Premier Value Nighttime Sleep-aid
6d24b6e1-7959-47c6-a09b-c7e00a437495
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Nighttime sleep-aid
Medication Information
Purpose
Nighttime sleep-aid
Description
Drug Facts
Uses
-
•for the relief of occasional sleeplessness
-
•reduces time to fall asleep if you have difficulty falling asleep
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.
Section 50566-9
Stop use and ask a doctor if sleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of serious underlying medical illness.
Section 50568-5
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers or any other sleep aid
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Directions
-
•take only one dose per day (24 hours) - see Overdose warning
-
•use dose cup or tablespoon
|
adults & children 12 yrs & over |
One Dose = 30 mL (2 tablespoons) at bed time if needed or as directed by a doctor |
Do Not Use
-
•for children under 12 years of age
-
•with any other product containing diphenhydramine, even one used on skin
-
•with other drugs that cause drowsiness such as antihistamines and nighttime cold/flu products
Overdose Warning
Taking more than directed can cause serious health problems. In case of overdose, get medical help or contact a Poison Control Center right away.
Other Information
-
•each 30 mL dose (2 tablespoons) contains: sodium 23 mg
-
•store at room temperature
-
•protect from light. Does not meet USP <671>.
Inactive Ingredients
anhydrous citric acid, carboxymethylcelluose sodium, FD&C blue 1, FD&C red 40, flavor, glycerin, potassium citrate, purified water, sodium benzoate, sorbitol, sucralose.
When Using This Product
-
•avoid alcoholic beverages and other drugs that cause drowsiness
-
•drowsiness will occur
-
•be careful when driving a motor vehicle or operating machinery
Ask A Doctor Before Use If You Have
-
•a breathing problem such as asthma, emphysema, or chronic bronchitis
-
•glaucoma
-
•difficulty in urination due to enlargement of the prostate gland
-
•heart disease
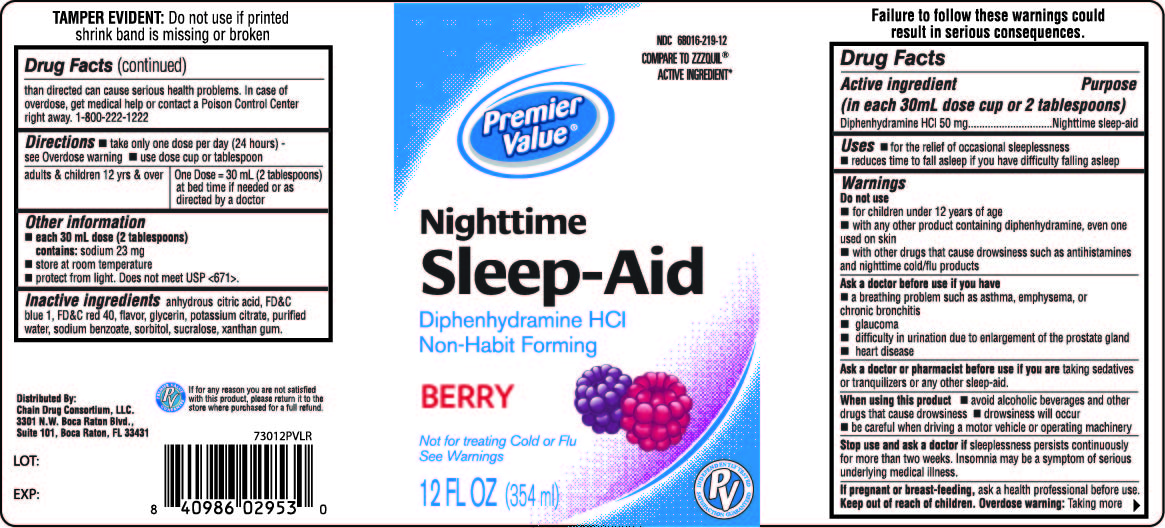
Principal Display Panel 354 Ml Bottle Label
Premier Value
Nighttime
Sleep-Aid
Diphenhydramine HCl /
-
•Non-Habit Forming
Berry Flavor
Not for treating Cold or Flu
See Warnings
12 FL OZ (354 ml)
Active Ingredients (in Each 30 Ml Dose Cup Or 2 Tablespoons)
Diphenhydramine HCl 50 mg
Structured Label Content
Uses
-
•for the relief of occasional sleeplessness
-
•reduces time to fall asleep if you have difficulty falling asleep
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 50566-9 (50566-9)
Stop use and ask a doctor if sleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of serious underlying medical illness.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers or any other sleep aid
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Nighttime sleep-aid
Directions
-
•take only one dose per day (24 hours) - see Overdose warning
-
•use dose cup or tablespoon
|
adults & children 12 yrs & over |
One Dose = 30 mL (2 tablespoons) at bed time if needed or as directed by a doctor |
Do Not Use (Do not use)
-
•for children under 12 years of age
-
•with any other product containing diphenhydramine, even one used on skin
-
•with other drugs that cause drowsiness such as antihistamines and nighttime cold/flu products
Overdose Warning (Overdose warning)
Taking more than directed can cause serious health problems. In case of overdose, get medical help or contact a Poison Control Center right away.
Other Information (Other information)
-
•each 30 mL dose (2 tablespoons) contains: sodium 23 mg
-
•store at room temperature
-
•protect from light. Does not meet USP <671>.
Inactive Ingredients (Inactive ingredients)
anhydrous citric acid, carboxymethylcelluose sodium, FD&C blue 1, FD&C red 40, flavor, glycerin, potassium citrate, purified water, sodium benzoate, sorbitol, sucralose.
When Using This Product (When using this product)
-
•avoid alcoholic beverages and other drugs that cause drowsiness
-
•drowsiness will occur
-
•be careful when driving a motor vehicle or operating machinery
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
-
•a breathing problem such as asthma, emphysema, or chronic bronchitis
-
•glaucoma
-
•difficulty in urination due to enlargement of the prostate gland
-
•heart disease
Principal Display Panel 354 Ml Bottle Label (PRINCIPAL DISPLAY PANEL - 354 ml Bottle Label)
Premier Value
Nighttime
Sleep-Aid
Diphenhydramine HCl /
-
•Non-Habit Forming
Berry Flavor
Not for treating Cold or Flu
See Warnings
12 FL OZ (354 ml)
Active Ingredients (in Each 30 Ml Dose Cup Or 2 Tablespoons) (Active ingredients (in each 30 mL dose cup or 2 tablespoons))
Diphenhydramine HCl 50 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:05.742023 · Updated: 2026-03-14T23:04:35.063520