactions resulting from topical or systemic administration of other drugs concomitantly with acyclovir cream. Due to minimal systemic absorption of acyclovir cream, systemic drug interactions are unlikely.
Storage and Handling
Each gram of acyclovir cream contains 50 mg (equivalent to 5% w/w) of acyclovir in an aqueous cream base. Acyclovir cream is supplied as follows: 2 g tubes (NDC 51672-1361-9). 5 g tubes (NDC 51672-1361-5).
How Supplied
Each gram of acyclovir cream contains 50 mg (equivalent to 5% w/w) of acyclovir in an aqueous cream base. Acyclovir cream is supplied as follows: 2 g tubes (NDC 51672-1361-9). 5 g tubes (NDC 51672-1361-5).
Medication Information
Warnings and Precautions
Only for topical use of recurrent HSV lesions on the external aspect of lips and the face. Acyclovir cream should not be applied on mucous membranes including in the eye or inside the mouth or nose. ( 5.1 ) There is a potential for irritation and contact sensitization. ( 5.2 )
Indications and Usage
Acyclovir cream is a herpes simplex virus (HSV) deoxynucleoside analogue DNA polymerase inhibitor indicated for the treatment of recurrent herpes labialis (cold sores) in immunocompetent adults and adolescents 12 years of age and older.
Dosage and Administration
Acyclovir cream should be applied 5 times per day for 4 days. Therapy should be initiated as early as possible following the onset of signs or symptoms of herpes labialis, i.e., during the prodrome or when lesions appear. For adolescents 12 years of age and older, the dosage is the same as in adults.
Contraindications
Acyclovir cream is contraindicated in patients with known hypersensitivity to acyclovir, valacyclovir, or any component of the formulation.
Adverse Reactions
The most common adverse reactions reported were local skin reactions at the application site. ( 6.1 ) Angioedema, anaphylaxis, contact dermatitis and eczema have been reported. ( 6.2 ) To report SUSPECTED ADVERSE REACTIONS, contact Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Clinical experience has identified no interactions resulting from topical or systemic administration of other drugs concomitantly with acyclovir cream. Due to minimal systemic absorption of acyclovir cream, systemic drug interactions are unlikely.
Storage and Handling
Each gram of acyclovir cream contains 50 mg (equivalent to 5% w/w) of acyclovir in an aqueous cream base. Acyclovir cream is supplied as follows: 2 g tubes (NDC 51672-1361-9). 5 g tubes (NDC 51672-1361-5).
How Supplied
Each gram of acyclovir cream contains 50 mg (equivalent to 5% w/w) of acyclovir in an aqueous cream base. Acyclovir cream is supplied as follows: 2 g tubes (NDC 51672-1361-9). 5 g tubes (NDC 51672-1361-5).
Description
Acyclovir cream is a herpes simplex virus (HSV) deoxynucleoside analogue DNA polymerase inhibitor indicated for the treatment of recurrent herpes labialis (cold sores) in immunocompetent adults and adolescents 12 years of age and older.
Section 42229-5
Risk Summary
Acyclovir is minimally absorbed systemically following topical route of administration, and maternal use is not expected to result in fetal exposure to the acyclovir cream [see Clinical Pharmacology (12.3)]. Experience with topical acyclovir use in pregnant women over several decades, based on published literature including observational studies, has not identified a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes .Animal reproduction studies with systemic exposure of acyclovir have been conducted. Refer to acyclovir prescribing information for additional details.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: January 2022 5235679 37 |
|
PATIENT INFORMATION
Acyclovir (ay sye' kloe vir) Cream |
|
Important information: Acyclovir cream is for use on cold sores on the lips and around the mouth only.
Acyclovir cream should not be used in your eyes, mouth, nose, or on your genitals. |
What is acyclovir cream?
|
| Do not use acyclovir cream if you areallergic to acyclovir, valacyclovir, or any of the ingredients in acyclovir cream. See the end of this leaflet for a complete list of ingredients in acyclovir cream. |
|
What should I tell my healthcare provider before using acyclovir cream?
Before using acyclovir cream, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
How should I use acyclovir cream?
|
|
What are the possible side effects of acyclovir cream?
The most common side effects of acyclovir cream are skin reactions at the treatment site and may include: dry or cracked lips, peeling, flaking or dryness of the skin, a burning or stinging feeling, and itching. These are not all the possible side effects of acyclovir cream.
|
How should I store acyclovir cream?
Keep acyclovir cream and all medicines out of the reach of children. |
|
General information about the safe and effective use of acyclovir cream
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use acyclovir cream for a condition for which it was not prescribed. Do not give acyclovir cream to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about acyclovir cream that is written for health professionals. |
|
What are the ingredients in acyclovir cream?
Active ingredient:acyclovir Inactive ingredients:cetostearyl alcohol, mineral oil, poloxamer 407, propylene glycol, sodium lauryl sulfate, water, and white petrolatum Mfd. by: Taro Pharmaceuticals Inc., Brampton, Ontario, Canada L6T 1C1
|
Section 44425-7
Store at 20° to 25°C (68° to 77°F)[see USP Controlled Room Temperature].
5.1 General
Acyclovir cream should only be applied on the affected external aspects of the lips and face in patients with herpes labialis. Because no data are available, application to human mucous membranes is not recommended. Acyclovir cream is intended for cutaneous use only and should not be used in the eye or inside the mouth or nose.
10 Overdosage
Overdosage by topical application of acyclovir cream is unlikely because of minimal systemic exposure [see Clinical Pharmacology (12.3)] . There is no information available for overdose.
11 Description
Acyclovir, a synthetic deoxynucleoside analogue active against herpes viruses. Acyclovir cream, 5% is a formulation for topical administration.
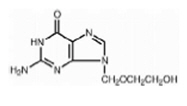
The chemical name of acyclovir is 2-amino-1,9-dihydro-9-[(2-hydroxyethoxy) methyl]-6 H-purin-6-one; it has the following structural formula:
Acyclovir is a white to off-white, crystalline powder with the molecular formula C 8H 11N 5O 3and a molecular weight of 225. The maximum solubility in water at 37°C is 2.5 mg/mL. The pKa's of acyclovir are 2.27 and 9.25.
Each gram of acyclovir cream, 5% contains 50 mg (equivalent to 5% w/w) of acyclovir and the following inactive ingredients: cetostearyl alcohol, mineral oil, poloxamer 407, propylene glycol, sodium lauryl sulfate, water, and white petrolatum.
8.4 Pediatric Use
An open-label, uncontrolled trial with acyclovir cream was conducted in 113 patients aged 12 to 17 years with recurrent herpes labialis. In this trial, therapy was applied using the same dosing regimen as in adults and subjects were followed for adverse events. The safety profile was similar to that observed in adults. Safety and effectiveness in pediatric patients less than 12 years of age have not been established.
8.5 Geriatric Use
Clinical studies of acyclovir cream did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. Systemic absorption of acyclovir after topical administration is minimal [see Clinical Pharmacology (12.3)] .
14.1 Adult Subjects
Acyclovir cream was evaluated in two double-blind, randomized, placebo (vehicle)-controlled trials for the treatment of recurrent herpes labialis. The average patient had five episodes of herpes labialis in the previous 12 months. In the first trial, the median age of subjects was 37 years (range 18 to 81 years), 74% were female, and 94% were Caucasian. In the second trial, median age of subjects was 38 years (range 18 to 87 years), 73% were female, and 94% were Caucasian. Subjects were instructed to initiate treatment within one hour of noticing signs or symptoms and continue treatment for four days, with application of study medication 5 times per day. In both studies, the mean duration of the recurrent herpes labialis episode was approximately one-half day shorter in the subjects treated with acyclovir cream (n = 682) compared with subjects treated with placebo (n = 703) for approximately 4.5 days versus 5 days, respectively. No significant difference was observed between subjects receiving acyclovir cream or placebo in the prevention of progression of cold sore lesions.
4 Contraindications
Acyclovir cream is contraindicated in patients with known hypersensitivity to acyclovir, valacyclovir, or any component of the formulation.
6 Adverse Reactions
- The most common adverse reactions reported were local skin reactions at the application site. ( 6.1)
- Angioedema, anaphylaxis, contact dermatitis and eczema have been reported. ( 6.2)
To report SUSPECTED ADVERSE REACTIONS, contact Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
Clinical experience has identified no interactions resulting from topical or systemic administration of other drugs concomitantly with acyclovir cream. Due to minimal systemic absorption of acyclovir cream, systemic drug interactions are unlikely.
12.3 Pharmacokinetics
A clinical pharmacology study was performed with acyclovir cream in adult volunteers to evaluate the percutaneous absorption of acyclovir. In this study, which included 6 male volunteers, the cream was applied to an area of 710 cm 2on the backs of the volunteers 5 times daily at intervals of 2 hours for a total of 4 days. The weight of cream applied and urinary excretion of acyclovir were measured daily. Plasma concentration of acyclovir was assayed 1 hour after the final application. The average daily urinary excretion of acyclovir was approximately 0.04% of the daily applied dose. Plasma acyclovir concentrations were below the limit of detection (0.01 µM) in 5 subjects and barely detectable (0.014 µM) in 1 subject. Systemic absorption of acyclovir from acyclovir cream is minimal in adults.
The systemic absorption of acyclovir following topical application of cream has not been evaluated in patients <18 years of age.
1 Indications and Usage
Acyclovir cream is a herpes simplex virus (HSV) deoxynucleoside analogue DNA polymerase inhibitor indicated for the treatment of recurrent herpes labialis (cold sores) in immunocompetent adults and adolescents 12 years of age and older.
14.2 Pediatric Subjects
An open-label, uncontrolled trial with acyclovir cream was conducted in 113 patients aged 12 to 17 years with recurrent herpes labialis. In this trial, therapy was applied using the same dosing regimen as in adults and subjects were followed for adverse events. The safety profile was similar to that observed in adults.
12.1 Mechanism of Action
Acyclovir is an antiviral drug active against α-herpesviruses [see Microbiology (12.4)] .
5.2 Contact Sensitization
Acyclovir cream has a potential for irritation and contact sensitization [see Adverse Reactions (6.1].
The effect of acyclovir cream has not been established in immunocompromised patients.
5 Warnings and Precautions
2 Dosage and Administration
Acyclovir cream should be applied 5 times per day for 4 days. Therapy should be initiated as early as possible following the onset of signs or symptoms of herpes labialis, i.e., during the prodrome or when lesions appear.
For adolescents 12 years of age and older, the dosage is the same as in adults.
3 Dosage Forms and Strengths
Each gram of acyclovir cream contains 50 mg (equivalent to 5% w/w) of acyclovir.
6.2 Postmarketing Experience
In addition to adverse events reported from clinical trials, the following events have been identified during postapproval use of acyclovir cream. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to acyclovir cream.
General: Angioedema, anaphylaxis.
Skin: Contact dermatitis, eczema.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug, and may not reflect the rates observed in practice.
In five double-blind, placebo-controlled trials, 1124 patients were treated with acyclovir cream and 1161 with placebo (vehicle) cream. Local application site reactions were reported by 5% of patients receiving acyclovir cream and 4% of patients receiving placebo. The most common adverse reactions at the site of topical application were dry lips, desquamation, dryness of skin, cracked lips, burning skin, pruritus, flakiness of skin, and stinging on skin; each adverse reaction occurred in less than 1% of patients receiving acyclovir cream and placebo. Three patients on acyclovir cream and one patient on placebo discontinued treatment due to an adverse event.
An additional study, enrolling 22 healthy adults, was conducted to evaluate the dermal tolerance of acyclovir cream compared with vehicle using single occluded and semi-occluded patch testing methodology. Both acyclovir cream and placebo showed a high and cumulative irritation potential. Another study, enrolling 251 healthy adults, was conducted to evaluate the contact sensitization potential of acyclovir cream using repeat insult patch testing methodology. Of 202 evaluable subjects, possible cutaneous sensitization reactions were observed in the same 4 (2%) subjects with both acyclovir cream and placebo, and these reactions to both acyclovir cream and placebo were confirmed in 3 subjects upon rechallenge. The sensitizing ingredient(s) has not been identified.
The safety profile in patients 12 to 17 years of age was similar to that observed in adults.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
Each gram of acyclovir cream contains 50 mg (equivalent to 5% w/w) of acyclovir in an aqueous cream base. Acyclovir cream is supplied as follows:
2 g tubes (NDC 51672-1361-9).
5 g tubes (NDC 51672-1361-5).
Principal Display Panel 5 G Tube Carton
NDC 51672-1361-5
5 g
Acyclovir
Cream 5%
USE ONLY FOR COLD SORES
Rx only
Keep this and all medications out of the reach of children.
TARO
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Systemic exposure following topical administration of acyclovir is minimal. Dermal carcinogenicity studies were not conducted. Results from the studies of carcinogenesis, mutagenesis and fertility are not included in the full prescribing information for acyclovir cream due to the minimal exposures of acyclovir that result from dermal application. Information on these studies is available in the full prescribing information for acyclovir capsules, tablets, and suspension and acyclovir for injection.
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
Acyclovir is minimally absorbed systemically following topical route of administration, and maternal use is not expected to result in fetal exposure to the acyclovir cream [see Clinical Pharmacology (12.3)]. Experience with topical acyclovir use in pregnant women over several decades, based on published literature including observational studies, has not identified a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes .Animal reproduction studies with systemic exposure of acyclovir have been conducted. Refer to acyclovir prescribing information for additional details.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: January 2022 5235679 37 |
|
PATIENT INFORMATION
Acyclovir (ay sye' kloe vir) Cream |
|
Important information: Acyclovir cream is for use on cold sores on the lips and around the mouth only.
Acyclovir cream should not be used in your eyes, mouth, nose, or on your genitals. |
What is acyclovir cream?
|
| Do not use acyclovir cream if you areallergic to acyclovir, valacyclovir, or any of the ingredients in acyclovir cream. See the end of this leaflet for a complete list of ingredients in acyclovir cream. |
|
What should I tell my healthcare provider before using acyclovir cream?
Before using acyclovir cream, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
How should I use acyclovir cream?
|
|
What are the possible side effects of acyclovir cream?
The most common side effects of acyclovir cream are skin reactions at the treatment site and may include: dry or cracked lips, peeling, flaking or dryness of the skin, a burning or stinging feeling, and itching. These are not all the possible side effects of acyclovir cream.
|
How should I store acyclovir cream?
Keep acyclovir cream and all medicines out of the reach of children. |
|
General information about the safe and effective use of acyclovir cream
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use acyclovir cream for a condition for which it was not prescribed. Do not give acyclovir cream to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about acyclovir cream that is written for health professionals. |
|
What are the ingredients in acyclovir cream?
Active ingredient:acyclovir Inactive ingredients:cetostearyl alcohol, mineral oil, poloxamer 407, propylene glycol, sodium lauryl sulfate, water, and white petrolatum Mfd. by: Taro Pharmaceuticals Inc., Brampton, Ontario, Canada L6T 1C1
|
Section 44425-7 (44425-7)
Store at 20° to 25°C (68° to 77°F)[see USP Controlled Room Temperature].
5.1 General
Acyclovir cream should only be applied on the affected external aspects of the lips and face in patients with herpes labialis. Because no data are available, application to human mucous membranes is not recommended. Acyclovir cream is intended for cutaneous use only and should not be used in the eye or inside the mouth or nose.
10 Overdosage (10 OVERDOSAGE)
Overdosage by topical application of acyclovir cream is unlikely because of minimal systemic exposure [see Clinical Pharmacology (12.3)] . There is no information available for overdose.
11 Description (11 DESCRIPTION)
Acyclovir, a synthetic deoxynucleoside analogue active against herpes viruses. Acyclovir cream, 5% is a formulation for topical administration.
The chemical name of acyclovir is 2-amino-1,9-dihydro-9-[(2-hydroxyethoxy) methyl]-6 H-purin-6-one; it has the following structural formula:
Acyclovir is a white to off-white, crystalline powder with the molecular formula C 8H 11N 5O 3and a molecular weight of 225. The maximum solubility in water at 37°C is 2.5 mg/mL. The pKa's of acyclovir are 2.27 and 9.25.
Each gram of acyclovir cream, 5% contains 50 mg (equivalent to 5% w/w) of acyclovir and the following inactive ingredients: cetostearyl alcohol, mineral oil, poloxamer 407, propylene glycol, sodium lauryl sulfate, water, and white petrolatum.
8.4 Pediatric Use
An open-label, uncontrolled trial with acyclovir cream was conducted in 113 patients aged 12 to 17 years with recurrent herpes labialis. In this trial, therapy was applied using the same dosing regimen as in adults and subjects were followed for adverse events. The safety profile was similar to that observed in adults. Safety and effectiveness in pediatric patients less than 12 years of age have not been established.
8.5 Geriatric Use
Clinical studies of acyclovir cream did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. Systemic absorption of acyclovir after topical administration is minimal [see Clinical Pharmacology (12.3)] .
14.1 Adult Subjects
Acyclovir cream was evaluated in two double-blind, randomized, placebo (vehicle)-controlled trials for the treatment of recurrent herpes labialis. The average patient had five episodes of herpes labialis in the previous 12 months. In the first trial, the median age of subjects was 37 years (range 18 to 81 years), 74% were female, and 94% were Caucasian. In the second trial, median age of subjects was 38 years (range 18 to 87 years), 73% were female, and 94% were Caucasian. Subjects were instructed to initiate treatment within one hour of noticing signs or symptoms and continue treatment for four days, with application of study medication 5 times per day. In both studies, the mean duration of the recurrent herpes labialis episode was approximately one-half day shorter in the subjects treated with acyclovir cream (n = 682) compared with subjects treated with placebo (n = 703) for approximately 4.5 days versus 5 days, respectively. No significant difference was observed between subjects receiving acyclovir cream or placebo in the prevention of progression of cold sore lesions.
4 Contraindications (4 CONTRAINDICATIONS)
Acyclovir cream is contraindicated in patients with known hypersensitivity to acyclovir, valacyclovir, or any component of the formulation.
6 Adverse Reactions (6 ADVERSE REACTIONS)
- The most common adverse reactions reported were local skin reactions at the application site. ( 6.1)
- Angioedema, anaphylaxis, contact dermatitis and eczema have been reported. ( 6.2)
To report SUSPECTED ADVERSE REACTIONS, contact Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
Clinical experience has identified no interactions resulting from topical or systemic administration of other drugs concomitantly with acyclovir cream. Due to minimal systemic absorption of acyclovir cream, systemic drug interactions are unlikely.
12.3 Pharmacokinetics
A clinical pharmacology study was performed with acyclovir cream in adult volunteers to evaluate the percutaneous absorption of acyclovir. In this study, which included 6 male volunteers, the cream was applied to an area of 710 cm 2on the backs of the volunteers 5 times daily at intervals of 2 hours for a total of 4 days. The weight of cream applied and urinary excretion of acyclovir were measured daily. Plasma concentration of acyclovir was assayed 1 hour after the final application. The average daily urinary excretion of acyclovir was approximately 0.04% of the daily applied dose. Plasma acyclovir concentrations were below the limit of detection (0.01 µM) in 5 subjects and barely detectable (0.014 µM) in 1 subject. Systemic absorption of acyclovir from acyclovir cream is minimal in adults.
The systemic absorption of acyclovir following topical application of cream has not been evaluated in patients <18 years of age.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Acyclovir cream is a herpes simplex virus (HSV) deoxynucleoside analogue DNA polymerase inhibitor indicated for the treatment of recurrent herpes labialis (cold sores) in immunocompetent adults and adolescents 12 years of age and older.
14.2 Pediatric Subjects
An open-label, uncontrolled trial with acyclovir cream was conducted in 113 patients aged 12 to 17 years with recurrent herpes labialis. In this trial, therapy was applied using the same dosing regimen as in adults and subjects were followed for adverse events. The safety profile was similar to that observed in adults.
12.1 Mechanism of Action
Acyclovir is an antiviral drug active against α-herpesviruses [see Microbiology (12.4)] .
5.2 Contact Sensitization
Acyclovir cream has a potential for irritation and contact sensitization [see Adverse Reactions (6.1].
The effect of acyclovir cream has not been established in immunocompromised patients.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Acyclovir cream should be applied 5 times per day for 4 days. Therapy should be initiated as early as possible following the onset of signs or symptoms of herpes labialis, i.e., during the prodrome or when lesions appear.
For adolescents 12 years of age and older, the dosage is the same as in adults.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Each gram of acyclovir cream contains 50 mg (equivalent to 5% w/w) of acyclovir.
6.2 Postmarketing Experience
In addition to adverse events reported from clinical trials, the following events have been identified during postapproval use of acyclovir cream. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to acyclovir cream.
General: Angioedema, anaphylaxis.
Skin: Contact dermatitis, eczema.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug, and may not reflect the rates observed in practice.
In five double-blind, placebo-controlled trials, 1124 patients were treated with acyclovir cream and 1161 with placebo (vehicle) cream. Local application site reactions were reported by 5% of patients receiving acyclovir cream and 4% of patients receiving placebo. The most common adverse reactions at the site of topical application were dry lips, desquamation, dryness of skin, cracked lips, burning skin, pruritus, flakiness of skin, and stinging on skin; each adverse reaction occurred in less than 1% of patients receiving acyclovir cream and placebo. Three patients on acyclovir cream and one patient on placebo discontinued treatment due to an adverse event.
An additional study, enrolling 22 healthy adults, was conducted to evaluate the dermal tolerance of acyclovir cream compared with vehicle using single occluded and semi-occluded patch testing methodology. Both acyclovir cream and placebo showed a high and cumulative irritation potential. Another study, enrolling 251 healthy adults, was conducted to evaluate the contact sensitization potential of acyclovir cream using repeat insult patch testing methodology. Of 202 evaluable subjects, possible cutaneous sensitization reactions were observed in the same 4 (2%) subjects with both acyclovir cream and placebo, and these reactions to both acyclovir cream and placebo were confirmed in 3 subjects upon rechallenge. The sensitizing ingredient(s) has not been identified.
The safety profile in patients 12 to 17 years of age was similar to that observed in adults.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Each gram of acyclovir cream contains 50 mg (equivalent to 5% w/w) of acyclovir in an aqueous cream base. Acyclovir cream is supplied as follows:
2 g tubes (NDC 51672-1361-9).
5 g tubes (NDC 51672-1361-5).
Principal Display Panel 5 G Tube Carton (PRINCIPAL DISPLAY PANEL - 5 g Tube Carton)
NDC 51672-1361-5
5 g
Acyclovir
Cream 5%
USE ONLY FOR COLD SORES
Rx only
Keep this and all medications out of the reach of children.
TARO
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Systemic exposure following topical administration of acyclovir is minimal. Dermal carcinogenicity studies were not conducted. Results from the studies of carcinogenesis, mutagenesis and fertility are not included in the full prescribing information for acyclovir cream due to the minimal exposures of acyclovir that result from dermal application. Information on these studies is available in the full prescribing information for acyclovir capsules, tablets, and suspension and acyclovir for injection.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:00.708904 · Updated: 2026-03-14T22:28:47.714661