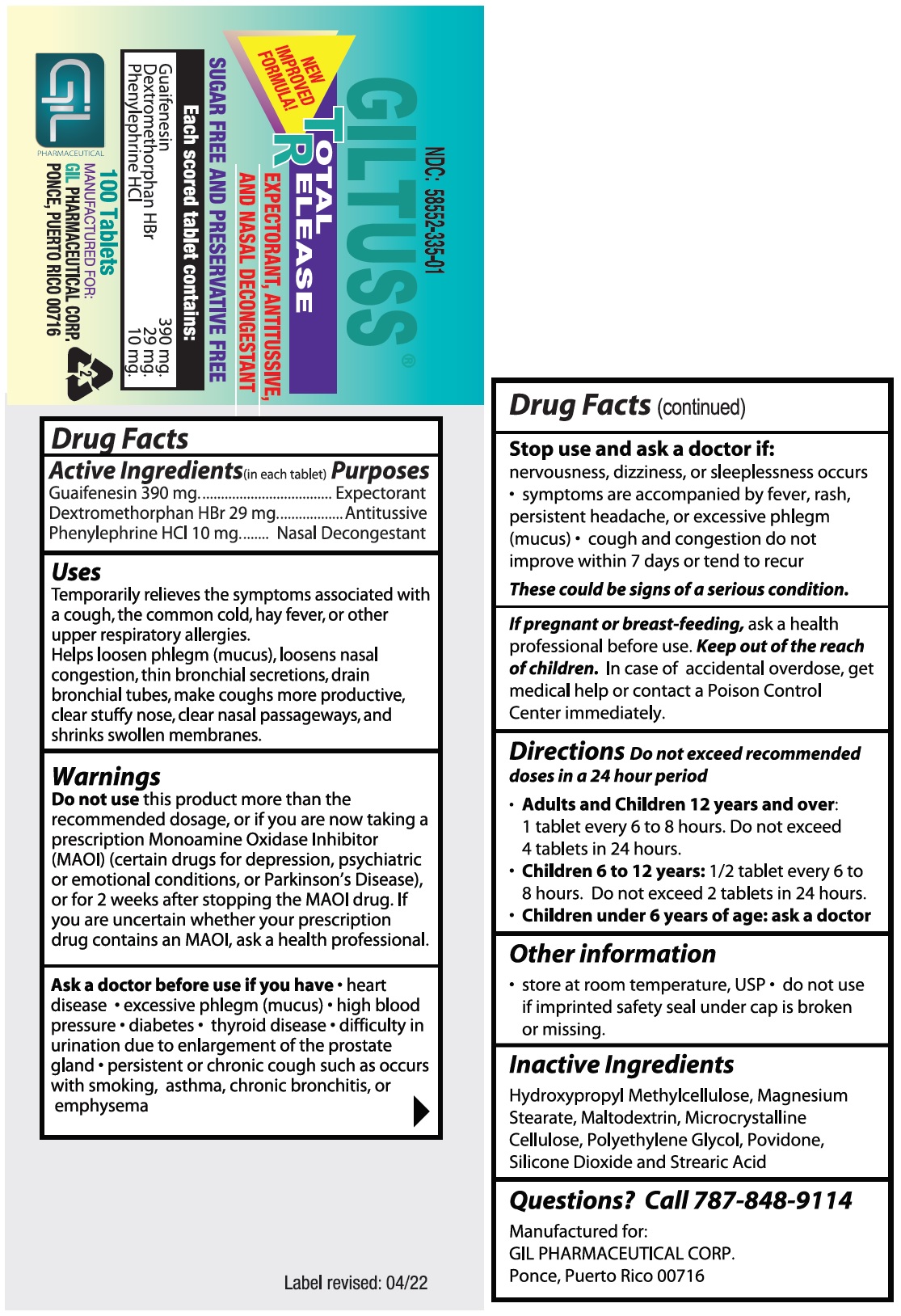
Giltuss ®
6cfefcd3-110a-4381-91cf-0f71407ac78e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients (in each tablet) Guaifenesin 390 mg. Dextromethorphan HBr 29 mg. Phenylephrine HCl 10 mg.
Purpose
Expectorant Antitussive Nasal Decongestant
Medication Information
Purpose
Expectorant Antitussive Nasal Decongestant
Description
Active Ingredients (in each tablet) Guaifenesin 390 mg. Dextromethorphan HBr 29 mg. Phenylephrine HCl 10 mg.
Uses
Temporarily relieves the symptoms associated with a cough, the common cold, hay fever, or other upper respiratory allergies.
Helps loosen phlegm (mucus), loosens nasal congestion, thin bronchial secretions, drain bronchial tubes, make coughs more productive, clear stuffy nose, clear nasal passageways, and shrinks swollen membranes.
Section 50565-1
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use . Keep out of the reach of children . In case of accidental overdose, get medical help or contact a Poison Control Center immediately.
Purposes
Expectorant
Antitussive
Nasal Decongestant
Warnings
Do not usethis product more than the recommended dosage, or if you are now taking a prescription Monoamine Oxidase Inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s Disease), or for 2 weeks after stopping the MAOI drug.
If you are uncertain whether your prescription drug contains an MAOI, ask a health professional.
Drug Facts
Active Ingredients (in each tablet)
Guaifenesin 390 mg.
Dextromethorphan HBr 29 mg.
Phenylephrine HCl 10 mg.
Directions
Do not exceed recommended doses in a 24 hour period
- Adults and Children 12 years and over:1 tablet every 6 to 8 hours. Do not exceed 4 tablets in 24 hours.
- Children 6 to 12 years:1/2 tablet every 6 to 8 hours. Do not exceed 2 tablets in 24 hours.
- Children under 6 years of age: ask a doctor.
Other Information
• store at room temperature, USP.
• do not use if imprinted safety seal under cap is broken or missing.
Inactive Ingredients
Hydroxypropyl Methylcellulose, Magnesium Stearate, Maltodextrin, Microcrystalline Cellulose, Polyethylene Glycol, Povidone, Silicone Dioxide and Stearic Acid.
Questions? Call 787 848 9114
Manufactured for:
GIL PHARMACEUTICAL CORP.
Ponce, Puerto Rico 00716
Label revised: 04/22
Stop Use and Ask A Doctor If:
• nervousness, dizziness, or sleeplessness occurs.
• symptoms are accompanied by fever, rash, persistent headache, or excessive phlegm (mucus).
• cough and congestion do not improve within 7 days or tend to recur.
These could be signs of a serious condition.
Ask A Doctor Before Use If You Have
• heart disease.
• excessive phlegm (mucus).
• high blood pressure.
• diabetes.
• thyroid disease.
• difficulty in urination due to enlargement of the prostate gland.
• persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema.
Package Label.principal Display Panel
Giltuss ®Total Release - NDC-58552-335-01 - 100's Bottle Label.
Structured Label Content
Uses
Temporarily relieves the symptoms associated with a cough, the common cold, hay fever, or other upper respiratory allergies.
Helps loosen phlegm (mucus), loosens nasal congestion, thin bronchial secretions, drain bronchial tubes, make coughs more productive, clear stuffy nose, clear nasal passageways, and shrinks swollen membranes.
Section 50565-1 (50565-1)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use . Keep out of the reach of children . In case of accidental overdose, get medical help or contact a Poison Control Center immediately.
Purposes
Expectorant
Antitussive
Nasal Decongestant
Warnings
Do not usethis product more than the recommended dosage, or if you are now taking a prescription Monoamine Oxidase Inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s Disease), or for 2 weeks after stopping the MAOI drug.
If you are uncertain whether your prescription drug contains an MAOI, ask a health professional.
Drug Facts
Active Ingredients (in each tablet)
Guaifenesin 390 mg.
Dextromethorphan HBr 29 mg.
Phenylephrine HCl 10 mg.
Directions
Do not exceed recommended doses in a 24 hour period
- Adults and Children 12 years and over:1 tablet every 6 to 8 hours. Do not exceed 4 tablets in 24 hours.
- Children 6 to 12 years:1/2 tablet every 6 to 8 hours. Do not exceed 2 tablets in 24 hours.
- Children under 6 years of age: ask a doctor.
Other Information (Other information)
• store at room temperature, USP.
• do not use if imprinted safety seal under cap is broken or missing.
Inactive Ingredients
Hydroxypropyl Methylcellulose, Magnesium Stearate, Maltodextrin, Microcrystalline Cellulose, Polyethylene Glycol, Povidone, Silicone Dioxide and Stearic Acid.
Questions? Call 787 848 9114 (Questions? Call 787-848-9114)
Manufactured for:
GIL PHARMACEUTICAL CORP.
Ponce, Puerto Rico 00716
Label revised: 04/22
Stop Use and Ask A Doctor If: (Stop use and ask a doctor if:)
• nervousness, dizziness, or sleeplessness occurs.
• symptoms are accompanied by fever, rash, persistent headache, or excessive phlegm (mucus).
• cough and congestion do not improve within 7 days or tend to recur.
These could be signs of a serious condition.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
• heart disease.
• excessive phlegm (mucus).
• high blood pressure.
• diabetes.
• thyroid disease.
• difficulty in urination due to enlargement of the prostate gland.
• persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema.
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
Giltuss ®Total Release - NDC-58552-335-01 - 100's Bottle Label.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:01.997851 · Updated: 2026-03-14T23:12:59.392851